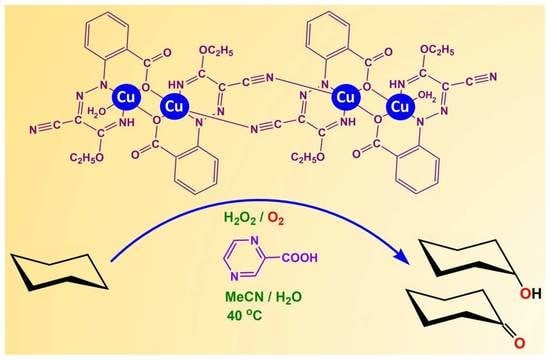
2.1. Catalytic Studies under Conventional Conditions
The results obtained under conventional conditions for the peroxidative oxidation of cyclohexane with hydrogen peroxide and employing the tetranuclear Cu(II) complex
1 as catalyst are summarized in
Table 1; those using the tetranuclear Cu(II) complex
2 are shown in
Table 2, and in
Table 3 the ones utilizing the mononuclear Cu(II) complexes
3 and
4 or the polynuclear Co(II/III) and Zn(II) complexes
5 and
6.
The experiments performed at room temperature with nitric acid as promoter and catalyst
1 (
Table 1, entries 1 to 4) have shown a slight increase in the total yield of oxygenated products upon increasing of the reaction time from 1 to 4 h; a further increase to 6 h had nearly no influence. Using PCA instead of HNO
3 led to an evident decrease in the yield, at room temperature (
Table 1, entry 5).
When the reaction temperature was increased to 40 °C in the presence of HNO
3 an overall rise in the total yield of products was observed with no significant changes associated with higher reaction times (
Table 1, entries 6–9) or higher temperature (
Table 1, entry 10). With PCA as promoter, a reaction time of 4 h at 40 °C gave the best results for this system (
Table 1, compare entries 11–13; total yield of 26% and TON = 52.0 in entry 13) but a marked decrease in the yield with a further increase of temperature to 50 °C (
Table 1, entry 14). Without any added acid promoter, the results were similar to those obtained with nitric acid (
Table 1, entries 15 and 16; compare with entries 9 and 10).
A study concerning the effect of the amount of H
2O in the system was carried out (
Table 1, entries 17–21). The successive replacement of CH
3CN by water resulted in lower total yields of oxygenated products from 16–18% to 10% although with a slight increase in the cyclohexanone production.
No significant changes were observed in the outcome of the system CyH/H
2O
2/HNO
3 at 50 °C when performing the reaction in uncapped glass tubes (thus with more O
2 available) or upon extending the reaction time from 4 to 6 h (
Table 1, entry 10 versus 22 and 23, respectively).
Typical copper(II) salts such as CuCl
2 and Cu(NO
3)
2·3H
2O worked only marginally as catalysts for the reaction under study, whatever the acid promoter was used (
Table 1, entries 24–27). The comparatively better results obtained with CuCl
2 were somehow expected since this Cu(II) salt has been widely employed as catalyst for oxidative coupling and oxygenation reactions in organic synthesis due to the formation of active multinuclear copper species in the presence of common coordinating solvents (such as acetonitrile) [
32,
33].
Concerning the tetranuclear Cu(II) complex
2 as catalyst (
Table 2, entries 1 to 8), PCA proved to be better acid promoter than HNO
3 both at 40 °C and 50 °C; at room temperature, only HNO
3 was effective (
Table 2, entry 2) although the amount of oxygenated products was still very limited (only 5% of cyclohexanol). The beneficial effect of increasing the temperature on the reaction yield was again observed.
The influence of H
2O amount in the outcome of the peroxidative oxidation of cyclohexane with hydrogen peroxide was similar to what was recorded for complex
1: as the amount of water in the system increased, there was a decrease in the total yield of oxygenated products (cyclohexanol + cyclohexanone) but, in contrast, the production of cyclohexanone slightly increased (
Table 2, entries 7 and 9–13).
Both mononuclear Cu(II) complexes
3 and
4 exhibited comparatively lower catalytic activities (
Table 3, entries 1–7 and 8–14, respectively); the best results were obtained with HNO
3 as acid promoter (PCA did not fulfill a promoting role) after 4 h at 50 °C (certify entries 6 and 13 in
Table 3: 8% total yield, TON = 16.0 and TOF = 4.0 h
−1 for
3, and 4% total yield, TON = 8.0 and TOF = 2.0 h
−1 for
4). A temperature effect on the reaction yield was marginally observed. Considering the catalytic activity per metal center (Cu
2+ ion), the obtained results are in the same order of magnitude as the ones obtained with the tetranuclear copper(II) complexes
1 and
2. No oxygenated products were detected when using the trinuclear Co(II/III) complex
5 or the tetranuclear Zn(II) complex
6 as catalysts (in this order,
Table 3 entries 15–16 and 17–18).
