1. Introduction
The relationship between natural and cultural landscapes becomes tangible in material culture through the manyfold technological solutions to production aims. It is a continuous dynamic interplay, where landscapes influence technology and, in turn, technology shapes the landscapes. From the end of the 18th century, significant consequences have resulted from the use of fossil fuels and the shift from artisanal to industrial production, which caused the persistent increase in the CO
2 concentration in the atmosphere. Such a human impact on the Earth’s natural systems is proposed to be recognized by the introduction of a new epoch: Anthropocene [
1,
2].
In this framework, the rapid growth of urban areas for the past three centuries has led to a significant increase in the production of lime and cement for construction and infrastructure, further increasing the CO2 emissions in the atmosphere, generated by the calcination of carbonates present in the raw materials. The rate of this escalation was locally different but contributed overall to the departure from the natural equilibrium of CO2 concentration in the atmosphere, leading to the current state of global warming and climate change.
As for the cultural landscape, the transition from stones, bricks and lime mortars to reinforced concrete technology gradually occurred between the second half of the 19th century and the first half of the 20th century [
3,
4]. This transition not only had an environmental impact but also had consequences on the material culture, since the new generations of artisans lost the knowledge of the lime’s production cycle, and progressively learned to handle the new material [
5].
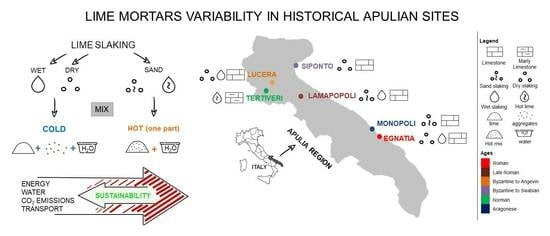
Insights on the evolution in time and space of the combination of natural and cultural aspects in traditional building materials in Apulia are provided by the historical sources and the archaeological and archaeometrical literature.
The carbonatic geological substratum characterizes the landscape and the material culture of Apulia. The large availability of limestone outcropping throughout the territory has promoted its use as raw material both as building and ornamental stone over time.
In addition, a parallel exploitation of limestone is documented for mortars used for building and artistic purposes. Previous studies have outlined the Apulian tradition in lime production [
6,
7,
8,
9,
10,
11,
12]. An idea of the impact of lime production in the economy of Apulia in the beginning of the 20th century is provided by the Regional Annals of Industrial Statistics that reports 248 active lime kilns in 1903 [
12].
Although the calcination and following slaking of pure limestone supply an aerial calcium binder, the local tradition is characterized by the use of technological expedients and technical choices that successfully attempt to overcome the lack of hydraulic elements in the largely available limestone.
The most widespread evidence of this trend is recorded in mortars of some archaeological and historical-artistic sites in the region, where the hydraulic character is ensured by the presence of cocciopesto [
13,
14,
15] or sandy clays [
16].
Less frequent in Apulia are examples of the use of pozzolanic sand rich in volcanic ash [
17], clayed sediments [
18] and
terra rossa (residual karst clay) [
19], all composed of aluminosilicates which confer hydraulic features to the mortar.
In the field of historical buildings, the wide use of lime as a binder in various contexts of application emerges from a series of case studies covering a chronological span from the 4th century BCE to the 15th century CE.
In this article, a selection of case studies about lime mortars from different sites and periods in Apulia were taken as examples of the complexity of lime mortar technology in the past and this poses insights and comparisons with current design trends for new inorganic binders.
In order to make the discussion of technological aspects clearer, a geological overview of Apulia and a presentation of the case studies are provided, as well as a recall of the production cycle of lime.
2. Geological Background
The Apulia (southern Italy) region (
Figure 1) is characterized by a lithological variety connected to its geological evolution, as it represents the foreland area of the southern Apennines chain system [
20,
21] and includes three geological domains: Avampaese Apulo (Gargano promontory, Murge plateau and Serre Salentine), Bradanic through (Tavoliere delle Puglie plain and Premurgian valley) and southern Apennines chain (Daunian mountains) [
21].
The region is characterized by a 6 km thick sedimentary succession consisting of Mesozoic shallow-water limestones and dolostones [
21] and Cenozoic and Neozoic deposits composed of carbonate rocks cropping out in tectonic depressions along the margins of the three main blocks, Gargano, Murge and Salento [
22,
23,
24,
25].
The Mesozoic sedimentary succession is made up of fine-grained limestones and dolomites including intercalations of red and grey clays. The Mesozoic shallow-water succession is composed of the Calcare di Bari and Calcare di Altamura Fms [
26] cropping out in the Gargano promontory and in the Murge and Salento areas.
The Cenozoic and Neozoic deposits are represented by an Oligo-Miocene open-shelf carbonate unit cropping out in Gargano and Salento covered by a Middle Pliocene-Lower Pleistocene shallow-marine carbonate unit which crops out at the margin of Gargano, Murge and Salento [
27,
28] in turn overlapped by Middle-Upper Pleistocene shallow marine carbonate and a mixed unit cropping out at the margin of Murge and Salento [
22]. The Oligo-Miocene unit is composed of biocalcarenites and biocalcirudites belonging to the Calcareniti di Porto Badisco (Late Oligocene), Pietra leccese (Late Burdigalian-Lower Messinian) and Calcareniti di Andrano (Late Miocene) Formations [
29,
30,
31]. The Middle Pliocene-Lower Pleistocene unit consists of calcarenites and calcirudites referred to the Calcarenite di Gravina Fm [
32], whereas the Middle-Upper Pleistocene unit is composed of mixed sands (Argille Subappennine) and calcarenites (Calcarenite di Gravina Fm) belonging to the Terraced Marine Deposits Fm [
22].
Figure 1.
Simplified geological map of Apulia and positions of the sites discussed in the text [
33], modified after Pieri et al. [
23].
Figure 1.
Simplified geological map of Apulia and positions of the sites discussed in the text [
33], modified after Pieri et al. [
23].
In the Gargano promontory, the Apulian carbonate platform consists of meso-cenozoic stratigraphic units deposited in different carbonate depositional environments, from shallow water to basin [
29,
33,
34,
35]. Several Cretaceous slope and basinal units such as the Maiolica Fm, Mattinata Fm and Scaglia Fm, and middle Eocene Peschici Fm formations contain nodular cherts exploited in prehistory [
36].
The outer sector of the Southern Apennines is made up of Langhian-Messinian turbidites of the Irpinian basin (i.e., Serra Palazzo Fm, Faeto Flysch Fm, and Marne argillose del Toppo Capuana Fm) and constitutes the Daunia tectonic unit formations [
37,
38,
39,
40].
The lower member of the Serra Palazzo Fm is composed of coarse grained siliciclastic turbidites (upper Burdigalian-Langhian), with intercalations of pelite. The upper member (Serravallian) is constituted by fine grained turbidites of calcareous and siliciclastic composition, with interbedded emipelagic limestones and marls [
40]. The Faeto Flysch Fm (Langhian–Serravallian), consists of beds and banks of calcarenites, calcirudites, calcilutites, calcareous marls and marly clays, conformably lying on the Lagonegro units (Cretaceous–Early Burdigalian). The dark gray, marls and marly clay of the Toppo Capuana Fm (Late Tortonian–Early Messinian) are the upper part of the succession [
40].
