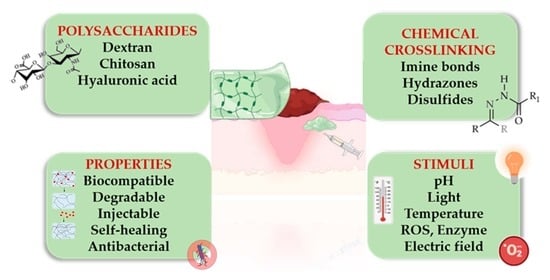
Stimuli-Responsive Polysaccharide Hydrogels and Their Composites for Wound Healing Applications
Abstract
:1. Introduction
2. Chemical Functionalization of Polysaccharides
2.1. Oxidation of Polysaccharides
2.2. Modification of Chitin and Chitosan
2.2.1. Quaternization of Chitosan

2.2.2. Carboxyalkylation of Chitosan
2.2.3. Etherification of Chitosan
3. Stimuli-Responsive Polysaccharide-Based Hydrogels
3.1. pH-Responsive Polysaccharide Hydrogels
3.2. Redox-Responsive Polysaccharide Hydrogels
3.3. Photo-Responsive Polysaccharide Hydrogels
3.4. Dual Responsive Hydrogels
3.4.1. pH- and Temperature-Responsive Polysaccharide Hydrogels
3.4.2. pH- and ROS-Responsive Polysaccharide Hydrogels
3.4.3. pH- and Electro-Responsive Polysaccharide Hydrogels
4. Polysaccharide-Peptide Composites with Cell Instructive/Responsive Properties
5. Applications in Wound Healing/Dressings
5.1. pH-Responsive Polysaccharide Hydrogels
5.2. Thermoresponsive Polysaccharide Hydrogels
5.3. Enzyme-Responsive Polysaccharide Hydrogels
5.4. Redox-Responsive Polysaccharide Hydrogels
5.5. Photo-Responsive Polysaccharide Hydrogels
6. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| Abbreviation | Name |
| ROS | reactive oxygen species |
| GSH | glutathione |
| CS | chitosan |
| QCS | quaternized chitosan |
| HBCS | hydroxybutyl chitosan |
| CMCS | carboxymethyl chitosan |
| GTMAC | glycidyl trimethyl ammonium chloride |
| OHA | oxidized hyaluronic acid |
| PEG | poly(ethylene glycol) |
| HPC | hydroxypropyl cellulose |
| OCAPS | octa (γ-chloroammoniumpropyl) silsesquioxane |
| rGO | reduced graphene oxide |
| TEOS | tetraethyl orthosilicate |
| OD | oxidized dextran |
| HTCC | N-(2-hydroxypropyl)-3-trimethylammonium chitosan chloride |
| HA-ADH | dihydrazide hyaluronic acid |
| BBH | berberine |
| EGF | epidermal growth factor |
| PEDOT: PSS | poly(3, 4-ethylenedioxythiophene):poly(styrene sulfonate) |
| PF127-CHO | aldehyde pluronic F127 |
| OD-DA | oxidized dextran |
| DFO | deferoxamine |
| CECS | carboxyethyl chitosan |
| ADH | adipic acid dihydrazide |
| AEDa | Aminoethyl disulfide |
| β-CD | β-cyclodextrin |
| HA-CD | HA conjugated with β-cyclodextrin |
| HA-Azo | HA conjugated with trans-azobenzene |
| APu | aldehyde pullulan |
| PEI | polyethylenimine |
| ADSC | adipose mesenchymal stem cell |
| HPCH | hydroxypropyl chitin |
| TA | tannic acid |
| ALG-BA | alginate backbone |
| CHOL | cholesterol |
| CP | chitosan-g-polyaniline |
| RGD | arginine-glycine-aspartate |
| CM-TM-CS | carboxymethyl-trimethyl chitosan |
| GelMA | gelatin methacrylamide |
| MMPs | matrix metalloproteinases |
| HIF-1α | factor-1 alpha |
| VEGF | vascular endothelial growth factor |
| SS | sisomicin sulfate |
| AgNPs | silver nanoparticles |
| PB | poly(vinyl alcohol)-borax |
| OCMC-DA | dopamine grafted oxidized carboxymethyl cellulose |
| EPL | ε-poly-L-lysine |
| KGF | keratinocyte growth factor |
| HP | heparin-modified poloxamer |
| FEP | fluorinated ethylene propylene |
| EDTA | ethylenediaminetetraacetic acid |
| ZIF-8 | zeolite imidazole framework-8 |
| PEG-TK | PEG-thioketal |
| BA | boric acid |
| HA | hyaluronic acid |
| NIR | Near infrared |
| WS2-NS2 | tungsten disulphide nanosheets |
References
- Reinke, J.M.; Sorg, H. Wound Repair and Regeneration. Eur. Surg. Res. 2012, 49, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Kruse, C.R.; Nuutila, K.; Lee, C.C.Y.; Kiwanuka, E.; Singh, M.; Caterson, E.J.; Eriksson, E.; Sørensen, J.A. The External Microenvironment of Healing Skin Wounds. Wound Repair Regen. 2015, 23, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Laurens, N.; Koolwijk, P.; De Maat, M.P.M. Fibrin Structure and Wound Healing. J. Thromb. Haemost. 2006, 4, 932–939. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Liu, W.; Li, N.; Wang, M.; Liang, B.; Ullah, I.; Luis Neve, A.; Feng, Y.; Chen, H.; Shi, C. Design and Development of Polysaccharide Hemostatic Materials and Their Hemostatic Mechanism. Biomater. Sci. 2017, 5, 2357–2368. [Google Scholar] [CrossRef]
