Recent Trends in SERS-Based Plasmonic Sensors for Disease Diagnostics, Biomolecules Detection, and Machine Learning Techniques
Abstract
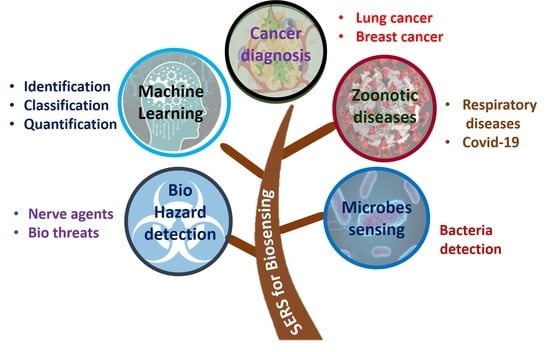
:1. Introduction
2. SERS for Disease Diagnosis
2.1. Cancer Diagnosis and Theranostics
2.1.1. Lung Cancer
2.1.2. Breast Cancer
2.1.3. Miscellaneous
2.2. SARS-CoV-2 and Other Respiratory Diseases
3. SERS-Based Detection of Microorganisms
3.1. Bacteria Sensing
3.2. Sensing of Biohazardous Molecules for Homeland Security
4. Machine Learning in SERS-Based Biosensing
4.1. Introduction to Machine Learning
4.2. Identification
4.3. Quantification
4.4. Classification
5. Conclusions and Scope
Author Contributions
Funding
Conflicts of Interest
References
- Stewart, M.E.; Anderton, C.R.; Thompson, L.B.; Maria, J.; Gray, S.K.; Rogers, J.A.; Nuzzo, R.G. Nanostructured Plasmonic Sensors. Chem. Rev. 2008, 108, 494–521. [Google Scholar] [CrossRef]
- Costanzo, H.; Gooch, J.; Frascione, N. Nanomaterials for Optical Biosensors in Forensic Analysis. Talanta 2023, 253, 123945. [Google Scholar] [CrossRef]
- Chen, G.; Chen, Y.; Huang, W.; Shi, Y. Plasmonic Nanobiosensors for Detection of Different Targets. In Proceedings of the Second International Conference on Medical Imaging and Additive Manufacturing (ICMIAM 2022), Xiamen, China, 25–27 February 2022. [Google Scholar] [CrossRef]
- Sadani, K.; Nag, P.; Thian, X.Y.; Mukherji, S. Enzymatic Optical Biosensors for Healthcare Applications. Biosens. Bioelectron. X 2022, 12, 100278. [Google Scholar] [CrossRef]
- Erkmen, C.; Selcuk, O.; Unal, D.N.; Kurbanoglu, S.; Uslu, B. Layer-by-Layer Modification Strategies for Electrochemical Detection of Biomarkers. Biosens. Bioelectron. X 2022, 12, 100270. [Google Scholar] [CrossRef]
- Spillman, W.B. Fiber Optic Biosensors; Elsevier: Amsterdam, The Netherlands, 2011; Volume 3, ISBN 9780470126844. [Google Scholar]
- Kazanskiy, N.L.; Khonina, S.N.; Butt, M.A.; Kaźmierczak, A.; Piramidowicz, R. State-of-the-Art Optical Devices for Biomedical Sensing Applications—A Review. Electronics 2021, 10, 973. [Google Scholar] [CrossRef]
- Ramirez, J.C.; Grajales García, D.; Maldonado, J.; Fernández-Gavela, A. Current Trends in Photonic Biosensors: Advances towards Multiplexed Integration. Chemosensors 2022, 10, 398. [Google Scholar] [CrossRef]
- Chadha, U.; Bhardwaj, P.; Agarwal, R.; Rawat, P.; Agarwal, R.; Gupta, I.; Panjwani, M.; Singh, S.; Ahuja, C.; Selvaraj, S.K.; et al. Recent Progress and Growth in Biosensors Technology: A Critical Review. J. Ind. Eng. Chem. 2022, 109, 21–51. [Google Scholar] [CrossRef]
- Dutta, G. Next-Generation Nanobiosensor Devices for Point-of-Care Diagnostics; Springer: Singapore, 2023; ISBN 9789811971303. [Google Scholar] [CrossRef]
- Ahangari, A.; Mahmoodi, P.; Mohammadzadeh, A. Advanced Nano Biosensors for Rapid Detection of Zoonotic Bacteria. Biotechnol. Bioeng. 2022, 120, 41–56. [Google Scholar] [CrossRef]
- Taha, B.A.; Al Mashhadany, Y.; Bachok, N.N.; Ashrif, A.; Bakar, A.; Hafiz Mokhtar, M.H.; Dzulkefly Bin Zan, M.S.; Arsad, N. Detection of COVID-19 Virus on Surfaces Using Photonics: Challenges and Perspectives. Diagnostics 2021, 11, 1119. [Google Scholar] [CrossRef]
- Soma, V.R.; Podagatlapalli, G.K.; Hamad, S.; Mechanics, F. Ultrafast Laser Ablation in Liquids for Nanomaterials and Applications. J. Nanosci. Nanotechnol. 2014, 14, 1364–1388. [Google Scholar] [CrossRef]
- Soler, M.; Lechuga, L.M. Principles, Technologies, and Applications of Plasmonic Biosensors. J. Appl. Phys. 2021, 129, 111102. [Google Scholar] [CrossRef]
- Barbillon, G. Plasmonics and Its Applications. Materials 2019, 12, 1502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Michaels, A.M.; Jiang, J.; Brus, L. Ag Nanocrystal Junctions as the Site for Surface-Enhanced Raman Scattering of Single Rhodamine 6G Molecules. J. Phys. Chem. B 2000, 104, 11965–11971. [Google Scholar] [CrossRef] [Green Version]
- Golightly, R.S.; Doering, W.E.; Natan, M.J. Surface-Enhanced Raman Spectroscopy and Homeland Security: A Perfect Match? ACS Nano 2009, 3, 2859–2869. [Google Scholar] [CrossRef] [PubMed]
- Vendamani, V.S.; Beeram, R.; Nageswara Rao, S.V.S.; Pathak, A.P.; Soma, V.R. Trace Level Detection of Explosives and Pesticides Using Robust, Low-Cost, Free-Standing Silver Nanoparticles Decorated Porous Silicon. Opt. Express 2021, 29, 30045. [Google Scholar] [CrossRef]
- Liu, C.; Xu, D.; Dong, X.; Huang, Q. A Review: Research Progress of SERS-Based Sensors for Agricultural Applications. Trends Food Sci. Technol. 2022, 128, 90–101. [Google Scholar] [CrossRef]
- Zhang, D.; Pu, H.; Huang, L.; Sun, D.W. Advances in Flexible Surface-Enhanced Raman Scattering (SERS) Substrates for Nondestructive Food Detection: Fundamentals and Recent Applications. Trends Food Sci. Technol. 2021, 109, 690–701. [Google Scholar] [CrossRef]
- Li, Y.; Liu, X.; Lin, Z. Recent Developments and Applications of Surface Plasmon Resonance Biosensors for the Detection of Mycotoxins in Foodstuffs. Food Chem. 2012, 132, 1549–1554. [Google Scholar] [CrossRef]
- Dies, H.; Raveendran, J.; Escobedo, C.; Docoslis, A. Rapid Identification and Quantification of Illicit Drugs on Nanodendritic Surface-Enhanced Raman Scattering Substrates. Sens. Actuators B Chem. 2018, 257, 382–388. [Google Scholar] [CrossRef]
- Vendamani, V.S.; Beeram, R.; Rao, S.V.S.N.; Rao, S.V. Protocol for Designing AuNP-Capped Ag Dendrites as Surface-Enhanced Raman Scattering Sensors for Trace Molecular Detection Protocol for Designing AuNP-Capped Ag Dendrites as Surface-Enhanced Raman Scattering Sensors for Trace Molecular Detection. STAR Protoc. 2023, 4, 102068. [Google Scholar] [CrossRef]
- He, L.; Rodda, T.; Haynes, C.L.; Deschaines, T.; Strother, T.; Diez-Gonzalez, F.; Labuza, T.P. Detection of a Foreign Protein in Milk Using Surface-Enhanced Raman Spectroscopy Coupled with Antibody-Modified Silver Dendrites. Anal. Chem. 2011, 83, 1510–1513. [Google Scholar] [CrossRef]
- Jebakumari, K.A.E.; Murugasenapathi, N.K. Engineered Two-Dimensional Nanostructures as SERS Substrates for Biomolecule Sensing: A Review. Biosensors 2023, 13, 102. [Google Scholar] [CrossRef]
- Bantz, K.C.; Meyer, A.F.; Wittenberg, N.J.; Im, H.; Kurtuluş, Ö.; Lee, S.H.; Lindquist, N.C.; Oh, S.H.; Haynes, C.L. Recent Progress in SERS Biosensing. Phys. Chem. Chem. Phys. 2011, 13, 11551–11567. [Google Scholar] [CrossRef] [PubMed]
- Vendamani, V.S.; Nageswara Rao, S.V.S.; Venugopal Rao, S.; Kanjilal, D.; Pathak, A.P. Three-Dimensional Hybrid Silicon Nanostructures for Surface Enhanced Raman Spectroscopy Based Molecular Detection. J. Appl. Phys. 2018, 123, 014301. [Google Scholar] [CrossRef] [Green Version]
- Huang, Z.; Zhang, A.; Zhang, Q.; Cui, D. Nanomaterial-Based SERS Sensing Technology for Biomedical Application. J. Mater. Chem. B 2019, 7, 3755–3774. [Google Scholar] [CrossRef]
- Szaniawska, A.; Kudelski, A. Applications of Surface-Enhanced Raman Scattering in Biochemical and Medical Analysis. Front. Chem. 2021, 9, 664134. [Google Scholar] [CrossRef]
- Chen, Y.; An, Q.; Teng, K.; Liu, C.; Sun, F.; Li, G. Application of SERS in In-Vitro Biomedical Detection. Chem. Asian J. 2022, 18, e202201194. [Google Scholar] [CrossRef]
- Hegde, M.; Pai, P.; Gangadhar Shetty, M.; Sundara Babitha, K. Gold Nanoparticle Based Biosensors for Rapid Pathogen Detection: A Review. Environ. Nanotechnol. Monit. Manag. 2022, 18, 100756. [Google Scholar] [CrossRef]
- Fleischmann, M.; Hendra, P.J.; McQuillan, A.J. Raman Spectra of Pyridine Adsorbed at a Silver Electrode. Chem. Phys. Lett. 1974, 26, 163–166. [Google Scholar] [CrossRef]
- Jeanmaire, D.L.; Duyne, R.P.V.A.N. Surface Raman spectroelectrochemistry: Part I. Heterocyclic, aromatic, and aliphatic amines adsorbed on the anodized silver electrode. J. Electroanal. Chem. Interfacial Electrochem. 1977, 84, 1–20. [Google Scholar] [CrossRef]
- Albrecht, M.G.; Creighton, J.A. Anomalously Intense Raman Spectra of Pyridine at a Silver Electrode. J. Am. Chem. Soc. 1977, 99, 5215–5217. [Google Scholar] [CrossRef]
- Pilot, R.; Signorini, R.; Durante, C.; Orian, L.; Bhamidipati, M.; Fabris, L. A Review on Surface-Enhanced Raman Scattering. Biosensors 2019, 9, 57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Le Ru, E.; Etchegoin, P. Principles of Surface Enhanced Raman Spectroscopy and Related Plasmonic Effects; Elseveir: Amsterdam, The Netherlands, 2008. [Google Scholar]
- Etchegoin, P.G.; Ru, E.C.L. Basic Electromagnetic Theory of SERS. In Surface Enhanced Raman Spectroscopy: Analytical, Biophysical and Life Science Applications; John Wiley & Sons: Hoboken, NJ, USA, 2011; pp. 1–37. ISBN 9783527325672. [Google Scholar]
- Le Ru, E.C.; Blackie, E.; Meyer, M.; Etchegoint, P.G. Surface Enhanced Raman Scattering Enhancement Factors: A Comprehensive Study. J. Phys. Chem. C 2007, 111, 13794–13803. [Google Scholar] [CrossRef]
- Sharma, B.; Frontiera, R.R.; Henry, A.I.; Ringe, E.; Van Duyne, R.P. SERS: Materials, Applications, and the Future. Mater. Today 2012, 15, 16–25. [Google Scholar] [CrossRef]
- Li, Q.; Huo, H.; Wu, Y.; Chen, L.; Su, L.; Zhang, X.; Song, J.; Yang, H. Design and Synthesis of SERS Materials for In Vivo Molecular Imaging and Biosensing. Adv. Sci. 2023, 2023, 2202051. [Google Scholar] [CrossRef]
- Israelsen, N.D.; Hanson, C.; Vargis, E. Nanoparticle Properties and Synthesis Effects on Surface-Enhanced Raman Scattering Enhancement Factor: An Introduction. Sci. World J. 2015, 2015, 124582. [Google Scholar] [CrossRef] [Green Version]
- Wang, A.X.; Kong, X. Review of Recent Progress of Plasmonic Materials and Nano-Structures for Surface-Enhanced Raman Scattering. Materials 2015, 8, 3024–3052. [Google Scholar] [CrossRef]
- Moram, S.S.B.; Byram, C.; Soma, V.R. Gold-Nanoparticle- and Nanostar-Loaded Paper-Based SERS Substrates for Sensing Nanogram-Level Picric Acid with a Portable Raman Spectrometer. Bull. Mater. Sci. 2020, 43, 53. [Google Scholar] [CrossRef]
- Zhang, Z.; Guan, R.; Li, J. Engineering Rational SERS Nanotags for Parallel Detection of Multiple Cancer Circulating Biomarkers. Chemosensors. 2023, 11, 110. [Google Scholar] [CrossRef]
- Pilot, R.; Massari, M. Silver Nanoparticle Aggregates: Wavelength Dependence of Their SERS Properties in the First Transparency Window of Biological Tissues. Chem. Phys. Impact 2021, 2, 100014. [Google Scholar] [CrossRef]
- Zhang, Y.; Mi, X.; Tan, X.; Xiang, R. Recent Progress on Liquid Biopsy Analysis Using Surface-Enhanced Raman Spectroscopy. Theranostics 2019, 9, 491–525. [Google Scholar] [CrossRef] [PubMed]
- Aitekenov, S.; Sultangaziyev, A.; Ilyas, A.; Dyussupova, A.; Boranova, A.; Gaipov, A.; Bukasov, R. Surface-Enhanced Raman Spectroscopy (SERS) for Protein Determination in Human Urine. Sens. Bio-Sens. Res. 2022, 38, 100535. [Google Scholar] [CrossRef]
- Akgönüllü, S.; Denizli, A. Recent Advances in Optical Biosensing Approaches for Biomarkers Detection. Biosens. Bioelectron. X 2022, 12, 100269. [Google Scholar] [CrossRef]
- Lussier, F.; Thibault, V.; Charron, B.; Wallace, G.Q.; Masson, J.F. Deep Learning and Artificial Intelligence Methods for Raman and Surface-Enhanced Raman Scattering. TrAC Trends Anal. Chem. 2020, 124, 115796. [Google Scholar] [CrossRef]
- Lin, X.; Lin, D.; Chen, Y.; Lin, J.; Weng, S.; Song, J.; Feng, S. High Throughput Blood Analysis Based on Deep Learning Algorithm and Self-Positioning Super-Hydrophobic SERS Platform for Non-Invasive Multi-Disease Screening. Adv. Funct. Mater. 2021, 31, 2103382. [Google Scholar] [CrossRef]
- Breuch, R.; Klein, D.; Siefke, E.; Hebel, M.; Herbert, U.; Wickleder, C.; Kaul, P. Differentiation of Meat-Related Microorganisms Using Paper-Based Surface-Enhanced Raman Spectroscopy Combined with Multivariate Statistical Analysis. Talanta 2020, 219, 121315. [Google Scholar] [CrossRef]
- Ilkhani, H.; Hughes, T.; Li, J.; Zhong, C.J.; Hepel, M. Nanostructured SERS-Electrochemical Biosensors for Testing of Anticancer Drug Interactions with DNA. Biosens. Bioelectron. 2016, 80, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Leong, S.X.; Leong, Y.X.; Tan, E.X.; Sim, H.Y.F.; Koh, C.S.L.; Lee, Y.H.; Chong, C.; Ng, L.S.; Chen, J.R.T.; Pang, D.W.C.; et al. Noninvasive and Point-of-Care Surface-Enhanced Raman Scattering (SERS)-Based Breathalyzer for Mass Screening of Coronavirus Disease 2019 (COVID-19) under 5 Min. ACS Nano 2022, 16, 2629–2639. [Google Scholar] [CrossRef]
