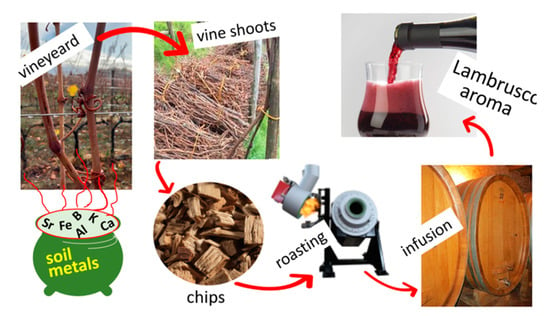
Exploring the Mineral Composition of Grapevine Canes for Wood Chip Applications in Alcoholic Beverage Production to Enhance Viticulture Sustainability
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Standards
2.2. Samples Preparation
2.3. Proximate Composition
2.4. Macerative Solvent Extraction
2.5. ICP-OES Analysis
2.6. Statistical Analysis
3. Results and Discussion
3.1. Proximate Composition of Grapevine Canes
3.2. Effects of Roasting on Sample Mass Loss
3.3. EtOH Extraction Yield
3.4. ICP-OES Analysis of Debarked Grapevine Cane Samples
- i.
- These metals are generally involved in protein–enzyme systems that ensure the physiological viability of the initial plant structures. The elevated roasting temperature is likely responsible for disrupting the original metal complex–chelate system present in the woody matrix, leading to the release of poorly soluble metal oxides and a subsequent reduction in their concentrations, as detected by the ICP-OES analysis of the extracts.
- ii.
- It is important to highlight the significance of B in this context, as its concentration reduction with increasing roasting temperature may be attributed to the covalent molecular characteristics of metal oxides and other compounds, which render them highly volatile. Consequently, boron can be lost through thermal stress during roasting.
- iii.
- It is worth noting that the release of metal oxides during roasting can be influenced not only by temperature but also by the presence of other compounds and the chemical environment. For instance, the interaction between metals and polyphenolic compounds, such as tannins, can affect the stability and solubility of the metal species. These interactions can be complex and depend on factors, such as pH, oxidation-reduction potential, and the specific polyphenolic profile of grapevine cane extracts. At elevated temperatures, tannins undergo various reactions, including disruption of their molecular structures and the formation of degradation compounds. These changes can affect the ability of tannins to bind metal ions and form stable complexes. The breakdown of tannin–metal complexes may facilitate the release of metal ions and subsequent formation of less soluble metal oxides or hydroxides.
- iv.
- Additionally, the two metal elements Ni and Bi exhibited opposing trends in the two cultivars, as depicted in Figure 2.
- v.
- The main mineral elements detected in the roasted grapevine canes were K, Mg, and Ca, which exhibited an increasing concentration with roasting temperature. These minerals are essential for maintaining the optimal health and functioning of the human body. Potassium is involved in various physiological processes, including fluid balance, nerve function, and muscle contraction [56,57]. Its ability to regulate blood pressure by counteracting the effects of sodium promotes cardiovascular health. In addition, potassium contributes to bone health and may reduce the risk of kidney stones. Magnesium plays a vital role in more than 300 enzymatic reactions in the body, making it vital for numerous physiological functions [58,59,60]. It is involved in energy production, nerve function, muscle relaxation, and the synthesis of DNA and proteins. Magnesium also supports a healthy heart rhythm, promotes bone health, and helps regulate blood sugar levels. Calcium is known for its role in building and maintaining strong bones and teeth [61]. They are also involved in muscle function, nerve transmission, and blood clotting [62]. Calcium plays a crucial role in maintaining a normal heart rhythm and blood pressure. Additionally, it has been linked to a reduced risk of colorectal cancer and may help in weight management. Therefore, the high content of these minerals in ethanolic extracts allows for the enrichment of beverages and enological products aged with infused chips from a nutritional perspective. By incorporating these minerals into the aging process, the resulting beverages can offer health benefits beyond their flavor and aroma profiles. It meets the increasing demand of consumers to incorporate high nutrient levels with an adequate amount of essential minerals into their normal diet, preferably from plant sources [53].