- Eming, S.A.; Krieg, T.; Davidson, J.M. Inflammation in Wound Repair: Molecular and Cellular Mechanisms. J. Investig. Dermatol. 2007, 127, 514–525. [Google Scholar] [CrossRef] [Green Version]
- Dunnill, C.; Patton, T.; Brennan, J.; Barrett, J.; Dryden, M.; Cooke, J.; Leaper, D.; Georgopoulos, N.T. Reactive Oxygen Species (ROS) and Wound Healing: The Functional Role of ROS and Emerging ROS-Modulating Technologies for Augmentation of the Healing Process. Int. Wound J. 2017, 14, 89–96. [Google Scholar] [CrossRef]
- Gao, Z.; Golland, B.; Tronci, G.; Thornton, P.D. A Redox-Responsive Hyaluronic Acid-Based Hydrogel for Chronic Wound Management. J. Mater. Chem. B 2019, 7, 7494–7501. [Google Scholar] [CrossRef]
- Mallinjoud, P.; Villemin, J.-P.; Mortada, H.; Polay Espinoza, M.; Desmet, F.-O.; Samaan, S.; Chautard, E.; Tranchevent, L.-C.; Auboeuf, D. Endothelial, Epithelial, and Fibroblast Cells Exhibit Specific Splicing Programs Independently of Their Tissue of Origin. Genome Res. 2014, 24, 511–521. [Google Scholar] [CrossRef] [Green Version]
- Farasati Far, B.; Naimi-Jamal, M.R.; Safaei, M.; Zarei, K.; Moradi, M.; Yazdani Nezhad, H. A Review on Biomedical Application of Polysaccharide-Based Hydrogels with a Focus on Drug Delivery Systems. Polymers 2022, 14, 5432. [Google Scholar] [CrossRef]
- Ribeiro, D.M.L.; Júnior, A.R.C.; de Macedo, G.H.R.V.; Chagas, V.L.; Silva, L.D.S.; da Silva Cutrim, B.; Santos, D.M.; Soares, B.L.L.; Zagmignan, A.; de Miranda, R.d.C.M.; et al. Polysaccharide-Based Formulations for Healing of Skin-Related Wound Infections: Lessons from Animal Models and Clinical Trials. Biomolecules 2020, 10, 63. [Google Scholar] [CrossRef] [Green Version]
- Kaliva, M.; Kavasi, R.; Chatzinikolaidou, M.; Vamvakaki, M. Book chapter 4.01 Polysaccharides and Applications in Regenerative Medicine. In Comprehensive Glycoscience (Second Edition); Elsevier: Amsterdam, The Netherlands, 2021; pp. 1–33. ISBN 9780128194751. [Google Scholar] [CrossRef]
- Farahani, M.; Shafiee, A. Wound Healing: From Passive to Smart Dressings. Adv. Healthc. Mater. 2021, 10, 2100477. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Xu, F.J. Rational Design and Latest Advances of Polysaccharide-Based Hydrogels for Wound Healing. Biomater. Sci. 2020, 8, 2084–2101. [Google Scholar] [CrossRef] [PubMed]
- Gholamali, I. Stimuli-Responsive Polysaccharide Hydrogels for Biomedical Applications: A Review. Regen. Eng. Transl. Med. 2021, 7, 91–114. [Google Scholar] [CrossRef]
- Mukherjee, I. Recent Development of Polysaccharide-Derived Hydrogel: Properties, Stimuli-Responsiveness and Bioapplications. ChemRxiv. Cambridge Cambridge Open Engag. 2022. [Google Scholar] [CrossRef]
- Rial-Hermida, M.I.; Rey-Rico, A.; Blanco-Fernandez, B.; Carballo-Pedrares, N.; Byrne, E.M.; Mano, J.F. Recent Progress on Polysaccharide-Based Hydrogels for Controlled Delivery of Therapeutic Biomolecules. ACS Biomater. Sci. Eng. 2021, 7, 4102–4127. [Google Scholar] [CrossRef]
- Taylor, D.L.; in het Panhuis, M. Self-Healing Hydrogels. Adv. Mater. 2016, 28, 9060–9093. [Google Scholar] [CrossRef]
- Li, Z.; Zhou, Y.; Li, T.; Zhang, J.; Tian, H. Stimuli-Responsive Hydrogels: Fabrication and Biomedical Applications. VIEW 2022, 3, 20200112. [Google Scholar] [CrossRef]
- He, Y.; Li, Y.; Sun, Y.; Zhao, S.; Feng, M.; Xu, G.; Zhu, H.; Ji, P.; Mao, H.; He, Y.; et al. A Double-Network Polysaccharide-Based Composite Hydrogel for Skin Wound Healing. Carbohydr. Polym. 2021, 261, 117870. [Google Scholar] [CrossRef]
- Wang, X.; Xu, P.; Yao, Z.; Fang, Q.; Feng, L.; Guo, R.; Cheng, B. Preparation of Antimicrobial Hyaluronic Acid/Quaternized Chitosan Hydrogels for the Promotion of Seawater-Immersion Wound Healing. Front. Bioeng. Biotechnol. 2019, 7, 360. [Google Scholar] [CrossRef]
- Mao, Q.; Hoffmann, O.; Yu, K.; Lu, F.; Lan, G.; Dai, F.; Shang, S.; Xie, R. Self-Contracting Oxidized Starch/Gelatin Hydrogel for Noninvasive Wound Closure and Wound Healing. Mater. Des. 2020, 194, 108916. [Google Scholar] [CrossRef]