The employment of CH
3CN/H
2O solvent mixtures is essential to solubilize both substrate and catalyst and the mild alkane oxidation does not proceed effectively only in water or acetonitrile as sole solvents. The acetonitrile was chosen due to: (i) its miscibility with water and ability to solubilize substrate, catalysts and products; (ii) the high stability towards oxidation under the applied reaction conditions (contrasting with solvents like methanol, ethanol or acetone); (iii) its coordination ability; and (iv) the best results previously achieved upon utilization of this solvent [
2,
15].
Complexes 1 and 2 are readily soluble in the acidic CH3CN/H2O medium, yielding green and greenish yellow solutions, respectively, with a slight solid residue in suspension; throughout the oxidation process, a minor change in the color of the solutions was observed and upon reaction completion greenish solutions with a small amount of a dark flocculated precipitate were obtained.
The mononuclear Cu(II) compounds 3 and 4 exhibit a higher solubility in the chosen medium thus affording almost clear yellow and greenish yellow solutions, respectively, that became paler and completely clear after the peroxidative oxidation reaction.
Finally, regarding the multinuclear Co(II/III) and Zn(II) complexes 5 and 6, there was a shift from dark green and yellow solutions, comprising a slight solid residue in suspension, to green (with a residual amount of a flocculated precipitate) and completely clear yellow solutions, respectively.
The presence of an acid promoter proved to be essential for achieving better results. Its role as co-catalyst can be associated with: (i) promotion of proton transfer steps; (ii) catalyst activation by unsaturation of the Cu(II) centers, upon ligand protonation; (iii) enhancement of oxidative properties of the catalysts and H
2O
2 (acids can also be oxidants themselves); (iv) facilitation of the formation of peroxo complexes; and (v) preventing the decomposition of H
2O
2 to water and oxygen (thus suppressing eventual catalase activity in acidic medium) [
2,
15].
As reported for other copper(II) catalysts [
1,
2,
10], the oxidation of cyclohexane is expected to follow a radical mechanism (
Scheme 4) via metal-promoted formation of the hydroxyl radical (HO
·) from H
2O
2, which acts as H-abstractor from cyclohexane (CyH) to give cyclohexyl radical (Cy
·). The reaction of this radical with O
2 forms the peroxyl radical CyOO
· which can further react with a LCu(I) species to yield the corresponding anion which is converted into the main primary product—cyclohexyl hydroperoxide—by protonation. CyOOH undergoes copper-assisted decomposition giving the corresponding alkoxyl CyO
· and alkylperoxyl CyOO
· radicals; the former is converted to the alcohol (CyOH) by H-abstraction from the substrate, and the latter dismutates to yield both the alcohol and the ketone (Cy′O).
2.2. Catalytic Studies in scCO2 or with MW Irradiation
The results obtained in supercritical carbon dioxide medium or under microwave irradiation are summarized in
Table 4 and
Table 5, respectively.
The use of scCO
2 in the system cyclohexane/H
2O
2/HNO
3/complex
1 at 40 °C led to the total yield of oxygenated compounds of 7% for the reaction time of 4 h (it was only 1% for 2 h) (
Table 4, entries 3 and 2, respectively). Increasing the temperature to 50 °C and reaction time to 6 h led to a further enhancement of the yield of total oxygenated compounds to 17%. These values are slightly below those obtained under conventional conditions and similar reaction parameters (e.g., compare with
Table 1, entry 9: 18% after 6 h at 40 °C). A lower CO
2 pressure led to a worse yield of products (
Table 4, compare entries 6 and 7).
The introduction of scCO
2 in the system cyclohexane/H
2O
2/PCA/complex
2 at 50 °C and 4 h did not result in a better performance, since no oxygenated products were detected (
Table 4, entry 9 versus
Table 2, entry 8—20%, TON = 40.0 and TOF = 10.0 h
−1). However, the use of nitric acid in combination with scCO
2 resulted in a slight increase in the reaction yield and catalytic parameters (21%, TON = 42.0, TOF = 10.5 h
−1—
Table 4, entry 8 vs. 16%, TON = 32.0, TOF = 8.0 h
−1—
Table 2, entry 7).
