1. Introduction
Gene silencing using synthetic oligonucleotides such as a small-interfering RNA (siRNA) or an antisense oligonucleotide (ASO) is a promising approach for suppressing the expression of pathogenic proteins in cells/tissues. Since these oligonucleotides are easily degraded by enzymes and easily eliminated from the systemic circulation by renal clearance, nano-sized drug delivery systems (DDS) are useful for protection from degradation and the control of their pharmacokinetics [
1]. In addition, the rational design of DDS could allow for more efficient endosomal escape. One of the most intensely investigated delivery systems is a lipid nanoparticle (LNP), which consists of artificial materials such as pH-sensitive cationic lipids or ionizable lipids [
2]. In 2018, an LNP-based oligonucleotide therapeutic (ONPATTRO
®) was approved as the first siRNA therapeutic by the Food and Drug Administration (FDA) and the European Medicines Agency (EMA) [
3]. Based on this success, the use of an LNP-type DDS promises to further contribute to the development of siRNA therapeutics/gene therapy.
An important function of ionizable lipids is the pH-sensing ability of their tertiary amine groups. The neutral surface charge of the LNP at physiological pH is beneficial for avoiding undesired interactions with biomolecules. Once taken up by cells, the protonation of the tertiary amine groups in the acidic environment in endosomes results in the development of a cationic charge to the surface of the LNP. The cationic charge facilitates interactions between the LNP and the endosomal membrane, which promotes the subsequent escape of the oligonucleotides from the endosomal degradation pathway to the cytoplasm.
To achieve efficient gene regulation, oligonucleotides must be delivered to the cytoplasm. The release of oligonucleotides from the DDS is an important step in intracellular trafficking as well as in endosomal escape. To facilitate the release of the oligonucleotides, bio-degradable functional units are incorporated into the materials of the DDS [
4]. The disulfide bond is frequently used in the design of bio-degradable materials in order to accelerate the release of oligonucleotides [
5,
6,
7]. Since the intracellular concentration of glutathione (GSH, γ-Glu-Cys-Gly), a naturally produced reducing agent is, at most, 1000-fold higher intracellularly than in the extracellular environment, the disulfide bond can be selectively cleaved in the cytoplasm [
8]. The rate of cleavage of a reduction-sensitive fluorescent probe that was directly microinjected into the cytoplasm indicated that the reaction reaches completion within approximately 2 min in the cell [
9]. This spatial selectivity and high reactivity of the disulfide bond makes it suitable for use as a trigger that activates the destabilization of the DDS and the subsequent intracellular release of oligonucleotides.
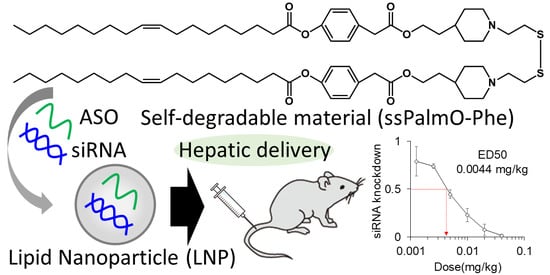
For the delivery of oligonucleotides/genes, we previously developed an SS-cleavable and pH-activated lipid-like material (ssPalm) as a component of the LNP (LNP
ssPalm) [
10,
11,
12]. ssPalm contains two tertiary amine residues with fatty acid scaffolds that are bridged via a disulfide bond. These functional units are integrated into one molecule for facilitating both endosomal escape and the release of the loaded nucleic acids into the cytoplasm. We recently developed ssPalmO-Phe as a self-degradable derivative of ssPalm that is self-degraded in the intraparticle space by a specific hydrolytic reaction (
Figure 1) [
13]. The key functional units in its structure are phenyl esters that are located between the tertiary amines and fatty acid scaffolds. Since the phenyl esters are susceptible to the hydrolysis mediated by a thiol group, the intraparticle accumulation of thiol groups after the reduction of LNP
ssPalm triggers the subsequent hydrolysis of the phenyl esters. The hydrolysis results in the dissociation of the tertiary amines and hydrophobic scaffolds from the LNP. Of note, it was unexpectedly discovered that the insertion of an aromatic ring facilitates endosomal escape by enhancing membrane destabilizing activity. These features of LNP
ssPalmO-Phe function synergistically to promote the delivery of the loaded oligonucleotides/genes to the cytoplasm.