- Kang, M.; Oderinde, O.; Han, X.; Fu, G.; Zhang, Z. Development of Oxidized Hydroxyethyl Cellulose-Based Hydrogel Enabling Unique Mechanical, Transparent and Photochromic Properties for Contact Lenses. Int. J. Biol. Macromol. 2021, 183, 1162–1173. [Google Scholar] [CrossRef] [PubMed]
- Rahmat, D.; Müller, C.; Barthelmes, J.; Shahnaz, G.; Martien, R.; Bernkop-Schnürch, A. Thiolated Hydroxyethyl Cellulose: Design and in Vitro Evaluation of Mucoadhesive and Permeation Enhancing Nanoparticles. Eur. J. Pharm. Biopharm. 2013, 83, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Wang, C.; Lai, R.C.; Su, K.; Zhang, F.; Wang, D. An Improved Injectable Polysaccharide Hydrogel: Modified Gellan Gum for Long-Term Cartilage Regenerationin Vitro. J. Mater. Chem. 2009, 19, 1968–1977. [Google Scholar] [CrossRef]
- Bacelar, A.H.; Silva-Correia, J.; Oliveira, J.M.; Reis, R.L. Recent Progress in Gellan Gum Hydrogels Provided by Functionalization Strategies. J. Mater. Chem. B 2016, 4, 6164–6174. [Google Scholar] [CrossRef] [Green Version]
- Ijaz, M.; Matuszczak, B.; Rahmat, D.; Mahmood, A.; Bonengel, S.; Hussain, S.; Huck, C.W.; Bernkop-Schnürch, A. Synthesis and Characterization of Thiolated β-Cyclodextrin as a Novel Mucoadhesive Excipient for Intra-Oral Drug Delivery. Carbohydr. Polym. 2015, 132, 187–195. [Google Scholar] [CrossRef]
- Kobayashi, M.; Urayama, T.; Suzawa, I.; Takagi, S.; Matsuda, K.; Ichishima, E. Cyclodextrin–Dialdehyde Prepared by Periodate Oxidation. Agric. Biol. Chem. 1988, 52, 2695–2702. [Google Scholar] [CrossRef]
- Zhao, X.; Wu, H.; Guo, B.; Dong, R.; Qiu, Y.; Ma, P.X. Antibacterial Anti-Oxidant Electroactive Injectable Hydrogel as Self-Healing Wound Dressing with Hemostasis and Adhesiveness for Cutaneous Wound Healing. Biomaterials 2017, 122, 34–47. [Google Scholar] [CrossRef]
- Sun, M.; Wang, T.; Pang, J.; Chen, X.; Liu, Y. Hydroxybutyl Chitosan Centered Biocomposites for Potential Curative Applications: A Critical Review. Biomacromolecules 2020, 21, 1351–1367. [Google Scholar] [CrossRef]
- Hao, Y.; Zhao, W.; Zhang, H.; Zheng, W.; Zhou, Q. Carboxymethyl Chitosan-Based Hydrogels Containing Fibroblast Growth Factors for Triggering Diabetic Wound Healing. Carbohydr. Polym. 2022, 287, 119336. [Google Scholar] [CrossRef]
- Li, X.; Weng, Y.; Kong, X.; Zhang, B.; Li, M.; Diao, K.; Zhang, Z.; Wang, X.; Chen, H. A Covalently Crosslinked Polysaccharide Hydrogel for Potential Applications in Drug Delivery and Tissue Engineering. J. Mater. Sci. Mater. Med. 2012, 23, 2857–2865. [Google Scholar] [CrossRef]
- Xue, C.; Xu, X.; Zhang, L.; Liu, Y.; Liu, S.; Liu, Z.; Wu, M.; Shuai, Q. Self-Healing/PH-Responsive/Inherently Antibacterial Polysaccharide-Based Hydrogel for a Photothermal Strengthened Wound Dressing. Colloids Surf. B Biointerfaces 2022, 218, 112738. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Chen, B.; Li, M.; He, J.; Yin, Z.; Guo, B. Injectable Antimicrobial Conductive Hydrogels for Wound Disinfection and Infectious Wound Healing. Biomacromolecules 2020, 21, 1841–1852. [Google Scholar] [CrossRef] [PubMed]
- Andreica, B.-I.; Cheng, X.; Marin, L. Quaternary Ammonium Salts of Chitosan. A Critical Overview on the Synthesis and Properties Generated by Quaternization. Eur. Polym. J. 2020, 139, 110016. [Google Scholar] [CrossRef]
- Jain, A.; Gulbake, A.; Shilpi, S.; Jain, A.; Hurkat, P.; Jain, S.K. A New Horizon in Modifications of Chitosan: Syntheses and Applications. Crit. Rev. Ther. Drug Carrier Syst. 2013, 30, 91–181. [Google Scholar] [CrossRef] [PubMed]
- Mourya, V.K.; Inamdara, N.; Ashutosh Tiwari, N. Carboxymethyl Chitosan And Its Applications. Adv. Mater. Lett. 2010, 1, 11–33. [Google Scholar] [CrossRef]
- Li, J.; Yu, F.; Chen, G.; Liu, J.; Li, X.-L.; Cheng, B.; Mo, X.-M.; Chen, C.; Pan, J.-F. Moist-Retaining, Self-Recoverable, Bioadhesive, and Transparent in Situ Forming Hydrogels To Accelerate Wound Healing. ACS Appl. Mater. Interfaces 2020, 12, 2023–2038. [Google Scholar] [CrossRef]