- Bharati, M.S.S.; Soma, V.R. Flexible SERS Substrates for Hazardous Materials Detection: Recent Advances. Opto-Electron. Adv. 2021, 4, 210048. [Google Scholar] [CrossRef]
- Ali, A.; Nettey-Oppong, E.E.; Effah, E.; Yu, C.Y.; Muhammad, R.; Soomro, T.A.; Byun, K.M.; Choi, S.H. Miniaturized Raman Instruments for SERS-Based Point-of-Care Testing on Respiratory Viruses. Biosensors 2022, 12, 590. [Google Scholar] [CrossRef]
- Mejía-Salazar, J.R.; Oliveira, O.N. Plasmonic Biosensing. Chem. Rev. 2018, 118, 10617–10625. [Google Scholar] [CrossRef]
- Han, X.; Liu, K.; Sun, C. Plasmonics for Biosensing. Materials 2019, 12, 1411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shrivastav, A.M.; Cvelbar, U.; Abdulhalim, I. A Comprehensive Review on Plasmonic-Based Biosensors Used in Viral Diagnostics. Commun. Biol. 2021, 4, 70. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Ren, Z.H.; Zhao, W.M.; Wang, L.; Yan, X.; Zhu, A.S.; Qiu, F.M.; Zhang, K.K. Research Advances on Surface Plasmon Resonance Biosensors. Nanoscale 2022, 14, 564–591. [Google Scholar] [CrossRef] [PubMed]
- Homola, J. Present and Future of Surface Plasmon Resonance Biosensors. Anal. Bioanal. Chem. 2003, 377, 528–539. [Google Scholar] [CrossRef]
- Piliarik, M.; Vaisocherová, H.; Homola, J. Surface Plasmon Resonance Biosensing. In Biosensors and Biodetection; Humana Press: Totowa, NJ, USA, 2009; pp. 65–88. [Google Scholar] [CrossRef]
- Hong, Y.; Huh, Y.M.; Yoon, D.S.; Yang, J. Nanobiosensors Based on Localized Surface Plasmon Resonance for Biomarker Detection. J. Nanomater. 2012, 2012, 759830. [Google Scholar] [CrossRef] [Green Version]
- Unser, S.; Bruzas, I.; He, J.; Sagle, L. Localized Surface Plasmon Resonance Biosensing: Current Challenges and Approaches. Sensors 2015, 15, 15684–15716. [Google Scholar] [CrossRef]
- Brolo, A.G. Plasmonics for Future Biosensors. Nat. Photonics 2012, 6, 709–713. [Google Scholar] [CrossRef]
- Liu, J.; Jalali, M.; Mahshid, S.; Wachsmann-Hogiu, S. Are Plasmonic Optical Biosensors Ready for Use in Point-of-Need Applications? Analyst 2020, 145, 364–384. [Google Scholar] [CrossRef] [Green Version]
- Moore, T.J.; Moody, A.S.; Payne, T.D.; Sarabia, G.M.; Daniel, A.R.; Sharma, B. In Vitro and in Vivo Sers Biosensing for Disease Diagnosis. Biosensors 2018, 8, 46. [Google Scholar] [CrossRef] [Green Version]
- Alvarez-Puebla, R.A.; Liz-Marzán, L.M. SERS-Based Diagnosis and Biodetection. Small 2010, 6, 604–610. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Liu, S.; Song, X.; Wang, H.; Wang, J.; Wang, Y.; Huang, J.; Yu, J. Robust and Universal SERS Sensing Platform for Multiplexed Detection of Alzheimer’s Disease Core Biomarkers Using PAapt-AuNPs Conjugates. ACS Sens. 2019, 4, 2140–2149. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Cho, S.; Kim, M.; Jung, Y.S. Carboxylic Acid-Functionalized, Graphitic Layer-Coated Three-Dimensional SERS Substrate for Label-Free Analysis of Alzheimer’s Disease Biomarkers. Nano Lett. 2020, 20, 2576–2584. [Google Scholar] [CrossRef]
- Dang, H.; Joung, Y.; Jeong, C.; Jeon, C.S.; Pyun, S.H.; Park, S.-G.; Choo, J. Nanoplasmonic Assay Platforms for Reproducible SERS Detection of Alzheimer’s Disease Biomarker. Bull. Korean Chem. Soc. 2023, 2023, 1. [Google Scholar] [CrossRef]
- Momenpour, A.; Lima, P.D.A.; Chen, Y.-A.; Tzeng, C.-R.; Tsang, B.K.; Anis, H. Surface-Enhanced Raman Scattering for the Detection of Polycystic Ovary Syndrome. Biomed. Opt. Express 2018, 9, 801. [Google Scholar] [CrossRef] [PubMed]
- Lyandres, O.; Shah, N.C.; Yonzon, C.R.; Walsh, J.T.; Glucksberg, M.R.; Van Duyne, R.P. Real-Time Glucose Sensing by Surface-Enhanced Raman Spectroscopy in Bovine Plasma Facilitated by a Mixed Decanethiol/Mercaptohexanol Partition Layer. Anal. Chem. 2005, 77, 6134–6139. [Google Scholar] [CrossRef]
- Qi, G.; Jia, K.; Fu, C.; Xu, S.; Xu, W. A Highly Sensitive SERS Sensor for Quantitative Analysis of Glucose Based on the Chemical Etching of Silver Nanoparticles. J. Opt. 2015, 17, 114020. [Google Scholar] [CrossRef]
- Rong, Z.; Xiao, R.; Xing, S.; Xiong, G.; Yu, Z.; Wang, L.; Jia, X.; Wang, K.; Cong, Y.; Wang, S. SERS-Based Lateral Flow Assay for Quantitative Detection of C-Reactive Protein as an Early Bio-Indicator of a Radiation-Induced Inflammatory Response in Nonhuman Primates. Analyst 2018, 143, 2115–2121. [Google Scholar] [CrossRef]
- Li, B.; Wu, Y.; Wang, Z.; Xing, M.; Xu, W.; Zhu, Y.; Du, P.; Wang, X.; Yang, H. Non-Invasive Diagnosis of Crohn’s Disease Based on SERS Combined with PCA-SVM. Anal. Methods 2021, 13, 5264–5273. [Google Scholar] [CrossRef]
- Xu, H.; Bjerneld, E.J.; Käll, M.; Börjesson, L. Spectroscopy of Single Hemoglobin Molecules by Surface Enhanced Raman Scattering. Phys. Rev. Lett. 1999, 83, 4357–4360. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization Report. Available online: https://www.who.int/news-room/fact-sheets/detail/cancer (accessed on 30 January 2023).
- Blanco-Formoso, M.; Alvarez-Puebla, R.A. Cancer Diagnosis through Sers and Other Related Techniques. Int. J. Mol. Sci. 2020, 21, 2253. [Google Scholar] [CrossRef] [Green Version]
- Guerrini, L.; Pazos-Perez, N.; Garcia-Rico, E.; Alvarez-Puebla, R. Cancer Characterization and Diagnosis with SERS-Encoded Particles. Cancer Nanotechnol. 2017, 8, 5. [Google Scholar] [CrossRef]
- Kaur, B.; Kumar, S.; Kaushik, B.K. Recent Advancements in Optical Biosensors for Cancer Detection. Biosens. Bioelectron. 2022, 197, 113805. [Google Scholar] [CrossRef]
- Thenrajan, T.; Wilson, J. Biosensors for Cancer Theranostics. Biosens. Bioelectron. X 2022, 12, 100232. [Google Scholar] [CrossRef]
- Falkowski, P.; Lukaszewski, Z.; Gorodkiewicz, E. Potential of Surface Plasmon Resonance Biosensors in Cancer Detection. J. Pharm. Biomed. Anal. 2021, 194, 113802. [Google Scholar] [CrossRef]
- Fu, Q.; Zhang, X.; Song, J.; Yang, H. Plasmonic Gold Nanoagents for Cancer Imaging and Therapy. View 2021, 2, 20200149. [Google Scholar] [CrossRef]
- Azzouz, A.; Hejji, L.; Kim, K.H.; Kukkar, D.; Souhail, B.; Bhardwaj, N.; Brown, R.J.C.; Zhang, W. Advances in Surface Plasmon Resonance–Based Biosensor Technologies for Cancer Biomarker Detection. Biosens. Bioelectron. 2022, 197, 113767. [Google Scholar] [CrossRef] [PubMed]
- Sugumaran, S.; Jamlos, M.F.; Ahmad, M.N.; Bellan, C.S.; Schreurs, D. Nanostructured Materials with Plasmonic Nanobiosensors for Early Cancer Detection: A Past and Future Prospect. Biosens. Bioelectron. 2018, 100, 361–373. [Google Scholar] [CrossRef] [PubMed]
- Fattahi, Z.; Khosroushahi, A.Y.; Hasanzadeh, M. Recent Progress on Developing of Plasmon Biosensing of Tumor Biomarkers: Efficient Method towards Early Stage Recognition of Cancer. Biomed. Pharmacother. 2020, 132, 110850. [Google Scholar] [CrossRef] [PubMed]
- Bellassai, N.; D’Agata, R.; Jungbluth, V.; Spoto, G. Surface Plasmon Resonance for Biomarker Detection: Advances in Non-Invasive Cancer Diagnosis. Front. Chem. 2019, 7, 570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Usman, F.; Dennis, J.O.; Aljameel, A.I.; Ali, M.K.M.; Aldaghri, O.; Ibnaouf, K.H.; Zango, Z.U.; Beygisangchin, M.; Alsadig, A.; Meriaudeau, F. Plasmonic Biosensors for the Detection of Lung Cancer Biomarkers: A Review. Chemosensors 2021, 9, 326. [Google Scholar] [CrossRef]
- Yin, B.; Ho, W.K.H.; Xia, X.; Chan, C.K.W.; Zhang, Q.; Ng, Y.M.; Lam, C.Y.K.; Cheung, J.C.W.; Wang, J.; Yang, M.; et al. A Multilayered Mesoporous Gold Nanoarchitecture for Ultraeffective Near-Infrared Light-Controlled Chemo/Photothermal Therapy for Cancer Guided by SERS Imaging. Small 2023, 2023, 2206762. [Google Scholar] [CrossRef] [PubMed]
- Constantinou, M.; Hadjigeorgiou, K.; Abalde-cela, S.; Andreou, C. Label-Free Sensing with Metal Nanostructure-Based Surface- Enhanced Raman Spectroscopy for Cancer Diagnosis. ACS Appl. Nano Mater. 2022, 5, 12276–12299. [Google Scholar] [CrossRef] [PubMed]
- Rajput, S.; Pink, D.; Findlay, S.; Woolner, E.; Lewis, J.D.; McDermott, M.T. Application of Surface-Enhanced Raman Spectroscopy to Guide Therapy for Advanced Prostate Cancer Patients. ACS Sens. 2022, 7, 827–838. [Google Scholar] [CrossRef]
- Avci, E.; Yilmaz, H.; Sahiner, N.; Tuna, B.G.; Cicekdal, M.B.; Eser, M.; Basak, K.; Altıntoprak, F.; Zengin, I.; Dogan, S.; et al. Label-Free Surface Enhanced Raman Spectroscopy for Cancer Detection. Cancers 2022, 14, 5021. [Google Scholar] [CrossRef]
- Guerrini, L.; Alvarez-puebla, R.A. Surface-Enhanced Raman Spectroscopy in Cancer Diagnosis, Prognosis and Monitoring. Cancers 2019, 11, 748. [Google Scholar] [CrossRef] [Green Version]
- Pollap, A.; Paweł, S. Recent Advances in Sandwich SERS Immunosensors for Cancer Detection. Int. J. Mol. Sci. 2022, 23, 4740. [Google Scholar] [CrossRef]
- Vendrell, M.; Maiti, K.K.; Dhaliwal, K.; Chang, Y.T. Surface-Enhanced Raman Scattering in Cancer Detection and Imaging. Trends Biotechnol. 2013, 31, 249–257. [Google Scholar] [CrossRef]
- Moisoiu, V.; Iancu, S.D.; Stefancu, A.; Moisoiu, T.; Pardini, B.; Dragomir, M.P.; Crisan, N.; Avram, L.; Crisan, D.; Andras, I.; et al. SERS Liquid Biopsy: An Emerging Tool for Medical Diagnosis. Colloids Surfaces B Biointerfaces 2021, 208, 112064. [Google Scholar] [CrossRef]
- Shanmugasundaram, K.B.; Li, J.; Sina, A.I.; Wuethrich, A.; Trau, M. Toward Precision Oncology: SERS Microfluidic Systems for Multiplex Biomarker Analysis in Liquid Biopsy. Mater. Adv. 2022, 3, 1459–1471. [Google Scholar] [CrossRef]
- Song, C.Y.; Yang, Y.J.; Yang, B.Y.; Sun, Y.Z.; Zhao, Y.P.; Wang, L.H. An Ultrasensitive SERS Sensor for Simultaneous Detection of Multiple Cancer-Related MiRNAs. Nanoscale 2016, 8, 17365–17373. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.N.; Crawford, B.M.; Norton, S.J.; Vo-Dinh, T. Direct and Label-Free Detection of MicroRNA Cancer Biomarkers Using SERS-Based Plasmonic Coupling Interference (PCI) Nanoprobes. J. Phys. Chem. B 2019, 123, 10245–10251. [Google Scholar] [CrossRef]
- Guerrini, L.; Garcia-Rico, E.; O’loghlen, A.; Giannini, V.; Alvarez-Puebla, R.A. Surface-Enhanced Raman Scattering (Sers) Spectroscopy for Sensing and Characterization of Exosomes in Cancer Diagnosis. Cancers. 2021, 13, 2179. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.U.; Kim, S.; Sim, S.J. SERS-Based Nanoplasmonic Exosome Analysis: Enabling Liquid Biopsy for Cancer Diagnosis and Monitoring Progression. BioChip J. 2020, 14, 231–241. [Google Scholar] [CrossRef]
- Vo-Dinh, T.; Allain, L.R.; Stokes, D.L. Cancer Gene Detection Using Surface-Enhanced Raman Scattering (SERS). J. Raman Spectrosc. 2002, 33, 511–516. [Google Scholar] [CrossRef]
- Zhu, D.; Li, A.; Di, Y.; Wang, Z.; Shi, J.; Ni, X.; Wang, Y. Interference-Free SERS Nanoprobes for Labeling and Imaging of MT1-MMP in Breast Cancer Cells. Nanotechnology 2022, 33, 115702. [Google Scholar] [CrossRef]
- Lin, J.; Chen, R.; Feng, S.; Pan, J.; Li, Y.; Chen, G.; Cheng, M.; Huang, Z.; Yu, Y.; Zeng, H. A Novel Blood Plasma Analysis Technique Combining Membrane Electrophoresis with Silver Nanoparticle-Based SERS Spectroscopy for Potential Applications in Noninvasive Cancer Detection. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 655–663. [Google Scholar] [CrossRef]
- Fabris, L. SERS Tags: The Next Promising Tool for Personalized Cancer Detection? ChemNanoMat 2016, 2, 249–258. [Google Scholar] [CrossRef]
- Davis, R.M.; Campbell, J.L.; Burkitt, S.; Qiu, Z.; Kang, S.; Mehraein, M.; Miyasato, D.; Salinas, H.; Liu, J.T.C.; Zavaleta, C. A Raman Imaging Approach Using CD47 Antibody-Labeled SERS Nanoparticles for Identifying Breast Cancer and Its Potential to Guide Surgical Resection. Nanomaterials 2018, 8, 953. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Wang, Z.; Zong, S.; Song, C.; Zhang, R.; Cui, Y. Distinguishing Breast Cancer Cells Using Surface-Enhanced Raman Scattering. Anal. Bioanal. Chem. 2012, 402, 1093–1100. [Google Scholar] [CrossRef]
- Dinish, U.S.; Balasundaram, G.; Chang, Y.T.; Olivo, M. Actively Targeted in Vivo Multiplex Detection of Intrinsic Cancer Biomarkers Using Biocompatible SERS Nanotags. Sci. Rep. 2014, 4, 4075. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, H.; Gao, X.; Xu, C.; Liu, D. SERS Tags for Biomedical Detection and Bioimaging. Theranostics 2022, 12, 1870–1903. [Google Scholar] [CrossRef] [PubMed]
- Report on Lung Cancer. Available online: https://www.chestnet.org/newsroom/chest-news/2020/07/world-lung-cancer-day-2020-fact-sheet (accessed on 30 January 2023).
