The Quick Removal of Toxic Dye Molecules by an Efficient Adsorptive BiOI/Bi2MoO6 Heterostructure
Abstract
:1. Introduction
2. Results and Discussion
2.1. XRD, FTIR, and XPS Analyses
2.2. Morphology and Surface Characterization
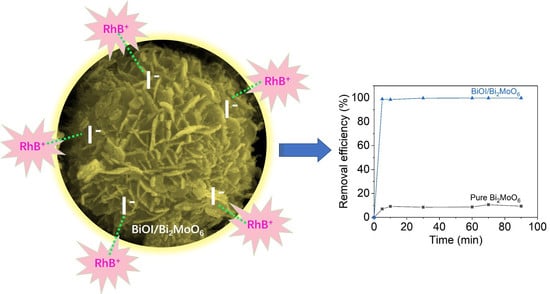
2.3. Toxic Dyes Adsorption Performance by BOI/BMO
2.4. Adsorption Kinetics and Isotherms
2.5. Thermodynamic Analysis
2.6. The Stability of BOI2/BMO
3. Materials and Methods
3.1. Materials
3.2. Synthesis of Pure Bi2MoO6, BiOI, and Heterostructure BiOI/Bi2MoO6
3.3. Composition, Structure, and Morphology Characterization
3.4. Adsorption of RhB and Other Dyes by the Bi-Based Adsorbents
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zhao, G.Y.; Liu, L.J.; Li, J.R.; Liu, Q. Efficient removal of dye MB: Through the combined action of adsorption and photodegradation from NiFe2O4/Ag3PO4. J. Alloys Compd. 2016, 664, 169–174. [Google Scholar] [CrossRef]
- Kant, R. Textile dyeing industry an environmental hazard. Nat. Sci. 2012, 4, 22–26. [Google Scholar] [CrossRef] [Green Version]
- Solís, M.; Solís, A.; Pérez, H.I.; Manjarrez, N.; Flores, M. Microbial decolouration of azo dyes: A review. Process Biochem. 2012, 47, 1723–1748. [Google Scholar] [CrossRef]
- Kropf, C.; Segner, H.; Fent, K. ABC transporters and xenobiotic defense systems in early life stages of rainbow trout (Oncorhynchus mykiss). Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2016, 185–186, 45–56. [Google Scholar] [CrossRef] [Green Version]
- Mohanty, L.; Sundar Pattanayak, D.; Singhal, R.; Pradhan, D.; Kumar Dash, S. Enhanced photocatalytic degradation of rhodamine B and malachite green employing BiFeO3/g-C3N4 nanocomposites: An efficient visible-light photocatalyst. Inorg. Chem. Commun. 2022, 138, 109286. [Google Scholar] [CrossRef]
- Rafique, M.A.; Jamal, A.; Afzal, G.; Abrar, S.; Kiran, S.; Nosheen, S.; Hussain, H.M.H.; Majeed, T. Photocatalytic mediated remediation of synthetic dyes effluent using zero-valent iron: A comparative study. Desalin. Water Treat. 2021, 237, 284–291. [Google Scholar] [CrossRef]
- Xu, M.Y.; Deng, Y.C.; Li, S.H.; Zheng, J.Y.; Liu, J.Y.; Tremblay, P.L.; Zhang, T. Bacterial cellulose flakes loaded with Bi2MoO6 nanoparticles and quantum dots for the photodegradation of antibiotic and dye pollutants. Chemosphere 2023, 312, 137249. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, R.; Lee, N.Y. Adsorptive removal of organic pollutant methylene blue using polysaccharide-based composite hydrogels. Chemosphere 2022, 286, 131890. [Google Scholar] [CrossRef] [PubMed]
- Iwuozor, K.O.; Ighalo, J.O.; Emenike, E.C.; Ogunfowora, L.A.; Igwegbe, C.A. Adsorption of methyl orange: A review on adsorbent performance. Curr. Res. Green Sustain. Chem. 2021, 4, 100179. [Google Scholar] [CrossRef]
- Al-Gheethi, A.A.; Azhar, Q.M.; Senthil Kumar, P.; Yusuf, A.A.; Al-Buriahi, A.K.; Radin Mohamed, R.M.S.; Al-Shaibani, M.M. Sustainable approaches for removing Rhodamine B dye using agricultural waste adsorbents: A review. Chemosphere 2022, 287, 132080. [Google Scholar] [CrossRef]
