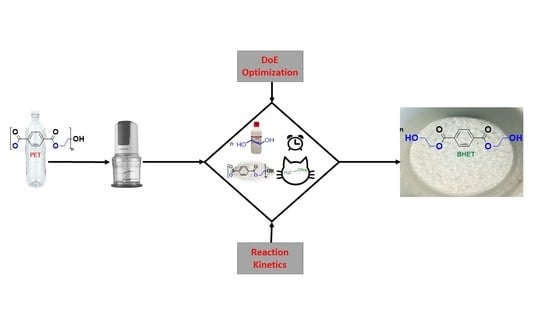
Optimization and Kinetic Evaluation for Glycolytic Depolymerization of Post-Consumer PET Waste with Sodium Methoxide
Abstract
:1. Introduction
Research Significance
2. Materials and Methods
2.1. Materials
2.2. Depolymerization of POSTC-PET Waste
2.3. Product Characterization
3. Results and Discussion
3.1. Impact of Catalyst and Selection of Reaction Temperature
3.2. Analysis of Glycolysed Products
3.3. Design of Experiments (DoE)
3.3.1. Development of Regression Model from Design Expert
3.3.2. Interactive Effects
3.3.3. Model Optimization
3.4. Performance Comparison for Sodium Methoxide
3.5. Thermodynamic and Kinetic Evaluation
3.5.1. Van’t Hoff Plot
3.5.2. Reaction Kinetics
3.5.3. Apparent Activation Energy
4. Conclusions
5. Recommendation and Outlook
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Suhaimi, N.A.S.; Muhamad, F.; Abd Razak, N.A.; Zeimaran, E. Recycling of polyethylene terephthalate wastes: A review of technologies, routes, and applications. Polym. Eng. Sci. 2022, 62, 2355–2375. [Google Scholar] [CrossRef]
- Volmajer Valh, J.; Stopar, D.; Selaya Berodia, I.; Erjavec, A.; Šauperl, O.; Fras Zemljič, L. Economical Chemical Recycling of Complex PET Waste in the Form of Active Packaging Material. Polymers 2022, 14, 3244. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, W.A. New advances in poly(ethylene terephthalate) polymerization and degradation. Polym. Int. 2002, 51, 923–930. [Google Scholar] [CrossRef]
- Statista. PET Global Production 2020|Statista. Available online: https://www.statista.com/statistics/650191/global-polyethylene-terephthalate-production-outlook/ (accessed on 21 April 2022).
- Benyathiar, P.; Kumar, P.; Carpenter, G.; Brace, J.; Mishra, D.K. Polyethylene Terephthalate (PET) Bottle-to-Bottle Recycling for the Beverage Industry: A Review. Polymers 2022, 14, 2366. [Google Scholar] [CrossRef] [PubMed]
- Statista. PET Global Bottle Production 2021|Statista. Available online: https://www.statista.com/statistics/723191/production-of-polyethylene-terephthalate-bottles-worldwide/ (accessed on 8 August 2022).
- Nabid, M.R.; Bide, Y.; Jafari, M. Boron nitride nanosheets decorated with Fe3O4 nanoparticles as a magnetic bifunctional catalyst for post-consumer PET wastes recycling. Polym. Degrad. Stab. 2019, 169, 108962. [Google Scholar] [CrossRef]
- Tuna, Ö.; Bal, A.; Güçlü, G. Investigation of the effect of hydrolysis products of postconsumer polyethylene terephthalate bottles on the properties of alkyd resins. Polym. Eng. Sci. 2013, 53, 176–182. [Google Scholar] [CrossRef]
- Al-Sabagh, A.M.; Yehia, F.Z.; Eshaq, G.; Rabie, A.M.; ElMetwally, A.E. Greener routes for recycling of polyethylene terephthalate. Egypt. J. Pet. 2016, 25, 53–64. [Google Scholar] [CrossRef] [Green Version]
- Mishra, S.; Zope, V.; Goje, A. Kinetics and thermodynamics of hydrolytic depolymerization of poly(ethylene terephthalate) at high pressure and temperature. J. Appl. Polym. Sci. 2003, 90, 3303–3309. [Google Scholar] [CrossRef]
- Grause, G.; Handa, T.; Kameda, T.; Mizoguchi, T.; Yoshioka, T. Effect of temperature management on the hydrolytic degradation of PET in a calcium oxide filled tube reactor. Chem. Eng. J. 2011, 166, 523–528. [Google Scholar] [CrossRef]
- Pang, K.; Kotek, R.; Tonelli, A. Review of conventional and novel polymerization processes for polyesters. Prog. Polym. Sci. 2006, 31, 1009–1037. [Google Scholar] [CrossRef]