In this work, case studies relative to different geological substrata were considered (
Table 1), to highlight the technological differences induced by the availability of raw materials. Egnazia and Monopoli [
41] are both located on the Adriatic coast of Apulia, on regressive calcarenites of the Pleistocene age, lying unconformably on the Calcare di Bari Fm (Cenomanian-Turonian). The necropolis of Lamapopoli lies SE of Canosa di Puglia on the northern border of Murge plateau, close to the Ofanto river valley. Catacombs were dug into the upper portion of the Calcarenite di Gravina Fm (Plio-Plistocene), also in this case unconformably overlying the Calcare di Bari Fm [
42]. Moving toward the north, Siponto is placed on the marine terraced deposits of the Gulf of Manfredonia (
Figure 1) and is close to the outcrop of Calcarenite di Gravina Fm (Pliocene) and the Calcare di Bari Fm (Kimmeridgian-Titonian) [
43,
44]. The sites of Lucera and Tertiveri occur on the top of hills in the Tavoliere plain, where the fluvial deposits of Sintema di Lucera Fm (Pleistocene) crop out [
45].
3. Lime Technology
The functioning of lime as a binder is the result of the transformation of carbonate rocks through a production process based on three main steps: calcination, hydration and carbonation.
Although the traditional process seems to be theoretically simple and one-sided, each of these transformation steps can undergo different changes and the production cycle can be diverted to obtain specific features of the finished product (
Figure 2), a reason that justifies the large variability of lime used throughout the centuries [
46,
47,
48,
49].
3.1. Raw Materials
The first action is the choice of raw material to be calcined, as the technological characteristics of the finished product mainly depend on it. In the case of extremely pure limestones (CaCO
3 + MgCO
3 > 95 wt%), pure air lime (CL) can be obtained with the maximum flexibility in use (
Figure 2).
If the raw material is a carbonate rock which contains or is composed exclusively of dolomite (dolostone), its transformation is due to a different reaction and the finished product is a lime with variable composition and providing weak hydraulic properties [
47]. The proportion of MgCO
3 in limestone is used today to classify the different types of magnesian limestone [
47,
50]. The use of magnesian lime (DL) is attested in the past, demonstrated by the presence of hydromagnesite as a hardening product [
5,
51].
When the choice of the raw material falls on marly limestone composed of 6.5–20 wt% clays, the production process is obviously characterized by different temperatures and times and the finished product is natural hydraulic lime (NHL) with particular hardening properties under water [
52,
53,
54]. For natural hydraulic lime is intended a binder obtained from the calcination of a siliceous or marly limestone in a temperature range between 900 and 1250 °C. Artificial hydraulic limes are obtained by mixing together air (CL) or natural hydraulic lime (NHL) and aggregates with pozzolanic activity (e.g., pozzolana, cocciopesto, glass, clays, plant ash). According to the modern classification, non-natural hydraulic limes used in preindustrial times could be considered as formulated limes (FL) [
55].
3.2. Calcination
The activation of the binding properties of lime occurs via calcination, i.e., the endothermic decomposition of carbonates (1). Numerous studies have demonstrated that calcite decomposes at a distinct border between the CaCO
3 and CaO phases and that this boundary advances steadily in the direction of the particle’s center. This entails the topotactic formation of nanometric CaO crystals and the collapse of the calcite crystal structure occurs in various stages [
53,
56,
57,
58].
Heat is first transferred to the surface of calcite particles by radiation and convection. Heating of calcite particles first produces a reduction in crystalline size, which is a precursor to decomposition. Next, heat is transferred through the layer of CaO that has already separated to the reaction zone. This is followed by the breakdown of carbonates on the reaction surface, which produces CaO and CO
2. Finally, CO
2 diffuses from the porous layer of CaO to the particle’s exterior surface, reaching the kiln’s atmosphere [
53].
At 1 atm of pressure, the transformation of calcite to CaO starts at 600 °C and the reaction takes place between 800 and 850 °C through a topotactic mechanism [
56].
Calcination occurred in preindustrial times with permanent or temporary structures. A vertical kiln, which is technically a shaft furnace, is a more effective way to calcinate limestone than a wind-blown wood fire or a typical pottery kiln heated at 900–1000 °C [
59]. An elongated vertical shape would have promoted a natural draft and better heat distribution in the kiln. The operation of the kiln lasted several days, recharging the combustible.
Periodic kilns were the rule in preindustrial times, with different variants documented in time and space [
12,
48,
60].
A simple and traditional method involves limestone blocks stacked in a shallow pit in a large mound or “clamp” with alternate layers of fuel (such as wood or coal). The entire structure was then covered with clay or turf to hinder heat loss. This process was less controlled than kiln burning and often resulted in lower-quality lime.
In flare kilns, a cylindrical structure contained a stack of limestone arranged above a domed vault with a firing chamber at the base. The fuel was lit at the bottom, and the heat rose through the stack, calcining the limestone. The process was more controlled than clamp kilns, allowing for better-quality quicklime. Cato in early Roman times (165 BCE) described how to build such a kiln.
Overburning of the lime at the bottom of the load would be unavoidable, with consequent formation overburnt relics (see below). If the temperature/time curve of the calcination is not sufficient to decompose calcite, lime nodules made of unreacted calcite core forms.
3.3. Lime Hydration
The next step of the production cycle is slaking, i.e., exothermic hydration of quicklime (2). The methods for slaking lime differs based on the lime’s composition and intended application. Once the calcined stones cool and are extracted from the burning structure, hydration begins with the steam present in the air and start to swell. Slaking may occur in three main ways: (1) in an excess of water; (2) in a shortfall of water; (3) in combination with aggregate and water [
61]. In the first two cases the hydration reaction occurs before the mix with aggregate (cold mix), whereas in the third case the reaction unfolds during the mix preparation (hot mix, see below).
Overburnt or underburnt limestone relics are difficult to hydrate and should be removed before slaking.
When quicklime is hydrated in excess of water, one part of it is spread in a basin and mixed with two to three parts of water. To control the rate of the reaction, water can be added gradually or cover the mix to retain heat. Generally, the reaction takes some minutes to a few tens of minutes. In the traditional production process, a variable excess of water within the slaked lime is preserved to produce slaked lime, lime milk, lime water, as needed [
62].
To obtain lime in powder form, the following two ways are historically known: (1) dipping in water for few seconds a basket full of small pieces of quicklime; (2) spraying water on a spread mass of quicklime.
When the slaking involves quicklime also containing MgO (periclase) in turn produced by the calcination of magnesian limestones, the binder includes Mg(OH)
2 (brucite) and excess insoluble MgO together with Ca(OH)
2 (portlandite). Magnesian limes slake slower and require longer periods, have a less energetic slaking reaction, and expand less due to their higher magnesium content, compared to fat limes which double their volume during the process [
63].
In this phase, in case of hydraulic limes, the amount of water is extremely important as it should allow the quicklime to be hydrated without the occurrence of premature reactions with aluminates and silicates [
64].
In some European and North Italian regions, recurrent is the custom to avoid or limit the hydration of quicklime and to keep it away from moisture until its used and simultaneously slaked. Different hypotheses on traditional slaking methods have been advanced [
65,
66] and almost all of these considered the blending of sand aggregate and water in the hot lime to start the hydration reaction and to produce mortar concurrently.