- Deng, Y.C.; Xu, M.Y.; Jiang, X.Y.; Wang, J.T.; Tremblay, P.L.; Zhang, T. Versatile iodine-doped BiOCl with abundant oxygen vacancies and (110) crystal planes for enhanced pollutant photodegradation. Environ. Res. 2023, 216, 114808. [Google Scholar] [CrossRef]
- Saravanan, S.; Carolin, C.F.; Kumar, P.S.; Chitra, B.; Rangasamy, G. Biodegradation of textile dye Rhodamine-B by Brevundimonas diminuta and screening of their breakdown metabolites. Chemosphere 2022, 308, 136266. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.B.; Zhang, J.L.; Wu, S.Y.; Zhou, Z.C. Efficient coagulation and removal of aqueous graphene oxide by different dyes. Desalin. Water Treat. 2021, 217, 339–349. [Google Scholar] [CrossRef]
- Yen Doan, T.H.; Minh Chu, T.P.; Dinh, T.D.; Nguyen, T.H.; Tu Vo, T.C.; Nguyen, N.M.; Nguyen, B.H.; Nguyen, T.A.; Pham, T.D. Adsorptive removal of rhodamine B using novel adsorbent-based surfactant-modified alpha alumina nanoparticles. J. Anal. Methods Chem. 2020, 2020, 6676320. [Google Scholar] [CrossRef]
- Du, H.L.; Zhang, Y.S.; Jiang, H.R.; Wang, H. Adsorption of rhodamine B on polyvinyl chloride, polystyrene, and polyethylene terephthalate microplastics in aqueous environments. Environ. Technol. Innov. 2022, 27, 102495. [Google Scholar] [CrossRef]
- Singh, A.; Singh, A.K.; Liu, J.; Kumar, A. Syntheses, design strategies, and photocatalytic charge dynamics of metal–organic frameworks (MOFs): A catalyzed photo-degradation approach towards organic dyes. Catal. Sci. Technol. 2021, 11, 3946–3989. [Google Scholar] [CrossRef]
- Dong, X.; Li, Y.; Li, D.; Liao, D.; Qin, T.; Prakash, O.; Kumar, A.; Liu, J. A new 3D 8-connected Cd(II) MOF as a potent photocatalyst for oxytetracycline antibiotic degradation. CrystEngComm 2022, 24, 6933–6943. [Google Scholar] [CrossRef]
- Zheng, M.; Chen, J.; Zhang, L.; Cheng, Y.; Lu, C.; Liu, Y.; Singh, A.; Trivedi, M.; Kumar, A.; Liu, J. Metal organic frameworks as efficient adsorbents for drugs from wastewater. Mater. Today Commun. 2022, 31, 103514. [Google Scholar]
- Du, X.C.; Zhu, J.H.; Quan, Z.J.; Wang, X.C. Adsorption of rhodamine B by organic porous materials rich in nitrogen, oxygen, and sulfur heteroatoms. N. J. Chem. 2021, 45, 3448–3453. [Google Scholar] [CrossRef]
- Wang, X.H.; Chen, S.Q.; Sun, J.C.; Zhang, D.H.; Yan, Z.H.; Xu, X.Y.; Song, J.Q. Synthesis of large pore sized mesoporous carbon using alumina-templated strategy for high-performance RhB removal. Microporous Mesoporous Mater. 2021, 318, 110993. [Google Scholar] [CrossRef]
- Li, R.Z.; Chen, H.Y.; Xiong, J.R.; Xu, X.Y.; Cheng, J.J.; Liu, X.Y.; Liu, G. A mini review on bismuth-based z-scheme photocatalysts. Materials 2020, 13, 5057. [Google Scholar] [CrossRef]