3.5. ICP-OES Analysis of Grapevine Wood Samples
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Khajuria, A.; Atienza, V.A.; Chavanich, S.; Henning, W.; Islam, I.; Kral, U.; Liu, M.; Liu, X.; Murthy, I.K.; Oyedotun, T.D.T.; et al. Accelerating Circular Economy Solutions to Achieve the 2030 Agenda for Sustainable Development Goals. Circ. Econ. 2022, 1, 100001. [Google Scholar] [CrossRef]
- Rodriguez-Anton, J.M.; Rubio-Andrada, L.; Celemín-Pedroche, M.S.; Alonso-Almeida, M.D.M. Analysis of the Relations between Circular Economy and Sustainable Development Goals. Int. J. Sustain. Dev. World Ecol. 2019, 26, 708–720. [Google Scholar] [CrossRef]
- Belmonte-Ureña, L.J.; Plaza-Úbeda, J.A.; Vazquez-Brust, D.; Yakovleva, N. Circular Economy, Degrowth and Green Growth as Pathways for Research on Sustainable Development Goals: A Global Analysis and Future Agenda. Ecol. Econ. 2021, 185, 107050. [Google Scholar] [CrossRef]
- UN General Assembly. Transforming Our World: The 2030 Agenda for Sustainable Development; A/RES/70/1; UN: Geneva, Switzerland, 2015. [Google Scholar]
- Geisendorf, S.; Pietrulla, F. The Circular Economy and Circular Economic Concepts—A Literature Analysis and Redefinition. Thunderbird Int. Bus. Rev. 2018, 60, 771–782. [Google Scholar] [CrossRef]
- Hamam, M.; Chinnici, G.; Di Vita, G.; Pappalardo, G.; Pecorino, B.; Maesano, G.; D’Amico, M. Circular Economy Models in Agro-Food Systems: A Review. Sustainability 2021, 13, 3453. [Google Scholar] [CrossRef]
- Kumar, B.; Verma, P. Biomass-Based Biorefineries: An Important Architype towards a Circular Economy. Fuel 2021, 288, 119622. [Google Scholar] [CrossRef]
- Chiaraluce, G. Circular Economy in the Agri-Food Sector: A Policy Overview. Ital. Rev. Agric. Econ. 2021, 76, 53–60. [Google Scholar]
- Širá, E.; Kravčáková Vozárová, I.; Kotulič, R.; Dubravská, M. EU27 Countries’ Sustainable Agricultural Development toward the 2030 Agenda: The Circular Economy and Waste Management. Agronomy 2022, 12, 2270. [Google Scholar] [CrossRef]
- Maletti, L.; D’Eusanio, V.; Lancellotti, L.; Marchetti, A.; Pincelli, L.; Strani, L.; Tassi, L. Candying Process for Enhancing Pre-Waste Watermelon Rinds to Increase Food Sustainability. Future Foods 2022, 6, 100182. [Google Scholar] [CrossRef]
- D’Eusanio, V.; Maletti, L.; Marchetti, A.; Roncaglia, F.; Tassi, L. Volatile Aroma Compounds of Gavina® Watermelon (Citrullus lanatus L.) Dietary Fibers to Increase Food Sustainability. AppliedChem 2023, 3, 6. [Google Scholar] [CrossRef]
- Maletti, L.; D’Eusanio, V.; Durante, C.; Marchetti, A.; Tassi, L. VOCs Analysis of Three Different Cultivars of Watermelon (Citrullus lanatus L.) Whole Dietary Fiber. Molecules 2022, 27, 8747. [Google Scholar] [CrossRef]
- D’Eusanio, V.; Bertacchini, L.; Marchetti, A.; Mariani, M.; Pastorelli, S.; Silvestri, M.; Tassi, L. Rosaceae Nut-Shells as Sustainable Aggregate for Potential Use in Non-Structural Lightweight Concrete. Waste 2023, 1, 33. [Google Scholar] [CrossRef]
- Bakan, B.; Bernet, N.; Bouchez, T.; Boutrou, R.; Choubert, J.-M.; Dabert, P.; Duquennoi, C.; Ferraro, V.; García-Bernet, D.; Gillot, S.; et al. Circular Economy Applied to Organic Residues and Wastewater: Research Challenges. Waste Biomass Valorizat. 2022, 13, 1267–1276. [Google Scholar] [CrossRef]
- Palmieri, N.; Suardi, A.; Alfano, V.; Pari, L. Circular Economy Model: Insights from a Case Study in South Italy. Sustainability 2020, 12, 3466. [Google Scholar] [CrossRef] [Green Version]
- Sehnem, S.; Vazquez-Brust, D.; Pereira, S.C.F.; Campos, L.M.S. Circular Economy: Benefits, Impacts and Overlapping. Supply Chain Manag. Int. J. 2019, 24, 784–804. [Google Scholar] [CrossRef]
- Korhonen, J.; Honkasalo, A.; Seppälä, J. Circular Economy: The Concept and Its Limitations. Ecol. Econ. 2018, 143, 37–46. [Google Scholar] [CrossRef]
- Devesa-Rey, R.; Vecino, X.; Varela-Alende, J.L.; Barral, M.T.; Cruz, J.M.; Moldes, A.B. Valorization of Winery Waste vs. the Costs of Not Recycling. Waste Manag. 2011, 31, 2327–2335. [Google Scholar] [CrossRef]
- García-Galindo, D.; López, E.; Gómez, M.; Sebastián, F.; Gebresenbet, G.; Jirjis, R.; Kern, J.; Germer, S.; Pari, L.; Suardi, A.; et al. Europruning Project: Summary of Final Results. Available online: http://Www.Etaflorence.It/Proceedings/?Detail=13021 (accessed on 20 March 2023).
- Morone, P.; Koutinas, A.; Gathergood, N.; Arshadi, M.; Matharu, A. Food Waste: Challenges and Opportunities for Enhancing the Emerging Bio-Economy. J. Clean. Prod. 2019, 221, 10–16. [Google Scholar] [CrossRef]
- D’Eusanio, V.; Malferrari, D.; Marchetti, A.; Roncaglia, F.; Tassi, L. Waste By-Product of Grape Seed Oil Production: Chemical Characterization for Use as a Food and Feed Supplement. Life 2023, 13, 326. [Google Scholar] [CrossRef]
- Bordiga, M.; Travaglia, F.; Locatelli, M. Valorisation of Grape Pomace: An Approach That Is Increasingly Reaching Its Maturity—A Review. Int. J. Food Sci. Technol. 2019, 54, 933–942. [Google Scholar] [CrossRef]
- FAO Faostat: FAO Statistical Databases. Available online: https://www.fao.org/faostat/en/#data (accessed on 20 February 2023).
- Miglietta, P.P.; Morrone, D.; Lamastra, L. Water Footprint and Economic Water Productivity of Italian Wines with Appellation of Origin: Managing Sustainability through an Integrated Approach. Sci. Total Environ. 2018, 633, 1280–1286. [Google Scholar] [CrossRef] [PubMed]
- Christ, K.L.; Burritt, R.L. Critical Environmental Concerns in Wine Production: An Integrative Review. J. Clean. Prod. 2013, 53, 232–242. [Google Scholar] [CrossRef]
- Pari, L.; Alfano, V.; Garcia-Galindo, D.; Suardi, A.; Santangelo, E. Pruning Biomass Potential in Italy Related to Crop Characteristics, Agricultural Practices and Agro-Climatic Conditions. Energies 2018, 11, 1365. [Google Scholar] [CrossRef] [Green Version]
- International Organisation of Vine and Wine (OIV) World Wine Production Outlook. 2022. Available online: https://www.oiv.int/ (accessed on 30 January 2023).