- Li, Z.; Zhao, Y.; Liu, H.; Ren, M.; Wang, Z.; Wang, X.; Liu, H.; Feng, Y.; Lin, Q.; Wang, C.; et al. PH-Responsive Hydrogel Loaded with Insulin as a Bioactive Dressing for Enhancing Diabetic Wound Healing. Mater. Des. 2021, 210, 110104. [Google Scholar] [CrossRef]
- Qu, J.; Zhao, X.; Liang, Y.; Xu, Y.; Ma, P.X.; Guo, B. Degradable Conductive Injectable Hydrogels as Novel Antibacterial, Anti-Oxidant Wound Dressings for Wound Healing. Chem. Eng. J. 2019, 362, 548–560. [Google Scholar] [CrossRef]
- Biagini, G.; Bertani, A.; Muzzarelli, R.; Damadei, A.; DiBenedetto, G.; Belligolli, A.; Riccotti, G.; Zucchini, C.; Rizzoli, C. Wound Management with N-Carboxybutyl Chitosan. Biomaterials 1991, 12, 281–286. [Google Scholar] [CrossRef]
- Cai, Y.; Zhong, Z.; He, C.; Xia, H.; Hu, Q.; Wang, Y.; Ye, Q.; Zhou, J. Homogeneously Synthesized Hydroxybutyl Chitosans in Alkali/Urea Aqueous Solutions as Potential Wound Dressings. ACS Appl. Bio Mater. 2019, 2, 4291–4302. [Google Scholar] [CrossRef]
- Wang, Q.Q.; Kong, M.; An, Y.; Liu, Y.; Li, J.J.; Zhou, X.; Feng, C.; Li, J.; Jiang, S.Y.; Cheng, X.J.; et al. Hydroxybutyl Chitosan Thermo-Sensitive Hydrogel: A Potential Drug Delivery System. J. Mater. Sci. 2013, 48, 5614–5623. [Google Scholar] [CrossRef]
- Bi, S.; Hu, S.; Zhou, Z.; Kong, M.; Liu, Y.; Feng, C.; Cheng, X.; Chen, X. The Green and Stable Dissolving System Based on KOH/Urea for Homogeneous Chemical Modification of Chitosan. Int. J. Biol. Macromol. 2018, 120, 1103–1110. [Google Scholar] [CrossRef] [PubMed]
- Bi, B.; Ma, M.; Lv, S.; Zhuo, R.; Jiang, X. In-Situ Forming Thermosensitive Hydroxypropyl Chitin-Based Hydrogel Crosslinked by Diels-Alder Reaction for Three Dimensional Cell Culture. Carbohydr. Polym. 2019, 212, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Zhong, Y.; Jiang, X. Thermosensitive and PH-Responsive Tannin-Containing Hydroxypropyl Chitin Hydrogel with Long-Lasting Antibacterial Activity for Wound Healing. Carbohydr. Polym. 2020, 236, 116096. [Google Scholar] [CrossRef] [PubMed]
- Yuan, M.; Bi, B.; Huang, J.; Zhuo, R.; Jiang, X. Thermosensitive and Photocrosslinkable Hydroxypropyl Chitin-Based Hydrogels for Biomedical Applications. Carbohydr. Polym. 2018, 192, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Gao, M.; Boakye-Yiadom, K.O.; Ho, W.; Yu, W.; Xu, X.; Zhang, X.Q. An Intrinsically Bioactive Hydrogel with On-Demand Drug Release Behaviors for Diabetic Wound Healing. Bioact. Mater. 2021, 6, 4592–4606. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Meng, Y.; Shen, W.; Dou, J.; Liu, R.; Jin, Q.; Fang, S. PH-Responsive Injectable Polysaccharide Hydrogels with Self-Healing, Enhanced Mechanical Properties Based on POSS. React. Funct. Polym. 2021, 158, 104773. [Google Scholar] [CrossRef]
- Khan, M.U.A.; Abd Razaq, S.I.; Mehboob, H.; Rehman, S.; Al-Arjan, W.S.; Amin, R. Antibacterial and Hemocompatible Ph-Responsive Hydrogel for Skin Wound Healing Application: In Vitro Drug Release. Polymers 2021, 13, 3703. [Google Scholar] [CrossRef]
- Hoque, J.; Prakash, R.G.; Paramanandham, K.; Shome, B.R.; Haldar, J. Biocompatible Injectable Hydrogel with Potent Wound Healing and Antibacterial Properties. Mol. Pharm. 2017, 14, 1218–1230. [Google Scholar] [CrossRef]
- Guan, S.; Li, Y.; Cheng, C.; Gao, X.; Gu, X.; Han, X.; Ye, H. Manufacture of PH- and HAase-Responsive Hydrogels with on-Demand and Continuous Antibacterial Activity for Full-Thickness Wound Healing. Int. J. Biol. Macromol. 2020, 164, 2418–2431. [Google Scholar] [CrossRef]
- Qu, J.; Zhao, X.; Liang, Y.; Zhang, T.; Ma, P.X.; Guo, B. Antibacterial Adhesive Injectable Hydrogels with Rapid Self-Healing, Extensibility and Compressibility as Wound Dressing for Joints Skin Wound Healing. Biomaterials 2018, 183, 185–199. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Long, L.; Cao, J.; Zhang, S.; Wang, Y. Dual-Crosslinked Mussel-Inspired Smart Hydrogels with Enhanced Antibacterial and Angiogenic Properties for Chronic Infected Diabetic Wound Treatment via PH-Responsive Quick Cargo Release. Chem. Eng. J. 2021, 411, 128564. [Google Scholar] [CrossRef]