The maturation of these products, which, as suggested by Roman literary sources and according to other authors [
67,
68], could even last several years to obtain a very performant lime. It should take place in excess of water conditions and lead to a significant improvement in the plasticity characteristics of lime, due to the gradual transformation of portlandite habitus, from prism to plate-like crystals [
67]. Maturation cannot be applied to natural hydraulic limes because of the presence of hydraulic phases, which will harden in wet conditions [
47].
3.4. Mixing
The volume is typically used to quantify the components of the mixture. The required volume of lime for a given volume of aggregate is the basis for mix calculations. Ideally, the minimum amount of lime should be that which fills the pores between the aggregate’s grains. An insufficient lime/aggregate ratio will yield a harsh and unworkable mortar. To add water to improve workability, it will result in a weak mix, susceptible to shrinking.
Common on-site methods for preparing mortar include cold mixing, which involves slaking the lime content before mixing, avoiding the use of heat generated by slaking (cold mix), and using lime putty or hydrated lime powder.
Cold mixing involves the preparation of a lime putty or a powder blended with aggregates of different sorts and textures [
53,
69]. The grain size distribution and the morphometry required for a mortar is largely dependent on the intended mortar function. Studies show that mortars with a lower lime content have lower mechanical strength, while the richest (1:1 and 1:2) mortars may have higher open porosity and capillary coefficients in the short term (90 days). However, 1:1 mortars may achieve higher strength in the long term. Open porosity is variable, depending on kneading water and sand grain size distribution, but the critical pore size is consistent between 0.5 and 1 mm [
70,
71,
72].
Higher mixing water content and/or higher contents of lime may increase drying shrinkage in the first few hours. However, low binder contents may also result in higher shrinkage due to low deformation resistance [
47].
As in quicklime slaking, when quicklime is mixed with aggregate and water, the water/solid ratio will give different characteristics to the material. Pavia et al., in 2023 [
63], distinguished wet slaking (hot lime) from dry slaking (sand slaking). In the first case, the mortar is mixed with sand and quicklime, then water is added. Sufficient water is needed to prevent overheating, but too much can cause overwetting. The mortar can be used while it is still hot or stored for later use. In the second case, quicklime and wet sand are mixed and stored to slake, removing moisture from the sand and air. The dry mix is sieved to remove lime particles, and then mixed with water to create a mortar. The mortar is covered with waterproof sheeting and left to mature before use [
63].
Hot-mixed mortars are generally richer in lime than cold mixes due to the quantity of quicklime needed. A mix ratio of 1:3 quicklime and aggregates (B/A) will produce, after expansion, a mortar that is 1:1.4 or even 1:1. Many historic mortar samples that have been analyzed show a similar ratio with a high binder content. It would not have been possible to make such a rich combination using lime putty since the mortar would have been slinky. Several experimental works showed the variability of B/A ratios between 1:5 and 1:13 in hot-lime mortars [
63,
66].
The initial slaking process heats and dries sand grains, forming a good bond with lime paste. The temperature reached during this process depends on several factors, including the specific type of lime, the amount of water used, and the mixing conditions. A temperature increase of approximately 60 °C, or even higher, together with the high pH of the mix promote the pozzolanic reaction between lime and hydraulic materials, when present. Steam generated during the slaking process entrains air and improves pore interconnectivity, which facilitate moisture transfer and accommodate freeze/thaw cycles, enhancing durability [
63,
65,
66,
73].
Dolomitic lime in mortars typically increases mixing water demand, influenced by lime surface area and micropore volume. Dry dolomitic limes have greater fineness due to thinner, larger brucite particles [
50].
3.5. Setting and Hardening
The transition from the plastic to the rigid state of mortars (setting) occurs, providing characteristic shrinkage when the water/lime ratio is exceeded (
Figure 2). The water/solid ratio and the environmental conditions are very important to determine the initial physical mechanical properties of a mortar [
53,
74].
Compared to cold mixtures, the hot-lime mix stiffens more quickly due to heat. As a result, construction can proceed more quickly and walls can be raised higher without mortar leaking out due to the weight above [
63].
In air-hardening calcic limes, the carbonation reaction [
68] involves calcium hydroxide and carbon dioxide, which form small granules and agglomerates of calcium carbonate (0.2–1 µm) (3).
The carbonation process of magnesian limes also produces the formation of magnesian carbonates (MgCO
3) and hydroxycarbonates (4), which develop good binding properties, with weak hydraulicity [
50,
75,
76]
For natural hydraulic limes (NHL), produced from marly limestone, the hardening process encompasses the hydration of 2CaO·SiO
2 (belite) and 3CaO·Al
2O
3 (celite) to form insoluble calcium silicate hydrate (C-S-H) (5) and calcium aluminate hydrate (C-A-H) (6), together with the carbonation of Ca(OH)
2 (3) [
74,
77].
The hydraulicity properties can also be reached by the addition of cocciopesto (crushed ceramics), pozzolana or plant ash materials to the finished lime since they contain amorphous silica and alumina, very reactive in the alkaline lime medium [
78,
79,
80]. The reaction rim of cocciopesto and clay-bearing aggregate will develop the following reaction (7), assuming the presence of kaolinite as clay mineral [
80]:
It should be also considered that the heat produced during slaking would favor the pozzolanic reaction, assuming any reactive aggregates exist, thus increasing adhesion to the aggregate and improving resistance to freeze/thaw weathering thanks to relatively large interconnected pores formed during mixing [
63,
65].
3.6. Sustainability
Lime was used as a binder for construction since time immemorial to at least the first decades of the 20th century, when it was progressively replaced by cement, which showed promise as an undisputed construction binder though the last century. Since the 2000s, the concepts of sustainability and ecologic building have acquired a great centrality in the economic and politic agenda of developed countries, so there was renewed interest for lime as a green and sustainable binder. Nowadays, environmental sustainability and safety for human health are fundamental aspects in the choice of a binder for construction alongside technical properties. The former aspect is related to the possibility of supporting the market needs, environmental impacts related to the consumption of natural raw materials, energy consumption, emission of pollutant and discharge of wastes associated with the production processes. The second aspect is related to the concept of “living comfort“, which concerns the risk from the development and emission of gaseous, solid and liquid pollutants, as well as molds and bacteria within the living environment, but it also refers to wastes and pollutants generated from buildings during their use and though its entire life cycle.
To examine in sufficient detail the life cycle of a binder for construction, consolidation and restoration, the examination of the following five life stages of a product is essential: (1) the extraction of the raw material; (2) production (transformation of the raw material into the finished product); (3) processing and use on site; (4) employment (permanence in the building); (5) end of life (removal, demolition, disposal and reusing or recycling).
The environmental assessment of the product arises from the detailed analysis of these stages, based on criteria related to the damage to the ecosystem and to resource consumption (emissions, energy use, waste production, socioeconomic and cultural effects of using the product, lifetime and repairability). For the examination of a binder in this aspect, the binder itself must be evaluated as part of the construction but also in relation to other alternative types of binders.