- Baliga, S.; Wong, W.T. Depolymerization of poly(ethylene terephthalate) recycled from post-consumer soft-drink bottles. J. Polym. Sci. A Polym. Chem. 1989, 27, 2071–2082. [Google Scholar] [CrossRef]
- Ghaemy, M.; Mossaddegh, K. Depolymerisation of poly(ethylene terephthalate) fibre wastes using ethylene glycol. Polym. Degrad. Stab. 2005, 90, 570–576. [Google Scholar] [CrossRef]
- Fehér, Z.; Kiss, J.; Kisszékelyi, P.; Molnár, J.; Huszthy, P.; Kárpáti, L.; Kupai, J. Optimisation of PET glycolysis by applying recyclable heterogeneous organocatalysts. Green Chem. 2022, 24, 8447–8459. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, M.; Hwang, J.; Im, E.; Moon, G.D. Optimizing PET Glycolysis with an Oyster Shell-Derived Catalyst Using Response Surface Methodology. Polymers 2022, 14, 656. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.R.; Palekar, V.; Pingale, N. Zeolite catalyzed glycolysis of poly(ethylene terephthalate) bottle waste. J. Appl. Polym. Sci. 2008, 110, 501–506. [Google Scholar] [CrossRef]
- Sert, E.; Yılmaz, E.; Atalay, F.S. Chemical Recycling of Polyethlylene Terephthalate by Glycolysis Using Deep Eutectic Solvents. J. Polym. Env. 2019, 27, 2956–2962. [Google Scholar] [CrossRef]
- Al-Sabagh, A.M.; Yehia, F.Z.; Eissa, A.M.F.; Moustafa, M.E.; Eshaq, G.; Rabie, A.M.; ElMetwally, A.E. Cu- and Zn-acetate-containing ionic liquids as catalysts for the glycolysis of poly(ethylene terephthalate). Polym. Degrad. Stab. 2014, 110, 364–377. [Google Scholar] [CrossRef]
- Jeya, G.; Dhanalakshmi, R.; Anbarasu, M.; Vinitha, V.; Sivamurugan, V. A short review on latest developments in catalytic depolymerization of Poly (ethylene terephathalate) wastes. J. Indian Chem. Soc. 2022, 99, 100291. [Google Scholar] [CrossRef]
- Ghosal, K.; Nayak, C. Recent advances in chemical recycling of polyethylene terephthalate waste into value added products for sustainable coating solutions–hope vs. hype. Mater. Adv. 2022, 3, 1974–1992. [Google Scholar] [CrossRef]
- Xin, J.; Zhang, Q.; Huang, J.; Huang, R.; Jaffery, Q.Z.; Yan, D.; Zhou, Q.; Xu, J.; Lu, X. Progress in the catalytic glycolysis of polyethylene terephthalate. J. Environ. Manage. 2021, 296, 113267. [Google Scholar] [CrossRef]
- Shukla, S.R.; Kulkarni, K.S. Depolymerization of poly(ethylene terephthalate) waste. J. Appl. Polym. Sci. 2002, 85, 1765–1770. [Google Scholar] [CrossRef]
- López-Fonseca, R.; Duque-Ingunza, I.; de Rivas, B.; Arnaiz, S.; Gutiérrez-Ortiz, J.I. Chemical recycling of post-consumer PET wastes by glycolysis in the presence of metal salts. Polym. Degrad. Stab. 2010, 95, 1022–1028. [Google Scholar] [CrossRef]
- Mohsin, M.A.; Alnaqbi, M.A.; Busheer, R.M.; Haik, Y. Sodium Methoxide Catalyzed Depolymerization of Waste Polyethylene Terephthalate Under Microwave Irradiation. Catal. Ind. 2018, 10, 41–48. [Google Scholar] [CrossRef]
- Aziz, N.A.M.; Hamid, H.A.; Yunus, R.; Abbas, Z.; Omar, R.; Rashid, U.; Syam, A.M. Kinetics and thermodynamics of synthesis of palm oil-based trimethylolpropane triester using microwave irradiation. J. Saudi Chem. Soc. 2020, 24, 552–566. [Google Scholar] [CrossRef]
- Aguado, A.; Martínez, L.; Becerra, L.; Arieta-araunabeña, M.; Arnaiz, S.; Asueta, A.; Robertson, I. Chemical depolymerisation of PET complex waste: Hydrolysis vs. glycolysis. J. Mater. Cycles Waste Manag. 2014, 16, 201–210. [Google Scholar] [CrossRef]