- Zhao, W.; Li, Y.; Zhang, X.; Zhang, R.; Hu, Y.; Boyer, C.; Xu, F.J. Photo-Responsive Supramolecular Hyaluronic Acid Hydrogels for Accelerated Wound Healing. J. Control. Release 2020, 323, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Klouda, L.; Mikos, A.G. Thermoresponsive Hydrogels in Biomedical Applications. Eur. J. Pharm. Biopharm. 2008, 68, 34–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, M.; Wang, C.; Chen, M.; Xi, Y.; Cheng, W.; Mao, C.; Xu, T.; Zhang, X.; Lin, C.; Gao, W.; et al. Efficient Angiogenesis-Based Diabetic Wound Healing/Skin Reconstruction through Bioactive Antibacterial Adhesive Ultraviolet Shielding Nanodressing with Exosome Release. ACS Nano 2019, 13, 10279–10293. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Zhang, F.; Long, L.; Kong, Q.; Luo, R.; Wang, Y. Dual-Responsive Injectable Hydrogels Encapsulating Drug-Loaded Micelles for on-Demand Antimicrobial Activity and Accelerated Wound Healing. J. Control. Release 2020, 324, 204–217. [Google Scholar] [CrossRef]
- Qu, J.; Zhao, X.; Ma, P.X.; Guo, B. Injectable Antibacterial Conductive Hydrogels with Dual Response to an Electric Field and PH for Localized “Smart” Drug Release. Acta Biomater. 2018, 72, 55–69. [Google Scholar] [CrossRef]
- Deidda, G.; Jonnalagadda, S.V.R.; Spies, J.W.; Ranella, A.; Mossou, E.; Forsyth, V.T.; Mitchell, E.P.; Bowler, M.W.; Tamamis, P.; Mitraki, A. Self-Assembled Amyloid Peptides with Arg-Gly-Asp (RGD) Motifs As Scaffolds for Tissue Engineering. ACS Biomater. Sci. Eng. 2017, 3, 1404–1416. [Google Scholar] [CrossRef]
- Kumar, V.B.; Tiwari, O.S.; Finkelstein-Zuta, G.; Rencus-Lazar, S.; Gazit, E. Design of Functional RGD Peptide-Based Biomaterials for Tissue Engineering. Pharmaceutics 2023, 15, 345. [Google Scholar] [CrossRef]
- Rowley, J.A.; Madlambayan, G.; Mooney, D.J. Alginate Hydrogels as Synthetic Extracellular Matrix Materials. Biomaterials 1999, 20, 45–53. [Google Scholar] [CrossRef]
- Hansson, A.; Hashom, N.; Falson, F.; Rousselle, P.; Jordan, O.; Borchard, G. In Vitro Evaluation of an RGD-Functionalized Chitosan Derivative for Enhanced Cell Adhesion. Carbohydr. Polym. 2012, 90, 1494–1500. [Google Scholar] [CrossRef] [PubMed]
- Oki, Y.; Kirita, K.; Ohta, S.; Ohba, S.; Horiguchi, I.; Sakai, Y.; Ito, T. Switching of Cell Proliferation/Differentiation in Thiol-Maleimide Clickable Microcapsules Triggered by in Situ Conjugation of Biomimetic Peptides. Biomacromolecules 2019, 20, 2350–2359. [Google Scholar] [CrossRef] [PubMed]
- Ansari, S.; Pouraghaei Sevari, S.; Chen, C.; Sarrion, P.; Moshaverinia, A. RGD-Modified Alginate-GelMA Hydrogel Sheet Containing Gingival Mesenchymal Stem Cells: A Unique Platform for Wound Healing and Soft Tissue Regeneration. ACS Biomater. Sci. Eng. 2021, 7, 3774–3782. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, K.B.; Bidarra, S.J.; Oliveira, M.J.; Granja, P.L.; Barrias, C.C. Molecularly Designed Alginate Hydrogels Susceptible to Local Proteolysis as Three-Dimensional Cellular Microenvironments. Acta Biomater. 2011, 7, 1674–1682. [Google Scholar] [CrossRef]
- Suo, H.; Hussain, M.; Wang, H.; Zhou, N.; Tao, J.; Jiang, H.; Zhu, J. Injectable and PH-Sensitive Hyaluronic Acid-Based Hydrogels with On-Demand Release of Antimicrobial Peptides for Infected Wound Healing. Biomacromolecules 2021, 22, 3049–3059. [Google Scholar] [CrossRef]
- Gao, L.; Chen, J.; Feng, W.; Song, Q.; Huo, J.; Yu, L.; Liu, N.; Wang, T.; Li, P.; Huang, W. A Multifunctional Shape-Adaptive and Biodegradable Hydrogel with Hemorrhage Control and Broad-Spectrum Antimicrobial Activity for Wound Healing. Biomater. Sci. 2020, 8, 6930–6945. [Google Scholar] [CrossRef]
- Bennison, L.R.; Miller, C.N.; Summers, R.J.; Minnis, A.M.B.; Sussman, G.; McGuiness, W. The PH of Wounds during Healing and Infection: A Descriptive Literature Review. Wound Pract. Res. J. Aust. Wound Manag. Assoc. 2017, 25, 63–69. [Google Scholar]