- Wang, D.J.; Shen, H.D.; Guo, L.; Wang, C.; Fu, F. Porous BiOBr/Bi2MoO6 heterostructures for highly selective adsorption of methylene blue. ACS Omega 2016, 1, 566–577. [Google Scholar] [CrossRef] [Green Version]
- Yang, H.Y.; Zhao, S.Q.; Zhuang, X.J.; Asuha, S. Hydrothermal synthesis of Bi2WO6/mesoporous TiO2 nanocomposites and their adsorptive properties. J. Nanopart. Res. 2022, 24, 159. [Google Scholar] [CrossRef]
- Shah, A.H.; Yuan, C.; Hao, W.Y.; Gu, W.H.; Liang, S.Y.; Abideen, Z.U.; Wahid, F.; Teng, F. Adsorption kinetics of simulated mixture wastewaters over porous Bi2MoO6@BiOCl@MOF-199 heterostructure. J. Solid State Chem. 2022, 307, 122835. [Google Scholar] [CrossRef]
- Kong, D.F.; Li, W.J.; Yan, T.J.; Wang, Z.; Kong, D.S.; You, J.M. Preparation of novel BiVO4 nanofibers and their excellent adsorptive properties. Mater. Res. Bull. 2018, 105, 84–90. [Google Scholar] [CrossRef]
- Liu, H.J.; Chen, M.; Wei, D.D.; Ma, Y.Q.; Wang, F.L.; Zhang, Q.X.; Shi, J.L.; Zhang, H.; Peng, J.B.; Liu, G.G.; et al. Smart removal of dye pollutants via dark adsorption and light desorption at recyclable Bi2O2CO3 nanosheets interface. ACS Appl. Mater. Interfaces 2020, 12, 20490–20499. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.H.; Gu, W.H.; Abideen, Z.U.; Teng, F. Removal of chromium from aqueous solution by porous Bi2MoO6@BiOCl nanostructure. J. Solid State Chem. 2020, 292, 121719. [Google Scholar] [CrossRef]
- Li, Y.; Wang, Z.; Huang, B.; Dai, Y.; Zhang, X.; Qin, X. Synthesis of BiOBr-PVP hybrids with enhanced adsorption-photocatalytic properties. Appl. Surf. Sci. 2015, 347, 258–264. [Google Scholar] [CrossRef]
- Yu, H.B.; Jiang, L.B.; Wang, H.; Huang, B.B.; Yuan, X.Z.; Huang, J.H.; Zhang, J.; Zeng, G.M. Modulation of Bi2MoO6-based materials for photocatalytic water splitting and environmental application: A critical review. Small 2019, 15, 1901008. [Google Scholar] [CrossRef]
- Yang, L.; Du, C.Y.; Tan, S.Y.; Zhang, Z.; Song, J.H.; Su, Y.H.; Zhang, Y.; Wang, S.T.; Yu, G.L.; Chen, H.; et al. Improved photocatalytic properties of Fe(III) ion doped Bi2MoO6 for the oxidation of organic pollutants. Ceram. Int. 2021, 47, 5786–5794. [Google Scholar] [CrossRef]
- Di, J.; Xia, J.X.; Ji, M.X.; Li, H.P.; Xu, H.; Li, H.M.; Chen, R. The synergistic role of carbon quantum dots for the improved photocatalytic performance of Bi2MoO6. Nanoscale 2015, 7, 11433–11443. [Google Scholar] [CrossRef]
- Li, S.J.; Liu, Y.P.; Long, Y.Q.; Mo, L.Y.; Zhang, H.Q.; Liu, J.S. Facile synthesis of Bi2MoO6 microspheres decorated by CdS nanoparticles with efficient photocatalytic removal of levfloxacin antibiotic. Catalysts 2018, 8, 477. [Google Scholar] [CrossRef] [Green Version]
- Yan, T.; Sun, M.; Liu, H.Y.; Wu, T.T.; Liu, X.J.; Yan, Q.; Xu, W.G.; Du, B. Fabrication of hierarchical BiOI/Bi2MoO6 heterojunction for degradation of bisphenol A and dye under visible light irradiation. J. Alloys Compd. 2015, 634, 223–231. [Google Scholar] [CrossRef]