When microwave irradiation was employed as alternative to conventional heating, it was possible to achieve total yields of oxygenates up to 14% (for the tetranuclear Cu(II) complex
1—
Table 5, entry 2) and 13% (for compound
2—
Table 5, entry 6), although at a higher temperature (70 °C) but in quite a shorter reaction time (0.5 h), thus leading to the highest TOFs of 56.0 and 52.0 h
−1, respectively. These values, however, are below the best ones recorded for the experiments held in conventional conditions when using nitric acid as co-catalyst, namely: 20% for catalyst
1, at 40 °C and 1 h reaction time (
Table 1, entry 6) and 16% for catalyst
2, at 40 or 50 °C and 4 h reaction time (
Table 2, entries 4 and 7). On the other hand, the increase of the reaction time to 1 h or the temperature to 80 °C (
Table 5, entries 3–4 and 7–8) did not lead to higher yields of oxygenated products.
Therefore, a higher temperature under MW irradiation (70 °C) or a longer reaction time under conventional heating are essential to achieve better results concerning the peroxidative oxidation of cyclohexane into a mixture of cyclohexanol and cyclohexanone.
2.3. Comparison with Other Copper Catalytic Systems
The most relevant results concerning other Cu(II) catalytic systems employed in the peroxidative oxidation of cyclohexane by hydrogen peroxide are summarized in
Table 6.
The highest total yields of oxygenates obtained in this work when using the tetranuclear Cu(II) complexes 1 and 2 as catalysts (0.005 mmol) and HNO
3 as co-catalyst (0.025 mmol), under conventional conditions, namely: (i) 20% (with TON = 40.0) at 40 °C and 1 h for 1 (
Table 1, entry 6); (ii) 16% (with TON = 32.0) at 40 °C and 4 h for 2 (
Table 2, entry 4), are slightly below the one recorded for the dicopper(II) complex [Cu
2(H
2O)
2(μ-L)
2], containing the basic form of the hydrazone-derived species H
2L = 3-(2-hydroxy-4-nitrophenylhydrazo)pentane-2,4-dione, in CH
3CN/aqueous H
2O
2, which afforded a total yield of 27% under mild conditions (25 °C, 6 h) (
Table 6, entry 1) although employing higher catalyst, promoter and oxidant loadings (0.02 mmol [Cu
2(H
2O)
2(μ-L)
2], 0.2 mmol HNO
3 and 10 mmol H
2O
2); therefore a lower TON of 13.3 was recorded [
26].
However, taking into consideration non-polymeric homometallic Cu(II) complexes with other types of ligands, the performance of the compounds 1 and 2 in the peroxidative oxidation of cyclohexane by aqueous H
2O
2 is quite different. Indeed, several tested tri- and tetra-copper(II) compounds with aminopolyalcoholates ligands exhibited total product yields up to 39% [
2] after 6 h reaction time at room temperature in acetonitrile, being the highest values achieved with [Cu
4(O)(tea)
4(BOH)
4][BF
4]
2 (39%) and [Cu
3(H
2tea)
2(poba)
2(H
2O)]·4H
2O (37%) (where H
3tea = triethanolamine; H
2poba = 4-oxybenzoic acid) [
34] (
Table 6, entries 2 and 3). The dinuclear copper(II) compound [{Cu(Hdea)(H
2dea)}
2(H
2pma)]·3H
2O (H
2dea = diethanolamine; H
4pma = pyromellitic acid), encompassing a carboxylate ligand as well, proved to be an efficient catalyst concerning the mild peroxidative oxidation of cyclohexane in acetonitrile/water medium at 50 °C with a maximum yield of products of 34%, in the presence of trifluoroacetic acid (TFA) as a co-catalyst [
10,
35] (
Table 6, entry 4).
Two tetranuclear Cu(II) complexes having carboxylate ligands and Schiff bases, instead of aminoalcoholates, also provided an interesting outcome concerning the peroxidative oxidation of cyclohexane (
Table 6, entries 5 and 6): [Cu
4(O)(L
3)
2(CH
3COO)
4] (where HL
3 = 4-methyl-2,6-bis(2-bromoethyliminomethyl)phenol) yielded 36% of oxygenates upon reaction for 6 h at room temperature in an aqueous acidic (HNO
3) medium [
36]; [(CuL
7)
2(cdc)]
2·2H
2O (HL
7 = 2-(2-pyridylmethyleneamino)benzenesulfonic acid; H
2cdc = cyclohexane-1,4-dicarboxylic acid) afforded yields in the 29–31% range in the absence of the promoter TFA or in its presence, in aqueous CH
3CN medium at 50 °C [
10,
37].