- Zheng, C.; An, X.; Gong, J. Novel PH Sensitive N-Doped Carbon Dots with Both Long Fluorescence Lifetime and High Quantum Yield. RSC Adv. 2015, 5, 32319–32322. [Google Scholar] [CrossRef]
- Omidi, M.; Yadegari, A.; Tayebi, L. Wound Dressing Application of PH-Sensitive Carbon Dots/Chitosan Hydrogel. RSC Adv. 2017, 7, 10638–10649. [Google Scholar] [CrossRef] [Green Version]
- Ji, H.; Dong, K.; Yan, Z.; Ding, C.; Chen, Z.; Ren, J.; Qu, X. Bacterial Hyaluronidase Self-Triggered Prodrug Release for Chemo-Photothermal Synergistic Treatment of Bacterial Infection. Small 2016, 12, 6200–6206. [Google Scholar] [CrossRef]
- Fouda, M.M.G.; Abdel-Mohsen, A.M.; Ebaid, H.; Hassan, I.; Al-Tamimi, J.; Abdel-Rahman, R.M.; Metwalli, A.; Alhazza, I.; Rady, A.; El-Faham, A.; et al. Wound Healing of Different Molecular Weight of Hyaluronan; in-Vivo Study. Int. J. Biol. Macromol. 2016, 89, 582–591. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Seidi, F.; Li, C.; Wan, Z.; Jin, Y.; Song, J.; Xiao, H. Antimicrobial/Biocompatible Hydrogels Dual-Reinforced by Cellulose as Ultrastretchable and Rapid Self-Healing Wound Dressing. Biomacromolecules 2021, 22, 1654–1663. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.-L.; Xu, J.; Shen, B.-X.; Zhang, S.-S.; Jin, B.-H.; Zhu, Q.-Y.; ZhuGe, D.-L.; Wu, X.-Q.; Xiao, J.; Zhao, Y.-Z. Dual Regulations of Thermosensitive Heparin–Poloxamer Hydrogel Using ε-Polylysine: Bioadhesivity and Controlled KGF Release for Enhancing Wound Healing of Endometrial Injury. ACS Appl. Mater. Interfaces 2017, 9, 29580–29594. [Google Scholar] [CrossRef]
- Wang, Q.; He, Y.; Zhao, Y.; Xie, H.; Lin, Q.; He, Z.; Wang, X.; Li, J.; Zhang, H.; Wang, C.; et al. A Thermosensitive Heparin-Poloxamer Hydrogel Bridges AFGF to Treat Spinal Cord Injury. ACS Appl. Mater. Interfaces 2017, 9, 6725–6745. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Wildhaber, B.E.; Teitelbaum, D.H. Keratinocyte Growth Factor Improves Epithelial Function after Massive Small Bowel Resection. J. Parenter. Enter. Nutr. 2003, 27, 198–206. [Google Scholar] [CrossRef]
- Ernst, M.; John, T.; Guenther, M.; Wagner, C.; Schaefer, U.F.; Lehr, C.-M. A Model for the Transient Subdiffusive Behavior of Particles in Mucus. Biophys. J. 2017, 112, 172–179. [Google Scholar] [CrossRef] [Green Version]
- Kaczmarek, B. Tannic Acid with Antiviral and Antibacterial Activity as A Promising Component of Biomaterials—A Minireview. Materials 2020, 13, 3224. [Google Scholar] [CrossRef]
- Fan, H.; Wang, J.; Zhang, Q.; Jin, Z. Tannic Acid-Based Multifunctional Hydrogels with Facile Adjustable Adhesion and Cohesion Contributed by Polyphenol Supramolecular Chemistry. ACS Omega 2017, 2, 6668–6676. [Google Scholar] [CrossRef]
- Qi, X.; Mao, W.; Gao, Y.; Chen, Y.; Chen, Y.; Zhao, C.; Li, N.; Wang, C.; Yan, M.; Lin, C.; et al. Chemical Characteristic of an Anticoagulant-Active Sulfated Polysaccharide from Enteromorpha Clathrata. Carbohydr. Polym. 2012, 90, 1804–1810. [Google Scholar] [CrossRef]
- Guo, S.-C.; Tao, S.-C.; Yin, W.-J.; Qi, X.; Yuan, T.; Zhang, C.-Q. Exosomes Derived from Platelet-Rich Plasma Promote the Re-Epithelization of Chronic Cutaneous Wounds via Activation of YAP in a Diabetic Rat Model. Theranostics 2017, 7, 81–96. [Google Scholar] [CrossRef] [Green Version]
- Hu, L.; Wang, J.; Zhou, X.; Xiong, Z.; Zhao, J.; Yu, R.; Huang, F.; Zhang, H.; Chen, L. Exosomes Derived from Human Adipose Mensenchymal Stem Cells Accelerates Cutaneous Wound Healing via Optimizing the Characteristics of Fibroblasts. Sci. Rep. 2016, 6, 32993. [Google Scholar] [CrossRef] [Green Version]