- OIV. State of the World Vine and Wine Sector 2021. Available online: https://Www.Oiv.Int/Sites/Default/Files/Documents/Eng-State-of-the-World-Vine-and-Wine-Sector-April-2022-V6_0.Pdf (accessed on 10 April 2023).
- Segré, A.; White, G.B. Marketing Italian Wine in the U.S. Market a Case Study of Cantine Riunite. In Developments in Agricultural Economics; Vine and Wine Economy; Botos, E.P., Ed.; Elsevier: Amsterdam, The Netherlands, 1991; Volume 8, pp. 245–259. [Google Scholar]
- Mura, R.; Chiriacò, M.; Vicentini, F. Economic and Environmental Outcomes of a Sustainable and Circular Approach: Case Study of an Italian Wine-Producing Firm. J. Bus. Res. 2022, 154, 113300. [Google Scholar] [CrossRef]
- Callejón, R.M.; Torija, M.J.; Mas, A.; Morales, M.L.; Troncoso, A.M. Changes of Volatile Compounds in Wine Vinegars during Their Elaboration in Barrels Made from Different Woods. Food Chem. 2010, 120, 561–571. [Google Scholar] [CrossRef]
- Herrero, P.; Sáenz-Navajas, M.P.; Avizcuri, J.M.; Culleré, L.; Balda, P.; Antón, E.C.; Ferreira, V.; Escudero, A. Study of Chardonnay and Sauvignon Blanc Wines from D.O.Ca Rioja (Spain) Aged in Different French Oak Wood Barrels: Chemical and Aroma Quality Aspects. Food Res. Int. 2016, 89, 227–236. [Google Scholar] [CrossRef]
- Lancellotti, L.; Ulrici, A.; Sighinolfi, S.; Marchetti, A. Chemical Characterization Of Commercial Balsamic Vinegar Glaze. J. Food Compos. Anal. 2020, 94, 103620. [Google Scholar] [CrossRef]
- Dumitriu, G.-D.; De Lerma, N.L.; Cotea, V.V.; Zamfir, C.-I.; Peinado, R.A. Effect of Aging Time, Dosage and Toasting Level of Oak Chips on the Color Parameters, Phenolic Compounds and Antioxidant Activity of Red Wines (Var. Fetească Neagră). Eur. Food Res. Technol. 2016, 242, 2171–2180. [Google Scholar] [CrossRef]
- Kayaoğlu, M.; Bayram, M.; Topuz, S. Effect of Oak Chips Addition on the Phenolic Composition of Grape Vinegar in Fermentation Process. J. Food Meas. Charact. 2022, 16, 3106–3116. [Google Scholar] [CrossRef]
- Vichi, S.; Santini, C.; Natali, N.; Riponi, C.; López-Tamames, E.; Buxaderas, S. Volatile and Semi-Volatile Components of Oak Wood Chips Analysed by Accelerated Solvent Extraction (ASE) Coupled to Gas Chromatography–Mass Spectrometry (GC–MS). Food Chem. 2007, 102, 1260–1269. [Google Scholar] [CrossRef]
- O’Sullivan, M. Sensory Properties of Beverage Products (Alcoholic and Nonalcoholic). In Handbook for Sensory and Consumer-Driven New Poduct Development Innovative Technologies for the Food and Beverage Industry; Woodhead Publishing: Sawston, UK, 2017; Volume 13, pp. 281–304. [Google Scholar]
- D’Eusanio, V.; Genua, F.; Marchetti, A.; Morelli, L.; Tassi, L. Characterization of Some Stilbenoids Extracted from Two Cultivars of Lambrusco—Vitis Vinifera Species: An Opportunity to Valorize Pruning Canes for a More Sustainable Viticulture. Molecules 2023, 28, 4074. [Google Scholar] [CrossRef]
- Gorena, T.; Saez, V.; Mardones, C.; Vergara, C.; Winterhalter, P.; von Baer, D. Influence of Post-Pruning Storage on Stilbenoid Levels in Vitis Vinifera L. Canes. Food Chem. 2014, 155, 256–263. [Google Scholar] [CrossRef]
- Lambert, C.; Richard, T.; Renouf, E.; Bisson, J.; Waffo-Téguo, P.; Bordenave, L.; Ollat, N.; Mérillon, J.-M.; Cluzet, S. Comparative Analyses of Stilbenoids in Canes of Major Vitis Vinifera L. Cultivars. J. Agric. Food Chem. 2013, 61, 11392–11399. [Google Scholar] [CrossRef]
- Sah, R.N.; Miller, R.O. Spontaneous Reaction for Acid Dissolution of Biological Tissues in Closed Vessels. Anal. Chem. 1992, 64, 230–233. [Google Scholar] [CrossRef]
- Durante, C.; Cocchi, M.; Lancellotti, L.; Maletti, L.; Marchetti, A.; Roncaglia, F.; Sighinolfi, S.; Tassi, L. Analytical Concentrations of Some Elements in Seeds and Crude Extracts from Aesculus Hippocastanum, by ICP-OES Technique. Agronomy 2021, 11, 47. [Google Scholar] [CrossRef]
- Official Journal of the European Union Regulation (EC) No 1107/2009 of the European Parliament and of the Council of 21 October 2009 Concerning the Placing of Plant Protection Products on the Market and Repealing Council Directives 79/117/EEC and 91/414/EEC 2009. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2009:309:0001:0050:en:PDF (accessed on 13 July 2023).