When considering the lime production process, it emerges that the calcination stage (limestone decomposition) and the consumption of fuel, electricity and limestone quarry are the most important factors for the environmental impact of a lime plant [
81,
82]. Approximately 60% of the total CO
2eq emissions can be attributed to the chemical reaction of decomposition [
83,
84], 39% are assigned to the fuel combustion and 1% to the electricity consumption at the plant. In terms of global warming, the kg CO
2eq per kg Hydrated Lime (HL) produced was estimated to be equal to 0.94 [
82]. Furthermore, it was estimated that the land occupation of the mineral extraction site is approximately 35% less than the impact factor assigned to the production of heat and electricity (on average), so it emerges that the major environmental impacts in the production of hydrated lime are related to energy consumption [
82]. As such, several studies outlined the benefits of using renewable energies rather than fossil fuels to reduce the environmental impacts of lime productions of approximately 20% [
81,
82]. Further benefits in CO
2 emissions reduction (from 20 to 40%) could be addressed by using last-generation lime kilns and/or CO
2 separation processes in existing kilns [
85].
Concerning CO
2 emissions, which are one of the most serious environmental threads of our time [
86] due to their effect on climate change, it can be outlined that when considering the lime cycle as whole, the CO
2 emissions related to the chemical reaction of calcite decomposition is totally recovered during the carbonatation of Ca-hydrate, contrary to other hydraulic binders, cement in particular, where the industry nowadays contributes to 5–7% to the global carbon emissions [
86,
87].
Considering the employment of lime as a construction material, it has several green attributes compared to other binders, due to the living comfort it gives to buildings and environmental performances. Lime contains no volatile organic compounds (VOCs), petrochemicals, lead, or other contaminants (unless included as additives to modify the material properties). Lime-based products allow water vapor to be dissipated, preventing the build-up of condensation. In addition, the alkalinity of lime helps to inhibit the growth of mold and other pathogens. These factors contribute to a healthier internal environment. The relatively high vapor permeability of lime allows moisture to move through it. The absorption and evaporation of moisture from the material helps regulate humidity within a building and diffuse penetrating water, subsequently protecting the structure from moisture-associated damage [
88]. Therefore, air lime mortars optimally control the diffusion of the air and vapor inside the walls, and plasters and paints formulated with aerial lime prevent the condensation phenomena and maintain the correct and healthy humidity in internal environments.
Finally, concerning the durability, contrary to what is generally believed, lime-based constructions and products, when carefully designed and diligently executed, have proved to be extremely long-lived and, furthermore, in the case of dismantling, they can be reused or reintegrated into nature in completely harmless ways [
47]. For these and numerous other reasons, aerial lime plays an important role in the field of sustainable and healthy construction.
4. Case Studies
The selection of case studies considered here are useful to show the variability of raw materials and technologies identified in Apulia by the authors in the last decade. Some of them were published [
15,
16,
19,
89] or presented in archaeometric conferences [
90,
91].
4.1. Egnazia
Egnazia, also known as Gnathia, was an ancient city on the Adriatic coast between Monopoli and Fasano (
Figure 1). Its history spans from the bronze age to late antiquity, with well-preserved Roman and early Christian architecture [
92,
93,
94]. Archaeometric work covers a key urban area, intended for residential use from the 2nd century BCE. The site contains bedding mortars and plasters from the Augustan and Imperial Ages, as well as a large house atrium with prestigious architectural solutions.
The petrographical (POM), mineralogical (XRPD) and chemical (XRF) analyses of 69 samples of bedding mortars (43), floors (2) and plasters (24) of the thermal baths of the Forum and rich residences aimed at verifying continuity and changes in craft skills during periods of increased building activity between the Roman imperial and Late Roman periods.
4.2. Lamapopoli
The Late Antique necropolis of Canosa was a complex archaeological reality, shaped by family hypogea and a funerary area. It was located on Via Traiana, near agricultural exploitation and rural structures. Over the course of five centuries, a large sepulchral settlement developed in Lamapopoli, synchronizing with Canosa’s role as a provincial capital and diocesan center [
95].
The POM, XRPD, XRF and SEM-EDS analyses conducted on 20 mortars from the hypogea A, C, F, G and Saggio V [
96] and from hypogea F and H [
15] revealed different ways of proceeding in the preparation and in the laying of the plasters, according to the function and period.
4.3. Tertiveri
Tertiveri, a frontier city founded by Byzantines, controlled the Tavoliere territory alongside other cities like Lucera, Ascoli, Bovino, Civitate, Dragonara, Fiorentino, Montecorvino, Troia and Biccari. Archaeological investigations reveal a tower house and a suffragan diocese. Tertiveri was a strategic outpost for Muslim rebels, leading to a Muslim enclave. A bishop was present until 1450, but this may have been occasional due to suppression and Lucera’s unified status [
97,
98,
99].
In this paper, the lime mortars of the tower are considered. Fourteen samples were analyzed by POM, XRPD, SEM-EDS and XRF.
4.4. Siponto
Siponto, a Roman colony founded in the 2nd century CE, became an episcopal see in the 5th century CE and was a rich center of a gastaldato. Despite being abandoned in the 13th century, it still retains its important port role. Recent archaeological investigations in 2000 revealed a medieval town portion with two large outdoor spaces, a small church, and a series of large buildings [
100].
The 82 mortar samples taken from excavated structures of the medieval town of Sipontum underwent to petrographical analysis (POM), X-ray powder diffraction (XRPD) and X-ray fluorescence spectrometry (XRF), in order to investigate aspects of construction technology in relation to the functional and architectural value of the building structures, as well as to correlate chronologically similar masonry structures, not easily datable and not in physical contact, verifying the hypothesis advanced by the analysis of the stratigraphic sequence. They refer to a Palatiata domus cum turre, to residential buildings and to a production plant, probably used for wheat processing for family use.
The petrographical investigation of the lime mortars showed five aggregate petrofacies and textures. The presence of semi-worked mortars allowed the correlation with the mortars of the structures and pointed to identification of at least three building phases within the archaeological structure to present investigated [
89].
4.5. Lucera
The fortress of Lucera, built by Frederick II between 1223 and 1233, was a significant Muslim settlement for Saracen rebels [
101]. The original structure had a tower shape with four wings and 32 rooms. The area was occupied by Angevins between 1269 and 1283, with archaeological investigations revealing intense building activity and the construction of dwellings for French and Provençal settlers [
102,
103,
104].
The analysis showed the use of fine clayey sand as aggregate in bedding mortars of both the Angevin’s fortification wall and the rectangular residential buildings [
101,
105].
Mortar samplings were taken for the structures identified in the Swabian Palatium referable to the phases prior to the building’s earliest constructive layout. These samplings referred to three structural elements: the inner SE wall, the outer SW wall and a subterranean cistern.
4.6. Monopoli
Monopoli, similarly to other Apulian cities such as Otranto and Gallipoli, has defensive walls that resemble fortresses. The city has undergone various construction phases over time, with archaeological evidence revealing continuity in urban occupation from prehistory to the present. The city’s defensive concept evolved with innovations, such as towers and castle construction. Frederick II of Swabia included Monopoli in his empire’s reorganizational work, completing the Norman wall. The defensive structure was not significantly changed until the 15th century when the castle was demolished by revolt. The walls underwent minor changes until the 18th century [
105].