- Mao, Y.; Sun, Y.; Xue, J.; Lu, W.; Cao, X. Ultra-Sensitive and High Efficiency Detection of Multiple Non-Small Cell Lung Cancer-Related MiRNAs on a Single Test Line in Catalytic Hairpin Assembly-Based SERS-LFA Strip. Anal. Chim. Acta 2021, 1178, 338800. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Liu, Y.; Ran, M.; Lu, D.; Cao, X.; Wang, Y. SERS Platform Based on Bimetallic Au-Ag Nanowires-Decorated Filter Paper for Rapid Detection of MiR-196ain Lung Cancer Patients Serum. J. Chem. 2020, 2020, 5073451. [Google Scholar] [CrossRef]
- Cao, X.; Sun, Y.; Mao, Y.; Ran, M.; Liu, Y.; Lu, D.; Bi, C. Rapid and Sensitive Detection of Dual Lung Cancer-Associated MiRNA Biomarkers by a Novel SERS-LFA Strip Coupling with Catalytic Hairpin Assembly Signal Amplification. J. Mater. Chem. C 2021, 9, 3661–3671. [Google Scholar] [CrossRef]
- Cao, X.; Mao, Y.; Gu, Y.; Ge, S.; Lu, W.; Gu, Y.; Li, Z. Highly Sensitive and Simultaneous Detection of CtDNAs Related to Non-Small Cell Lung Cancer in Serum Using a Catalytic Hairpin Assembly Strategy in a SERS Microfluidic Chip. J. Mater. Chem. B 2022, 10, 6194–6206. [Google Scholar] [CrossRef]
- Cao, X.; Ge, S.; Zhou, X.; Mao, Y.; Sun, Y.; Lu, W.; Ran, M. A Dual-Signal Amplification Strategy Based on Pump-Free SERS Microfluidic Chip for Rapid and Ultrasensitive Detection of Non-Small Cell Lung Cancer-Related Circulating Tumour DNA in Mice Serum. Biosens. Bioelectron. 2022, 205, 114110. [Google Scholar] [CrossRef]
- Ye, L.P.; Hu, J.; Liang, L.; Zhang, C.Y. Surface-Enhanced Raman Spectroscopy for Simultaneous Sensitive Detection of Multiple MicroRNAs in Lung Cancer Cells. Chem. Commun. 2014, 50, 11883–11886. [Google Scholar] [CrossRef]
- Guo, T.; Li, W.; Qian, L.; Yan, X.; Cui, D.; Zhao, J.; Ni, H.; Zhao, X.; Zhang, Z.; Li, X.; et al. Highly-Selective Detection of EGFR Mutation Gene in Lung Cancer Based on Surface Enhanced Raman Spectroscopy and Asymmetric PCR. J. Pharm. Biomed. Anal. 2020, 190, 113522. [Google Scholar] [CrossRef]
- Shin, H.; Oh, S.; Hong, S.; Kang, M.; Kang, D.; Ji, Y.G.; Choi, B.H.; Kang, K.W.; Jeong, H.; Park, Y.; et al. Early-Stage Lung Cancer Diagnosis by Deep Learning-Based Spectroscopic Analysis of Circulating Exosomes. ACS Nano 2020, 14, 5435–5444. [Google Scholar] [CrossRef]
- Lei, J.; Yang, D.; Li, R.; Dai, Z.X.; Zhang, C.; Yu, Z.; Wu, S.; Pang, L.; Liang, S.; Zhang, Y. Label-Free Surface-Enhanced Raman Spectroscopy for Diagnosis and Analysis of Serum Samples with Different Types Lung Cancer. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 261, 120021. [Google Scholar] [CrossRef]
- Wang, Z.; Hong, Y.; Yan, H.; Luo, H.; Zhang, Y.; Li, L.; Lu, S.; Chen, Y.; Wang, D.; Su, Y.; et al. Fabrication of Optoplasmonic Particles through Electroless Deposition and the Application in SERS-Based Screening of Nodule-Involved Lung Cancer. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 279, 121483. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Dong, Y.; Zhu, W.; Xie, D.; Zhao, Y.; Yang, D.; Li, M. Ultrasensitive Detection of Circulating Tumor DNA of Lung Cancer via an Enzymatically Amplified SERS-Based Frequency Shift Assay. ACS Appl. Mater. Interfaces 2019, 11, 18145–18152. [Google Scholar] [CrossRef] [PubMed]
- Jonak, S.T.; Liu, Z.; Liu, J.; Li, T.; D’Souza, B.V.; Schiaffino, A.; Oh, S.; Xie, Y.-H. Analyzing Bronchoalveolar Fluid Derived Small Extracellular Vesicles Using Single-Vesicle SERS for Non-Small Cell Lung Cancer Detection. Sens. Diagn. 2023, 2, 90–99. [Google Scholar] [CrossRef]
- Park, J.; Hwang, M.; Choi, B.; Jeong, H.; Jung, J.H.; Kim, H.K.; Hong, S.; Park, J.H.; Choi, Y. Exosome Classification by Pattern Analysis of Surface-Enhanced Raman Spectroscopy Data for Lung Cancer Diagnosis. Anal. Chem. 2017, 89, 6695–6701. [Google Scholar] [CrossRef]
- Wen, H.; Wang, H.; Hai, J.; He, S.; Chen, F.; Wang, B. Photochemical Synthesis of Porous CuFeSe 2 /Au Heterostructured Nanospheres as SERS Sensor for Ultrasensitive Detection of Lung Cancer Cells and Their Biomarkers. ACS Sustain. Chem. Eng. 2019, 7, 5200–5208. [Google Scholar] [CrossRef]
- Qiao, X.; Su, B.; Liu, C.; Song, Q.; Luo, D.; Mo, G.; Wang, T. Selective Surface Enhanced Raman Scattering for Quantitative Detection of Lung Cancer Biomarkers in Superparticle@MOF Structure. Adv. Mater. 2018, 30, 1702275. [Google Scholar] [CrossRef]
- Perumal, J.; Lee, P.; Dev, K.; Lim, H.Q.; Dinish, U.S.; Olivo, M. Machine Learning Assisted Real-Time Label-Free SERS Diagnoses of Malignant Pleural Effusion Due to Lung Cancer. Biosensors 2022, 12, 940. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Liu, X.; Man, B.; Yang, C.; Zhang, C.; Liu, M.; Zhang, Y.; Liu, L.; Chen, C. Label-Free and Stable Serum Analysis Based on Ag-NPs/PSi Surface-Enhanced Raman Scattering for Noninvasive Lung Cancer Detection. Biomed. Opt. Express 2018, 9, 4345. [Google Scholar] [CrossRef]
- Zhang, K.; Hao, C.; Huo, Y.; Man, B.; Zhang, C.; Yang, C.; Liu, M.; Chen, C. Label-Free Diagnosis of Lung Cancer with Tissue-Slice Surface-Enhanced Raman Spectroscopy and Statistical Analysis. Lasers Med. Sci. 2019, 34, 1849–1855. [Google Scholar] [CrossRef]
- Chon, H.; Lee, S.; Yoon, S.Y.; Chang, S.I.; Lim, D.W.; Choo, J. Simultaneous Immunoassay for the Detection of Two Lung Cancer Markers Using Functionalized SERS Nanoprobes. Chem. Commun. 2011, 47, 12515–12517. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Gao, Y.; Lu, Y.; Zhang, H.; Cai, C. High Specific Detection and Near-Infrared Photothermal Therapy of Lung Cancer Cells with High SERS Active Aptamer-Silver-Gold Shell-Core Nanostructures. Analyst 2013, 138, 6501–6510. [Google Scholar] [CrossRef]
- Chen, Y.W.; Liu, T.Y.; Chen, P.J.; Chang, P.H.; Chen, S.Y. A High-Sensitivity and Low-Power Theranostic Nanosystem for Cell SERS Imaging and Selectively Photothermal Therapy Using Anti-EGFR-Conjugated Reduced Graphene Oxide/Mesoporous Silica/AuNPs Nanosheets. Small 2016, 12, 1458–1468. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ye, X.; Xu, G.; Jin, X.; Luan, M.; Lou, J.; Wang, L.; Huang, C.; Ye, J. Identification and Distinction of Non-Small-Cell Lung Cancer Cells by Intracellular SERS Nanoprobes. RSC Adv. 2016, 6, 5401–5407. [Google Scholar] [CrossRef]
- Huang, Y.; Xie, T.; Zou, K.; Gu, Y.; Yang, G.; Zhang, F.; Qu, L.L.; Yang, S. Ultrasensitive SERS Detection of Exhaled Biomarkers of Lung Cancer Using a Multifunctional Solid Phase Extraction Membrane. Nanoscale 2021, 13, 13344–13352. [Google Scholar] [CrossRef]
- Cai, C.; Liu, Y.; Li, J.; Wang, L.; Zhang, K. Serum Fingerprinting by Slippery Liquid-Infused Porous SERS for Non-Invasive Lung Cancer Detection. Analyst 2022, 147, 4426–4432. [Google Scholar] [CrossRef]
- Sivashanmugan, K.; Huang, W.L.; Lin, C.H.; Liao, J.D.; Lin, C.C.; Su, W.C.; Wen, T.C. Bimetallic Nanoplasmonic Gap-Mode SERS Substrate for Lung Normal and Cancer-Derived Exosomes Detection. J. Taiwan Inst. Chem. Eng. 2017, 80, 149–155. [Google Scholar] [CrossRef]
- Qian, K.; Wang, Y.; Hua, L.; Chen, A.; Zhang, Y. New Method of Lung Cancer Detection by Saliva Test Using Surface-Enhanced Raman Spectroscopy. Thorac. Cancer 2018, 9, 1556–1561. [Google Scholar] [CrossRef]
- Yang, T.; Guo, X.; Wu, Y.; Wang, H.; Fu, S.; Wen, Y.; Yang, H. Facile and Label-Free Detection of Lung Cancer Biomarker in Urine by Magnetically Assisted Surface-Enhanced Raman Scattering. ACS Appl. Mater. Interfaces 2014, 6, 20985–20993. [Google Scholar] [CrossRef]
- Breast Cancer Report. Available online: https://www.cancer.org/cancer/breast-cancer/about/how-common-is-breast-cancer.html (accessed on 30 January 2023).
- Moisoiu, T.; Iancu, S.D.; Burghelea, D.; Dragomir, M.P.; Iacob, G.; Stefancu, A.; Cozan, R.G.; Antal, O.; Bálint, Z.; Muntean, V.; et al. SERS Liquid Biopsy Profiling of Serum for the Diagnosis of Kidney Cancer. Biomedicines 2022, 10, 233. [Google Scholar] [CrossRef]
- Kim, S.; Kim, T.G.; Lee, S.H.; Kim, W.; Bang, A.; Moon, S.W.; Song, J.; Shin, J.H.; Yu, J.S.; Choi, S. Label-Free Surface-Enhanced Raman Spectroscopy Biosensor for On-Site Breast Cancer Detection Using Human Tears. ACS Appl. Mater. Interfaces 2020, 12, 7897–7904. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, R.A.R.; Lataliza, A.A.B.; Raposo, N.R.B.; Costa, L.A.S.; Sant’Ana, A.C. Insights on the Transport of Tamoxifen by Gold Nanoparticles for MCF-7 Breast Cancer Cells Based on SERS Spectroscopy. Colloids Surf. B Biointerfaces 2018, 170, 712–717. [Google Scholar] [CrossRef] [PubMed]
- Nargis, H.F.; Nawaz, H.; Bhatti, H.N.; Jilani, K.; Saleem, M. Comparison of Surface Enhanced Raman Spectroscopy and Raman Spectroscopy for the Detection of Breast Cancer Based on Serum Samples. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 246, 119034. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Su, X.; Wen, Y.; Zheng, C.; Li, M. Artificial Intelligent Label-Free SERS Profiling of Serum Exosomes for Breast Cancer Diagnosis and Postoperative Assessment. Nano Lett. 2022, 22, 7910–7918. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Truong, V.X.; Li, M. Real-Time Intraoperative Surface-Enhanced Raman Spectroscopy-Guided Thermosurgical Eradication of Residual Microtumors in Orthotopic Breast Cancer. Nano Lett. 2021, 21, 3066–3074. [Google Scholar] [CrossRef]
- Xiao, L.; Harihar, S.; Welch, D.R.; Zhou, A. Imaging of Epidermal Growth Factor Receptor on Single Breast Cancer Cells Using Surface-Enhanced Raman Spectroscopy. Anal. Chim. Acta 2014, 843, 73–82. [Google Scholar] [CrossRef] [Green Version]
- Liang, L.; Shen, Y.; Zhang, J.; Xu, S.; Xu, W.; Liang, C.; Han, B. Identification of Breast Cancer through Spectroscopic Analysis of Cell-Membrane Sialic Acid Expression. Anal. Chim. Acta 2018, 1033, 148–155. [Google Scholar] [CrossRef]
- Hernández-Arteaga, A.; de Jesús Zermeño Nava, J.; Kolosovas-Machuca, E.S.; Velázquez-Salazar, J.J.; Vinogradova, E.; José-Yacamán, M.; Navarro-Contreras, H.R. Diagnosis of Breast Cancer by Analysis of Sialic Acid Concentrations in Human Saliva by Surface-Enhanced Raman Spectroscopy of Silver Nanoparticles. Nano Res. 2017, 10, 3662–3670. [Google Scholar] [CrossRef]
- Han, Y.; Qiang, L.; Gao, Y.; Gao, J.; He, Q.; Liu, H.; Han, L.; Zhang, Y. Large-Area Surface-Enhanced Raman Spectroscopy Substrate by Hybrid Porous GaN with Au/Ag for Breast Cancer MiRNA Detection. Appl. Surf. Sci. 2021, 541, 148456. [Google Scholar] [CrossRef]
- Yarbakht, M.; Nikkhah, M.; Moshaii, A.; Weber, K.; Matthäus, C.; Cialla-May, D.; Popp, J. Simultaneous Isolation and Detection of Single Breast Cancer Cells Using Surface-Enhanced Raman Spectroscopy. Talanta 2018, 186, 44–52. [Google Scholar] [CrossRef]
- Zheng, Z.; Wu, L.; Li, L.; Zong, S.; Wang, Z.; Cui, Y. Simultaneous and Highly Sensitive Detection of Multiple Breast Cancer Biomarkers in Real Samples Using a SERS Microfluidic Chip. Talanta 2018, 188, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Hameed, M.K.; Parambath, J.B.M.; Gul, M.T.; Khan, A.A.; Park, Y.; Han, C.; Mohamed, A.A. Arylated Gold Nanostars Aided SERS Study of Breast Cancer Cells. Appl. Surf. Sci. 2022, 583, 152504. [Google Scholar] [CrossRef]
- Kapara, A.; Brunton, V.G.; Graham, D.; Faulds, K. Characterisation of Estrogen Receptor Alpha (ERα) Expression in Breast Cancer Cells and Effect of Drug Treatment Using Targeted Nanoparticles and SERS. Analyst 2020, 145, 7225–7233. [Google Scholar] [CrossRef] [PubMed]
- Kapara, A.; Brunton, V.; Graham, D.; Faulds, K. Investigation of Cellular Uptake Mechanism of Functionalised Gold Nanoparticles into Breast Cancer Using SERS. Chem. Sci. 2020, 11, 5819–5829. [Google Scholar] [CrossRef]
- Lee, S.; Chon, H.; Lee, J.; Ko, J.; Chung, B.H.; Lim, D.W.; Choo, J. Rapid and Sensitive Phenotypic Marker Detection on Breast Cancer Cells Using Surface-Enhanced Raman Scattering (SERS) Imaging. Biosens. Bioelectron. 2014, 51, 238–243. [Google Scholar] [CrossRef]
- Choi, N.; Dang, H.; Das, A.; Sim, M.S.; Chung, I.Y.; Choo, J. SERS Biosensors for Ultrasensitive Detection of Multiple Biomarkers Expressed in Cancer Cells. Biosens. Bioelectron. 2020, 164, 112326. [Google Scholar] [CrossRef]
- Meng, S.; Chen, R.; Xie, J.; Li, J.; Cheng, J.; Xu, Y.; Cao, H.; Wu, X.; Zhang, Q.; Wang, H. Surface-Enhanced Raman Scattering Holography Chip for Rapid, Sensitive and Multiplexed Detection of Human Breast Cancer-Associated MicroRNAs in Clinical Samples. Biosens. Bioelectron. 2021, 190, 113470. [Google Scholar] [CrossRef]
- Weng, S.; Lin, D.; Lai, S.; Tao, H.; Chen, T.; Peng, M.; Qiu, S.; Feng, S. Highly Sensitive and Reliable Detection of MicroRNA for Clinically Disease Surveillance Using SERS Biosensor Integrated with Catalytic Hairpin Assembly Amplification Technology. Biosens. Bioelectron. 2022, 208, 114236. [Google Scholar] [CrossRef]
- Li, Y.; Qi, X.; Lei, C.; Qifeng, Q.; Zhang, S. Simultaneous SERS Detection and Imaging of Two Biomarkers on the Cancer Cell Surface by Self-Assembly of Branched DNA-Gold Nanoaggregates. Chem. Commun. 2014, 50, 9907–9909. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.U.; Kim, W.H.; Lee, H.S.; Park, K.H.; Sim, S.J. Quantitative and Specific Detection of Exosomal MiRNAs for Accurate Diagnosis of Breast Cancer Using a Surface-Enhanced Raman Scattering Sensor Based on Plasmonic Head-Flocked Gold Nanopillars. Small 2019, 15, 1804968. [Google Scholar] [CrossRef]
- Zhong, Q.; Zhang, K.; Huang, X.; Lu, Y.; Zhao, J.; He, Y.; Liu, B. In Situ Ratiometric SERS Imaging of Intracellular Protease Activity for Subtype Discrimination of Human Breast Cancer. Biosens. Bioelectron. 2022, 207, 114194. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Liao, M.; Chen, Y.; Shan, B.; Li, M. Surface-Enhanced Raman Spectroscopy (SERS) Nanoprobes for Ratiometric Detection of Cancer Cells. J. Mater. Chem. B 2019, 7, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Kang, S.; Khan, A.; Ruttner, G.; Leigh, S.Y.; Murray, M.; Abeytunge, S.; Peterson, G.; Rajadhyaksha, M.; Dintzis, S.; et al. Quantitative Molecular Phenotyping with Topically Applied SERS Nanoparticles for Intraoperative Guidance of Breast Cancer Lumpectomy. Sci. Rep. 2016, 6, 21242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Q.; Ma, R.; Zhang, Y.; Zhao, J.; Wang, Y.; Xu, Z. Dual-Aptamer-Assisted Ratiometric SERS Biosensor for Ultrasensitive and Precise Identification of Breast Cancer Exosomes. ACS Sens. 2023. [Google Scholar] [CrossRef]