While ssPalmO-Phe was originally developed for the delivery of in vitro-transcribed messenger RNA (mRNA), we hypothesized that the self-degradation-driven release of the nucleic acids would also improve the efficiency of delivery of the other types of oligonucleotides. However, it is now recognized that the optimal lipid composition of the LNP is dependent on the type of nucleic acids being delivered [
14,
15]. In this study, we explored the optimal composition of LNP
ssPalmO-Phe for the hepatic delivery of siRNA by monitoring the membrane destabilizing ability of the system (LNP
ssPalmO-Phe–siRNA), and the resulting LNP formulation was also then used for ASO delivery (LNP
ssPalmO-Phe–ASO).
2. Materials and Methods
2.1. Animal Experiments
For the in vivo analysis of LNPssPalmO-Phe–siRNA, C57BL6/J mice (male, 4–6 weeks of age) and ICR mice (male, 6–7 weeks of age) were purchased from Japan SLC, Inc (Shizuoka, Japan). The experimental protocols were reviewed and approved by the Chiba University Animal Care Committee in accordance with the Guide for Care and Use of Laboratory Animals. Ethical approval codes issued from the committee for this research include: 30–41. For the in vivo analysis of LNPssPalmO-Phe–ASO, C57BL/6J mice (male, 5 weeks of age) were purchased from Charles River Laboratories Japan, Inc (Yokohama, Japan). The experimental procedures were reviewed and approved by the Institutional Animal Care and Use Committee of LSI Medience Corporation (Ibaraki, Japan). The ethical approval codes issued by the committee were 2020-0002 and 2020-0065.
2.2. Materials
Detailed information from suppliers is listed in the Supplementary Material
Table S1. The synthesis of ssPalmO-Ben (a non-degradable counterpart of ssPalmO-Phe) has been described in a previous manuscript [
13]. ssPalmO-Phe (Product # COATSOME
® SS-OP), ssPalmO-P4C2 (Product # COATSOME
® SS-OC), ssPalmE-P4C2 (Product # COATSOME
® SS-EC), 1,2-dioleoyl-sn-glycero-3-phosphatidylcholine (DOPC; Product # COATSOME
® MC-8181), 1,2-distearyol-sn-glycero-3-phosphatidylcholine (DSPC; Product # COATSOME
® MC-8080), and 1-(monomethoxy polyethyleneglycol2000)2,3-dimyristoylglycerol (DMG-PEG2000; Product # SUNBRIGHT
® GM-020) were manufactured by NOF CORPORATION (Kanagawa, Japan). The suffix “P4C2” refers to 4-ethylpiperdine moieties that were incorporated as head groups into the structure. siRNA was purchased from Hokkaido System Science Co., Ltd. (Hokkaido, Japan). The sequence of the siRNA against factor VII (siFVII) has been reported previously [
11]. A gapmer ASO targeting ApoB and one targeting Nr3c1 (also known as a glucocorticoid receptor) were purchased from Gene Design, Inc. (Osaka, Japan). The sequences of these ASOs, named ASOap and ASOgr, respectively, are provided in the
Supplementary Materials Table S2. Cholesterol and 6-(p-toluidino)-2-naphthalenesulfonic acid sodium salt (TNS) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Amicon Ultra-4-100K centrifugal units and Amicon Ultra-15-100K centrifugal units were purchased form Merck Millipore (Darmstadt, Germany). Phosphate buffered saline (PBS) without Ca
2+ and Mg
2+ (PBS minus; PBS(−)) was purchased from NACALAI TESQUE, INC. (Kyoto, Japan). Ultrapure
TM Distilled water was purchased from Thermo Fisher Scientific (Waltham, MA, USA). Otsuka normal saline was purchased from Otsuka Pharmaceutical (Tokyo, Japan). All other reagents and chemicals were commercially available and were used without further purification.