- Matsuura, I.; Schuit, G.C.A. Adsorption and reaction of adsorbed species on Bi2MoO6 catalyst: Influence on sintering and of temperature of reduction. J. Catal. 1972, 25, 314–325. [Google Scholar] [CrossRef]
- Zhou, G.L.; Tian, Z.X.; Sun, H.G.; Zhang, J.M.; Zhao, H.Y.; Li, P.; Sun, H.Y. Understanding the photocatalytic mechanisms of the BiOI/Bi2MoO6 and BiOCl/Bi2MoO6 heterostructures: First-principles study. J. Phys. Chem. Solids 2020, 146, 109577. [Google Scholar] [CrossRef]
- Wang, S.M.; Guan, Y.; Wang, L.P.; Zhao, W.; He, H.; Xiao, J.; Yang, S.G.; Sun, C. Fabrication of a novel bifunctional material of BiOI/Ag3VO4 with high adsorption–photocatalysis for efficient treatment of dye wastewater. Appl. Catal. B Environ. 2015, 168–169, 448–457. [Google Scholar] [CrossRef]
- Ma, F.Q.; Yao, J.W.; Zhang, Y.F.; Wei, Y. Oxygen vacancy promoting adsorption property of BiOI microspheres modified with SDS. Chin. Chem. Lett. 2018, 29, 1689–1691. [Google Scholar] [CrossRef]
- Ren, K.X.; Zhang, K.; Liu, J.; Luo, H.; Huang, Y.B.; Yu, X.B. Controllable synthesis of hollow/flower-like BiOI microspheres and highly efficient adsorption and photocatalytic activity. CrystEngComm 2012, 14, 4384–4390. [Google Scholar] [CrossRef]
- Phuruangrat, A.; Ekthammathat, N.; Kuntalue, B.; Dumrongrojthanath, P.; Thongtem, S.; Thongtem, T. Hydrothermal synthesis, characterization, and optical properties of Ce doped Bi2MoO6 nanoplates. J. Nanomater. 2014, 2014, 934165. [Google Scholar]
- Holzwarth, U.; Gibson, N. The Scherrer equation versus the “Debye-Scherrer equation”. Nat. Nanotechol. 2011, 6, 534. [Google Scholar] [CrossRef]
- Li, W.T.; Zheng, Y.F.; Yin, H.Y.; Song, X.C. Heterojunction BiOI/Bi2MoO6 nanocomposite with much enhanced photocatalytic activity. J. Nanopart. Res. 2015, 17, 271. [Google Scholar] [CrossRef]
- Dumrongrojthanath, P.; Phuruangrat, A.; Thongtem, S.; Thongtem, T. Hydrothermal synthesis and characterization of visible light-driven I-doped Bi2MoO6 photocatalyst. J. Iran. Chem. Soc. 2018, 16, 733–739. [Google Scholar] [CrossRef]
- Qin, L.; Li, Y.; Liang, F.; Li, L.; Lan, Y.; Li, Z.; Lu, X.; Yang, M.; Ma, D. A microporous 2D cobalt-based MOF with pyridyl sites and open metal sites for selective adsorption of CO2. Microporous Mesoporous Mater. 2022, 341, 112098. [Google Scholar] [CrossRef]
- Liu, Z.; Liu, X.Q.; Yu, C.L.; Wei, L.F.; Ji, H.B. Fabrication and characterization of I doped Bi2MoO6 microspheres with distinct performance for removing antibiotics and Cr(VI) under visible light illumination. Sep. Purif. Technol. 2020, 247, 116951. [Google Scholar] [CrossRef]
- Sin, J.C.; Lam, S.M.; Zeng, H.; Lin, H.; Li, H.; Tham, K.O.; Mohamed, A.R.; Lim, J.W.; Qin, Z.Z. Magnetic NiFe2O4 nanoparticles decorated on N-doped BiOBr nanosheets for expeditious visible light photocatalytic phenol degradation and hexavalent chromium reduction via a Z-scheme heterojunction mechanism. Appl. Surf. Sci. 2021, 559, 559–562. [Google Scholar] [CrossRef]