- Shen, L.S.N.; Du, Y.; Wei, N.; Li, Q.; Li, S.M.; Sun, T.M.; Xu, S.; Wang, H.; Man, X.X.; Han, B. SERS Studies on Normal Epithelial and Cancer Cells Derived from Clinical Breast Cancer Specimens. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 237, 118364. [Google Scholar] [CrossRef]
- Lin, Y.; Gao, S.; Zheng, M.; Tang, S.; Lin, K.; Xie, S.; Yu, Y.; Lin, J. A Microsphere Nanoparticle Based-Serum Albumin Targeted Adsorption Coupled with Surface-Enhanced Raman Scattering for Breast Cancer Detection. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 261, 120039. [Google Scholar] [CrossRef]
- Lin, Y.; Gao, J.; Tang, S.; Zhao, X.; Zheng, M.; Gong, W.; Xie, S.; Gao, S.; Yu, Y.; Lin, J. Label-Free Diagnosis of Breast Cancer Based on Serum Protein Purification Assisted Surface-Enhanced Raman Spectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 263, 120234. [Google Scholar] [CrossRef]
- Moisoiu, V.; Socaciu, A.; Stefancu, A.; Iancu, S.D.; Boros, I.; Alecsa, C.D.; Rachieriu, C.; Chiorean, A.R.; Eniu, D.; Leopold, N.; et al. Breast Cancer Diagnosis by Surface-Enhanced Raman Scattering (SERS) of Urine. Appl. Sci. 2019, 9, 806. [Google Scholar] [CrossRef] [Green Version]
- Akbar, S.; Majeed, M.I.; Nawaz, H.; Rashid, N.; Tariq, A.; Hameed, W.; Shakeel, S.; Dastgir, G.; Bari, R.Z.A.; Iqbal, M.; et al. Surface-Enhanced Raman Spectroscopic (SERS) Characterization of Low Molecular Weight Fraction of the Serum of Breast Cancer Patients with Principal Component Analysis (PCA) and Partial Least Square-Discriminant Analysis (PLS-DA). Anal. Lett. 2022, 55, 1588–1604. [Google Scholar] [CrossRef]
- Feng, S.; Huang, S.; Lin, D.; Chen, G.; Xu, Y.; Li, Y.; Huang, Z.; Pan, J.; Chen, R.; Zeng, H. Surface-Enhanced Raman Spectroscopy of Saliva Proteins for the Noninvasive Differentiation of Benign and Malignant Breast Tumors. Int. J. Nanomed. 2015, 10, 537–547. [Google Scholar] [CrossRef] [Green Version]
- Iancu, S.D.; Cozan, R.G.; Stefancu, A.; David, M.; Moisoiu, T.; Moroz-Dubenco, C.; Bajcsi, A.; Chira, C.; Andreica, A.; Leopold, L.F.; et al. SERS Liquid Biopsy in Breast Cancer. What Can We Learn from SERS on Serum and Urine? Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 273, 120992. [Google Scholar] [CrossRef]
- Lin, X.; Jia, X.; Lin, J.Y.; Wu, P.H.; Weng, Y.; Feng, S. A Comparative Study Based on Serum SERS Spectra in and on the Coffee Ring for High Precision Breast Cancer Detection. J. Raman Spectrosc. 2022, 53, 1371–1379. [Google Scholar] [CrossRef]
- Știufiuc, G.F.; Toma, V.; Buse, M.; Mărginean, R.; Morar-Bolba, G.; Culic, B.; Tetean, R.; Leopold, N.; Pavel, I.; Lucaciu, C.M.; et al. Solid Plasmonic Substrates for Breast Cancer Detection by Means of SERS Analysis of Blood Plasma. Nanomaterials 2020, 10, 1212. [Google Scholar] [CrossRef] [PubMed]
- Cervo, S.; Mansutti, E.; Del Mistro, G.; Spizzo, R.; Colombatti, A.; Steffan, A.; Sergo, V.; Bonifacio, A. SERS Analysis of Serum for Detection of Early and Locally Advanced Breast Cancer. Anal. Bioanal. Chem. 2015, 407, 7503–7509. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Wang, Y.; Wang, T.; Zhu, Y.; Lin, X.; Lin, Y.; Feng, S. Metabolite Profiling of Human Blood by Surface-Enhanced Raman Spectroscopy for Surgery Assessment and Tumor Screening in Breast Cancer. Anal. Bioanal. Chem. 2020, 412, 1611–1618. [Google Scholar] [CrossRef] [PubMed]
- Gao, N.; Wang, Q.; Tang, J.; Yao, S.; Li, H.; Yue, X.; Fu, J.; Zhong, F.; Wang, T.; Wang, J. Non-Invasive SERS Serum Detection Technology Combined with Multivariate Statistical Algorithm for Simultaneous Screening of Cervical Cancer and Breast Cancer. Anal. Bioanal. Chem. 2021, 413, 4775–4784. [Google Scholar] [CrossRef]
- Vargas-Obieta, E.; Martínez-Espinosa, J.C.; Martínez-Zerega, B.E.; Jave-Suárez, L.F.; Aguilar-Lemarroy, A.; González-Solís, J.L. Breast Cancer Detection Based on Serum Sample Surface Enhanced Raman Spectroscopy. Lasers Med. Sci. 2016, 31, 1317–1324. [Google Scholar] [CrossRef]
- Ma, X.; Xiong, H.; Guo, J.; Liu, Z.; Han, Y.; Liu, M. Label-Free Breast Cancer Detection and Classification by Convolutional Neural Network-Based on Exosomes Surface-Enhanced Raman Scattering. J. Innov. Opt. Health Sci. 2022, 2022, 2244001. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, J.; Shanmugasundaram, K.B.; Yeo, B.; Möller, A.; Wuethrich, A.; Lin, L.L.; Trau, M. Tracking Drug-Induced Epithelial–Mesenchymal Transition in Breast Cancer by a Microfluidic Surface-Enhanced Raman Spectroscopy Immunoassay. Small 2020, 16, 1905614. [Google Scholar] [CrossRef]
- Zhu, J.; Zhou, J.; Guo, J.; Cai, W.; Liu, B.; Wang, Z.; Sun, Z. Surface-Enhanced Raman Spectroscopy Investigation on Human Breast Cancer Cells. Chem. Cent. J. 2013, 7, 37. [Google Scholar] [CrossRef] [Green Version]
- Brozek-Pluska, B.; Kopec, M.; Surmacki, J. Surface-Enhanced Raman Spectroscopy Analysis of Human Breast Cancer via Silver Nanoparticles: An Examination of Fabrication Methods. J. Spectrosc. 2018, 2018, 4893274. [Google Scholar] [CrossRef]
- Narayanan, N.; Kim, J.H.; Santhakumar, H.; Joseph, M.M.; Karunakaran, V.; Shamjith, S.; Saranya, G.; Sujai, P.T.; Jayasree, R.S.; Barman, I.; et al. Nanotheranostic Probe Built on Methylene Blue Loaded Cucurbituril [8] and Gold Nanorod: Targeted Phototherapy in Combination with SERS Imaging on Breast Cancer Cells. J. Phys. Chem. B 2021, 125, 13415–13424. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Chen, L.; Xia, Y.; Xing, J.; Li, Z.; Qian, Q.; Wang, Y.; Wu, A.; Zeng, L.; Zhou, Y. Bioconjugation of Gold Nanobipyramids for SERS Detection and Targeted Photothermal Therapy in Breast Cancer. ACS Biomater. Sci. Eng. 2017, 3, 608–618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeng, L.; Pan, Y.; Wang, S.; Wang, X.; Zhao, X.; Ren, W.; Lu, G.; Wu, A. Raman Reporter-Coupled Agcore@Aushell Nanostars for in Vivo Improved Surface Enhanced Raman Scattering Imaging and Near-Infrared-Triggered Photothermal Therapy in Breast Cancers. ACS Appl. Mater. Interfaces 2015, 7, 16781–16791. [Google Scholar] [CrossRef] [PubMed]
- Xinyue, L.; Keshavarz, M.; Panagiotis, K.; Roddan, A.; Yeatman, E.; Thompson, A. SERS Detection of Breast Cancer-Derived Exosomes Using a Nanostructured Pt-Black Template. Adv. Sens. Res. 2023, 2023, 2200039. [Google Scholar] [CrossRef]
- Pramanik, A.; Mayer, J.; Patibandla, S.; Gates, K.; Gao, Y.; Davis, D.; Seshadri, R.; Ray, P.C. Mixed-Dimensional Heterostructure Material-Based SERS for Trace Level Identification of Breast Cancer-Derived Exosomes. ACS Omega 2020, 5, 16602–16611. [Google Scholar] [CrossRef]
- Li, G.; Zhu, N.; Zhou, J.; Kang, K.; Zhou, X.; Ying, B.; Yi, Q.; Wu, Y. A Magnetic Surface-Enhanced Raman Scattering Platform for Performing Successive Breast Cancer Exosome Isolation and Analysis. J. Mater. Chem. B 2021, 9, 2709–2716. [Google Scholar] [CrossRef]
- Yang, Z.; Su, H.S.; You, E.M.; Liu, S.; Li, Z.; Zhang, Y. High Uniformity and Enhancement Au@AgNS 3D Substrates for the Diagnosis of Breast Cancer. ACS Omega 2022, 7, 15223–15230. [Google Scholar] [CrossRef]
- Wang, X.P.; Walkenfort, B.; König, M.; König, L.; Kasimir-Bauer, S.; Schlücker, S. Fast and Reproducible ISERS Microscopy of Single HER2-Positive Breast Cancer Cells Using Gold Nanostars as SERS Nanotags. Faraday Discuss. 2017, 205, 377–386. [Google Scholar] [CrossRef]
- Chen, Z.; Shen, X.; Chen, S.; Dai, K. Gastric Cancer Prewarning and Early Diagnosis System; Springer: Berlin/Heidelberg, Germany, 2017; ISBN 9789402409499. [Google Scholar]
- Hunter, R.A.; Asare-Werehene, M.; Mandour, A.; Tsang, B.K.; Anis, H. Determination of Chemoresistance in Ovarian Cancer by Simultaneous Quantification of Exosomes and Exosomal Cisplatin with Surface Enhanced Raman Scattering. Sens. Actuators B Chem. 2022, 354, 131237. [Google Scholar] [CrossRef]
- Moothanchery, M.; Perumal, J.; Mahyuddin, A.P.; Singh, G.; Choolani, M.; Olivo, M. Rapid and Sensitive Detection of Ovarian Cancer Biomarker Using a Portable Single Peak Raman Detection Method. Sci. Rep. 2022, 12, 12459. [Google Scholar] [CrossRef]
- Sarkar, S.; Gogoi, M.; Mahato, M.; Joshi, A.B.; Baruah, A.J.; Kodgire, P.; Boruah, P. Biosensors for Detection of Prostate Cancer: A Review. Biomed. Microdevices 2022, 24, 32. [Google Scholar] [CrossRef]
- Turan, E.; Zengin, A.; Suludere, Z.; Kalkan, N.Ö.; Tamer, U. Construction of a Sensitive and Selective Plasmonic Biosensor for Prostate Specific Antigen by Combining Magnetic Molecularly-Imprinted Polymer and Surface-Enhanced Raman Spectroscopy. Talanta 2022, 237, 122926. [Google Scholar] [CrossRef]
- Haroon, M.; Tahir, M.; Nawaz, H.; Majeed, M.I.; Al-Saadi, A.A. Surface-Enhanced Raman Scattering (SERS) Spectroscopy for Prostate Cancer Diagnosis: A Review. Photodiagn. Photodyn. Ther. 2022, 37, 102690. [Google Scholar] [CrossRef] [PubMed]
- Ashrafizadeh, M.; Aghamiri, S.; Tan, S.C.; Zarrabi, A.; Sharifi, E.; Rabiee, N.; Kadumudi, F.B.; Pirouz, A.D.; Delfi, M.; Byrappa, K.; et al. Nanotechnological Approaches in Prostate Cancer Therapy: Integration of Engineering and Biology. Nano Today 2022, 45, 101532. [Google Scholar] [CrossRef]
- Gaba, F.; Tipping, W.J.; Salji, M.; Faulds, K.; Graham, D.; Leung, H.Y. Raman Spectroscopy in Prostate Cancer: Techniques, Applications and Advancements. Cancers. 2022, 14, 1535. [Google Scholar] [CrossRef]
- Pandey, A.; Sarkar, S.; Pandey, S.K.; Srivastava, A. Silica Nanospheres Coated Silver Islands as an Effective Opto-Plasmonic SERS Active Platform for Rapid and Sensitive Detection of Prostate Cancer Biomarkers. Molecules 2022, 27, 7821. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Zhu, Y.Y.; Wang, M.L. Surface-Enhanced Raman Spectroscopy of Gastric Cancer Serum with Gold Nanoparticles/Silicon Nanowire Arrays. Optik 2016, 127, 7902–7907. [Google Scholar] [CrossRef]
- Ito, H.; Inoue, H.; Hasegawa, K.; Hasegawa, Y.; Shimizu, T.; Kimura, S.; Onimaru, M.; Ikeda, H.; Kudo, S. ei Use of Surface-Enhanced Raman Scattering for Detection of Cancer-Related Serum-Constituents in Gastrointestinal Cancer Patients. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 599–608. [Google Scholar] [CrossRef]
- Ge, S.; Ran, M.; Mao, Y.; Sun, Y.; Zhou, X.; Li, L.; Cao, X. A Novel DNA Biosensor for the Ultrasensitive Detection of DNA Methyltransferase Activity Based on a High-Density ‘Hot Spot’ SERS Substrate and Rolling Circle Amplification Strategy. Analyst 2021, 146, 5326–5336. [Google Scholar] [CrossRef]
- Feng, S.; Chen, R.; Lin, J.; Pan, J.; Wu, Y.; Li, Y.; Chen, J.; Zeng, H. Gastric Cancer Detection Based on Blood Plasma Surface-Enhanced Raman Spectroscopy Excited by Polarized Laser Light. Biosens. Bioelectron. 2011, 26, 3167–3174. [Google Scholar] [CrossRef]
- Pan, H.; Dong, Y.; Gong, L.; Zhai, J.; Song, C.; Ge, Z.; Su, Y.; Zhu, D.; Chao, J.; Su, S.; et al. Sensing Gastric Cancer Exosomes with MoS2-Based SERS Aptasensor. Biosens. Bioelectron. 2022, 215, 114553. [Google Scholar] [CrossRef]
- Liu, Z.; Li, T.; Wang, Z.; Liu, J.; Huang, S.; Min, B.H.; An, J.Y.; Kim, K.M.; Kim, S.; Chen, Y.; et al. Gold Nanopyramid Arrays for Non-Invasive Surface-Enhanced Raman Spectroscopy-Based Gastric Cancer Detection via SEVs. ACS Appl. Nano Mater. 2022, 5, 12506–12517. [Google Scholar] [CrossRef]
- Gayoung, E.; Hongki, K.; Ahreum, H.; Hye-Young, S.; Yuna, C.; Jeong, M.; Donghyeong, K.; Miyeon, L.; Eun-Kyung, L.; Jinyoung, J.; et al. Nanogap-Rich Au Nanowire SERS Sensor for Ultrasensitive Telomerase Activity Detection. Adv. Funct. Mater. 2017, 27, 1701832. [Google Scholar] [CrossRef]
- Chen, Y.; Cheng, S.; Zhang, A.; Song, J.; Chang, J.; Wang, K.; Gao, G.; Zhang, Y.; Li, S.; Liu, H.; et al. Salivary Analysis Based on Surface Enhanced Raman Scattering Sensors Distinguishes Early and Advanced Gastric Cancer Patients from Healthy Persons. J. Biomed. Nanotechnol. 2018, 14, 1773–1784. [Google Scholar] [CrossRef]
- Cao, X.; Ge, S.; Hua, W.; Zhou, X.; Lu, W.; Gu, Y.; Li, Z.; Qian, Y. A Pump-Free and High-Throughput Microfluidic Chip for Highly Sensitive SERS Assay of Gastric Cancer-Related Circulating Tumor DNA via a Cascade Signal Amplification Strategy. J. Nanobiotechnol. 2022, 20, 271. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, Y.; Pan, F.; Liu, J.; Wang, K.; Zhang, C.; Cheng, S.; Lu, L.; Zhang, W.; Zhang, Z.; et al. Breath Analysis Based on Surface-Enhanced Raman Scattering Sensors Distinguishes Early and Advanced Gastric Cancer Patients from Healthy Persons. ACS Nano 2016, 10, 8169–8179. [Google Scholar] [CrossRef]
- Huang, L.; Zhu, Y.; Xu, C.; Cai, Y.; Yi, Y.; Li, K.; Ren, X.; Jiang, D.; Ge, Y.; Liu, X.; et al. Noninvasive Diagnosis of Gastric Cancer Based on Breath Analysis with a Tubular Surface-Enhanced Raman Scattering Sensor. ACS Sens. 2022, 7, 1439–1450. [Google Scholar] [CrossRef]
- Cao, D.; Lin, H.; Liu, Z.; Qiu, J.; Ge, S.; Hua, W.; Cao, X.; Qian, Y.; Xu, H.; Zhu, X. PCA-TLNN-Based SERS Analysis Platform for Label-Free Detection and Identification of Cisplatin-Treated Gastric Cancer. Sens. Actuators B Chem. 2023, 375, 132903. [Google Scholar] [CrossRef]
- Guo, L.; Li, Y.; Huang, F.; Dong, J.; Li, F.; Yang, X.; Zhu, S.; Yang, M. Identification and Analysis of Serum Samples by Surface-Enhanced Raman Spectroscopy Combined with Characteristic Ratio Method and PCA for Gastric Cancer Detection. J. Innov. Opt. Health Sci. 2019, 12, 1950003. [Google Scholar] [CrossRef] [Green Version]
- Ma, J.; Zhou, H.; Gong, L.; Liu, S.; Zhou, Z.; Mao, W.; Zheng, R. Distinction of Gastric Cancer Tissue Based on Surface-Enhanced Raman Spectroscopy. Opt. Health Care Biomed. Opt. V 2012, 8553, 855328. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, Y.; Chen, G.; Zheng, X.; He, C.; Feng, S.; Lin, X.; Chen, R.; Zeng, H. Discrimination of Gastric Cancer from Normal by Serum RNA Based on Surface-Enhanced Raman Spectroscopy (SERS) and Multivariate Analysis. Med. Phys. 2012, 39, 5664–5668. [Google Scholar] [CrossRef] [Green Version]