2.3. Preparation of siRNA-Encapsulating LNP by a Microfluidic Device (siRNA)
For the preparation of the siRNA-encapsulating LNP used in “Effects of DOPC and cholesterol on hepatic knockdown efficiency” by an invasive lipid nanoparticle production (iLiNP) device [
16], siFVII was diluted to a concentration of 7.5 μg/mL in 20 mM malic acid/NaOH buffer (600 μL, pH 3.0, with 30 mM NaCl). The lipid–ethanol solution was prepared at a concentration of 2.5 mM (400 μL). These solutions were mixed using the iLiNP device (total flow rate: 1 mL/min; flow ratio of water/ethanol: 3/2 (
v/v)) by a syringe pump (Pump 33 Dual Drive System, Harvard Apparatus, Cambridge, MA, USA). The siRNA–lipid mixture (1 mL in total) was recovered and diluted with 3 mL of PBS(−). For the preparation of the siRNA-encapsulating LNP used in “Effects of the amount of PEG–lipid used” by NanoAssemblr
TM (Vancouver, BC, Canada), siFVII was diluted at a concentration of 0.69 μg/mL in 20 mM malic acid/NaOH buffer (650 μL, pH 3.0, with 30 mM NaCl). The lipid–ethanol solution was prepared at a concentration of 1.93 mM total lipids (350 μL). These solutions were mixed using a NanoAssemblr
TM (total flow rate: 20 mL/min; flow ratio of water/ethanol: 6.5/3.5 (
v/v); total volume: 1 mL). The lipid–siRNA mixture (1 mL) was recovered and diluted with 3 mL of PBS(−). For the preparation of the siRNA-encapsulating LNP used in “Effects of the lipid/RNA ratio” and “Hemolytic activity in the presence of serum proteins” by a NanoAssemblr
TM, siFVII was diluted at a concentration of 1.25 μg/mL in 20 mM malic acid/NaOH buffer (900 μL, pH 3.0, with 30 mM NaCl). The lipid–ethanol solution was prepared at a concentration of 4.5 mM total lipid (100 μL). These solutions were mixed using the NanoAssemblr
TM (total flow rate: 20 mL/min; flow ratio of water/ethanol: 9/1 (
v/v); total volume: 1 mL). The siRNA–lipid mixture (1 mL) was recovered and diluted with 3 mL of PBS(−).
In all of these prepared samples, the external solution was replaced with PBS(−) by ultrafiltration using Amicon Ultra-4-100K (Darmstadt, Germany) centrifugal units (25 °C, 1000×
g). The particle solution was diluted to an adequate concentration in PBS(−) before administration. Encapsulation efficiency and recovery of the siRNA were obtained by Quant-IT
TM RiboGreen
TM assay. The concentration of total siRNA and non-encapsulated siRNA were quantified after the LNP samples had been treated with Triton X-100 and without Triton X-100, as previously described [
11]. The encapsulation efficiency was calculated as follows:
2.4. Preparation of ASO-Encapsulating LNP by a Microfluidic Device
ASO was diluted to a concentration of 0.167 mg/mL in 20 mM malic acid/NaOH buffer (2400 μL, pH 3.0, with 30 mM NaCl). The lipid–ethanol solution was prepared at a concentration of 12.5 mM of total lipid (800 μL). These solutions were mixed using a NanoAssemblrTM (total flow rate: 12 mL/min; flow ratio of water/ethanol: 3/1 (v/v)). The ASO–lipid mixture (3.2 mL in total) was recovered and diluted with 12.8 mL of PBS(−). The external solution was replaced by ultrafiltration with UltrapureTM distilled water using Amicon Ultra-15-100K centrifugal units (25 °C, 1000× g). The particle solution was diluted to an adequate concentration with Otsuka normal saline before administration. The recovery of the ASO was determined by a Quant-ITTM OliGreenTM assay. The OliGreenTM assay was performed according to the manufacture’s protocol in the presence of 0.1% of Triton X-100. The calibration curve was generated using 125–1000 ng/mL ASO. Since the OliGreenTM assay of the LNP–ASO in the absence of Triton X-100 did not produce reliable fluorescence, the encapsulation efficiency of the ASO was not calculated.