The results of the petrographical, mineralogical and chemical analyses on 22 samples of mortars taken from the Aragonese portions of the city walls of Monopoli (“Bastione di S. Maria” and “Bastione di Pappacenere”) point to two different constructive phases within the supposed Aragonese portions of the city walls, while the use of cocciopesto shows the need to give hydraulic properties to the mortar.
Each sampling site has mortars which can be classed in the two petrographical groups. The first group features
terra rossa concretions and calcareous and quartz sand (B/A~1:2). In both groups, the aggregate has seriate grain size distribution [
19].
5. Analytical Methods
The analytical methods used in the case studies considered here are polarized optical microscopy (POM), scanning electron microscopy coupled with energy dispersive spectroscopy (SEM-EDS) and X-ray powder diffraction (XRPD).
POM observations were carried out on thin sections through a Axioskop 40 POL (Zeiss, Oberkochen, Germany) polarizing optical microscope. Images were acquired with a DS-Fi1c CCD camera (Nikon, Tokyo, Japan) with an associated Nikon Digital Sight DS-U2 controller unit. The abundance of the aggregate and macroporosity was obtained by visual estimation using comparison charts [
106].
Scanning electron microscope (SEM-EDS) observations were made on thin sections, previously fixed on aluminum specimen holders and metallized with graphite. A SEM EVO-50XVP (LEO) (Zeiss, Oberkochen, Germany), equipped with an AZTEC (Oxford Instruments, Abingdon, UK) EDS microanalysis system with SD X-Max
N detector (80 mm
2), was used. The accuracy of the analytical data was verified using various standards produced by Micro-Analysis Consultants Ltd. (St. Ives, UK). The working voltage was 15 kV and the beam current between 250 and 400 pA. Spectra acquisitions lasted 50 s, with counts ranging from 25,000 to 30,000. Chemical maps were acquired with a dwell time of 100 µs, a counting time of 10 min and a resolution of 2048. The correction of X-ray intensity was performed following Pouchou and Pichoir [
107]. Analytical precision (σ) was 0.5% for concentrations > 15 wt.%, 1% for concentrations of approximately 5 wt.%, and up to approximately 30% for concentrations near the detection limit. Different Micro-Analysis Consultants Ltd. (UK) mineral standards were used to check the accuracy of the analytical data.
The petrographic investigation took into account the composition, volume percentage, and grain size distribution of the aggregate. The texture (i.e., microcrystalline, cryptocrystalline) and structure (e.g., homogeneous and nodular) of the binder as well as the presence of fine earthy inclusions were noted. As for the porosity, the range of the pores’ roundness (0–3) was indicated for each sample. An estimation of the degree of homogeneity of the mortar was also provided.
EDS analyses of approximately 20 × 20 µm
2 of the binder-rich portions of the mortars were used to determine the chemical composition and calculate the hydraulicity index (HI) of the lime.
Particular attention was paid to the identification of the different types of lime nodules occurring in the mortars. Several authors faced the identification of lime nodules or lumps in mortars [
108,
109,
110,
111]. In this paper, a description and classification key was provided to help with understanding mortar technology. In
Figure 3, three categories are distinguished: (1) calcination, (2) slaking and (3) mixing nodules. A selection of photomicrographs taken to the analyzed mortars documenting the different types of lime nodules considered here are shown in
Figure 4. In the first case, two subcategories are possible according to the heating dynamics of limestone, which yielded under- or overburnt relics. Incomplete calcination (i.e., underburning) yields clasts of limestone with possible preservation of rock fabric and heat-induced texture reduction from macro- to cryptocrystalline (
Figure 4a,b). When the temperature/time curve of calcination exceeds the optimal combination, the new-formed quicklime particles tend to sinterize and form lumps (
Figure 4c). Although, in both cases, they do not react with water, the former may have a rim of quicklime that tends to hydrate on contact with water. Slaking nodules concern quicklime lumps that are not fully hydrated because of insufficient amounts of water and/or time of reaction and/or homogenization. They may indicate that shrinkage fissuration occurred during setting (
Figure 4d) or include calcination relics (
Figure 4e). The mixing phase of mortar preparation may yield nodules constituted by both lime and aggregates as a consequence of poor homogenization and/or a low amount of water (
Figure 4f). In this phase, the formation of nested nodules is also possible, when slaking nodules, or more rarely slaking nodules bearing calcination relics, occur inside the mixing ones.
Table 2 shows the main features of the lime nodules, whereas the implications of these features are explained in
Section 3.
The powdered samples of mortars were analyzed with a PANalytical X’Pert pro MDS powder diffractometer (Malvern, PANalytical, Almelo, The Netherlands), with a PANalytical X’Celerator detector (Malvern, PANalytical, Almelo, The Netherlands). Approximately 1 g of powder from each sample was investigated by means of X-ray powder diffraction (XRPD) in the region of 2θ = 2°–65°. The X-ray tube was operated at 40 kV and 40 mA. All XRPD spectra were processed with the X-Pert Highscore software (PANalytical, version 3.0), with a PDF-2 reference database (ICDD) for identification of inorganic phases. The diffraction peaks of the XRPD spectra were compared to a JCPDS-ICDD diffraction chart and the crystalline phases thereby identified.
6. Results
6.1. Geomaterials
6.1.1. Stones for Calcination
Laboratory analyses on lime mortars from different areas and periods in Apulia showed the use of low-Mg limestones in central Apulia and also siliceous limestones in the subapennine area. SEM-EDS analyses on calcination relics or slaking nodules (
Figure 5) provided the composition of the limestones used to produce lime (
Table 3). In particular, the lime of Egnazia was obtained from the Calcare di Bari Fm. HI is below 0.1 and the obtained lime can be classified as air lime. Only in the case of Tertiveri are there petrographic (
Figure 4b) and chemical evidence (HI ≈ 0.2) of the use of siliceous limestone for calcination. An example of bulk mineralogical composition of these mortars is shown
Figure 6c. Together with XRPD peaks of quartz and feldspars that are part of the aggregate and those of calcite as both aggregate and carbonation products, and poorly crystalline phases in the ranges between 7 and 9 °2Θ and between 11 and 13 °2Θ can be identified. Considering the chemical and petrographic features of these mortars, the probable phases associated with these peaks are the hardening products originated by the hydration of the C
2S of the feebly hydraulic lime (CSH) and the hydrated products generated by the interaction of reactive silica and alumina in the aggregate with lime (CSH, CAH) [
112,
113]. Residual peaks of clay minerals as reacting parts of the aggregate, other than chert, may be present (
Figure 7d,e).
The results obtained give some clues about the procurement of limestone and the processing before calcination. Egnazia, Monopoli, Lamapopoli and Siponto are close to the outcrops of the Calcare di Bari Fm, but the recycling of compact limestone blocks of the constructions is possible. Late Roman recycling of marbles for lime production in Egnazia were attested by Cassano [
92]. The circulation of quicklime would have been the most convenient solution in many of the cases discussed here.
6.1.2. Aggregates
The types of aggregates identified in the analyzed mortars show a variability preferentially dictated by the function of the mortar. As a whole, natural and artificial aggregates can be distinguished.