- Dorosh, O.; Fernandes, V.C.; Moreira, M.M.; Delerue-Matos, C. Occurrence of Pesticides and Environmental Contaminants in Vineyards: Case Study of Portuguese Grapevine Canes. Sci. Total Environ. 2021, 791, 148395. [Google Scholar] [CrossRef]
- Rayne, S.; Karacabey, E.; Mazza, G. Grape Cane Waste as a Source of Trans-Resveratrol and Trans-Viniferin: High-Value Phytochemicals with Medicinal and Anti-Phytopathogenic Applications. Ind. Crops Prod. 2008, 27, 335–340. [Google Scholar] [CrossRef]
- Mendívil, M.A.; Muñoz, P.; Morales, M.P.; Juárez, M.C.; García-Escudero, E. Chemical Characterization of Pruned Vine Shoots from La Rioja (Spain) for Obtaining Solid Bio-Fuels. J. Renew. Sustain. Energy 2013, 5, 033113. [Google Scholar] [CrossRef]
- Courregelongue, M.; Shinkaruk, S.; Prida, A.; Darriet, P.; Pons, A. Identification and Distribution of New Impact Aldehydes in Toasted Oak Wood (Quercus Petraea). J. Agric. Food Chem. 2022, 70, 11667–11677. [Google Scholar] [CrossRef]
- Corzo-Martínez, M.; Corzo, N.; Villamiel, M.; del Castillo, M.D. Browning Reactions. In Food Biochemistry and Food Processing; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2012; pp. 56–83. ISBN 978-1-118-30803-5. [Google Scholar]
- Pereira, V.; Albuquerque, F.M.; Ferreira, A.C.; Cacho, J.; Marques, J.C. Evolution of 5-Hydroxymethylfurfural (HMF) and Furfural (F) in Fortified Wines Submitted to Overheating Conditions. Food Res. Int. 2011, 44, 71–76. [Google Scholar] [CrossRef]
- U.S. FDA (Food and Drug Administration) Exploratory Data on Furan in Food: Individual Food Products. 2008. Available online: https://www.fda.gov/food/process-contaminants-food/exploratory-data-furan-food (accessed on 21 March 2023).
- Knutsen, H.K.; Alexander, J.; Barregård, L.; Bignami, M.; Brüschweiler, B.; Ceccatelli, S.; Cottrill, B.; Dinovi, M.; Edler, L.; et al.; EFSA Panel on Contaminants in the Food Chain (CONTAM) Risks for Public Health Related to the Presence of Furan and Methylfurans in Food. EFSA J. 2017, 15, e05005. [Google Scholar] [CrossRef] [PubMed]
- EU. Council Regulation (EC) No 479/2008 of 29 April 2008 on the Common Organisation of the Market in Wine, Amending Regulations (EC) No 1493/1999 and Following. Off. J. Eur. Union 2008, 148, 1–61. [Google Scholar]
- Çetin, E.S.; Altinöz, D.; Tarçan, E.; Göktürk Baydar, N. Chemical Composition of Grape Canes. Ind. Crops Prod. 2011, 34, 994–998. [Google Scholar] [CrossRef]
- Delgado-Torre, M.P.; Ferreiro-Vera, C.; Priego-Capote, F.; Pérez-Juan, P.M.; Luque de Castro, M.D. Comparison of Accelerated Methods for the Extraction of Phenolic Compounds from Different Vine-Shoot Cultivars. J. Agric. Food Chem. 2012, 60, 3051–3060. [Google Scholar] [CrossRef] [PubMed]
- Romain, C.; Gaillet, S.; Carillon, J.; Vidé, J.; Ramos, J.; Izard, J.-C.; Cristol, J.-P.; Rouanet, J.-M. Vineatrol and Cardiovascular Disease: Beneficial Effects of a Vine-Shoot Phenolic Extract in a Hamster Atherosclerosis Model. J. Agric. Food Chem. 2012, 60, 11029–11036. [Google Scholar] [CrossRef]
- Aburto, N.J.; Hanson, S.; Gutierrez, H.; Hooper, L.; Elliott, P.; Cappuccio, F.P. Effect of Increased Potassium Intake on Cardiovascular Risk Factors and Disease: Systematic Review and Meta-Analyses. BMJ 2013, 346, f1378. [Google Scholar] [CrossRef] [Green Version]
- WHO. WHO Guideline: Potassium Intake for Adults and Children; WHO: Geneva, Switzerland, 2012. [Google Scholar]