- Stoski, A.; Viante, M.F.; Nunes, C.S.; Muniz, E.C.; Felsner, M.L.; Almeida, C.A.P. Oligomer production through glycolysis of poly(ethylene terephthalate): Effects of temperature and water content on reaction extent. Polym. Int. 2016, 65, 1024–1030. [Google Scholar] [CrossRef]
- Katoch, S.; Sharma, V.; Kundu, P.P.; Bera, M.B. Optimization of PET Glycolysis Process by Response Surface Methodological Approach: A Two-Component Modelling Using Glycolysis Time and Temperature. ISRN Polym. Sci. 2012, 2012, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.-H.; Chen, C.-Y.; Lo, Y.-W.; Mao, C.-F.; Liao, W.-T. Studies of glycolysis of poly(ethylene terephthalate) recycled from postconsumer soft-drink bottles. II. Factorial experimental design. J. Appl. Polym. Sci. 2001, 80, 956–962. [Google Scholar] [CrossRef]
- Chen, C.-H. Study of glycolysis of poly(ethylene terephthalate) recycled from postconsumer soft-drink bottles. III. Further investigation. J. Appl. Polym. Sci. 2003, 87, 2004–2010. [Google Scholar] [CrossRef]
- Van-Pham, D.-T.; Le, Q.-H.; Lam, T.-N.; Nguyen, C.-N.; Sakai, W. Four-factor optimization for PET glycolysis with consideration of the effect of sodium bicarbonate catalyst using response surface methodology. Polym. Degrad. Stab. 2020, 179, 109257. [Google Scholar] [CrossRef]
- ChemicalBook. Ethylene Glycol|107-21-1. Available online: https://www.chemicalbook.com/ChemicalProductProperty_EN_CB7852707.htm (accessed on 30 October 2022).
- Yue, H.; Zhao, Y.; Ma, X.; Gong, J. Ethylene glycol: Properties, synthesis, and applications. Chem. Soc. Rev. 2012, 41, 4218–4244. [Google Scholar] [CrossRef]
- Yunita, I.; Putisompon, S.; Chumkaeo, P.; Poonsawat, T.; Somsook, E. Effective catalysts derived from waste ostrich eggshells for glycolysis of post-consumer PET bottles. Chem. Pap. 2019, 73, 1547–1560. [Google Scholar] [CrossRef]
- Froidevaux, V.; Negrell, C.; Caillol, S.; Pascault, J.-P.; Boutevin, B. Biobased Amines: From Synthesis to Polymers; Present and Future. Chem. Rev. 2016, 116, 14181–14224. [Google Scholar] [CrossRef] [PubMed]
- Lima, G.R.; Monteiro, W.F.; Ligabue, R.; Santana, R.M.C. Titanate Nanotubes as New Nanostrutured Catalyst for Depolymerization of PET by Glycolysis Reaction. Mat. Res. 2017, 20, 588–595. [Google Scholar] [CrossRef] [Green Version]
- Xi, G.; Lu, M.; Sun, C. Study on depolymerization of waste polyethylene terephthalate into monomer of bis(2-hydroxyethyl terephthalate). Polym. Degrad. Stab. 2005, 87, 117–120. [Google Scholar] [CrossRef]
- Guclu, G.; Kasgoz, A.; Ozbudak, S.; Ozgumus, S.; Orbay, M. Glycolysis of poly(ethylene terephthalate) wastes in xylene. J. Appl. Polym. Sci. 1998, 69, 2311–2319. [Google Scholar] [CrossRef]
- Silva, C.V.G.; Silva Filho, E.A.d.; Uliana, F.; Jesus, L.F.R.d.; Melo, C.V.P.d.; Barthus, R.C.; Rodrigues, J.G.A.; Vanini, G. PET glycolysis optimization using ionic liquid [Bmin]ZnCl3 as catalyst and kinetic evaluation. Polímeros 2018, 28, 450–459. [Google Scholar] [CrossRef] [Green Version]
- Veregue, F.R.; Pereira da Silva, C.T.; Moisés, M.P.; Meneguin, J.G.; Guilherme, M.R.; Arroyo, P.A.; Favaro, S.L.; Radovanovic, E.; Girotto, E.M.; Rinaldi, A.W. Ultrasmall Cobalt Nanoparticles as a Catalyst for PET Glycolysis: A Green Protocol for Pure Hydroxyethyl Terephthalate Precipitation without Water. ACS Sust. Chem. Eng. 2018, 6, 12017–12024. [Google Scholar] [CrossRef]
- Al-Sabagh, A.M.; Yehia, F.Z.; Eissa, A.-M.M.F.; Moustafa, M.E.; Eshaq, G.; Rabie, A.-R.M.; ElMetwally, A.E. Glycolysis of Poly(ethylene terephthalate) Catalyzed by the Lewis Base Ionic Liquid [Bmim][OAc]. Ind. Eng. Chem. Res. 2014, 53, 18443–18451. [Google Scholar] [CrossRef]