- Feng, S.Y.; Pan, J.J.; Wu, Y.A.; Lin, D.; Chen, Y.P.; Xi, G.Q.; Lin, J.Q.; Chen, R. Study on Gastric Cancer Blood Plasma Based on Surface-Enhanced Raman Spectroscopy Combined with Multivariate Analysis. Sci. China Life Sci. 2011, 54, 828–834. [Google Scholar] [CrossRef] [Green Version]
- Aslam, M.A.; Xue, C.; Wang, K.; Chen, Y.; Zhang, A.; Cai, W.; Ma, L.; Yang, Y.; Sun, X.; Liu, M.; et al. SVM Based Classification and Prediction System for Gastric Cancer Using Dominant Features of Saliva. Nano Biomed. Eng. 2020, 12, 1–13. [Google Scholar] [CrossRef]
- Aslam, M.A.; Xue, C.; Liu, M.; Wang, K.; Cui, D. Classification and Prediction of Gastric Cancer from Saliva Diagnosis Using Artificial Neural Network. Eng. Lett. 2020, 29, 2. [Google Scholar]
- Avram, L.; Iancu, S.D.; Stefancu, A.; Moisoiu, V.; Colnita, A.; Marconi, D.; Donca, V.; Buzdugan, E.; Craciun, R.; Leopold, N.; et al. SERS-Based Liquid Biopsy of Gastrointestinal Tumors Using a Portable Raman Device Operating in a Clinical Environment. J. Clin. Med. 2020, 9, 212. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Yang, T.; Li, S.; Wang, D.; Song, Y.; Yu, K. Different Classification Algorithms and Serum Surface Enhanced Raman Spectroscopy for Noninvasive Discrimination of Gastric Diseases. J. Raman Spectrosc. 2016, 47, 917–925. [Google Scholar] [CrossRef]
- Li, S.X.; Zhang, Y.J.; Zeng, Q.Y.; Li, L.F.; Guo, Z.Y.; Liu, Z.M.; Xiong, H.L.; Liu, S.H. Potential of Cancer Screening with Serum Surface-Enhanced Raman Spectroscopy and a Support Vector Machine. Laser Phys. Lett. 2014, 11, 065603. [Google Scholar] [CrossRef]
- Moisoiu, T.; Dragomir, M.P.; Iancu, S.D.; Schallenberg, S.; Birolo, G.; Ferrero, G.; Burghelea, D.; Stefancu, A.; Cozan, R.G.; Licarete, E.; et al. Combined MiRNA and SERS Urine Liquid Biopsy for the Point-of-Care Diagnosis and Molecular Stratification of Bladder Cancer. Mol. Med. 2022, 28, 39. [Google Scholar] [CrossRef]
- Gao, S.; Lin, Y.; Zhao, X.; Gao, J.; Xie, S.; Gong, W.; Yu, Y.; Lin, J. Label-Free Surface Enhanced Raman Spectroscopy Analysis of Blood Serum via Coffee Ring Effect for Accurate Diagnosis of Cancers. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 267, 120605. [Google Scholar] [CrossRef]
- Meng, C.; Li, H.; Chen, C.; Wu, W.; Gao, J.; Lai, Y.; Ka, M.; Zhu, M.; Lv, X.; Chen, F.; et al. Serum Raman Spectroscopy Combined with Gaussian—Convolutional Neural Network Models to Quickly Detect Liver Cancer Patients. Spectrosc. Lett. 2022, 55, 79–90. [Google Scholar] [CrossRef]
- Ni, J.-T.; Huang, M.-Y.; Ji, W.; Wang, L.; Sun, T.-D. Recent Advances in Surface-Enhanced Raman Scattering for Liver Cancer Detection. Chin. J. Anal. Chem. 2022, 50, 100180. [Google Scholar] [CrossRef]
- Zhang, Q.; Hou, D.; Wen, X.; Xin, M.; Li, Z.; Wu, L.; Pathak, J.L. Gold Nanomaterials for Oral Cancer Diagnosis and Therapy: Advances, Challenges, and Prospects. Mater. Today Bio 2022, 15, 100333. [Google Scholar] [CrossRef] [PubMed]
- Zadeh, F.A.; Shahhosseini, E.; Rasoolzadegan, S.; Özbolat, G.; Farahbod, F. Au Nanoparticles in the Diagnosis and Treatment of Ovarian Cancer: A New Horizon in the Personalized Medicine. Nanomed. Res. J. 2022, 7, 1–18. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Du, Q.; Cao, D.; Lu, X.; Meng, Z. Sensitive SERS Detection of Oral Squamous Cell Carcinoma-Related MiRNAs in Saliva via a Gold Nanohexagon Array Coupled with Hybridization Chain Reaction Amplification. Anal. Methods 2022, 14, 4563–4575. [Google Scholar] [CrossRef]
- Fălămaș, A.; Rotaru, H.; Hedeșiu, M. Surface-Enhanced Raman Spectroscopy (SERS) Investigations of Saliva for Oral Cancer Diagnosis. Lasers Med. Sci. 2020, 35, 1393–1401. [Google Scholar] [CrossRef]
- Wang, K.; Qiu, Y.; Wu, C.; Wen, Z.N.; Li, Y. Surface-Enhanced Raman Spectroscopy and Multivariate Analysis for the Diagnosis of Oral Squamous Cell Carcinoma. J. Raman Spectrosc. 2023. [Google Scholar] [CrossRef]
- Lin, Y.; Lin, J.; Zheng, M.; Gong, W.; Li, H.; Shu, Z.; Du, W.; Gao, S.; Yu, Y. Quantitative and Direct Serum Albumin Detection by Label-Free SERS Using Tunable Hydroxyapatite Nanostructure for Prostate Cancer Detection. Anal. Chim. Acta 2022, 1221, 340101. [Google Scholar] [CrossRef]
- Zhao, J.; Wang, J.; Liu, Y.; Han, X.X.; Xu, B.; Ozaki, Y.; Zhao, B. Detection of Prostate Cancer Biomarkers via a SERS-Based Aptasensor. Biosens. Bioelectron. 2022, 216, 114660. [Google Scholar] [CrossRef]
- Lu, Y.; Zhan, C.; Yu, L.; Yu, Y.; Jia, H.; Chen, X.; Zhang, D.; Gao, R. Multifunctional Nanocone Array as Solid Immunoassay Plate and SERS Substrate for the Early Diagnosis of Prostate Cancer on Microfluidic Chip. Sens. Actuators B Chem. 2023, 376, 133046. [Google Scholar] [CrossRef]
- Munteanu, V.C.; Munteanu, R.A.; Gulei, D.; Mărginean, R.; Schițcu, V.H.; Onaciu, A.; Toma, V.; Știufiuc, G.F.; Coman, I.; Știufiuc, R.I. New Insights into the Multivariate Analysis of SER Spectra Collected on Blood Samples for Prostate Cancer Detection: Towards a Better Understanding of the Role Played by Different Biomolecules on Cancer Screening: A Preliminary Study. Cancers 2022, 14, 3227. [Google Scholar] [CrossRef] [PubMed]
- Stefancu, A.; Moisoiu, V.; Couti, R.; Andras, I.; Rahota, R.; Crisan, D.; Pavel, I.E.; Socaciu, C.; Leopold, N.; Crisan, N. Combining SERS Analysis of Serum with PSA Levels for Improving the Detection of Prostate Cancer. Nanomedicine 2018, 13, 2455–2467. [Google Scholar] [CrossRef] [Green Version]
- Liyanage, T.; Alharbi, B.; Quan, L.; Esquela-Kerscher, A.; Slaughter, G. Plasmonic-Based Biosensor for the Early Diagnosis of Prostate Cancer. ACS Omega 2022, 7, 2411–2418. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Xu, Q.; Lin, Y.; Du, W.; Bai, X.; Gao, J.; Li, T.; Huang, Y.; Yu, Y.; Wu, X.; et al. Label-free surface-enhanced Raman spectroscopy detectionof prostate cancer combined with multivariate statistical algorithm. J. Raman Spectrosc. 2022, 53, 1861–1870. [Google Scholar] [CrossRef]
- Sayyadi, N.; Justiniano, I.; Wang, Y.; Zheng, X.; Zhang, W.; Jiang, L.; Polikarpov, D.M.; Willows, R.D.; Gillatt, D.; Campbell, D.; et al. Detection of Rare Prostate Cancer Cells in Human Urine Offers Prospect of Non-Invasive Diagnosis. Sci. Rep. 2022, 12, 18452. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Lin, S.; Zhang, H.; Liang, L.; Shen, S. Methods of Respiratory Virus Detection: Advances towards Point-of-Care for Early Intervention. Micromachines 2021, 12, 697. [Google Scholar] [CrossRef] [PubMed]
- Omidifar, N.; Lankarani, K.B.; Moghadami, M.; Shokripour, M.; Chashmpoosh, M.; Mousavi, S.M.; Hashemi, S.A.; Gholami, A. Different Laboratory Diagnosis Methods of COVID-19: A Systematic Review. Arch. Clin. Infect. Dis. 2021, 16, e110667. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Hashemi, S.A.; Rahmanian, V.; Kalashgrani, M.Y. Highly Sensitive Flexible SERS-Based Sensing Platform for Detection of Biosensors Highly Sensitive Flexible SERS-Based Sensing Platform for Detection of COVID-19. Biosensors 2022, 12, 466. [Google Scholar] [CrossRef]
- Stöckel, S.; Kirchhoff, J.; Neugebauer, U.; Rösch, P.; Popp, J. The Application of Raman Spectroscopy for the Detection and Identification of Microorganisms. J. Raman Spectrosc. 2016, 47, 89–109. [Google Scholar] [CrossRef]
- Soler, M.; Estevez, M.C.; Cardenosa-Rubio, M.; Astua, A.; Lechuga, L.M. How Nanophotonic Label-Free Biosensors Can Contribute to Rapid and Massive Diagnostics of Respiratory Virus Infections: COVID-19 Case. ACS Sens. 2020, 5, 2663–2678. [Google Scholar] [CrossRef]
- Iravani, S. Nano- And Biosensors for the Detection of SARS-CoV-2: Challenges and Opportunities. Mater. Adv. 2020, 1, 3092–3103. [Google Scholar] [CrossRef]
- Zhang, D.; Zhang, X.; Ma, R.; Deng, S.; Wang, X.; Wang, X.; Zhang, X.; Huang, X.; Liu, Y.; Li, G.; et al. Ultra-Fast and Onsite Interrogation of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) in Waters via Surface Enhanced Raman Scattering (SERS). Water Res. 2021, 200, 117243. [Google Scholar] [CrossRef] [PubMed]
- Joung, Y.; Kim, K.; Lee, S.; Chun, B.-S.; Lee, S.; Hwang, J.; Choi, S.; Kang, T.; Lee, M.-K.; Chen, L.; et al. Rapid and Accurate On-Site Immunodiagnostics of Highly Contagious Severe Acute Respiratory Syndrome Coronavirus 2 Using Portable Surface-Enhanced Raman Scattering-Lateral Flow Assay Reader. ACS Sens. 2022, 7, 3470–3480. [Google Scholar] [CrossRef] [PubMed]
- Saviñon-Flores, F.; Méndez, E.; López-Castaños, M.; Carabarin-Lima, A.; López-Castaños, K.A.; González-Fuentes, M.A.; Méndez-Albores, A. A Review on Sers-Based Detection of Human Virus Infections: Influenza and Coronavirus. Biosensors 2021, 11, 66. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.Y.; Nam, J.S.; Yang, S.E.; Shin, H.; Jang, Y.H.; Bae, G.U.; Kang, T.; Lim, K.I.; Choi, Y. Identification of Newly Emerging Influenza Viruses by Surface-Enhanced Raman Spectroscopy. Anal. Chem. 2015, 87, 11652–11659. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Wang, Z.; Liu, H.; Perea-López, N.; Ranasinghe, J.C.; Bepete, G.; Minns, A.M.; Rossi, R.M.; Lindner, S.E.; Huang, S.X.; et al. Understanding the Excitation Wavelength Dependence and Thermal Stability of the SARS-CoV-2 Receptor-Binding Domain Using Surface-Enhanced Raman Scattering and Machine Learning. ACS Photonics 2022, 9, 2963–2972. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, B.; Murray, J.; Haverstick, J.; Chen, X.; Tripp, R.A.; Zhao, Y. Rapid and Quantitative Detection of Respiratory Viruses Using Surface-Enhanced Raman Spectroscopy and Machine Learning. Biosens. Bioelectron. 2022, 217, 114721. [Google Scholar] [CrossRef]
- Ye, J.; Yeh, Y.; Xue, Y.; Wang, Z.; Zhang, N.; Liu, H.; Zhang, K.; Ricker, R.; Yu, Z.; Roder, A. Accurate Virus Identi Fi Cation with Interpretable Raman Signatures by Machine Learning. Proc. Natl. Acad. Sci. USA 2022, 199, e2118836119. [Google Scholar] [CrossRef] [PubMed]
- Carlomagno, C.; Bertazioli, D.; Gualerzi, A.; Picciolini, S.; Banfi, P.I.; Lax, A.; Messina, E.; Navarro, J.; Bianchi, L.; Caronni, A.; et al. COVID-19 Salivary Raman Fingerprint: Innovative Approach for the Detection of Current and Past SARS-CoV-2 Infections. Sci. Rep. 2021, 11, 4943. [Google Scholar] [CrossRef]
- Zavyalova, E.; Ambartsumyan, O.; Zhdanov, G.; Gribanyov, D.; Gushchin, V.; Tkachuk, A.; Rudakova, E.; Nikiforova, M.; Kuznetsova, N.; Popova, L.; et al. Sers-Based Aptasensor for Rapid Quantitative Detection of Sars-Cov-2. Nanomaterials 2021, 11, 1394. [Google Scholar] [CrossRef]
- Hwang, C.S.H.; Lee, S.; Lee, S.; Kim, H.; Kang, T.; Lee, D.; Jeong, K.H. Highly Adsorptive Au-TiO2 Nanocomposites for the SERS Face Mask Allow the Machine-Learning-Based Quantitative Assay of SARS-CoV-2 in Artificial Breath Aerosols. ACS Appl. Mater. Interfaces 2022, 14, 54550–54557. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Jiang, S.; Wang, X.; Dong, T.; Wang, Y.; Li, D.; Gao, X.; Qu, Z.; Li, Y. A Novel Enhanced Substrate for Label-Free Detection of SARS-CoV-2 Based on Surface-Enhanced Raman Scattering. Sens. Actuators B Chem. 2022, 359, 131568. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wang, C.; Wang, X.; Wang, K.; Zhu, Y.; Rong, Z.; Wang, W.; Xiao, R.; Wang, S. Magnetic SERS Strip for Sensitive and Simultaneous Detection of Respiratory Viruses. ACS Appl. Mater. Interfaces 2019, 11, 19495–19505. [Google Scholar] [CrossRef]
- Zhang, D.; Huang, L.; Liu, B.; Ge, Q.; Dong, J.; Zhao, X. Rapid and Ultrasensitive Quantification of Multiplex Respiratory Tract Infection Pathogen via Lateral Flow Microarray Based on SERS Nanotags. Theranostics 2019, 9, 4849–4859. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Peng, Y.; Lin, C.; Long, L.; Hu, J.; He, J.; Zeng, H.; Huang, Z.; Li, Z.Y.; Tanemura, M.; et al. Human ACE2-Functionalized Gold “Virus-Trap” Nanostructures for Accurate Capture of SARS-CoV-2 and Single-Virus SERS Detection. Nano-Micro Lett. 2021, 13, 109. [Google Scholar] [CrossRef]
- Peng, Y.; Lin, C.; Long, L.; Masaki, T.; Tang, M.; Yang, L.; Liu, J.; Huang, Z.; Li, Z.; Luo, X.; et al. Charge-Transfer Resonance and Electromagnetic Enhancement Synergistically Enabling MXenes with Excellent SERS Sensitivity for SARS-CoV-2 S Protein Detection. Nano-Micro Lett. 2021, 13, 52. [Google Scholar] [CrossRef]
- Gu, M.M.; Guan, P.C.; Xu, S.S.; Li, H.M.; Kou, Y.C.; Lin, X.D.; Kathiresan, M.; Song, Y.; Zhang, Y.J.; Jin, S.Z.; et al. Ultrasensitive Detection of SARS-CoV-2 S Protein with Aptamers Biosensor Based on Surface-Enhanced Raman Scattering. J. Chem. Phys. 2023, 158, 024203. [Google Scholar] [CrossRef]
- Lim, J.Y.; Nam, J.S.; Shin, H.; Park, J.; Song, H.I.; Kang, M.; Lim, K.I.; Choi, Y. Identification of Newly Emerging Influenza Viruses by Detecting the Virally Infected Cells Based on Surface Enhanced Raman Spectroscopy and Principal Component Analysis. Anal. Chem. 2019, 91, 5677–5684. [Google Scholar] [CrossRef]
- Eom, G.; Hwang, A.; Kim, H.; Yang, S.; Lee, D.K.; Song, S.; Ha, K.; Jeong, J.; Jung, J.; Lim, E.K.; et al. Diagnosis of Tamiflu-Resistant Influenza Virus in Human Nasal Fluid and Saliva Using Surface-Enhanced Raman Scattering. ACS Sens. 2019, 4, 2282–2287. [Google Scholar] [CrossRef]
- Zhang, M.; Li, X.; Pan, J.; Zhang, Y.; Zhang, L.; Wang, C.; Yan, X.; Liu, X.; Lu, G. Ultrasensitive Detection of SARS-CoV-2 Spike Protein in Untreated Saliva Using SERS-Based Biosensor. Biosens. Bioelectron. 2021, 190, 113421. [Google Scholar] [CrossRef]
- Yadav, S.; Sadique, M.A.; Ranjan, P.; Kumar, N.; Singhal, A.; Srivastava, A.K.; Khan, R. Sers Based Lateral Flow Immunoassay for Point-of-Care Detection of Sars-Cov-2 in Clinical Samples. ACS Appl. Bio Mater. 2021, 4, 2974–2995. [Google Scholar] [CrossRef]
- Chen, S.; Meng, L.; Wang, L.; Huang, X.; Ali, S.; Chen, X.; Yu, M.; Yi, M.; Li, L.; Chen, X.; et al. SERS-Based Lateral Flow Immunoassay for Sensitive and Simultaneous Detection of Anti-SARS-CoV-2 IgM and IgG Antibodies by Using Gap-Enhanced Raman Nanotags. Sens. Actuators B Chem. 2021, 348, 130706. [Google Scholar] [CrossRef]