- Yang, Z.X.; Wang, R.Q.; Xu, L.J.; Liu, C.L.; Cheng, Y.; Jiang, Z.; Liu, Y.L.; Zhang, T.; Li, J.Y.; Liu, X.Z. Highly efficient flower-like Dy3+-doped Bi2MoO6 photocatalyst under simulated sunlight: Design, fabrication and characterization. Opt. Mater. 2021, 116, 111–114. [Google Scholar] [CrossRef]
- Sun, X.M.; Lu, J.; Wu, J.; Guan, D.Y.; Liu, Q.Z.; Yan, N.Q. Enhancing photocatalytic activity on gas-phase heavy metal oxidation with self-assembled BiOI/BiOCl microflowers. J. Colloid Interface Sci. 2019, 546, 32–42. [Google Scholar] [CrossRef]
- Xia, S.Q.; Jia, R.Y.; Feng, F.; Xie, K.; Li, H.X.; Jing, D.F.; Xu, X.T. Effect of solids retention time on antibiotics removal performance and microbial communities in an A/O-MBR process. Bioresour. Technol. 2012, 106, 36–43. [Google Scholar] [CrossRef]
- Tang, W.H.; Zhang, Y.L.; Guo, H.G.; Liu, Y. Heterogeneous activation of peroxymonosulfate for bisphenol AF degradation with BiOI0.5Cl0.5. RSC Adv. 2019, 9, 14060–14071. [Google Scholar] [CrossRef] [Green Version]
- Sun, G.T.; Shi, J.W.; Mao, S.M.; Ma, D.D.; He, C.; Wang, H.K.; Cheng, Y.H. Dodecylamine coordinated tri-arm CdS nanorod wrapped in intermittent ZnS shell for greatly improved photocatalytic H2 evolution. Chem. Eng. J. 2022, 429, 132382. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, F.; Xiao, X.; Zuo, X.X.; Nan, J.M. Microwave synthesis of iodine-doped bismuth oxychloride microspheres for the visible light photocatalytic removal of toxic hydroxyl-contained intermediates of parabens: Catalyst synthesis, characterization, and mechanism insight. Environ. Sci. Pollut. Res. Int. 2019, 26, 28871–28883. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.W.; Huang, H.W.; Yu, S.X.; Dong, F.; Zhang, Y.H. A self-sacrifice template route to iodine modified BiOIO3: Band gap engineering and highly boosted visible-light active photoreactivity. Phys. Chem. Chem. Phys. 2016, 18, 7851–7859. [Google Scholar] [CrossRef]
- Zhang, K.; Zhang, D.Q.; Liu, J.; Ren, K.X.; Luo, H.; Peng, Y.J.; Li, G.S.; Yu, X.B. A novel nanoreactor framework of iodine-incorporated BiOCl core–shell structure: Enhanced light-harvesting system for photocatalysis. CrystEngComm 2012, 14, 700–707. [Google Scholar] [CrossRef]
- Singh, S.; Sahoo, R.K.; Shinde, N.M.; Yun, J.M.; Mane, R.S.; Chung, W.; Kim, K.H. Asymmetric faradaic assembly of Bi2O3 and MnO2 for a high-performance hybrid electrochemical energy storage device. RSC Adv. 2019, 9, 32154–32164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, H.P.; Liu, J.Y.; Hou, W.G.; Du, N.; Zhang, R.J.; Tao, X.T. Synthesis and characterization of g-C3N4/Bi2MoO6 heterojunctions with enhanced visible light photocatalytic activity. Appl. Catal. B Environ. 2014, 160–161, 89–97. [Google Scholar] [CrossRef]
- Xu, Y.S.; Zhang, W.D. Monodispersed Ag3PO4 nanocrystals loaded on the surface of spherical Bi2MoO6 with enhanced photocatalytic performance. Dalton Trans. 2013, 42, 1094–1101. [Google Scholar] [CrossRef]