- Dong, J.-Y.; Xun, P.; He, K.; Qin, L.-Q. Magnesium Intake and Risk of Type 2 Diabetes: Meta-Analysis of Prospective Cohort Studies. Diabetes Care 2011, 34, 2116–2122. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.J.; Xun, P.; Liu, K.; Loria, C.; Yokota, K.; Jacobs, D.R., Jr.; He, K. Magnesium Intake in Relation to Systemic Inflammation, Insulin Resistance, and the Incidence of Diabetes. Diabetes Care 2010, 33, 2604–2610. [Google Scholar] [CrossRef] [Green Version]
- Fan, X.; Rudel, R.; Berger, M.; Hershman, M.; Seres, D.S. General Nutritional Principles. In Yamada’s Textbook of Gastroenterology; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2022; pp. 376–407. ISBN 978-1-119-60020-6. [Google Scholar]
- Bolland, M.J.; Leung, W.; Tai, V.; Bastin, S.; Gamble, G.D.; Grey, A.; Reid, I.R. Calcium Intake and Risk of Fracture: Systematic Review. BMJ 2015, 351, h4580. [Google Scholar] [CrossRef] [Green Version]
- Cormick, G.; Belizán, J.M. Calcium Intake and Health. Nutrients 2019, 11, 1606. [Google Scholar] [CrossRef] [Green Version]
- Sánchez-Gómez, R.; Zalacain, A.; Alonso, G.L.; Salinas, M.R. Vine-Shoot Waste Aqueous Extracts for Re-Use in Agriculture Obtained by Different Extraction Techniques: Phenolic, Volatile, and Mineral Compounds. J. Agric. Food Chem. 2014, 62, 10861–10872. [Google Scholar] [CrossRef]
- Du Toit, W.J. Micro-Oxygenation, Oak Alternatives and Added Tannins and Wine Quality. In Managing Wine Quality; Woodhead Publishing Series in Food Science Technology and Nutrition; Reynolds, A.G., Ed.; Woodhead Publishing: Sawston, UK, 2010; pp. 226–254. ISBN 978-1-84569-798-3. [Google Scholar]
- Durguti, V.Y.; Aliu, S.; Laha, F.; Feka, F. Determination of Iron, Copper and Zinc in the Wine by FAAS. Emerg. Sci. J. 2020, 4, 411–417. [Google Scholar] [CrossRef]
- Malavolti, M.; Fairweather-Tait, S.J.; Malagoli, C.; Vescovi, L.; Vinceti, M.; Filippini, T. Lead Exposure in an Italian Population: Food Content, Dietary Intake and Risk Assessment. Food Res. Int. 2020, 137, 109370. [Google Scholar] [CrossRef]
- Satarug, S.; Gobe, C.; Vesey, A.; Phelps, K.R. Cadmium and Lead Exposure, Nephrotoxicity, and Mortality. Toxics 2020, 8, 86. [Google Scholar] [CrossRef]
- Satarug, S. Dietary Cadmium Intake and Its Effects on Kidneys. Toxics 2018, 6, 15. [Google Scholar] [CrossRef] [Green Version]
- Kim, K.; Melough, M.M.; Vance, T.M.; Noh, H.; Koo, S.I.; Chun, O.K. Dietary Cadmium Intake and Sources in the US. Nutrients 2019, 11, 2. [Google Scholar] [CrossRef] [Green Version]
- Volpe, M.G.; La Cara, F.; Volpe, F.; De Mattia, A.; Serino, V.; Petitto, F.; Zavalloni, C.; Limone, F.; Pellecchia, R.; De Prisco, P.P.; et al. Heavy Metal Uptake in the Enological Food Chain. Food Chem. 2009, 117, 553–560. [Google Scholar] [CrossRef]
- Krstić, B.; Stanković, D.; Igić, R.; Nikolic, N. The Potential of Different Plant Species for Nickel Accumulation. Biotechnol. Biotechnol. Equip. 2007, 21, 431–436. [Google Scholar] [CrossRef] [Green Version]
- Prabagar, S.; Dharmadasa, R.M.; Lintha, A.; Thuraisingam, S.; Prabagar, J. Accumulation of Heavy Metals in Grape Fruit, Leaves, Soil and Water: A Study of Influential Factors and Evaluating Ecological Risks in Jaffna, Sri Lanka. Environ. Sustain. Indic. 2021, 12, 100147. [Google Scholar] [CrossRef]
- Cempel, M.; Nikel, G. Nickel: A Review of Its Sources and Environmental Toxicology. Pol. J. Environ. Stud. 2006, 15, 375–382. [Google Scholar]
- Waterhouse, A.L.; Laurie, V.F. Oxidation of Wine Phenolics: A Critical Evaluation and Hypotheses. Am. J. Enol. Vitic. 2006, 57, 306–313. [Google Scholar] [CrossRef]