- Wang, H.; Liu, Y.; Li, Z.; Zhang, X.; Zhang, S.; Zhang, Y. Glycolysis of poly(ethylene terephthalate) catalyzed by ionic liquids. Eur. Polym. J. 2009, 45, 1535–1544. [Google Scholar] [CrossRef]
- Kratofil Krehula, L.; Hrnjak-Murgić, Z.; Jelenčić, J.; Andričić, B. Evaluation of Poly(ethylene-terephthalate) Products of Chemical Recycling by Differential Scanning Calorimetry. J. Polym. Env. 2009, 17, 20–27. [Google Scholar] [CrossRef]
- Chamoli, S. ANN and RSM approach for modeling and optimization of designing parameters for a V down perforated baffle roughened rectangular channel. Alex. Eng. J. 2015, 54, 429–446. [Google Scholar] [CrossRef] [Green Version]
- Rana, A.G.; Minceva, M. Analysis of Photocatalytic Degradation of Phenol with Exfoliated Graphitic Carbon Nitride and Light-Emitting Diodes Using Response Surface Methodology. Catalysts 2021, 11, 898. [Google Scholar] [CrossRef]
- Goje, A.S.; Mishra, S. Chemical Kinetics, Simulation, and Thermodynamics of Glycolytic Depolymerization of Poly(ethylene terephthalate) Waste with Catalyst Optimization for Recycling of Value Added Monomeric Products: 29. Macromol. Mater. Eng. 2003, 288, 326–336. [Google Scholar] [CrossRef]
- Sodium Methoxide | Sigma-Aldrich, 2023.000Z. Available online: https://www.sigmaaldrich.com/DE/en/search/sodium-methoxide?focus=products&page=1&perpage=30&sort=relevance&term=sodium%20methoxide&type=product (accessed on 19 January 2023).
- Cobalt Acetate | Sigma-Aldrich, 2023.000Z. Available online: https://www.sigmaaldrich.com/DE/en/search/cobalt-acetate?focus=products&page=1&perpage=30&sort=relevance&term=cobalt%20acetate&type=product (accessed on 19 January 2023).
- Zinc Acetate | Sigma-Aldrich, 2023.000Z. Available online: https://www.sigmaaldrich.com/DE/en/search/zinc-acetate?focus=products&page=1&perpage=30&sort=relevance&term=zinc%20acetate&type=product (accessed on 19 January 2023).
- López-Fonseca, R.; Duque-Ingunza, I.; de Rivas, B.; Flores-Giraldo, L.; Gutiérrez-Ortiz, J.I. Kinetics of catalytic glycolysis of PET wastes with sodium carbonate. Chem. Eng. J. 2011, 168, 312–320. [Google Scholar] [CrossRef]







| Independent Variable | Symbol | Coded Levels | |||
|---|---|---|---|---|---|
| Actual (Vi) | Coded (Ci) | −1 (L) | 0 (M) | +1 (H) | |
| PET:MeONa (mol/mol) | A | C1 | 50 | 100 | 150 |
| EG:PET (mol/mol) | B | C2 | 3 | 5 | 7 |
| Reaction time, (h) | C | C3 | 2 | 4 | 6 |
| Particle size, (mm) | D | C4 | 0.25 | 0.625 | 1 |
| Response | PET Conversion (X) |
|---|---|
| R2 | 0.9552 |
| Adjusted R2 | 0.9103 |
| Predicted R2 | 0.7770 |
| Adequate Precision (>4) | 19.288 |
| Lack of Fit (p-value) | 1.73 |
| Significant terms | A, B, C, AB, AC, AD, C2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Javed, S.; Fisse, J.; Vogt, D. Optimization and Kinetic Evaluation for Glycolytic Depolymerization of Post-Consumer PET Waste with Sodium Methoxide. Polymers 2023, 15, 687. https://doi.org/10.3390/polym15030687
Javed S, Fisse J, Vogt D. Optimization and Kinetic Evaluation for Glycolytic Depolymerization of Post-Consumer PET Waste with Sodium Methoxide. Polymers. 2023; 15(3):687. https://doi.org/10.3390/polym15030687
Chicago/Turabian StyleJaved, Saqib, Jonas Fisse, and Dieter Vogt. 2023. "Optimization and Kinetic Evaluation for Glycolytic Depolymerization of Post-Consumer PET Waste with Sodium Methoxide" Polymers 15, no. 3: 687. https://doi.org/10.3390/polym15030687