- Baek, K.; Liang, J.; Lim, W.T.; Zhao, H.; Kim, D.H.; Kong, H. In Situ Assembly of Antifouling/Bacterial Silver Nanoparticle-Hydrogel Composites with Controlled Particle Release and Matrix Softening. ACS Appl. Mater. Interfaces 2015, 7, 15359–15367. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Luu, Y.K.; Chang, C.; Fang, D.; Hsiao, B.S.; Chu, B.; Hadjiargyrou, M. Incorporation and Controlled Release of a Hydrophilic Antibiotic Using Poly(Lactide-Co-Glycolide)-Based Electrospun Nanofibrous Scaffolds. J. Control. Release 2004, 98, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Tian, R.; Qiu, X.; Yuan, P.; Lei, K.; Wang, L.; Bai, Y.; Liu, S.; Chen, X. Fabrication of Self-Healing Hydrogels with On-Demand Antimicrobial Activity and Sustained Biomolecule Release for Infected Skin Regeneration. ACS Appl. Mater. Interfaces 2018, 10, 17018–17027. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, Y.; Wang, X.; Ji, S.; Tian, S.; Wu, H.; Luo, P.; Fang, H.; Wang, L.; Wu, G.; Xiao, S.; et al. Mepenzolate Bromide Promotes Diabetic Wound Healing by Modulating Inflammation and Oxidative Stress. Am. J. Transl. Res. 2016, 8, 2738–2747. [Google Scholar] [PubMed]
- Wu, C.; Belenda, C.; Leroux, J.-C.; Gauthier, M.A. Interplay of Chemical Microenvironment and Redox Environment on Thiol–Disulfide Exchange Kinetics. Chem.-A Eur. J. 2011, 17, 10064–10070. [Google Scholar] [CrossRef]
- Zhu, Y.; Yao, Z.; Liu, Y.; Zhang, W.; Geng, L.; Ni, T. Incorporation of ROS-Responsive Substance P-Loaded Zeolite Imidazolate Framework-8 Nanoparticles into a Ca2+-Cross-Linked Alginate/Pectin Hydrogel for Wound Dressing Applications. Int. J. Nanomed. 2020, 15, 333–346. [Google Scholar] [CrossRef] [Green Version]
- Bertoni, S.; Liu, Z.; Correia, A.; Martins, J.P.; Rahikkala, A.; Fontana, F.; Kemell, M.; Liu, D.; Albertini, B.; Passerini, N.; et al. PH and Reactive Oxygen Species-Sequential Responsive Nano-in-Micro Composite for Targeted Therapy of Inflammatory Bowel Disease. Adv. Funct. Mater. 2018, 28, 1806175. [Google Scholar] [CrossRef]
- Chung, M.-F.; Chia, W.-T.; Wan, W.-L.; Lin, Y.-J.; Sung, H.-W. Controlled Release of an Anti-Inflammatory Drug Using an Ultrasensitive ROS-Responsive Gas-Generating Carrier for Localized Inflammation Inhibition. J. Am. Chem. Soc. 2015, 137, 12462–12465. [Google Scholar] [CrossRef]
- Šoltés, L.; Mendichi, R.; Kogan, G.; Schiller, J.; Stankovská, M.; Arnhold, J. Degradative Action of Reactive Oxygen Species on Hyaluronan. Biomacromolecules 2006, 7, 659–668. [Google Scholar] [CrossRef]
- Yang, N.; Zhu, M.; Xu, G.; Liu, N.; Yu, C. A Near-Infrared Light-Responsive Multifunctional Nanocomposite Hydrogel for Efficient and Synergistic Antibacterial Wound Therapy and Healing Promotion. J. Mater. Chem. B 2020, 8, 3908–3917. [Google Scholar] [CrossRef] [PubMed]
- Tracy, L.E.; Minasian, R.A.; Caterson, E.J. Extracellular Matrix and Dermal Fibroblast Function in the Healing Wound. Adv. Wound Care 2014, 5, 119–136. [Google Scholar] [CrossRef] [PubMed]
- McCarty, S.M.; Percival, S.L. Proteases and Delayed Wound Healing. Adv. Wound Care 2013, 2, 438–447. [Google Scholar] [CrossRef] [PubMed]











| Natural Polysaccharides | Advantages | Disadvantages |
|---|---|---|
| Chitosan/chitin | Facile modification methods, Versatility of functionalization, Inherent antibacterial activity, low cost | Performances of different batches greatly vary Non-soluble at neutral conditions Non-FDA approved Weak mechanical properties |
| Hyaluronic acid | Biocompatible, biodegradable, FDA-approved Inherent antioxidative properties Facile modification methods Versatility of functionalization High water-holding capacity | Weak mechanical properties Poor tissue-adhesion Fast degradation rates Expensive |
| Dextran | Biocompatible, biodegradable, FDA-approved Resistance to protein adsorption Facile modification methods Versatility of functionalization High water holding/absorbing capacity Low cost | Weak mechanical properties Poor tissue adhesion |
| Sodium alginate | Biocompatible, water-soluble Facile modification methods Low cost | Weak mechanical properties |
| Cellulose | Good biocompatibility, non-toxic, good mechanical properties | Solubility issues Non-adherent |
| Natural Polysaccharide | Composite/Active Agent | Synthetic Pathway/ Crosslinking | Stimuli- Responsiveness | Wound Healing Application | Clinical and Non-Clinical Application | Reference |
|---|---|---|---|---|---|---|