2.5. Evaluation of Surface pKa by TNS Assay
For the 6-(p-Toluidino)-2-naphthalenesulfonyl chloride (TNS) assay, 20 mM citric acid/NaOH buffer (with 150 mM NaCl, pH 3.0, 3.5, 4.0, 4.5, 5.0, 5.5), 20 mM sodium dihydrogen phosphate/NaOH buffer (with 150 mM NaCl, pH 6.0, 6.4, 6.8, 7.2, 7.6, 8.0), and 20 mM Tris/HCl buffer (with 150 mM NaCl pH8.5, 9.0, 9.5, 10.0) were prepared. TNS was dissolved at 0.6 mM in water as a stock solution. In the wells of a 96-well black plate, 2 μL of the TNS solution, 12 μL of the LNP solution (0.5 mM total lipid) and 186 μL of the each of the buffers above were mixed. After shaking the incubation mixture (400 rpm, 10 min), the fluorescence of the TNS (Ex: 321/Em: 447) was measured. The apparent pKa of the surface was calculated as the pH at which the LNP showed 50% of the maximum fluorescence.
2.6. Hemolysis Assay
pH-dependent hemolytic activity was used as an index of endosomal escape efficiency [
17,
18]. Phosphate buffered saline with malic acid (PMBS) buffer was prepared by dissolving DL-malic acid with PBS(−) to a concentration of 20 mM. The pH was then adjusted to pH 5.5, pH 6.5, and pH7.4 with NaOH solution. Whole blood from ICR mice was collected from the inferior vena cava in the presence of 0.5 μL of heparin sodium (5000 U/5 mL). Red blood cells were purified by washing the blood (1 mL) in 9 mL of PBS(−). The blood was centrifuged (4 °C, 400×
g, 5 min) and the supernatant was discarded by aspiration. Washing was repeated 5 times to completely remove serum proteins. The red blood cells were then incubated with the LNP at pH 5.5, pH 6.5, and pH 7.4. The final concentration of the total lipid was from 1.56 μM to 400 μM. For hemolysis assays in the presence of serum proteins, the serum was collected and the protein concentration was quantified using the BCA Protein Assay kit (TAKARA Bio, Inc., Shiga, Japan) according to the manufacture’s protocol. The serum (10–60 μg of protein; final concentration: 20–120 μg/mL) was added to the red blood cells and the hemolysis assay was conducted.
2.7. FVII Assay
siFVII-encapsulating LNPssPalmO-Phe was diluted to the appropriate concentrations with PBS(−) and administered to C57BL6/J mice intravenously at the indicated dose. The volume was adjusted to 10 mL/kg. At 24 h after injection, 400 μL of blood was collected in the presence of 0.5 μL of heparin sodium (5000 U/5 mL) and stored on ice until used. The plasma concentration of FVII was determined using a colorimetric Biophen VII assay kit (Hyphen Biomed) according to the manufacturer’s protocol. The standard curve for Factor VII plasma levels was generated using plasma collected from non-treated mice. The baseline expression level of FVII was represented by the PBS-treated group.
2.8. Evaluation of Hepatotoxicity of LNPssPalmO-Phe
LNPssPalmO-Phe was diluted to the appropriate concentrations with saline and intravenously administered to C57BL6/J mice at the indicated dose. The volume was adjusted to 10 mL/kg. At 96 h after injection, 500 μL of blood was collected and blood samples were processed to obtain serum, which was then stored below −80 °C until used. The level of serum aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were measured using a biochemical autoanalyzer (DRI-CHEM 7000, Fujifilm Co., Tokyo, Japan, or TBA-2000FR, Canon Medical Systems Co., Tochigi, Japan).
2.9. Evaluation of the Knockdown Efficiency of ASOs
ASOap, ASOgr, ASOap-encapsulating LNP
ssPalmO-Phe and ASOgr-encapsulating LNP
ssPalmO-Phe were diluted to the appropriate concentrations with saline and administered to C57BL6/J mice intravenously at the indicated dose. The volume was adjusted to 10 mL/kg. At 96 h after injection, livers were isolated from mice and stored in RNAlater RNA Stabilization Reagent (Qiagen, Valencia, CA, USA) overnight. Livers were homogenized using a BioMasher II (Nippi, Tokyo, Japan). Total RNA was isolated from mouse liver tissues using the RNeasy Mini Kit (Qiagen, Valencia, CA, USA) according to the manufacturer’s protocol. qRT-PCR was performed using a One Step TB Green PrimeScript PLUS RT-PCR Kit (TAKARA Bio, Inc., Shiga, Japan) and analyzed with a 7500 Fast Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). The primers used in this study are shown in the
Supplementary Materials. The level of target gene expression was normalized to that of mouse Glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Hepatotoxicity evaluation was performed as described above.