- Cebrián-Tarancón, C.; Fernández-Roldán, F.; Alonso, G.L.; Salinas, R.M. Classification of Vine-shoots for Use as Enological Additives. J. Sci. Food Agric. 2022, 102, 724–731. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Gómez, R.; Zalacain, A.; Alonso, G.L.; Salinas, M.R. Effect of Toasting on Non-Volatile and Volatile Vine-Shoots Low Molecular Weight Phenolic Compounds. Food Chem. 2016, 204, 499–505. [Google Scholar] [CrossRef] [PubMed]


| Ancellotta | Salamino | |
|---|---|---|
| Grapevine plants sampled after grape harvest | 20 | 20 |
| Total mass of collected grapevine wood | ~6 kg | ~6 kg |
| Roasting of the debarked grapevine cane samples at 8 temperatures ranging from 120 to 260 °C | 4 replicates for each roasting temperature | 4 replicates for each roasting temperature |
| Macerative solvent extraction of tannic fraction in EtOH at 80 °C, for roasted grapevine chips at each temperature in the range of 120–260 °C | 4 replicates | 4 replicates |
| Mineralization of dry tannic extracts for each sample of roasted grapevine chips (wet method) | 4 replicates | 4 replicates |
| Total metal content of grapevine canes (dry method) | 4 replicates | 4 replicates |
| Ancellotta | Salamino | Ref (Mendívil et al. [46]) | |
|---|---|---|---|
| Moisture% (at 105 °C) | 16.2 ± 0.2 | 22.9 ± 0.3 | 48.3–55.5 (51.5) |
| Forced drying% (at 120 °C) | 26.7 ± 0.2 | 38.4 ± 0.2 | - |
| C% * | 46.1 ± 0.3 | 45.6 ± 0.4 | 45.7–47.5 (46.3) |
| H% * | 6.94 ± 0.08 | 6.94 ± 0.09 | 5.18–6.27 (6.0) |
| N% * | 0.58 ± 0.03 | 0.46 ± 0.04 | 0.68–0.82 (0.8) |
| S% * | <0.1 | <0.1 | 0.04–0.06 (0.05) |
| O% *,# | 43.1 ± 0.4 | 45.2 ± 0.5 | - |
| Ash% | 3.21 ± 0.05 | 3.07 ± 0.06 | 2.36–3.82 (3.0) |
| Ancellotta | Salamino | Sample Appearance | |
|---|---|---|---|
| t (°C) | −Δm% | −Δm% | |
| 105 | 16.21 ± 0.24 | 22.90 ± 0.27 | No significant color change, typical odor of dried wood |
| 120 | 26.67 ± 0.23 | 38.38 ± 0.21 | Slight color change, pleasant scent, not specific |
| 140 | 34.11 ± 0.21 | 44.29 ± 0.24 | Slight color change, very light roasting aroma |
| 160 | 36.51 ± 0.38 | 45.09 ± 0.35 | Mild color change, light roasting aroma |
| 180 | 37.06 ± 0.29 | 46.77 ± 0.33 | Moderate color change, medium roasting aroma |
| 200 | 37.95 ± 0.30 | 48.17 ± 0.36 | Significant color change, medium-strong roasting aroma |
| 220 | 40.77 ± 0.24 | 51.18 ± 0.39 | Significant color change, strong roasting aroma |
| 240 | 47.40 ± 0.32 | 58.27 ± 0.37 | Very significant color change, dark roasting aroma |
| 260 | 55.58 ± 0.38 | 66.68 ± 0.41 | Dark black color, typical odor of semi-carbonized wood |
| Ancellotta | Salamino | |
|---|---|---|
| t (°C) | Extraction Yield g/100 g * | Extraction Yield g/100 g * |
| 120 | 2.02 ± 0.07 a | 2.09 ± 0.11 a |
| 140 | 1.89 ± 0.08 a | 2.06 ± 0.10 a |
| 160 | 1.85 ± 0.09 a | 2.08 ± 0.14 a |
| 180 | 1.91 ± 0.10 a | 2.13 ± 0.15 a |
| 200 | 1.99 ± 0.09 a | 2.15 ± 0.13 a |
| 220 | 1.97 ± 0.09 a | 2.15 ± 0.11 a |
| 240 | 1.48 ± 0.08 b | 1.66 ± 0.12 b |
| 260 | 1.12 ± 0.10 c | 1.04 ± 0.15 c |
| Anc120 | Anc140 | Anc160 | Anc180 | Anc200 | Anc220 | Anc240 | |
|---|---|---|---|---|---|---|---|
| Al | 3.74 ± 0.94 a | 4.43 ± 0.91 a | 4.85 ± 0.91 a | 5.52 ± 0.94 a | 5.92 ± 0.83 a | 6.47 ± 0.83 a | 7.15 ± 0.90 a |
| B | 7.79 ± 0.95 a | 7.21 ± 0.88 a | 6.98 ± 0.88 a | 6.41 ± 0.91 a | 6.18 ± 0.96 a | 5.25 ± 0.95 ab | 3.82 ± 0.72 b |
| Ba | 4.57 ± 0.14 a | 4.17 ± 0.12 ab | 3.65 ± 0.09 bcd | 3.55 ± 0.10 bd | 3.02 ± 0.64 cd | 1.83 ± 0.49 e | 0.34 ± 0.09 f |