- Liu, H.; Dai, E.; Xiao, R.; Zhou, Z.; Zhang, M.; Bai, Z.; Shao, Y.; Qi, K.; Tu, J.; Wang, C.; et al. Development of a SERS-Based Lateral Flow Immunoassay for Rapid and Ultra-Sensitive Detection of Anti-SARS-CoV-2 IgM/IgG in Clinical Samples. Sens. Actuators B Chem. 2021, 329, 129196. [Google Scholar] [CrossRef] [PubMed]
- Antoine, D.; Mohammadi, M.; Vitt, M.; Dickie, J.M.; Jyoti, S.S.; Tilbury, M.A.; Johnson, P.A.; Wawrousek, K.E.; Wall, J.G. Rapid, Point-of-Care ScFv-SERS Assay for Femtogram Level Detection of SARS-CoV-2. ACS Sens. 2022, 7, 866–873. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Luo, Y.; Song, Y.; Zhu, Q.; Xu, T.; Zhang, X. One-Click Investigation of Shape Influence of Silver Nanostructures on SERS Performance for Sensitive Detection of COVID-19. Anal. Chim. Acta 2022, 1234, 340523. [Google Scholar] [CrossRef]
- Kim, W.; Kim, S.; Han, J.; Kim, T.G.; Bang, A.; Choi, H.W.; Min, G.E.; Shin, J.H.; Moon, S.W.; Choi, S. An Excitation Wavelength-Optimized, Stable SERS Biosensing Nanoplatform for Analyzing Adenoviral and AstraZeneca COVID-19 Vaccination Efficacy Status Using Tear Samples of Vaccinated Individuals. Biosens. Bioelectron. 2022, 204, 114079. [Google Scholar] [CrossRef] [PubMed]
- Karunakaran, V.; Joseph, M.M.; Yadev, I.; Sharma, H.; Shamna, K.; Saurav, S.; Sreejith, R.P.; Anand, V.; Beegum, R.; Regi David, S.; et al. A Non-Invasive Ultrasensitive Diagnostic Approach for COVID-19 Infection Using Salivary Label-Free SERS Fingerprinting and Artificial Intelligence. J. Photochem. Photobiol. B Biol. 2022, 234, 112545. [Google Scholar] [CrossRef]
- Shanmukh, S.; Jones, L.; Driskell, J.; Zhao, Y.; Dluhy, R.; Tripp, R.A. Rapid and Sensitive Detection of Respiratory Virus Molecular Signatures Using a Silver Nanorod Array SERS Substrate. Nano Lett. 2006, 6, 2630–2636. [Google Scholar] [CrossRef] [PubMed]
- Dluhy, R.A.; Shanmukh, S.; Jones, L.; Zhao, Y.P.; Driskell, J.D.; Tripp, R.A. Identification and Classification of Respiratory Syncytial Virus (RSV) Strains by Surface-Enhanced Raman Spectroscopy and Multivariate Statistical Techniques. Anal. Bioanal. Chem. 2008, 390, 1551–1555. [Google Scholar] [CrossRef]
- Huang, J.; Wen, J.; Zhou, M.; Ni, S.; Le, W.; Chen, G.; Wei, L.; Zeng, Y.; Qi, D.; Pan, M.; et al. On-Site Detection of SARS-CoV-2 Antigen by Deep Learning-Based Surface-Enhanced Raman Spectroscopy and Its Biochemical Foundations. Anal. Chem. 2021, 93, 9174–9182. [Google Scholar] [CrossRef] [PubMed]
- Yeh, Y.J.; Le, T.N.; Hsiao, W.W.W.; Tung, K.L.; Ostrikov, K.; Chiang, W.H. Plasmonic Nanostructure-Enhanced Raman Scattering for Detection of SARS-CoV-2 Nucleocapsid Protein and Spike Protein Variants. Anal. Chim. Acta 2023, 1239, 340651. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Wang, C.; Zheng, S.; Yang, X.; Han, H.; Dai, Y.; Xiao, R. Simultaneously Ultrasensitive and Quantitative Detection of Influenza A Virus, SARS-CoV-2, and Respiratory Syncytial Virus via Multichannel Magnetic SERS-Based Lateral Flow Immunoassay. Nanomed. Nanotechnol. Biol. Med. 2023, 47, 102624. [Google Scholar] [CrossRef] [PubMed]
- Chisanga, M.; Williams, H.; Boudreau, D.; Pelletier, J.N.; Trottier, S. Label-Free SERS for Rapid Differentiation of SARS-CoV-2-Induced Serum Metabolic Profiles in Non-Hospitalized Adults. Anal. Chem. 2023, 95, 3638–3646. [Google Scholar] [CrossRef] [PubMed]
- Bacteria. Available online: https://microbiologysociety.org/why-microbiology-matters/what-is-microbiology/bacteria.html#:~:text=Bacteria%20are%20classified%20into%20five,)%20or%20corkscrew%20(spirochaetes) (accessed on 30 January 2023).
- Bacteria as Pathogens. Available online: https://sphweb.bumc.bu.edu/otlt/mph-modules/ph/ph709_infectiousagents/PH709_InfectiousAgents4.html#:~:text=While%20only%20about%205%25%20of,of%20human%20disease%20and%20death (accessed on 30 January 2023).
- Nanda, M.; Kumar, V.; Sharma, D.K. Multimetal Tolerance Mechanisms in Bacteria: The Resistance Strategies Acquired by Bacteria That Can Be Exploited to ‘Clean-up’ Heavy Metal Contaminants from Water. Aquat. Toxicol. 2019, 212, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Glick, B.R. Phytoremediation: Synergistic Use of Plants and Bacteria to Clean up the Environment. Biotechnol. Adv. 2003, 21, 383–393. [Google Scholar] [CrossRef]
- Kulshreshtha, A.; Agrawal, R.; Barar, M.; Saxena, S. A Review on Bioremediation of Heavy Metals in Contaminated Water. IOSR J. Environ. Sci. Toxicol. Food Technol. 2014, 8, 44–50. [Google Scholar] [CrossRef]
- Zhou, X.; Hu, Z.; Yang, D.; Xie, S.; Jiang, Z.; Niessner, R.; Haisch, C.; Zhou, H.; Sun, P. Bacteria Detection: From Powerful SERS to Its Advanced Compatible Techniques. Adv. Sci. 2020, 7, 2001739. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Kim, K.; Choi, N.; Wang, X.; Lee, J.; Jeon, J.H.; Rhie, G.E.; Choo, J. Highly Sensitive Detection of High-Risk Bacterial Pathogens Using SERS-Based Lateral Flow Assay Strips. Sens. Actuators B Chem. 2018, 270, 72–79. [Google Scholar] [CrossRef]
- Zhu, T.; Hu, Y.; Yang, K.; Dong, N.; Yu, M.; Jiang, N. A Novel SERS Nanoprobe Based on the Use of Core-Shell Nanoparticles with Embedded Reporter Molecule to Detect E. coli O157:H7 with High Sensitivity. Microchim. Acta 2018, 185, 30. [Google Scholar] [CrossRef]
- Chisanga, M.; Muhamadali, H.; Ellis, D.I.; Goodacre, R. Surface-Enhanced Raman Scattering (SERS) in Microbiology: Illumination and Enhancement of the Microbial World. Appl. Spectrosc. 2018, 72, 987–1000. [Google Scholar] [CrossRef]
- Kim, J.A.; Wales, D.J.; Thompson, A.J.; Yang, G.Z. Fiber-optic SERS probes fabricated using two-photon polymerization for rapid detection of bacteria. Adv. Opt. Mater. 2020, 8, 1901934. [Google Scholar] [CrossRef] [Green Version]
- Zhou, H.; Yang, D.; Ivleva, N.P.; Mircescu, N.E.; Niessner, R.; Haisch, C. SERS Detection of Bacteria in Water by in Situ Coating with Ag Nanoparticles. Anal. Chem. 2014, 86, 1525–1533. [Google Scholar] [CrossRef]
- Yang, Y.; Zeng, C.; Huang, J.; Wang, M.; Qi, W.; Wang, H.; He, Z. Specific and Quantitative Detection of Bacteria Based on Surface Cell Imprinted SERS Mapping Platform. Biosens. Bioelectron. 2022, 215, 114524. [Google Scholar] [CrossRef]
- Pearson, B.; Wang, P.; Mills, A.; Pang, S.; McLandsborough, L.; He, L. Innovative Sandwich Assay with Dual Optical and SERS Sensing Mechanisms for Bacterial Detection. Anal. Methods 2017, 9, 4732–4739. [Google Scholar] [CrossRef]
- Hudson, S.D.; Chumanov, G. Bioanalytical Applications of SERS (Surface-Enhanced Raman Spectroscopy). Anal. Bioanal. Chem. 2009, 394, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Mosier-Boss, P.A. Review on SERS of Bacteria. Biosensors 2017, 7, 51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jarvis, R.M.; Goodacre, R. Characterisation and Identification of Bacteria Using SERS. Chem. Soc. Rev. 2008, 37, 931–936. [Google Scholar] [CrossRef]
- Xia, J.; Li, W.; Sun, M.; Wang, H. Application of SERS in the Detection of Fungi, Bacteria and Viruses. Nanomaterials 2022, 12, 3572. [Google Scholar] [CrossRef]
- Wang, C.; Meloni, M.M.; Wu, X.; Zhuo, M.; He, T.; Wang, J.; Wang, C.; Dong, P. Magnetic Plasmonic Particles for SERS-Based Bacteria Sensing: A Review. AIP Adv. 2019, 9, 010701. [Google Scholar] [CrossRef] [Green Version]
- Efrima, S.; Zeiri, L. Understanding SERS of Bacteria. J. Raman Spectrosc. 2009, 40, 277–288. [Google Scholar] [CrossRef]
- Liu, H.B.; Du, X.J.; Zang, Y.X.; Li, P.; Wang, S. SERS-Based Lateral Flow Strip Biosensor for Simultaneous Detection of Listeria Monocytogenes and Salmonella Enterica Serotype Enteritidis. J. Agric. Food Chem. 2017, 65, 10290–10299. [Google Scholar] [CrossRef]
- Mungroo, N.A.; Oliveira, G.; Neethirajan, S. SERS Based Point-of-Care Detection of Food-Borne Pathogens. Microchim. Acta 2016, 183, 697–707. [Google Scholar] [CrossRef]
- Lin, H.Y.; Huang, C.H.; Hsieh, W.H.; Liu, L.H.; Lin, Y.C.; Chu, C.C.; Wang, S.T.; Kuo, I.T.; Chau, L.K.; Yang, C.Y. On-Line SERS Detection of Single Bacterium Using Novel SERS Nanoprobes and a Microfl Uidic Dielectrophoresis Device. Small 2014, 10, 4700–4710. [Google Scholar] [CrossRef]
- Wang, Y.; Lee, K.; Irudayaraj, J. Silver Nanosphere SERS Probes for Sensitive Identification of Pathogens. J. Phys. Chem. C 2010, 114, 16122–16128. [Google Scholar] [CrossRef]
- Witkowska, E.; Korsak, D.; Kowalska, A.; Janeczek, A.; Kamińska, A. Strain-Level Typing and Identification of Bacteria—A Novel Approach for SERS Active Plasmonic Nanostructures. Anal. Bioanal. Chem. 2018, 410, 5019–5031. [Google Scholar] [CrossRef] [Green Version]
- Pang, Y.; Wan, N.; Shi, L.; Wang, C.; Sun, Z.; Xiao, R.; Wang, S. Dual-Recognition Surface-Enhanced Raman Scattering (SERS)Biosensor for Pathogenic Bacteria Detection by Using Vancomycin-SERS Tags and Aptamer-Fe3O4@Au. Anal. Chim. Acta 2019, 1077, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Yin, Y.; Wu, H.; Hao, Z.; Li, J.; Wang, S.; Liu, Y. Integrated SERS Platform for Reliable Detection and Photothermal Elimination of Bacteria in Whole Blood Samples. Anal. Chem. 2021, 93, 1569–1577. [Google Scholar] [CrossRef]
- Zhou, Z.; Xiao, R.; Cheng, S.; Wang, S.; Shi, L.; Wang, C.; Qi, K.; Wang, S. A Universal SERS-Label Immunoassay for Pathogen Bacteria Detection Based on Fe3O4@Au-Aptamer Separation and Antibody-Protein A Orientation Recognition. Anal. Chim. Acta 2021, 1160, 338421. [Google Scholar] [CrossRef]
- Wang, J.; Wu, X.; Wang, C.; Rong, Z.; Ding, H.; Li, H.; Li, S.; Shao, N.; Dong, P.; Xiao, R.; et al. Facile Synthesis of Au-Coated Magnetic Nanoparticles and Their Application in Bacteria Detection via a SERS Method. ACS Appl. Mater. Interfaces 2016, 8, 19958–19967. [Google Scholar] [CrossRef]
- Wang, C.; Wang, J.; Li, M.; Qu, X.; Zhang, K.; Rong, Z.; Xiao, R.; Wang, S. A Rapid SERS Method for Label-Free Bacteria Detection Using Polyethylenimine-Modified Au-Coated Magnetic Microspheres and Au@Ag Nanoparticles. Analyst 2016, 141, 6226–6238. [Google Scholar] [CrossRef]
- Huang, L.; Sun, D.W.; Wu, Z.; Pu, H.; Wei, Q. Reproducible, Shelf-Stable, and Bioaffinity SERS Nanotags Inspired by Multivariate Polyphenolic Chemistry for Bacterial Identification. Anal. Chim. Acta 2021, 1167, 338570. [Google Scholar] [CrossRef] [PubMed]
- Bi, L.; Wang, X.; Cao, X.; Liu, L.; Bai, C.; Zheng, Q.; Choo, J.; Chen, L. SERS-Active Au@Ag Core-Shell Nanorod (Au@AgNR) Tags for Ultrasensitive Bacteria Detection and Antibiotic-Susceptibility Testing. Talanta 2020, 220, 121397. [Google Scholar] [CrossRef] [PubMed]
- Hunter, R.; Sohi, A.N.; Khatoon, Z.; Berthiaume, V.R.; Alarcon, E.I.; Godin, M.; Anis, H. Optofluidic Label-Free SERS Platform for Rapid Bacteria Detection in Serum. Sens. Actuators B Chem. 2019, 300, 126907. [Google Scholar] [CrossRef]
- Sivanesan, A.; Witkowska, E.; Adamkiewicz, W.; Dziewit, Ł.; Kamińska, A.; Waluk, J. Nanostructured Silver-Gold Bimetallic SERS Substrates for Selective Identification of Bacteria in Human Blood. Analyst 2014, 139, 1037–1043. [Google Scholar] [CrossRef]
- Witkowska, E.; Szymborski, T.; Kamińska, A.; Waluk, J. Polymer Mat Prepared via ForcespinningTM as a SERS Platform for Immobilization and Detection of Bacteria from Blood Plasma. Mater. Sci. Eng. C 2017, 71, 345–350. [Google Scholar] [CrossRef]
- Zhang, L.; Xu, J.; Mi, L.; Gong, H.; Jiang, S.; Yu, Q. Multifunctional Magnetic-Plasmonic Nanoparticles for Fast Concentration and Sensitive Detection of Bacteria Using SERS. Biosens. Bioelectron. 2012, 31, 130–136. [Google Scholar] [CrossRef]
- Krafft, B.; Tycova, A.; Urban, R.D.; Dusny, C.; Belder, D. Microfluidic device for concentration and SERS-based detection of bacteria in drinking water. Electrophoresis 2021, 42, 86–94. [Google Scholar] [CrossRef]
- Yang, E.; Li, D.; Yin, P.; Xie, Q.; Li, Y.; Lin, Q.; Duan, Y. A Novel Surface-Enhanced Raman Scattering (SERS) Strategy for Ultrasensitive Detection of Bacteria Based on Three-Dimensional (3D) DNA Walker. Biosens. Bioelectron. 2021, 172, 112758. [Google Scholar] [CrossRef]
- Wang, C.; Wang, C.; Li, J.; Tu, Z.; Gu, B.; Wang, S. Ultrasensitive and Multiplex Detection of Four Pathogenic Bacteria on a Bi-Channel Lateral Flow Immunoassay Strip with Three-Dimensional Membrane-like SERS Nanostickers. Biosens. Bioelectron. 2022, 214, 114525. [Google Scholar] [CrossRef]
- Kearns, H.; Goodacre, R.; Jamieson, L.E.; Graham, D.; Faulds, K. SERS Detection of Multiple Antimicrobial-Resistant Pathogens Using Nanosensors. Anal. Chem. 2017, 89, 12666–12673. [Google Scholar] [CrossRef] [Green Version]
- Gracie, K.; Correa, E.; Mabbott, S.; Dougan, J.A.; Graham, D.; Goodacre, R.; Faulds, K. Simultaneous Detection and Quantification of Three Bacterial Meningitis Pathogens by SERS. Chem. Sci. 2014, 5, 1030–1040. [Google Scholar] [CrossRef] [Green Version]
- Mosier-Boss, P.A.; Sorensen, K.C.; George, R.D.; Obraztsova, A. SERS Substrates Fabricated Using Ceramic Filters for the Detection of Bacteria. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2016, 153, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Walter, A.; März, A.; Schumacher, W.; Rösch, P.; Popp, J. Towards a Fast, High Specific and Reliable Discrimination of Bacteria on Strain Level by Means of SERS in a Microfluidic Device. Lab Chip 2011, 11, 1013–1021. [Google Scholar] [CrossRef]
- Wu, X.; Xu, C.; Tripp, R.A.; Huang, Y.W.; Zhao, Y. Detection and Differentiation of Foodborne Pathogenic Bacteria in Mung Bean Sprouts Using Field Deployable Label-Free SERS Devices. Analyst 2013, 138, 3005–3012. [Google Scholar] [CrossRef] [PubMed]
- Ankamwar, B.; Sur, U.K.; Das, P. SERS Study of Bacteria Using Biosynthesized Silver Nanoparticles as the SERS Substrate. Anal. Methods 2016, 8, 2335–2340. [Google Scholar] [CrossRef]
- Ciloglu, F.U.; Caliskan, A.; Saridag, A.M.; Kilic, I.H.; Tokmakci, M.; Kahraman, M.; Aydin, O. Drug-Resistant Staphylococcus Aureus Bacteria Detection by Combining Surface-Enhanced Raman Spectroscopy (SERS) and Deep Learning Techniques. Sci. Rep. 2021, 11, 18444. [Google Scholar] [CrossRef]
- Nerve Agents, BBC Report. Available online: https://www.bbc.com/news/uk-43431537 (accessed on 30 January 2023).