- Hou, J.H.; Jiang, K.; Shen, M.; Wei, R.; Wu, X.G.; Idrees, F.; Cao, C.B. Micro and nano hierachical structures of BiOI/activated carbon for efficient visible-light-photocatalytic reactions. Sci. Rep. 2017, 7, 11665. [Google Scholar] [CrossRef] [Green Version]
- Tang, Y.F.; Lin, T.P.; Jiang, C.J.; Zhao, Y.M.; Ai, S.J. Renewable adsorbents from carboxylate-modified agro-forestry residues for efficient removal of methylene blue dye. J. Phys. Chem. Solids 2021, 149, 149–151. [Google Scholar] [CrossRef]
- Wang, Y.T.; He, L.Y.; Dang, G.Y.; Li, H.; Li, X.L. Polypyrrole-functionalized magnetic Bi2MoO6 nanocomposites as a fast, efficient and reusable adsorbent for removal of ketoprofen and indomethacin from aqueous solution. J. Colloid Interface Sci. 2021, 592, 51–65. [Google Scholar] [CrossRef]
- Guo, Y.P.; Zhao, J.Z.; Zhang, H.; Yang, S.F.; Qi, J.R.; Wang, Z.C.; Xu, H.D. Use of rice husk-based porous carbon for adsorption of Rhodamine B from aqueous solutions. Dyes Pigm. 2005, 66, 123–128. [Google Scholar] [CrossRef]
- Oyetade, O.A.; Nyamori, V.O.; Martincigh, B.S.; Jonnalagadda, S.B. Effectiveness of carbon nanotube–cobalt ferrite nanocomposites for the adsorption of rhodamine B from aqueous solutions. RSC Adv. 2015, 5, 22724–22739. [Google Scholar] [CrossRef]
- Jiang, L.; Wen, Y.Y.; Zhu, Z.J.; Liu, X.F.; Shao, W. A Double cross-linked strategy to construct graphene aerogels with highly efficient methylene blue adsorption performance. Chemosphere 2021, 265, 129169. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.X.; Chen, Y.; Xue, M.; Ding, R.; Kang, Y.; Tremblay, P.L.; Zhang, T. Fast-growing cyanobacteria bio-embedded into bacterial cellulose for toxic metal bioremediation. Carbohydr. Polym. 2022, 295, 119881. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Wang, Y.H.; Cui, B.W.; Zhang, X.B.; Zhang, D.G.; Xu, X.Y. Preparation of MoS2 nanoflowers with rich active sites as an efficient adsorbent for aqueous organic dyes. N. J. Chem. 2020, 44, 4558–4567. [Google Scholar] [CrossRef]
- Tang, S.; Xia, D.S.; Yao, Y.; Chen, T.Y.; Sun, J.; Yin, Y.J.; Shen, W.; Peng, Y.X. Dye adsorption by self-recoverable, adjustable amphiphilic graphene aerogel. J. Colloid Interface Sci. 2019, 554, 682–691. [Google Scholar] [CrossRef]
- Godiya, C.B.; Kumar, S.; Xiao, Y.H. Amine functionalized egg albumin hydrogel with enhanced adsorption potential for diclofenac sodium in water. J. Hazard. Mater. 2020, 393, 122417. [Google Scholar] [CrossRef] [PubMed]
- Zamani, S.; Tabrizi, N.S. Removal of methylene blue from water by graphene oxide aerogel: Thermodynamic, kinetic, and equilibrium modeling. Res. Chem. Intermed. 2014, 41, 7945–7963. [Google Scholar] [CrossRef]
- Kinniburgh, D.G. General purpose adsorption isotherms. Environ. Sci. Technol. 1986, 20, 895–904. [Google Scholar] [CrossRef]