The thermal complex of Egnazia showed an articulated building stratigraphy with aggregates such as littoral sand, weathered calcarenite, cocciopesto and speleothemic calcite. The slight petrographic differences between the mortar’s groups containing calcareous sand suggest different sources. One case is alluvial, due to presence of calcareous rock fragments from Calcarenite di Gravina Fm and Calcare di Bari Fm, sometimes coated with red earth (
Figure 8a,c). In the other case, XRPD helped to discriminate littoral sand from weathered calcarenite thanks to the presence of aragonite, otherwise absent in carbonate older rocks (
Figure 6a,b). The use of cocciopesto is limited to the plasters of the thermal baths and to the floor of the domus (
Figure 8c). Spathic calcite of speleothemic origin was used exclusively in plasters, as well as straw in the preparation layer (
Figure 8a).
The use of cocciopesto was also attested in some of the lime mortars of Lamapopoli, both in plasters and in sealing mortars for sarcophagi (
Figure 8d). Generally, the rubble from the excavated fine calcarenite to obtain the cubicles was recycled in the plasters of the catacombs (
Figure 8e). The use of local alluvial deposits from a stream is also identified (
Figure 8f) [
96]. A fine clayey sand was also used as aggregate in the bedding mortars of the Angevin’s fortification walls and residential buildings of Lucera (
Figure 7a) [
16]. Cocciopesto was used to hydraulicize the lime plaster to line the cistern under Frederick’s II palace (
Figure 7b). In the same structure, there is a plaster layer showing the presence of straw ash (
Figure 7c).
In the Norman tower of Tertiveri, the use of a fluvial sand, with siliciclatic and carbonatic composition, was identified. In
Figure 7d,e, angular clasts of chert and well-rounded grains of marls show a reaction rim with the lime, formed during the pozzolanic reaction.
The Aragonese city walls of Monopoli show the use of red clay (
terra rossa) as a hydraulic aggregate (
Figure 7f), also adopted in modern rural constructions.
In the medieval town of Siponto, the use of aggregates is differentiated according to the value of the buildings. A fine alluvial sand is common in the bedding mortars of the popular dwellings (
Figure 9a). The Palatium has bedding mortars containing recycled waste from the cutting of the blocks of calcarenite used in the dressed walls (
Figure 9b). The infilling of the rubble masonry was obtained by mixing lime with earth, as for the lean bedding mortars of the ordinary residencies (
Figure 9c).
The use of pozzolana in lime concrete of the Roman harbors of Egnazia and Brindisi was demonstrated by Oleson et al. [
114].
Figure 9.
Types of aggregates identified in Siponto (
Table 4): (
a) very fine clayey sand (Group S); (
b) seriate clasts of calcarenite (Group K); (
c) combined use of fine sand and calcarenite rubbles (Group SK); (
d) cocciopesto (Group CP).
Figure 9.
Types of aggregates identified in Siponto (
Table 4): (
a) very fine clayey sand (Group S); (
b) seriate clasts of calcarenite (Group K); (
c) combined use of fine sand and calcarenite rubbles (Group SK); (
d) cocciopesto (Group CP).
6.2. Transformations
6.2.1. Calcination
Petrographic analysis plays a central role in identifying relics of calcination related to limestone and/or carbonized wood fuel. In
Figure 3, two types of calcination relics are distinguished: underburnt and overburnt rock relics. In the first case, the decomposition of carbonates is incomplete, and some optical and textural characteristics of the pristine rock are still detectable, as in the case of Siponto (
Figure 4a) and Tertiveri (
Figure 4b). In the second case, an overstep of temperature and/or long duration of heating cause sintering of quicklime particles formed (
Figure 4c). In both cases, they are not reactive in contact with water. The presence of charcoal in mortar is related to lime, and may suggest mobile calcination structure, where the bottom ash is more easily mixed up with the quicklime blocks. Small fragments of charcoal were identified in the mortars of all the sites (
Figure 8f).
6.2.2. Lime Hydration
As reported in
Figure 2, several modes of lime hydration and mixing are possible. One of the indicators of the slaking conditions is the presence/absence of lime lumps originated from incomplete hydration of quicklime. Although lime lumps can be visible at naked eyes, petrographic investigation on thin sections in transmitted light is necessary to correctly identify them. In
Table 3, some typical features of different lime nodules are provided.
Figure 4d,e show hydration nodules with typical shrinkage occurring during and after mortar setting.
Archaeological evidence of dry slaking was found in Siponto [
89], where stone tanks were used to slake quicklime covered by fine gravels to allow water infiltration and hinder air circulation and carbonation [
48]. The slaking nodules observed in the mortars of the two building phases mirror what was found archaeologically (
Table 5).
The plasters of Egnazia show rare hydration nodules, whereas they are more or less frequent in the other mortars, with charcoal.
Hydration nodules and charcoal are quite frequent in the mortars of Lamapopoli too.
Like in Egnazia, some difference in the quality of lime processing was observed between the bedding and plaster mortars of Lucera. As a whole, the bedding mortars show variable amounts of lime nodules and charcoal, while lining plaster in the cistern is essentially devoid of them.
In Siponto, hydration nodules are a common presence in the mortars, as well as charcoal.
The mortars of Tertiveri show the minor presence of slaking nodules, combined with rounded pores and reaction rims that exclude wet slaking before mixing (see below).
The wall belt of Monopoli also features mortars with hydration nodules and charcoal.
6.2.3. Mortar Preparation
Historical mortars also show the existence of unconventional mixing ratios compared to what is advised in historical sources and manuals [
115]. Important proxies of the mortar mixing are the lime/aggregate ratio (B/A), the homogenization degree of the paste, the amount of hydration nodules, the amount of primary pores and their roundness, and the amount of shrinkage (i.e., secondary) pores.
The plasters of Egnazia were prepared by mixing together lime putty with different types of aggregates. Littoral sand, weathered calcarenite or cocciopesto were used for preparation layers and spathic calcite for the finishing layers. In some cases, mineralized straw stalks were detected. They had the function to hinder the rapid drying of the plaster and to improve cohesion and stability [
47,
108]. Some differences were identified between the B/A of the mortars, generally correlated to the presence of lime nodules. In particular, the mortars with slaking nodules that exceed calcination relics have a higher B/A than plasters, i.e., 1:2 to 1:3. The lime used in the plaster is almost free of nodules. The same was observed in Lucera (
Table 4), where a different hydration was adopted for the lime of bedding mortars compared to that of plaster. The presence of plant ash in the lime plaster of the cistern in Lucera was oriented to confer hydraulicity to the lining, as was the use of cocciopesto (
Figure 7b,c). Such rare solution in Apulia is probably introduced by Saracen workers during the Swab period [
79].
The same care was not found in the plaster of Lamapopoli. In this case, sand slaking with different amounts of water was inferred after the presence of hydration nodules, rounded pores and high B/A [
15], as well as in the mortars of the Monopoli’s city walls [
19].