| Bi | 2.75 ± 0.93 a | 3.18 ± 0.85 a | 3.49 ± 0.79 a | 4.58 ± 0.85 ab | 5.27 ± 1.03 ac | 6.49 ± 1.10 bc | 8.37 ± 1.01 c |
| Ca | 18.2 ± 1.11 a | 19.9 ± 1.33 a | 20.5 ± 1.28 a | 21.4 ± 1.17 a | 22.8 ± 1.20 a | 23.6 ± 1.02 a | 24.1 ± 1.16 a |
| Cd | 0.25 ± 0.09 a | 0.32 ± 0.09 a | 0.40 ± 0.11 a | 0.45 ± 0.14 a | 0.52 ± 0.16 a | 0.54 ± 0.19 a | 0.70 ± 0.14 a |
| Co | 1.42 ± 0.17 a | 1.26 ± 0.18 a | 1.21 ± 0.19 a | 1.12 ± 0.18 a | 1.08 ± 0.20 a | 0.99 ± 0.22 a | 0.95 ± 0.18 a |
| Cr | 0.33 ± 0.09 a | 0.51 ± 0.08 ab | 0.56 ± 0.11 ab | 0.72 ± 0.11 ac | 0.78 ± 0.14 bd | 1.02 ± 0.16 cd | 1.23 ± 0.10 d |
| Cu | 3.11 ± 0.58 a | 3.49 ± 0.58 a | 3.71 ± 0.48 a | 4.25 ± 0.59 a | 4.83 ± 0.43 ab | 6.39 ± 0.68 bc | 7.75 ± 0.58 c |
| Fe | 0.05 ± 0.01 a | 0.08 ± 0.05 a | 0.12 ± 0.05 ab | 0.15 ± 0.04 ab | 0.21 ± 0.09 ab | 0.34 ± 0.11 bc | 0.61 ± 0.09 c |
| K | 29.0 ± 1.19 a | 30.1 ± 1.25 a | 31.4 ± 1.34 a | 32.2 ± 1.32 a | 33.8 ± 1.42 a | 34.5 ± 1.22 a | 35.7 ± 1.44 a |
| Mg | 11.5 ± 0.72 a | 11.9 ± 0.62 a | 12.3 ± 0.71 a | 12.9 ± 0.64 a | 13.7 ± 0.76 a | 14.4 ± 0.84 a | 15.1 ± 0.78 a |
| Mn | 0.69 ± 0.10 a | 0.59 ± 0.08 ab | 0.51 ± 0.10 ab | 0.46 ± 0.09 ab | 0.41 ± 0.19 ab | 0.33 ± 0.10 b | 0.30 ± 0.09 b |
| Ni | 4.13 ± 0.89 a | 5.47 ± 1.08 ab | 6.54 ± 1.06 abc | 7.38 ± 1.09 abc | 8.19 ± 1.28 bd | 10.1 ± 1.19 cd | 12.1 ± 1.12 d |
| P | 6.79 ± 0.22 a | 6.36 ± 0.28 ab | 5.93 ± 0.39 abc | 5.69 ± 0.28 bc | 5.21 ± 0.26 bcd | 4.90 ± 0.25 cd | 4.51 ± 0.29 d |
| Pb | 4.09 ± 1.01 a | 5.26 ± 1.14 ab | 5.66 ± 1.18 ab | 6.19 ± 1.18 ab | 6.96 ± 1.14 ab | 7.64 ± 1.02 ab | 9.23 ± 1.23 b |
| Sr | 0.54 ± 0.09 a | 0.46 ± 0.06 ab | 0.40 ± 0.06 abc | 0.34 ± 0.06 bcd | 0.22 ± 0.08 cd | 0.20 ± 0.07 cd | 0.19 ± 0.05 d |
| Zn | 0.19 ± 0.06 a | 0.36 ± 0.11 a | 0.53 ± 0.17 ab | 0.67 ± 0.14 ab | 1.17 ± 0.22 b | 2.01 ± 0.20 c | 3.28 ± 0.19 d |
| Sal120 | Sal140 | Sal160 | Sal180 | Sal200 | Sal220 | Sal240 | |
|---|---|---|---|---|---|---|---|
| Al | 3.39 ± 0.57 a | 4.27 ± 0.77 a | 4.45 ± 0.77 a | 5.03 ± 0.94 a | 5.75 ± 1.04 a | 5.75 ± 0.93 a | 6.14 ± 1.06 a |
| B | 5.67 ± 0.64 a | 4.95 ± 0.71 ab | 4.58 ± 0.49 abc | 4.16 ± 0.50 abc | 3.97 ± 0.40 bc | 3.45 ± 0.35 bc | 3.32 ± 0.47 c |
| Ba | 8.51 ± 0.90 a | 8.14 ± 0.88 ab | 7.99 ± 0.62 ab | 7.37 ± 1.04 ab | 6.21 ± 0.96 ab | 5.64 ± 1.13 bc | 3.44 ± 0.58 c |
| Bi | 9.85 ± 0.87 a | 9.13 ± 0.97 a | 8.97 ± 0.92 a | 8.47 ± 1.02 a | 8.31 ± 1.26 ab | 7.23 ± 1.21 ab | 5.70 ± 1.40 b |
| Ca | 21.5 ± 1.68 a | 22.3 ± 1.74 a | 23.7 ± 1.53 a | 24.4 ± 1.62 a | 25.6 ± 1.76 a | 26.3 ± 1.15 a | 27.7 ± 1.46 a |
| Cd | 0.10 ± 0.03 a | 0.28 ± 0.12 a | 0.31 ± 0.16 a | 0.41 ± 0.13 a | 0.43 ± 0.16 a | 0.52 ± 0.29 a | 0.55 ± 0.23 a |
| Co | 2.27 ± 0.25 a | 1.86 ± 0.29 ab | 1.54 ± 0.44 abc | 1.36 ± 0.28 bcd | 1.09 ± 0.26 bcd | 0.99 ± 0.28 cd | 0.58 ± 0.25 d |
| Cr | 0.17 ± 0.07 a | 0.39 ± 0.11 a | 0.58 ± 0.30 a | 0.61 ± 0.30 a | 0.75 ± 0.31 a | 0.88 ± 0.36 a | 0.89 ± 0.33 a |
| Cu | 3.47 ± 0.58 a | 4.12 ± 0.68 ab | 4.36 ± 0.91 ab | 5.27 ± 0.95 abc | 6.04 ± 0.93 bcd | 7.17 ± 0.94 cd | 9.19 ± 0.97 d |
| Fe | 0.08 ± 0.05 a | 0.11 ± 0.04 a | 0.21 ± 0.10 ab | 0.34 ± 0.19 ab | 0.44 ± 0.19 ab | 0.66 ± 0.18 b | 0.69 ± 0.29 b |
| K | 26.3 ± 0.97 a | 27.2 ± 0.98 ab | 28.5 ± 1.25 ab | 29.1 ± 0.85 ab | 30.2 ± 0.90 ab | 31.5 ± 0.95 b | 31.9 ± 1.02 b |
| Mg | 15.1 ± 0.71 a | 15.9 ± 0.74 ab | 16.3 ± 0.76 ab | 16.9 ± 0.81 ab | 17.3 ± 0.79 ab | 18.4 ± 0.56 b | 19.7 ± 0.62 b |
| Mn | 1.45 ± 0.32 a | 1.22 ± 0.21 ab | 1.05 ± 0.38 abc | 0.91 ± 0.31 abc | 0.70 ± 0.21 bc | 0.65 ± 0.19 bc | 0.40 ± 0.15 c |