- Wu, Z.B.; Zhong, H.; Yuan, X.Z.; Wang, H.; Wang, L.L.; Chen, X.H.; Zeng, G.M.; Wu, Y. Adsorptive removal of methylene blue by rhamnolipid-functionalized graphene oxide from wastewater. Water Res. 2014, 67, 330–344. [Google Scholar] [CrossRef]
- Ding, L.L.; Zou, B.; Gao, W.; Liu, Q.; Wang, Z.C.; Guo, Y.P.; Wang, X.F.; Liu, Y.H. Adsorption of Rhodamine-B from aqueous solution using treated rice husk-based activated carbon. Colloid Surf. A 2014, 446, 1–7. [Google Scholar] [CrossRef]
- Lian, L.L.; Guo, L.P.; Guo, C.J. Adsorption of Congo red from aqueous solutions onto Ca-bentonite. J. Hazard. Mater. 2009, 161, 126–131. [Google Scholar] [CrossRef]
- Zhao, G.X.; Li, J.X.; Ren, X.M.; Chen, C.L.; Wang, X.K. Few-layered graphene oxide nanosheets as superior sorbents for heavy metal ion pollution management. Environ. Sci. Technol. 2011, 45, 10454–10462. [Google Scholar] [CrossRef] [PubMed]
- Costa, L.N.; Nobre, F.X.; Lobo, A.O.; De Matos, J.M.E. Photodegradation of ciprofloxacin using Z-scheme TiO2/SnO2 nanostructures as photocatalyst. Environ. Nanotechnol. Monit. Manag. 2021, 16, 100466. [Google Scholar]
- Zhao, M.; Liu, P. Adsorption of methylene blue from aqueous solutions by modified expanded graphite powder. Desalination 2009, 249, 331–336. [Google Scholar] [CrossRef]
- Kaewmanee, T.; Wannapop, S.; Phuruangrat, A.; Thongtem, T.; Thongtem, S. Solvothermal synthesis of BiOBrxI1−x (x = 0.0–1.0) solid solutions used for adsorption and photodegradation of cationic and anionic dyes. Inorg. Chem. Commun. 2021, 134, 109054. [Google Scholar] [CrossRef]






| Absorbent | Time (min) | Removal Efficiency (%) | Adsorbent Dosage (mg) | [RhB] (mg/L) | Reaction Volume (mL) | Reference |
|---|---|---|---|---|---|---|
| BiOBr/Bi2MoO6 | 120 | 98.4 a | 20 | 30 | 20 | [22] |
| Bi2MoO6@BiOCl@MOF-199 | 10 | 30.0 b | 100 | 7.5 | 200 | [24] |
| MnFe2O4/Bi2MoO6/PPy | 120 | 54.9 b | 30 | 10 | 50 | [59] |
| Bi2MoO6@BiOCl | 8 | 94.0 b | 100 | 7 | 200 | [27] |
| BiOI/Bi2MoO6 (BOI2/BMO) | 5 | 98.9 b | 50 | 10 | 50 | This work |
| Temperature (K) | ΔG0 (kJ/mol) | ΔS0 (J/mol/K) | ΔH0 (kJ/mol) |
|---|---|---|---|
| 298 | −30.78 | 64.60 | −11.53 a |
| 308 | −32.62 | 68.47 | |
| 318 | −33.64 | 69.53 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Onyedika, T.E.; Xu, M.; Deng, Y.; Liu, Y.; Li, L.; Tremblay, P.-L.; Zhang, T. The Quick Removal of Toxic Dye Molecules by an Efficient Adsorptive BiOI/Bi2MoO6 Heterostructure. Catalysts 2023, 13, 457. https://doi.org/10.3390/catal13030457
Onyedika TE, Xu M, Deng Y, Liu Y, Li L, Tremblay P-L, Zhang T. The Quick Removal of Toxic Dye Molecules by an Efficient Adsorptive BiOI/Bi2MoO6 Heterostructure. Catalysts. 2023; 13(3):457. https://doi.org/10.3390/catal13030457
Chicago/Turabian StyleOnyedika, Tasie Ebenezer, Mengying Xu, Yichao Deng, Yang Liu, Lian Li, Pier-Luc Tremblay, and Tian Zhang. 2023. "The Quick Removal of Toxic Dye Molecules by an Efficient Adsorptive BiOI/Bi2MoO6 Heterostructure" Catalysts 13, no. 3: 457. https://doi.org/10.3390/catal13030457