In Siponto, several mixing solutions were identified. Slaking nodules and underburnt relics were detected in all the samples (
Table 4). A lean fine textured mortar is common in the buildings of the peripheral zone of the whole medieval urban area (group S). Group K consists of air lime-containing nodules and calcination relics mixed with prevalent lithic fragments of calcarenite subangular and the minor presence of quartz and chert. Group T also consists of air lime added to a siliciclastic and carbonate sand aggregate and earthy inclusions; the clay component covers the surface of some clasts of the aggregate, indicative of the same origin of both. In the mortars of this group, charcoal fragments are frequent. A combination of the compositional characters of these two groups can be recognized in the KT group, where trace amounts of charcoal are also present. The cocciopesto, detected only in the samples of group CP, refers to the samples from the plasters of the two basins from the I phase of the domus and from the pile of unused mortar, found in the II phase of the area’s construction site [
89]. In the SK group samples, there is an alluvial fine sand, dominated by quartz and calcite, compatible with more recent sediments in the area, mixed with the same crushed calcarenite of the group K aggregate and to frequent lime nodules and calcination relics.
Pore roundness, visible reaction rims around reactive aggregate grains, slaking nodules and shrinkage in the mortars of Tertiveri point to hot mixing.
7. Discussion
7.1. Complexity: What and How
According to Moles [
116],
complex refers to something that has many interconnected parts that belong to a few classes, whereas
complicated has to do with something constituted by elements belonging to several classes. After these definitions, the issue of lime mortar technology faced in this paper through the Apulian cases studies can be considered as a complex system, where many elements belonging to a few categories are properly connected to achieve a functional product.
The first class of elements is that of raw materials for calcination. Although dolostone and dolomitic limestones are largely available in Apulia, none of the studied contexts points to their use to yield high-Mg lime. An exception to the use of pure limestone is related to the site of Tertiveri, where the use of siliceous limestones coming from the Miocene turbidites of the outer thrusts of the southern Apennines.
Few archaeological and archaeometric data are available about the pyrotechnology used to calcinate the raw materials. None of the sites discussed here returned lime kilns. However, some insight can be obtained from the occurrence of underburnt relics and charcoal fragments. Their regular presence in the mortars suggest the use of periodic calcination structures (clamp kiln?), where efficient calcination occurred, and quicklime blocks easily mixed with fuel residues.
As for the hydration of quicklime, wet, dry and sand slaking were identified (
Table 1). Evidence of more cured preparation of lime was observed in the plasters, while bedding mortars show features recalling dry or sand slaking. In particular, the absence of charcoal and calcination relics in the plasters of Lucera and Egnazia are more probably related to their elimination during slaking than a consequence of a different pyrotechnology. Wet slaking is also inferred by the absence of hydration nodules, but the ageing of putty cannot be confirmed. Dry slaking is characterized by the presence of hydration nodules, due to insufficient water and/or homogenization. The bedding mortars of Egnazia, Siponto and Lucera present these features, together with a B/A of approximately 1:3 and a medium to low pore roundness. On the contrary, sand slaking for the mortars of Lamapopoli, Monopoli and Tertiveri is demonstrated by the higher B/A and pore roundness, other than the reaction rims around the reactive aggregate, slaking nodules and shrinkage in samples with higher water/lime ratio.
The comparison of mortars from these sites thus shows a multiplicity of combinations between possible variants at various stages of the operational chain of production not always taken into account in the historical and technological analysis of the built heritage.
Pure limestones with a low Mg content seems to be the preferred choice, although dolomitic limestones or dolostone are widely available in Apulia. The use of siliceous limetones appears only in the subapennine area.
While several examples of permanent lime kiln are documented in different areas of Apulia [
6,
12], this does not seem to be so common in ancient times. From the results obtained, temporary kilns appear to be the most widely used, and this seems to be in agreement with the absence of permanent kilns at the same sites. The issue of lime production should not be separated from the distribution of quicklime on the building sites, which would deserve more attention in the study of past construction sites.
One of the most relevant results obtained pertains to the modes of hydration of quicklime in the different sites. The identification of different hydration and mixing practices brings to light a further element of variability in the operational chain, determined by the function of mortar and the availability of water. In particular, contrary to what is observed today, the use of lime putty seems to be essentially related to plaster preparation, while dry or sand slaking was more diffused than expected in bedding mortars. If these latter cases allow for lower water consumption, a hot-lime mix recovers some of the hydration enthalpy to foster the pozzolanic reaction and improve the mechanical strength.
7.2. Sustainability Now and Then
Lime production has evolved over time and the actual technology can be considered the best outcome in terms of environmental performance since its design process is the product of the intuitive knowledge and empiric experience of ancient masons who developed ecological and economical strategies for exploiting the maximum potential of the available resources [
117]. From the analysis of Apulian case studies illustrated herein, several aspects about the sustainability of traditional lime technology come out:
Raw material availability and mobility. Overtime, the wide availability of limestones in Apulia represented a supporting factor to the sustainability of traditional local lime production, since none of the case studies pointed out long distance transport of raw materials, except for the use of pozzolana in the Roman harbors of Egnazia and Brindisi [
114]. As added value, the different types of limestone through the region resulted in limes having colors, textures and properties that characterize and enhance local identities. Lime has been and still it is sourced locally, reducing emissions related to transport of raw materials. This is a fundamental characteristic for a modern construction material as it reduces the carbon footprint of construction projects and enhances its sustainability. However, it is worth to noting that much depends on the responsible management of limestone quarries, local production and processing practices.
Lower carbon footprint. The calcination stage is the most impactful step of the lime production process related to both the dissolution of CaCO3 and fuel consumption. However, due to the lower calcination temperature of lime compared to other hydraulic binders, less fuel is required for its production and, consequently, a lower amount of CO2 is emitted into the atmosphere than hydraulic binders. It must be emphasized that the energy cost can be further reduced through the adoption of renewable energy sources or from the optimization of production processes. Furthermore, in lime cycle production, all the CO2 emitted during calcination is reabsorbed during the carbonation process and, consequently, removed from the environment.
Cold vs. hot lime. Petrographic investigation of mortars highlights the technological features of the frequent use of dry or sand slaked lime. This latter case, with variable water/lime ration, is referred as hot-lime mix and consists in the slaking of quicklime with moisture and wet sand in a ratio that will yield a dry mix or a plastic paste ready to be layered. According to scientific and historic records, the hot-lime method produces mortars with superior performance by taking advantage of the high slaking temperature which reduces the particle size and increases the surface area of the resultant hydrate, so increasing its reactivity. The choice between the hot and cold lime preparation methods for the investigated Apulian case study could be related to the specific application of empirical knowledge to reproduce and develop desired properties. Further, it may also be related to the local factors such as the availability of water, which may differ significantly according to the geological substratum and morphology in the Apulia region.
One-part mixes. Hot-lime mixing identified in historical mortars gives evidence of a technical practice able to save water and recycle part of the energy of the exothermic reaction of quicklime hydration for the development of several properties and improvement of technical features: (1) workability: hot-mixed limes produce sticky, workable mortars that adhere well to masonry; (2) hood fill: quicklime expands as it slakes, so the volume of a hot-mixed lime mortar increases during and, for a while, after mixing. When used hot, residual expansion can continue to an extent after the mortar is laid within the wall, allowing voids and joints to be well filled; (3) early stiffening: hot-mixed lime mortars take up water rapidly as they slake and so the mortar stiffens up quickly, allowing an efficient build rate [
65].
Self-healing capacity. In lime mortars, the spontaneous dissolution and recrystallization of Ca-bearing compounds, due to water circulation in open porosity, may fill cracks and voids eventually present and improve the seal and structural strength of the material, hence contributing to its durability.