- Lister, A.P.; Sellors, W.J.; Howle, C.R.; Mahajan, S. Raman Scattering Techniques for Defense and Security Applications. Anal. Chem. 2021, 93, 417–429. [Google Scholar] [CrossRef]
- Sadayoshi, O.H.B.U.; Yamashina, A.; Takasu, N.; Yamaguchi, T.; Murai, T.; Nakano, K.; Hinohara, S. Sarin poisoning on Tokyo subway. South. Med. J. 1997, 90, 587–593. [Google Scholar]
- Inscore, F.E.; Gift, A.D.; Maksymiuk, P.; Farquharson, S. Characterization of Chemical Warfare G-Agent Hydrolysis Products by Surface-Enhanced Raman Spectroscopy. Chem. Biol. Point Sens. Homel. Def. II 2004, 5585, 46. [Google Scholar] [CrossRef]
- Mukherjee, S.; Gupta, R.D. Organophosphorus Nerve Agents: Types, Toxicity, and Treatments. J. Toxicol. 2020, 2020, 3007984. [Google Scholar] [CrossRef]
- Saylan, Y.; Akgönüllü, S.; Denizli, A. Plasmonic Sensors for Monitoring Biological and Chemical Threat Agents. Biosensors 2020, 10, 142. [Google Scholar] [CrossRef] [PubMed]
- Primera-Pedrozo, O.M.; Jerez-Rozo, J.I.; De La Cruz-Montoya, E.; Luna-Pineda, T.; Pacheco-Londoño, L.C.; Hernández-Rivera, S.P. Nanotechnology-Based Detection of Explosives and Biological Agents Simulants. IEEE Sens. J. 2008, 8, 963–973. [Google Scholar] [CrossRef]
- Yan, F.; Vo-Dinh, T. Surface-Enhanced Raman Scattering Detection of Chemical and Biological Agents Using a Portable Raman Integrated Tunable Sensor. Sens. Actuators B Chem. 2007, 121, 61–66. [Google Scholar] [CrossRef]
- Pearman, W.F.; Fountain, A.W. Classification of Chemical and Biological Warfare Agent Simulants by Surface-Enhanced Raman Spectroscopy and Multivariate Statistical Techniques. Appl. Spectrosc. 2006, 60, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Hakonen, A.; Rindzevicius, T.; Schmidt, M.S.; Andersson, P.O.; Juhlin, L.; Svedendahl, M.; Boisen, A.; Käll, M. Detection of Nerve Gases Using Surface-Enhanced Raman Scattering Substrates with High Droplet Adhesion. Nanoscale 2016, 8, 1305–1308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Juhlin, L.; Mikaelsson, T.; Hakonen, A.; Stenbæk, M. Selective Surface-Enhanced Raman Scattering Detection of Tabun, VX and Cyclosarin Nerve Agents Using 4-Pyridine Amide Oxime Functionalized Gold Nanopillars Talanta Selective Surface-Enhanced Raman Scattering Detection of Tabun, VX and Cyclosarin Nerve A. Talanta 2020, 211, 120721. [Google Scholar] [CrossRef]
- Farquharson, S.; Gift, A.; Maksymiuk, P.; Inscore, F. Surface-Enhanced Raman Spectra of VX and Its Hydrolysis Products. Appl. Spectrosc. 2005, 59, 654–660. [Google Scholar] [CrossRef]
- Heleg-Shabtai, V.; Sharabi, H.; Zaltsman, A.; Ron, I.; Pevzner, A. Surface-Enhanced Raman Spectroscopy (SERS) for Detection of VX and HD in the Gas Phase Using a Hand-Held Raman Spectrometer. Analyst 2020, 145, 6334–6341. [Google Scholar] [CrossRef]
- Zhao, Q.; Liu, G.; Zhang, H.; Zhou, F.; Li, Y.; Cai, W. SERS-Based Ultrasensitive Detection of Organophosphorus Nerve Agents via Substrate’s Surface Modification. J. Hazard. Mater. 2017, 324, 194–202. [Google Scholar] [CrossRef]
- Spencer, K.M.; Sylvia, J.M.; Clauson, S.L.; Janni, J.A. Surface-Enhanced Raman as a Water Monitor for Warfare Agents. Vib. Spectrosc. Sens. Syst. 2002, 4577, 158. [Google Scholar] [CrossRef]
- Bertone, J.F.; Cordeiro, K.L.; Sylvia, J.M.; Spencer, K.M. A Nanoengineered Sensor to Detect Vibrational Modes of Warfare Agents/Explosives Using Surface-Enhanced Raman Scattering. Sens. Command. Control. Commun. Intell. Technol. Homel. Secur. Homel. Def. III 2004, 5403, 387. [Google Scholar] [CrossRef]
- Kim, Y.T.; Kim, D.; Park, S.; Zhexembekova, A.; Byeon, M.; Hong, T.E.; Lee, J.; Lee, C.Y. Aqueous Microlenses for Localized Collection and Enhanced Raman Spectroscopy of Gaseous Molecules. Adv. Opt. Mater. 2021, 9, 2101209. [Google Scholar] [CrossRef]
- Wu, J.; Zhu, Y.; Gao, J.; Chen, J.; Feng, J.; Guo, L.; Xie, J. A Simple and Sensitive Surface-Enhanced Raman Spectroscopic Discriminative Detection of Organophosphorous Nerve Agents. Anal. Bioanal. Chem. 2017, 409, 5091–5099. [Google Scholar] [CrossRef] [PubMed]
- Lafuente, M.; Berenschot, E.J.W.; Tiggelaar, R.M.; Mallada, R.; Tas, N.R.; Pina, M.P. 3D Fractals as SERS Active Platforms: Preparation and Evaluation for Gas Phase Detection of G-Nerve Agents. Micromachines 2018, 9, 60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goel, A.K. Anthrax: A Disease of Biowarfare and Public Health Importance. World J. Clin. Cases 2015, 3, 20. [Google Scholar] [CrossRef] [PubMed]
- Farrell, M.E.; Pellegrino, P.M. Army Relevant Biological Hazards Detection with Commercial SERS Substrates. Biosen. Nanomed. V 2012, 8460, 84600J. [Google Scholar] [CrossRef]
- Sajanlal, P.R.; Pradeep, T. Functional Hybrid Nickel Nanostructures as Recyclable SERS Substrates: Detection of Explosives and Biowarfare Agents. Nanoscale 2012, 4, 3427–3437. [Google Scholar] [CrossRef]
- Wang, T.; Dong, P.; Zhu, C.; Sha, P.; Gao, W.; Wu, Y.; Wu, X. Trace Detection of Anthrax Protective Antigens via a Competitive Method Based on Surface-Enhanced Raman Scattering. Sens. Actuators B Chem. 2021, 346, 130467. [Google Scholar] [CrossRef]
- Gao, R.; Ko, J.; Cha, K.; Ho Jeon, J.; Rhie, G.E.; Choi, J.; de Mello, A.J.; Choo, J. Fast and Sensitive Detection of an Anthrax Biomarker Using SERS-Based Solenoid Microfluidic Sensor. Biosens. Bioelectron. 2015, 72, 230–236. [Google Scholar] [CrossRef]
- Naqvi, T.K.; Bajpai, A.; Bharati, M.S.S.; Kulkarni, M.M.; Siddiqui, A.M.; Soma, V.R.; Dwivedi, P.K. Ultra-Sensitive Reusable SERS Sensor for Multiple Hazardous Materials Detection on Single Platform. J. Hazard. Mater. 2021, 407, 124353. [Google Scholar] [CrossRef]
- Sengupta, A.; Shende, C.; Farquharson, S.; Inscore, F. Detection of Bacillus Anthracis Spores Using Peptide Functionalized SERS-Active Substrates. Int. J. Spectrosc. 2012, 2012, 176851. [Google Scholar] [CrossRef] [Green Version]
- Yilmaz, M.; Senlik, E.; Biskin, E.; Yavuz, M.S.; Tamer, U.; Demirel, G. Combining 3-D Plasmonic Gold Nanorod Arrays with Colloidal Nanoparticles as a Versatile Concept for Reliable, Sensitive, and Selective Molecular Detection by SERS. Phys. Chem. Chem. Phys. 2014, 16, 5563–5570. [Google Scholar] [CrossRef]
- Li, B.; Wang, T.; Bai, W.; Su, Q.; Wu, X.; Dong, P. Label-Free and Rapid Detection of Anthrax Protective Antigen by Surface-Enhanced Raman Scattering on Au Nanorods. IEEE Sens. J. 2021, 21, 18425–18434. [Google Scholar] [CrossRef]
- Cheung, M.; Lee, W.W.Y.; Cowcher, D.P.; Goodacre, R.; Bell, S.E.J. SERS of Meso-Droplets Supported on Superhydrophobic Wires Allows Exquisitely Sensitive Detection of Dipicolinic Acid, an Anthrax Biomarker, Considerably below the Infective Dose. Chem. Commun. 2016, 52, 9925–9928. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Félix-Rivera, H.; González, R.; Rodríguez, G.D.M.; Primera-Pedrozo, O.M.; Ríos-Velázquez, C.; Hernández-Rivera, S.P. Improving SERS Detection of Bacillus Thuringiensis Using Silver Nanoparticles Reduced with Hydroxylamine and with Citrate Capped Borohydride. Int. J. Spectrosc. 2011, 2011, 989504. [Google Scholar] [CrossRef] [Green Version]
- FountainIII, A.W.; Pearman, W.F. Multivariate Statistical Classification of Surface Enhanced Raman Spectra of Chemical and Biological Warfare Agent Simulants. Chem. Biol. Sens. Ind. Environ. Secur. 2005, 5994, 180–193. [Google Scholar] [CrossRef]
- Arano-Martinez, J.A.; Martínez-González, C.L.; Salazar, M.I.; Torres-Torres, C. A Framework for Biosensors Assisted by Multiphoton Effects and Machine Learning. Biosensors 2022, 12, 710. [Google Scholar] [CrossRef]
- Luo, R.; Popp, J.; Bocklitz, T. Deep Learning for Raman Spectroscopy: A Review. Analytica 2022, 3, 287–301. [Google Scholar] [CrossRef]
- Ralbovsky, N.M.; Lednev, I.K. Towards Development of a Novel Universal Medical Diagnostic Method: Raman Spectroscopy and Machine Learning. Chem. Soc. Rev. 2020, 49, 7428–7453. [Google Scholar] [CrossRef]
- Ding, Y.; Sun, Y.; Liu, C.; Jiang, Q.Y.; Chen, F.; Cao, Y. SERS-Based Biosensors Combined with Machine Learning for Medical Application. ChemistryOpen 2023, 12, e202200192. [Google Scholar] [CrossRef]
- Cui, F.; Yue, Y.; Zhang, Y.; Zhang, Z.; Zhou, H.S. Advancing Biosensors with Machine Learning. ACS Sens. 2020, 5, 3346–3364. [Google Scholar] [CrossRef]
- Lv, R.; Wang, Z.; Ma, Y.; Li, W.; Tian, J. Machine Learning Enhanced Optical Spectroscopy for Disease Detection. J. Phys. Chem. Lett. 2022, 13, 9238–9249. [Google Scholar] [CrossRef]
- Schackart, K.E.; Yoon, J.Y. Machine Learning Enhances the Performance of Bioreceptor-Free Biosensors. Sensors 2021, 21, 5519. [Google Scholar] [CrossRef]
- Lussier, F.; Missirlis, D.; Spatz, J.P.; Masson, J.F. Machine-Learning-Driven Surface-Enhanced Raman Scattering Optophysiology Reveals Multiplexed Metabolite Gradients Near Cells. ACS Nano 2019, 13, 1403–1411. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.H.L.; Hong, B.; Rubin, S.; Fainman, Y. Machine Learning for Composition Analysis of SsDNA Using Chemical Enhancement in SERS. Biomed. Opt. Express 2020, 11, 5092. [Google Scholar] [CrossRef] [PubMed]
- Narla, L.M.; Rao, S.V. Identification of Metals and Alloys Using Color CCD Images of Laser-Induced Breakdown Emissions Coupled with Machine Learning. Appl. Phys. B Lasers Opt. 2020, 126, 113. [Google Scholar] [CrossRef]
- Beeram, R.; Banerjee, D.; Narlagiri, L.M.; Soma, V.R. Machine Learning for Rapid Quantification of Trace Analyte Molecules Using SERS and Flexible Plasmonic Paper Substrates. Anal. Methods 2022, 14, 1788–1796. [Google Scholar] [CrossRef]
- Murthy, N.L.; Abdul Salam, S.; Rao, S.V. Stand-off Femtosecond Laser Induced Breakdown Spectroscopy of Metals, Soil, Plastics and Classification Studies. In Proceedings of the 2019 Workshop on Recent Advances in Photonics (WRAP), Guwahati, India, 13–14 December 2019; pp. 1–3. [Google Scholar] [CrossRef]
- Boehmke, B.; Greenwell, B. Hands-On Machine Learning with R; Chapman and Hall/CRC: Boca Raton, FL, USA, 2019; ISBN 9781492032649. [Google Scholar]
- Li, D.; Zhang, Q.; Deng, B.; Chen, Y.; Ye, L. Rapid, Sensitive Detection of Ganciclovir, Penciclovir and Valacyclovir-Hydrochloride by Artificial Neural Network and Partial Least Squares Combined with Surface Enhanced Raman Spectroscopy. Appl. Surf. Sci. 2021, 539, 148224. [Google Scholar] [CrossRef]
- Boulesteix, A.L.; Strimmer, K. Partial Least Squares: A Versatile Tool for the Analysis of High-Dimensional Genomic Data. Brief. Bioinform. 2007, 8, 32–44. [Google Scholar] [CrossRef] [Green Version]
- Deng, W.; Huang, Z.; Zhang, J.; Xu, J. A Data Mining Based System for Transaction Fraud Detection. In Proceedings of the 2021 IEEE International Conference on Consumer Electronics and Computer Engineering (ICCECE), Guangzhou, China, 15–17 January 2021; pp. 542–545. [Google Scholar] [CrossRef]
- Fan, X.; Ming, W.; Zeng, H.; Zhang, Z.; Lu, H. Deep Learning-Based Component Identification for the Raman Spectra of Mixtures. Analyst 2019, 144, 1789–1798. [Google Scholar] [CrossRef]
- Zhou, H.; Xu, L.; Ren, Z.; Zhu, J.; Lee, C. Machine Learning-Augmented Surface-Enhanced Spectroscopy toward next-Generation Molecular Diagnostics. Nanoscale Adv. 2023, 5, 538–570. [Google Scholar] [CrossRef]
- Malinick, A.S.; Stuart, D.D.; Lambert, A.S.; Cheng, Q. Surface Plasmon Resonance Imaging (SPRi) in Combination with Machine Learning for Microarray Analysis of Multiple Sclerosis Biomarkers in Whole Serum. Biosens. Bioelectron. X 2022, 10, 100127. [Google Scholar] [CrossRef]