| Chitosan | Carbon dots | Solvent casting method | pH-monitoring | Antibacterial | In vivo (rats) | [70] |
| Quaternized chitosan and dopamine-oxidized dextran | Silver NPs and deferoxamine | Imine bonds and catechol-catechol adducts | pH-responsive | Antibacterial Angiogenic | In vivo (rats) | [53] |
| Oxidized hyaluronic acid and dihydrazide hyaluronic acid | Sisomicin sulfate or quaternized chitosan | Imine and acylhydrazonebonds | pH and enzyme-responsive | Antimicrobial | In vivo (mice) | [51] |
| Dopamine-grafted oxidized carboxymethyl cellulose | Poly(vinyl alcohol)-borax, neomycin | Hydrogen, borate ester, and imine bonds | pH-responsive | Antibacterial antioxidant | In vitro | [73] |
| Heparin | Poloxamer, Keratinocyte growth factor, ε-polylysine | Electrostatic interactions | Thermo-responsive | Wound healing of endometrial injury | In vivo (rats) | [74] |
| Hydroxypropyl chitin | Tannic acidFe3+ ions | Hydrogen and coordination bonds | Thermo-responsive pH-responsive | Antibacterial | In vivo (mice) | [45] |
| Hyaluronic acid | Ethylenediaminetetraacetic acid (EDTA)−Fe3+ complexes Platelet-derived growth factor | Coordination interactions | Enzyme-responsive | Antibacterial cutaneous regeneration | In vivo (mice) | [85] |
| Hyaluronic acid | Glutathione, Aminoethyl disulfide | Amide bonds | Redox-responsive | Wound monitoringantioxidant fibroblast growth | In vitro | [7] |
| Sodium alginate pectin | ZIF-8 nanoparticles calcium chloride, neuro peptide (SP), PEG-thioketal | Ionic cross-linking | Redox responsive | Wound healing acceleration | In vivo (mice) | [88] |
| Hyaluronic acid-cholesterol and alginate-boronic acid | Naproxen, amikacin | Boronic ester | Redox and pH-responsive | Wound healing acceleration, antibacterial, anti- inflammation | In vivo (rats) | [57] |
| Chitosan | Tungsten disulfide nanosheets (WS2-NS2), ciprofloxacin | Imine bonds | Photo-responsive | Antimicrobial, hemostaticantioxidant | In vivo (mice) | [92] |
| Hyaluronic acid- β-cyclodextrin and hyaluronic acid-azobenze | Epidermal growth factor | Host–guest interactions | Photo-responsive | Angiogenesis, granulation tissue formation | In vivo (mice) | [54] |
| Quaternized chitosan, and oxidized hyaluronic acid | Epidermal growth factor PEDOT:PSS, berberine | Imine bonds | pH-responsive | Antibacterial, wound healing acceleration | In vivo (mice) | [32] |
| Quaternized chitosan | Pluronic®F127 (PF127-CHO), curcumin | Imine bonds | pH-responsive | Antibacterial, hemostatic | In vivo (mice) | [52] |
| N-narboxyethyl chitosan, oxidized hyaluronic acid | Adipic acid dihydrazide, insulin | Acylhydrazones and imine bonds | pH-responsive | Diabetic wound healing, Re-epithelization Angiogenesis, | In vivo (rats) | [38] |
| N-carboxyethyl chitosan and oxidized hyaluronic acid | Polyaniline, amoxicillin | Imine bonds | pH and electro-responsive | antibacterial, antioxidant, angiogenesis, collagen disposition | In vivo (mice) | [39] |
| Oxidized hyaluronic acid | Antimicrobial peptide KK(SLKL)3KK | Imine bonds | pH-responsive | Antibacterial | In vivo (mice) | [66] |
| Oxidized pullulan | Polyethyleneimine, Pluronic F127 | Imine bonds | pH and thermo responsive | Antibacterial, hemostatic, stimulate angiogenesis, remodelling and re-epithelialization | In vivo (mice) | [56] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Psarrou, M.; Mitraki, A.; Vamvakaki, M.; Kokotidou, C. Stimuli-Responsive Polysaccharide Hydrogels and Their Composites for Wound Healing Applications. Polymers 2023, 15, 986. https://doi.org/10.3390/polym15040986
Psarrou M, Mitraki A, Vamvakaki M, Kokotidou C. Stimuli-Responsive Polysaccharide Hydrogels and Their Composites for Wound Healing Applications. Polymers. 2023; 15(4):986. https://doi.org/10.3390/polym15040986
Chicago/Turabian StylePsarrou, Maria, Anna Mitraki, Maria Vamvakaki, and Chrysoula Kokotidou. 2023. "Stimuli-Responsive Polysaccharide Hydrogels and Their Composites for Wound Healing Applications" Polymers 15, no. 4: 986. https://doi.org/10.3390/polym15040986