4. Discussion
In this study, the optimal composition of LNP
ssPalmO-Phe was explored in an attempt to achieve more efficient delivery of a series of oligonucleotides. The findings revealed that the optimal composition for siRNA delivery was ssPalmO-Phe/Chol = 70/30, and that for the ASO delivery, the optimal composition was ssPalmO-Phe/DOPC/Chol = 40/20/40, respectively. Both compositions contained an additional 1.5% of DMG-PEG2000. These compositions were different from the composition (ssPalmO-Phe/DOPC/Chol = 52.5/7.5/40) that was optimized for the delivery of mRNA [
13]. This observation strongly supports the current understanding that the composition of the LNP needs to be optimized for each therapeutic modality being considered [
14,
15]. The differences can be partially attributed to the chemical properties of each nucleic acid. The mRNA molecule is the most vulnerable molecule among these nucleic acids, since mRNA basically does not contain chemical modification in its phosphodiester backbone and ribose structure. The mRNA molecule can easily be degraded by nucleases in the extracellular environment. Hydrolysis of the backbone would result in complete loss of its function and would cause its subsequent degradation by nucleases. Thus, the incorporation of 7.5% DOPC and 40% cholesterol seems to be a prerequisite for protecting it by improving membrane integrity.
In the case of siRNA, the molecule is chemically more stable than mRNA, since its ribose structure has been artificially modified. However, for eliciting the maximum potency of the siRNA, it is important that the siRNA reaches the cytoplasm in double-stranded form [
28]. It is known that the optimal pKa of LNP systems for the hepatic delivery of siRNA was 6.2–6.5 [
24]. These observations suggest that the siRNA may need to be released in an intact form from the endosomal compartments at an early stage of the endocytosis process. The results shown in
Figure 5a suggest that the optimal composition of LNP
ssPalmO-Phe for the delivery of siRNA has a suitable membrane-destabilizing ability, since hemolytic activity was observed at pH 6.5. To achieve a high endosomal escape efficiency, the optimal composition for siRNA required a large amount of ionizable lipids (up to 70% of the total lipids).
As LNP
ssPalmO-Phe containing ASO is unstable, the ratio of DOPC needed to be increased to 20% of the total lipids. Although ASOap (13 mer) and ASOgr (14 mer) have a similar molecular size and charge, the Zeta-potential and ASO recovery ratio for LNP
ssPalmO-Phe–ASOap and LNP
ssPalmO-Phe–ASOgr were different (
Table 3). These differences can be partly explained by the structure of ASO. While siRNA and mRNA molecules have a rigid double-stranded structure as a result of inter- and intra-molecular base pairing, ASO has a flexible single-stranded structure. This indicates that the nucleobases of ASO, in addition to its phosphorothioate backbone, can interact with ssPalm. In other words, in the case of single-stranded ASO, the sequence and/or nucleobase content would have a profound effect on the properties of the LNP. This sequence dependency should be taken into account in the case of ASO delivery using DDS. Since the ASO itself was highly stable and relatively hydrophobic because of its chemically modified structure, it is expected that the ASO would reach the cytoplasm more readily once taken up by the cells. It is well known that a neutral LNP is taken up by hepatocytes via apolipoprotein E (ApoE) and low-density lipoprotein receptor (LDLR)-dependent endocytosis [
29]. This hepatic uptake mediated by these endogenous proteins most likely plays a key role in the hepatic delivery of ASO. To ensure efficient extravasation through the fenestra and efficient uptake by the liver cells, the use of a lipid composition that permits stable and small particles to be prepared is important for the delivery of ASO. Collectively, the lipid composition of the LNP should be determined by taking the critical steps for each nucleic acid into consideration: extracellular stability of the mRNA, endosomal escape of the siRNA, and cellular uptake of the ASO, respectively.