- Pradhan, P.; Guo, S.; Ryabchykov, O.; Popp, J.; Bocklitz, T.W. Deep Learning a Boon for Biophotonics? J. Biophotonics 2020, 13, e201960186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moon, G.; Lee, J.; Lee, H.; Yoo, H.; Ko, K.; Im, S.; Kim, D. Machine Learning and Its Applications for Plasmonics in Biology. Cell Rep. Phys. Sci. 2022, 3, 101042. [Google Scholar] [CrossRef]
- Sun, Y.; Shi, L.; Mi, L.; Guo, R.; Li, T. Recent Progress of SERS Optical Nanosensors for MiRNA Analysis. J. Mater. Chem. B 2020, 8, 5178–5183. [Google Scholar] [CrossRef] [PubMed]
- Raji, H.; Tayyab, M.; Sui, J.; Mahmoodi, S.R.; Javanmard, M. Biosensors and Machine Learning for Enhanced Detection, Stratification, and Classification of Cells: A Review. Biomed. Microdevices 2022, 24, 26. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Maity, S.; Mastrangelo, C.H. Nanostructures for biosensing, with a brief overview on Cancer Detection, IoT, and the Role of Machine Learning In Smart Biosensors. Sensors 2021, 21, 1253. [Google Scholar] [CrossRef]
- Beeram, R.; Vendamani, V.S.; Soma, V.R. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy Deep Learning Approach to Overcome Signal Fluctuations in SERS for Efficient On-Site Trace Explosives Detection. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2023, 289, 122218. [Google Scholar] [CrossRef] [PubMed]
- Moon, G.; Son, T.; Lee, H.; Kim, D. Deep Learning Approach for Enhanced Detection of Surface Plasmon Scattering. Anal. Chem. 2019, 91, 9538–9545. [Google Scholar] [CrossRef]
- Gupta, A.K.; Hsu, C.H.; Lai, C.S. Enhancement of the Au/ZnO-NA Plasmonic SERS Signal Using Principal Component Analysis as a Machine Learning Approach. IEEE Photonics J. 2020, 12, 1–11. [Google Scholar] [CrossRef]
- Vendamani, V.S.; Beeram, R.; Neethish, M.M.; Rao, S.V.S.N.; Rao, S.V. Wafer-Scale Silver Nanodendrites with Homogeneous Distribution of Gold Nanoparticles for Biomolecules Detection. iScience 2022, 25, 104849. [Google Scholar] [CrossRef]
- Erzina, M.; Trelin, A.; Guselnikova, O.; Dvorankova, B.; Strnadova, K.; Perminova, A.; Ulbrich, P.; Mares, D.; Jerabek, V.; Elashnikov, R.; et al. Precise Cancer Detection via the Combination of Functionalized SERS Surfaces and Convolutional Neural Network with Independent Inputs. Sens. Actuators B Chem. 2020, 308, 127660. [Google Scholar] [CrossRef]
- Wang, S.; Dong, H.; Shen, W.; Yang, Y.; Li, Z.; Liu, Y.; Wang, C.; Gu, B.; Zhang, L. Rapid SERS Identification of Methicillin-Susceptible and Methicillin-Resistant: Staphylococcus Aureus via Aptamer Recognition and Deep Learning. RSC Adv. 2021, 11, 34425–34431. [Google Scholar] [CrossRef]
- Kazemzadeh, M.; Hisey, C.L.; Dauros-Singorenko, P.; Swift, S.; Zargar-Shoshtari, K.; Xu, W.; Broderick, N.G.R. Label-Free Classification of Bacterial Extracellular Vesicles by Combining Nanoplasmonic Sensors with Machine Learning. IEEE Sens. J. 2022, 22, 1128–1137. [Google Scholar] [CrossRef]
- Dong, R.; Weng, S.; Yang, L.; Liu, J. Detection and Direct Readout of Drugs in Human Urine Using Dynamic Surface-Enhanced Raman Spectroscopy and Support Vector Machines. Anal. Chem. 2015, 87, 2937–2944. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Liang, S.; Li, Y.; Peng, Y.; Huang, Z.; Li, Z.; Yang, Y.; Luo, X. Localized Plasmonic Sensor for Direct Identifying Lung and Colon Cancer from the Blood. Biosens. Bioelectron. 2022, 211, 114372. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Lu, D.; Zhang, B.; You, R.; Chen, J.; Xu, H.; Lu, Y. Machine Learning—Assisted Internal Standard Calibration Label—Free SERS Strategy for Colon Cancer Detection. Anal. Bioanal. Chem. 2023. [Google Scholar] [CrossRef] [PubMed]
- Seifert, S.; Merk, V.; Kneipp, J. Identification of Aqueous Pollen Extracts Using Surface Enhanced Raman Scattering (SERS) and Pattern Recognition Methods. J. Biophotonics 2016, 9, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Hassoun, M.; Rüger, J.; Kirchberger-Tolstik, T.; Schie, I.W.; Henkel, T.; Weber, K.; Cialla-May, D.; Krafft, C.; Popp, J. A Droplet-Based Microfluidic Chip as a Platform for Leukemia Cell Lysate Identification Using Surface-Enhanced Raman Scattering. Anal. Bioanal. Chem. 2018, 410, 999–1006. [Google Scholar] [CrossRef] [PubMed]
- Mühlig, A.; Bocklitz, T.; Labugger, I.; Dees, S.; Henk, S.; Richter, E.; Andres, S.; Merker, M.; Stöckel, S.; Weber, K.; et al. LOC-SERS: A Promising Closed System for the Identification of Mycobacteria. Anal. Chem. 2016, 88, 7998–8004. [Google Scholar] [CrossRef] [PubMed]
- Bratchenko, L.A.; Al-Sammarraie, S.Z.; Tupikova, E.N.; Konovalova, D.Y.; Lebedev, P.A.; Zakharov, V.P.; Bratchenko, I.A. Analyzing the Serum of Hemodialysis Patients with End-Stage Chronic Kidney Disease by Means of the Combination of SERS and Machine Learning. Biomed. Opt. Express 2022, 13, 4926. [Google Scholar] [CrossRef] [PubMed]
- Gao, K.; Zhu, H.; Charron, B.; Mochizuki, T.; Dong, C.; Ding, H.; Cui, Y.; Lu, M.; Peng, W.; Zhu, S.; et al. Combining Dense Au Nanoparticle Layers and 2D Surface-Enhanced Raman Scattering Arrays for the Identification of Mutant Cyanobacteria Using Machine Learning. J. Phys. Chem. C 2022, 126, 9446–9455. [Google Scholar] [CrossRef]
- Ikponmwoba, E.; Ukorigho, O.; Moitra, P.; Pan, D.; Gartia, M.R.; Owoyele, O. A Machine Learning Framework for Detecting COVID-19 Infection Using Surface-Enhanced Raman Scattering. Biosensors 2022, 12, 589. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Jiménez, A.I.; Lyu, D.; Lu, Z.; Liu, G.; Ren, B. Surface-Enhanced Raman Spectroscopy: Benefits, Trade-Offs and Future Developments. Chem. Sci. 2020, 11, 4563–4577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hou, M.; Huang, Y.; Ma, L.; Zhang, Z. Quantitative Analysis of Single and Mix Food Antiseptics Basing on SERS Spectra with PLSR Method. Nanoscale Res. Lett. 2016, 11, 296. [Google Scholar] [CrossRef] [Green Version]
- Yan, S.; Liu, C.; Fang, S.; Ma, J.; Qiu, J.; Xu, D.; Li, L.; Yu, J.; Li, D.; Liu, Q. SERS-Based Lateral Flow Assay Combined with Machine Learning for Highly Sensitive Quantitative Analysis of Escherichia coli O157:H7. Anal. Bioanal. Chem. 2020, 412, 7881–7890. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.Q.; Thrift, W.J.; Bhattacharjee, A.; Ranjbar, S.; Gallagher, T.; Darvishzadeh-Varcheie, M.; Sanderson, R.N.; Capolino, F.; Whiteson, K.; Baldi, P.; et al. Longitudinal Monitoring of Biofilm Formation via Robust Surface-Enhanced Raman Scattering Quantification of Pseudomonas Aeruginosa -Produced Metabolites. ACS Appl. Mater. Interfaces 2018, 10, 12364–12373. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.M.; Herrman, T.J. Determination and Prediction of Fumonisin Contamination in Maize by Surface–Enhanced Raman Spectroscopy (SERS). Food Bioprocess Technol. 2016, 9, 588–603. [Google Scholar] [CrossRef]
- Kuligowski, J.; El-Zahry, M.R.; Sánchez-Illana, Á.; Quintás, G.; Vento, M.; Lendl, B. Surface Enhanced Raman Spectroscopic Direct Determination of Low Molecular Weight Biothiols in Umbilical Cord Whole Blood. Analyst 2016, 141, 2165–2174. [Google Scholar] [CrossRef] [Green Version]
- Tan, A.; Zhao, Y.; Sivashanmugan, K.; Squire, K.; Wang, A.X. Quantitative TLC-SERS Detection of Histamine in Seafood with Support Vector Machine Analysis. Food Control 2019, 103, 111–118. [Google Scholar] [CrossRef] [Green Version]
- Rahman, A.; Kang, S.; Wang, W.; Huang, Q.; Kim, I.; Vikesland, P.J. Lectin-Modified Bacterial Cellulose Nanocrystals Decorated with Au Nanoparticles for Selective Detection of Bacteria Using Surface-Enhanced Raman Scattering Coupled with Machine Learning. ACS Appl. Nano Mater. 2022, 5, 259–268. [Google Scholar] [CrossRef]
- Banaei, N.; Moshfegh, J.; Mohseni-Kabir, A.; Houghton, J.M.; Sun, Y.; Kim, B. Machine Learning Algorithms Enhance the Specificity of Cancer Biomarker Detection Using SERS-Based Immunoassays in Microfluidic Chips. RSC Adv. 2019, 9, 1859–1868. [Google Scholar] [CrossRef] [Green Version]
- Cheng, N.; Chen, D.; Lou, B.; Fu, J.; Wang, H. A Biosensing Method for the Direct Serological Detection of Liver Diseases by Integrating a SERS-Based Sensor and a CNN Classifier. Biosens. Bioelectron. 2021, 186, 113246. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Liang, B.; Zhang, J.; Hao, X.; Xu, X.; Chang, H.M.; Leung, P.C.K.; Tan, J. Raman Spectroscopy of Follicular Fluid and Plasma with Machine-Learning Algorithms for Polycystic Ovary Syndrome Screening. Mol. Cell. Endocrinol. 2021, 523, 111139. [Google Scholar] [CrossRef] [PubMed]
- Barucci, A.; D’Andrea, C.; Farnesi, E.; Banchelli, M.; Amicucci, C.; De Angelis, M.; Hwang, B.; Matteini, P. Label-Free SERS Detection of Proteins Based on Machine Learning Classification of Chemo-Structural Determinants. Analyst 2021, 146, 674–682. [Google Scholar] [CrossRef]
- Kazemzadeh, M.; Hisey, C.L.; Zargar-Shoshtari, K.; Xu, W.; Broderick, N.G.R. Deep Convolutional Neural Networks as a Unified Solution for Raman Spectroscopy-Based Classification in Biomedical Applications. Opt. Commun. 2022, 510, 127977. [Google Scholar] [CrossRef]
- Othman, N.H.; Yoot Lee, K.; Mohd Radzol, A.R.; Mansor, W.; Amanina Yusoff, N. PCA-Polynomial-ELM Model Optimal for Detection of NS1 Adulterated Salivary SERS Spectra. J. Phys. Conf. Ser. 2019, 1372, 012064. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, B.; Haverstick, J.; Ibtehaz, N.; Muszyński, A.; Chen, X.; Chowdhury, M.E.H.; Zughaier, S.; Zhao, Y. Differentiation and Classification of Bacterial Endotoxins Based on Surface Enhanced Raman Scattering and Advanced Machine Learning. Nanoscale 2022, 14, 8806–8817. [Google Scholar] [CrossRef]
- Lin, D.; Hsieh, C.L.; Hsu, K.C.; Liao, P.H.; Qiu, S.; Gong, T.; Yong, K.T.; Feng, S.; Kong, K.V. Geometrically Encoded SERS Nanobarcodes for the Logical Detection of Nasopharyngeal Carcinoma-Related Progression Biomarkers. Nat. Commun. 2021, 12, 3430. [Google Scholar] [CrossRef]
- Wang, G.; Lipert, R.J.; Jain, M.; Kaur, S.; Chakraboty, S.; Torres, M.P.; Batra, S.K.; Brand, R.E.; Porter, M.D. Detection of the Potential Pancreatic Cancer Marker MUC4 in Serum Using Surface-Enhanced Raman Scattering. Anal. Chem. 2011, 83, 2554–2561. [Google Scholar] [CrossRef] [Green Version]
- Lu, L.; Guan, S.; Guan, Y.; Hong, M. Dual-Modal Fluorescence-SERS Detection of Blood Glucose Engineered by Hierarchical Laser-Induced Micro/Nano Structures for Diabetes Screening. Adv. Mater. Interfaces 2022, 9, 2102532. [Google Scholar] [CrossRef]
- Sun, J.; Gong, L.; Wang, W.; Gong, Z.; Wang, D.; Fan, M. Surface-Enhanced Raman Spectroscopy for on-Site Analysis: A Review of Recent Developments. Luminescence 2020, 35, 808–820. [Google Scholar] [CrossRef] [PubMed]
- Kho, K.W.; Fu, C.Y.; Dinish, U.S.; Olivo, M. Clinical SERS: Are We There Yet? J. Biophotonics 2011, 4, 667–684. [Google Scholar] [CrossRef] [PubMed]











Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beeram, R.; Vepa, K.R.; Soma, V.R. Recent Trends in SERS-Based Plasmonic Sensors for Disease Diagnostics, Biomolecules Detection, and Machine Learning Techniques. Biosensors 2023, 13, 328. https://doi.org/10.3390/bios13030328
Beeram R, Vepa KR, Soma VR. Recent Trends in SERS-Based Plasmonic Sensors for Disease Diagnostics, Biomolecules Detection, and Machine Learning Techniques. Biosensors. 2023; 13(3):328. https://doi.org/10.3390/bios13030328
Chicago/Turabian StyleBeeram, Reshma, Kameswara Rao Vepa, and Venugopal Rao Soma. 2023. "Recent Trends in SERS-Based Plasmonic Sensors for Disease Diagnostics, Biomolecules Detection, and Machine Learning Techniques" Biosensors 13, no. 3: 328. https://doi.org/10.3390/bios13030328