| Ni | 8.93 ± 0.93 a | 7.07 ± 1.01 ab | 6.18 ± 0.99 bc | 5.73 ± 0.70 bc | 5.20 ± 0.71 bc | 4.52 ± 0.57 c | 2.10 ± 0.56 d |
| P | 5.95 ± 0.31 a | 5.63 ± 0.27 ab | 5.05 ± 0.29 bc | 4.51 ± 0.27 cd | 4.33 ± 0.25 cd | 4.03 ± 0.23 d | 3.92 ± 0.24 d |
| Pb | 2.71 ± 0.27 a | 3.34 ± 0.36 ab | 4.15 ± 0.39 bc | 4.81 ± 0.41 c | 5.27 ± 0.40 c | 6.89 ± 0.44 d | 7.97 ± 0.42 d |
| Sr | 1.22 ± 0.21 a | 0.85 ± 0.19 ab | 0.72 ± 0.15 bc | 0.60 ± 0.15 bd | 0.37 ± 0.11 cd | 0.32 ± 0.11 cd | 0.21 ± 0.10 d |
| Zn | 0.36 ± 0.12 a | 0.64 ± 0.14 ab | 0.71 ± 0.23 ab | 1.10 ± 0.27 ab | 1.38 ± 0.39 ab | 2.07 ± 0.62 b | 4.15 ± 1.14 c |
| Anc120 * | Sal120 * | Sánchez-Gómez et al. [63] | |
|---|---|---|---|
| Al | 3.74 ± 0.94 | 3.39 ± 0.57 | - |
| B | 7.79 ± 0.95 | 5.67 ± 0.64 | 0.04–0.09 (0.06) |
| Ba | 4.57 ± 0.14 | 8.51 ± 0.90 | - |
| Bi | 2.75 ± 0.93 | 9.85 ± 0.87 | - |
| Ca | 18.2 ± 1.11 | 21.5 ± 1.68 | 23.6–39.4 (31.9) |
| Cd | 0.25 ± 0.09 | 0.10 ± 0.03 | - |
| Co | 1.42 ± 0.17 | 2.27 ± 0.25 | - |
| Cr | 0.33 ± 0.09 | 0.17 ± 0.07 | - |
| Cu | 3.11 ± 0.58 | 3.47 ± 0.58 | 0.02–0.11 (0.06) |
| Fe | 0.05 ± 0.01 | 0.08 ± 0.05 | 0.03–0.14 (0.07) |
| K | 29.0 ± 1.19 | 26.3 ± 0.97 | 141.3–154.1 (146.9) |
| Mg | 11.5 ± 0.72 | 15.1 ± 0.71 | 25.5–31.7 (27.9) |
| Mn | 0.69 ± 0.10 | 1.45 ± 0.32 | 0.20–0.26 (0.24) |
| Ni | 4.13 ± 0.89 | 8.93 ± 0.93 | - |
| P | 6.79 ± 0.22 | 5.95 ± 0.31 | 22.9–32.4 (27.8) |
| Pb | 4.09 ± 1.01 | 2.71 ± 0.27 | - |
| Sr | 0.54 ± 0.09 | 1.22 ± 0.21 | - |
| Zn | 0.19 ± 0.06 | 0.36 ± 0.12 | 0.15–0.22 (0.17) |
| mg/100 g on Dry Basis | ||||
|---|---|---|---|---|
| Anc * | Sal * | Çetin et al. [53] | Mendívil et al. [46] | |
| Al | 13.04 ± 0.15 | 11.28 ± 0.19 | 1.5–3.7 (2.54) | |
| B | 7.81 ± 0.14 | 9.39 ± 0.18 | 0.8–1.8 (1.34) | |
| Ba | 5.64 ± 0.20 | 4.22 ± 0.16 | 0.3–0.8 (0.52) | |
| Bi | 4.50 ± 0.09 | 3.86 ± 0.08 | ||
| Ca | 319.2 ± 13.7 | 394.5 ± 12.4 | 633–1021 (756) | 450–890 (629) |
| Cd | 0.57 ± 0.03 | 0.44 ± 0.04 | <0.1 | |
| Co | 0.62 ± 0.04 | 0.51 ± 0.03 | ||
| Cr | 0.72 ± 0.03 | 0.87 ± 0.04 | <(0.1–0.5) | |
| Cu | 12.71 ± 0.31 | 14.73 ± 0.27 | 0.8–2.0 (1.21) | |
| Fe | 0.51 ± 0.04 | 0.46 ± 0.03 | 0.26–0.68 (0.36) | 1.8–4.3 (2.52) |
| K | 561.5 ± 24.6 | 485.1 ± 19.3 | 519–823 (632) | 510–870 (674) |
| Mg | 25.12 ± 2.3 | 40.03 ± 2.9 | 1.94–11.12 (4.72) | 120–200 (159) |
| Mn | 2.32 ± 0.11 | 2.30 ± 0.08 | 1.7–4.5 (2.66) | |
| Ni | 10.81 ± 0.19 | 10.21 ± 0.21 | <0.2 | |
| P | 37.38 ± 4.5 | 32.68 ± 4.1 | 42–93 (69) | 70–90 (81) |
| Pb | 4.46 ± 0.07 | 4.14 ± 0.08 | <1 | |
| Sr | 4.37 ± 0.11 | 3.78 ± 0.09 | ||
| Zn | 2.84 ± 0.08 | 2.18 ± 0.06 | 0.70–9.82 (1.48) | 0.9–2.4 (1.41) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Eusanio, V.; Genua, F.; Marchetti, A.; Morelli, L.; Tassi, L. Exploring the Mineral Composition of Grapevine Canes for Wood Chip Applications in Alcoholic Beverage Production to Enhance Viticulture Sustainability. Beverages 2023, 9, 60. https://doi.org/10.3390/beverages9030060
D’Eusanio V, Genua F, Marchetti A, Morelli L, Tassi L. Exploring the Mineral Composition of Grapevine Canes for Wood Chip Applications in Alcoholic Beverage Production to Enhance Viticulture Sustainability. Beverages. 2023; 9(3):60. https://doi.org/10.3390/beverages9030060
Chicago/Turabian StyleD’Eusanio, Veronica, Francesco Genua, Andrea Marchetti, Lorenzo Morelli, and Lorenzo Tassi. 2023. "Exploring the Mineral Composition of Grapevine Canes for Wood Chip Applications in Alcoholic Beverage Production to Enhance Viticulture Sustainability" Beverages 9, no. 3: 60. https://doi.org/10.3390/beverages9030060