The findings shown in
Figure 5a revealed that the incorporation of an aromatic ring improved endosomal escape efficiency. It was unexpectedly revealed that LNP
ssPalmO-Phe conferred resistance against the effect of serum proteins compared to the conventional ionizable lipids (
Figure 7 and
Figure S1). Since this resistance against serum proteins was also observed for ssPalmO-P4C2 (
Figure S2), we concluded that this property was conferred by the structural characteristics of ssPalm and not by the insertion of a phenyl ester. As the bond angle of the disulfide bond is located at the center of the chemical structure, the conformation of ssPalm was calculated as a linear form compared to that of conventional lipids with head–tail structures. The distribution of the angles formed by the two oleic acid scaffolds ranges from 30° to 150°, and for ssPalmO-P4C2, the most frequent angle was 140° (open conformation) [
13]. Thus, the surface of LNP
ssPalm might be hydrophobic compared to an LNP that is composed of conventional lipids. This difference in the surface properties of LNP
ssPalm would be expected to result in changes in molecular interactions and the quantity of the proteins on the surface of the particles. However, the detailed mechanisms of the resistance to serum proteins remains to be clarified.
The findings reported herein reveal that the encapsulation of ASO in LNP
ssPalmO-Phe significantly improved the knockdown efficiency of the preparation (
Figure 8). In parallel, the liver toxicity of the ASO was also observed at lower doses (
Figure 9). The dose for the total lipids for LNP
ssPalmO-Phe–ASOap and LNP
ssPalmO-Phe–ASOgr was 0.41–30 mg/kg and 0.14–10.1 mg/kg, respectively. Since there was no evidence of toxicity for the empty particle in the concentration range of 49–782 mg/kg total lipids (29–470 mg/kg ssPalmO-Phe), the toxicity observed for LNP
ssPalmO-Phe-ASO was due to the improved efficiency of the delivery of these toxic ASOs (
Figure 10). On the other hand, we previously reported that the administration of LNP
ssPalmO-Phe with an optimal composition for mRNA at a dose of more than 350 mg/kg total lipids (>260 mg/kg ssPalmO-Phe) induced an elevation in serum AST/ALT levels [
13]. These observations indicate that the liver toxicity of LNPs
sPalmO-Phe was not simply determined by the dose of the ssPalmO-Phe molecule, but also reflects the difference in the lipid composition. The ratio of ssPalmO-Phe/DOPC was different between the lipid compositions for ASO (ssPalmO-Phe/DOPC/Chol = 40/20/40) and mRNA (ssPalmO-Phe/DOPC/Chol = 52.5/7.5/40). It is generally thought that phospholipids in the LNP are aligned on the surface of the nanoparticle [
25]. Due to its amphiphilicity and overall cylindrical shape, these phosphatidylcholines confer stability to the surface of LNP
ssPalmO-Phe. Although the stability of a nanoparticle is important in maintaining its dispersibility, excess stabilization could result in poor transfection activity. On the other hand, a strong membrane destabilizing ability is also accompanied by the risk of adverse responses due to the leakage of the endosomal compartment into the cytoplasm. The destabilization of the endosome, also known as endosomolysis, results in cell death via apoptosis/necrosis depending on the severity of the membrane destabilization [
30,
31,
32]. This toxic event can be induced by exogenous molecules such as a crotamine derivatives and can activate a cathepsin–caspase axis [
33,
34].
Since the optimal composition of an LNP depends on the physicochemical/biological properties of each nucleic acid, it is difficult to present a general principle for the process of optimization for different types of nucleic acids. In the case of RNAs (siRNA and mRNA), endosomal escape is still considered to be a major barrier that inhibits cytoplasmic delivery. Thus, hemolytic activity, the index of endosomal escape efficiency, needs to be prioritized. Since the phospholipid content is difficult to determine using in vitro experiments, experimental verification in vivo would be needed to develop an optimal LNP. However, hepatic damage may also depend on the membrane destabilizing activity of the LNP. Thus, a parallel evaluation of the hemolytic activity and the hepatic toxicity is needed to confirm the efficacy and safety. On the other hand, ASO is a unique material because of its innate membrane permeability characteristics. In this case, stability in the circulation should be a more important parameter to be optimized for hepatic targeting, as avoiding renal clearance is the main purpose of the use of an LNP. Thus, the optimal composition depends on the combination of the ASO being used and its target.