Recycling. Lime-based product can be recycled in construction projects, reducing the demand for new resources and minimizing waste. Lime is also an economically advantageous product thanks to its excellent volume performance: used in plasters, to cover the same surface unit, its dosage is up to 50% lower than ordinary hydraulic binders.
From the above analysis, it emerges that lime technology offers several environmental and performance benefits. Nowadays, it is in fact considered more sustainable than most modern cement-based binders, at least from an environment point of view, although it can be more expensive, particularly when considering the initial material costs and processing. However, the long-term durability and environmental benefits offset these initial expenses. It is important to note that sustainable sourcing and responsible construction practices are crucial to fully realize these benefits.
The use of lime as a building material dates back thousands of years, until the introduction of OPC (Ordinary Portland Cement) in the middle of the 19th century. However, an important revival of lime technology started to occur in recent decades, when both the harmful effects of cement, especially on historical buildings, emerged [
118] and environmental and sustainable issues of OPC chain-production at a large scale became prominent.
In this scenario, the concept of sustainability also passes through the re-adoption of traditional materials and technologies requiring less energy. A plethora of recent studies are bringing new insight into the composition and durability of historic binders, mortars and concrete showing how delving into the past of binder science can inspire the development of modern environmentally friendly cementitious materials [
119].
A better understanding of ancient technologies such as that of traditional lime mortar production can thus help to recover practices or gain advantages in the development of contemporary production technologies. This has a special importance in the field of reparation and restoration of ancient constructions for which the lime binder has proven to bring several advantages with respect to modern binders, but the challenge is the re-introduction of lime at a larger scale in new building construction processes due to its ecological and sustainable virtues. As such, it is worth noting that the carbonation mechanism is at the base of a widespread investigated class of binders generally referred to as “carbonatable binders” [
120] or carbonation-hardening cements [
121], which gained attention in recent times against the backdrop of international movements towards a carbon-neutral world. Similarly, alkali-activated materials, sometimes also identified with the subcategory known with the name of geopolymers, represent a wide category of binders that use alkalis (usually in aqueous form) mixed with powdered aluminosilicate reactive precursors [
122,
123]. Both “carbonatable binders” and “alkali-activated materials” (or geopolymers) are not new technologies but are at the heart of human-built environment technologies, the former finding their basis in traditional lime technology, the latter in ancient hydraulic binders.
From the perspective of reducing the energy required for the exploitation, transport and transformation of primary resources, as well as to minimize waste production and reduce the landfill requirement for waste disposal, several trials to include anthropic wastes and industrial by-products as secondary resources in the formulation of these and other alternative non-Portland binders (calcium sulfoalminate-based and calcium aluminate cements, belite cements, magnesia silicate and magnesia phosphate cements, sorel cements, etc.) are under investigation [
121], in agreement with the practice of reusing and recycling, well consolidated in ancient times. Steel slag and magnesium slag, showing high carbonation reactivity, have been tested in the formulation of “carbonatable binders”, whereas several industrial by-products such as blast furnace slags, calcined clay sediments and coal combustion ashes, as well as any other waste material containing reactive aluminosilicate components (either Ca-poor or Ca-rich) can be used in the “alkaline-activation technology” [
122].
However, it should be stressed that the use of by-products and waste products in substitution of clinker in OPC is also a well-consolidated practice today to reduce CO
2 emissions associated with the clinker production. With this aim, several natural or waste-derived materials such as granulated blast furnace slag, fly ash, calcinated clays, natural pozzolans and finely ground limestone are included today as supplementary cementitious materials (SCMs) to replace the clinker in OPC formulations, thus reducing the amount of CO
2 emitted per mass of binder [
112,
123,
124].
8. Conclusions
The complexity emerging from the comparison between the six Apulian case studies on lime mortars is the consequence of the dynamic interplay of natural and cultural landscapes temporally stratified. The results of this study showed how natural surroundings and the climate of a region can influence the types of challenges that need technological solutions. If the availability of specific geomaterials can be considered a constant in the natural landscape, except when important phenomena (e.g., volcanic eruptions, sea level changes, and landslides) occurred, the availability and quality of biomass and water to perform all the steps of the operational chain are not secondary issues. In the frame of the ecology of production, place-specific resources are “filtered” by cultural preferences and identities, which determine the acceptance and integration of certain technologies into a society. This is particularly true in regions like Apulia, where, from prehistory to modernity, several populations and cultures have succeeded each other over time, leaving important evidence in material culture.
The case studies discussed here highlight the importance of a sound methodological approach to reconstruct the operational chain of mortar production. Instead, most studies today focus on the origin of components and less on how those components were processed and transformed to obtain a hardened mortar. This is because the potential information from the mineralogical and petrographic investigation of mortars is sometimes not adequately exploited. As shown here for the Apulian case studies, different processing of the same ingredients may lead to final products with different characteristics, awareness of which is not only valuable in the knowledge of the past but can provide useful technological solutions for the future. One of the most relevant results of this study is the evidence of different hydration modes of quicklime in the investigated sites. The identification of different hydration and mixing practices brings to light a further element of variability in the operational chain, determined by the function of mortar and the availability of water. Contrary to what is observed today, the use of lime putty was found to be essentially related to plaster preparation, while dry or sand slaking was more diffused than expected in bedding mortars. Specifically, the wet slaking preparation methodology was used for plasters of Lucera and Egnazia, dry slaking for the bedding mortars of Egnazia, Siponto and Lucera and sand slaking for the mortars of Lamapopoli, Monopoli and Tertiveri.
Recycling of stone working rubbles and crushed ceramics, previously documented, represents a further key aspect also pursued by today’s strategies for sustainability. The way of processing carried out deals with energy and resource sustainability other than the efficacy and performance of a product, i.e., dry or sand slaking allow for lower water consumption, and a hot-lime mix recovers some of the hydration enthalpy to foster the pozzolanic reaction and improve the mechanical strength. For this reason, the attention paid to the operational chain and performance over time not only deals with the past but also with the future. The case of lime mortars and concretes, which dominated the scene in building construction until the introduction of proto-Portland binders, is emblematic, because it provides us countless cases of use over a period of more than a thousand years. The CO2 cycle of lime mortar production has less impact on the environment compared to OPC, but of course they have different mechanical, physical and chemical characteristics, which make them not exchangeable. Therefore, even if a return to lime is not feasible, a better understanding of the production and curing processes of lime mortars of the past can help to recover empirical practices that have stood the test of time in contemporary production technologies. The implementation of hybrid cements and geopolymers, and the experimentation on one-part mixing are some examples of new materials and practices which are in relation with the ancient technologies.
Nowadays, the issue of environmental conservation is an inescapable quest of contemporary societies, which engage governments, research institutions, enterprises and civil society in actions oriented to develop technologies and behaviors with lower ecological footprints. However, many ancient cultures did develop practices and beliefs that aligned with sustainable living and a harmonious relationship with nature although the concepts of environmental conservation and sustainability, as we understand them today, were not explicitly articulated in the past in the same way. As a matter of fact, preindustrial production cycles did not result in a heavy impact on the environment as they were often shaped by practical considerations for survival in their specific ecological contexts. This could probably be one of the main lessons that we may learn from the past alongside several technological outcomes.