These remarkable results may arise from the presence of a good number of hydrophilic carboxylate (and sulfonate, in the case of [(CuL
7)
2(cdc)]
2·2H
2O) groups which are able to activate water molecules towards its significant role as a promoter for proton-shift steps involved in the formation of hydroxyl radicals from H
2O
2. Moreover, the multinuclear character of these complexes allows the presence, in proximity, of copper units in the oxidized (Cu
2+) and reduced (Cu
+) forms, thus favoring that key stage in the mechanism of the cyclohexane oxidation [
37].
In the absence of carboxylate ligands, some Schiff base Cu(II) complexes also displayed an interesting catalytic activity towards the peroxidative oxidation of C
6H
12 in acetonitrile/water medium, under mild conditions, although longer reaction times were employed. The dinuclear Cu(II) complex [Cu
2(L
1)
2(μ
2-Cl)Cl]·2.5H
2O (being HL
1 = 1-((2-hydroxyethylimino)methyl)naphthalen-2-ol) gave yields of oxygenates varying from 9 to 30.4% depending on the n(H
2O
2)/n(catalyst) ratio (from 100 to 500) upon reaction at 35 °C for 8 h in CH
3CN; however, when the reaction time was extended to 48 h, the yields increased from 15.5 to 48.2%, respectively [
38] (
Table 6, entry 7). With the mononuclear aroylhydrazone Cu(II) complex [Cu(H
2L)(NO
3)(H
2O)], comprising the deprotonated
keto form of the Schiff base (3,5-di-
tert-butyl-2-hydroxybenzylidene)-2-hydroxybenzohydrazide (H
3L), a maximum overall yield of 30% was attained after 24 h (at room temperature and without any additive) [
1,
39] (
Table 6, entry 8).
On the other hand, the Cu(II) compound with phenanthroline [CuCl(phen)
2]Cl·5H
2O (
Table 6, entry 9) afforded a remarkable total yield of 67% of oxygenated products at 70 °C in acetonitrile:water (3.5:1) solution; however, at a higher temperature range (50–70 °C), adipic acid was also formed in low quantities [
1,
40].
Recently, the heptanuclear complex [(Ph
5Si
5O
10)
2][PhCOO]
4Cu
7(EtOH)
6, comprising four benzoate ligands, was evaluated as a catalyst for the peroxidative oxidation of alkanes with H
2O
2 at 50 °C in acetonitrile (
Table 6, entry 10): using cyclohexane as substrate, a maximum yield of oxygenated products as high as 32% was achieved after 2 h [
41], thus stressing the key role of a higher nuclearity and the presence of activating ligands (such as carboxylates) for a better catalytic performance.
Another important factor related to a higher activity is the presence of the very strong basic O
2- ion in the complexes structure—due to its high affinity towards protons, the activation of the peroxide oxidant through proton abstraction would be enhanced resulting in more active systems. Indeed, the tetranuclear copper compounds [Cu
4(O)(tea)
4(BOH)
4][BF
4]
2 and [Cu
4(O)(L
3)
2(CH
3COO)
4] (
Table 6, entries 2 and 5, respectively) are amongst the most active ones (and under milder conditions—reactions at r.t.).
In some cases, the total yields of oxygenates (CyOH and Cy’O) were similar to the ones obtained for the tetranuclear Cu(II) complexes 1 and 2 (20 and 16%, respectively): (i) the C-scorpionate complex [CuCl
2{κ
3-HOCH
2C(pz)
3}] (Hpz = pyrazole) led to 23% of the yield of ketone + alcohol at room temperature, in an acidic-CH
3CN medium [
1] (
Table 6, entry 11); (ii) the compound [Cu(L)(bipy)]·MeOH, where H
2L =
o-[(
o-hydroxyphenyl)methylideneamino]benzenesulfonic acid and bipy = 2,2′-bipyridine, led to a 20% yield, after 2 h of reaction time at 60 °C in acetonitrile [
42] (
Table 6, entry 12); (iii) several Cu(II) complexes with barbiturate derivatives as ligands (formed upon reaction of 5-formylbarbiturates with primary aryl amines) afforded yields of the oxidation products in the range 11.5–22.1% after 6 h of reaction at 50 °C in acidic acetonitrile medium [
43] (
Table 6, entry 13).
Finally, concerning the employment of microwave irradiation, the Cu
I/II pyrazolate compounds [Cu
2(
N,N-3,5-(NO
2)
2pz)
2(PPh
3)
2] and [
trans-Cu
6(OH)
6(3,5-(CF
3)
2pz)
6] were successfully tested as catalysts for the MW-assisted neat oxidation of cyclohexane, affording significantly high yields (up to 58%) in rather short reaction times, but a different oxidant—
tert-butylhydroperoxide (TBHP)—was used [
1,
44].