4.1. Probiotic Properties
Probiotic characterization from plant sources has gained attention as it integrates human health benefits. The current study comprised 53 strains that were isolated to be co-cultured in tapai pulut
(Oryza Sativa L.) and intensively studied to evaluate their potential probiotics as an added value. The present study is consistent with previous studies on traditional fermented food known as tempoyak [
18,
19], in which LAB was involved in both traditional fermented foods.
L. plantarum is a dominant species of
Lactobacilli in vegetables and fruits such as cucumber and cassava [
20]. However, LAB isolated from different strains has different capabilities of becoming lactic starters/probiotics, as they depend on the temperature, time, pH, and medium for growth, including carbon and nitrogen sources [
21].
The tolerance to the harsh environment of the gastrointestinal tract is one of the main factors limiting probiotics’ use. Acid and bile salt tolerances are vital for probiotics to survive in the gut [
22]. The ability of potential probiotic strains to withstand the low-pH environment (at pH 2.0) of the stomach is the first limitation before they can reach and enter to colonize the host’s small intestine. The survival of the strains at pH 3.0 is considered an optimum acid tolerance in the stomach. Likewise, food digestion and ingestion provide a buffering effect that elevates the pH to 3.0 [
23]. Hence, this study examined the strains’ abilities to tolerate pH 3.0. Good resistance to low pH with some minor differences was depicted among the ten strains. These results from RB5 strains are aligned with those obtained from previous studies, which proves that resistance of
Lactobacillus strains of human or animal origin or fermented food, after exposure to pH, ranges from 1.0 to 3.0 because of strain-specific attitudes, and the surviving and resistance percentages of strains were higher at pH 3.0 [
24].
High bile salts (%) cause disorganization of the cell wall, oxidative stress, DNA damage, protein denaturation, and intracellular acidification [
25]. Therefore, it is necessary to examine the ability of the strains to withstand the bile salts through the probiotic screening process. The range of bile salt in the gut is between 3.5 to 4% after an hour of food ingestion, and it reduces gradually to around 3% [
26]. Instead, it has been proven that the amount of bile salt in percentage in the human small intestine is 3% (
w/
v). Hence, bile salts at 3% were used in this study to investigate the bile salts’ survivabilities of the strains. The results show that the ten strains showed good resistance when exposed to 3% bile salts. These findings match those observed in earlier studies [
27], where all nine isolated strains (CL3, PC2, TA8, PC4, AFSF2, FS3, RB5, CL8, and HML3) exhibited good tolerance to 3% bile salt. This study also confirmed that the
L. acidophilus group and
L. fermentum show tolerance to high bile salt.
At 40 °C, the survivability of the strains is more than 65%, which aligns with another study [
28] that studied three incubation temperatures of 37, 40, and 44 °C and three cold storage temperatures (2, 5, and 8 °C) on the probiotic’s survivability levels during the production of yogurt. It was shown that 20 days of fermentation at 37 °C and at a temperature of 2 °C of storage led to the highest survival rate of
L. acidophilus, whereas
Bifidobacteria survived optimally at 8 °C. It was noted that the survival levels of probiotics during fermentation at 40 °C was higher than 44 °C. This is because of the production of acetic acid in a large amount released by
Bifidobacteria species and the antagonistic effect of yogurt starter bacteria in the end product stage [
29]. The same study depicted that throughout the fermentation process at 37 °C and 44 °C, 6 h was required to detect and attain probiotic survival levels at the lowest and highest, respectively.
The presence of enzymes could be effective in the maintenance of probiotic cells [
30]. It has been reported that using pepsin and glucose in fermented samples positively supports potential probiotic survival through the reduction of oxidation [
31]. The mechanism of action occurs when the enzyme catalyzes the conversion of glucose to gluconic acid and hydrogen peroxide by engulfing the oxygen that threatens the survival of some probiotics in the fermented medium. The observed optimum concentrations of glucose oxidase and glucose are 62.32 and 4.35 mg/kg, respectively [
30]. In a separate study [
32] to compare the plastic packages with lower oxygen permeability and glucose oxidase for preserving the potential probiotics in the fermented product, yogurt, a reduction of oxygen level was observed in the probiotic counts (higher than 7 log CFU) without changes in the sensorial and organoleptic properties of the yogurt. Enzyme tolerance is a variable to determine the ability of the selected probiotics to function during the presence of enzymes used in modulating the human gut.
Antibiotic resistance phenotype is a criterion for assessing probiotic safety issues such as the presence of pathogens and health risk issues. The probiotics should not have a resistance to antibiotics because resistant strains that harbor acquired and transferable antibiotic resistance genes can transfer these genes to pathogenic microorganisms [
33]. In this study, all of the isolated strains are resistant to DA (clindamycin), E (Erythromycin), IPM (Imipenem), VA (Vancomycin), and GN (Gentamicin). However, all the strains are sensitive to C (Chloramphenicol), and all strains except for CL8 are sensitive to TE (tetracycline). Consistent with our results, in a study [
32], it was found that most
Lactobacillus species are intrinsically resistant to vancomycin, which indicates their safety [
34]. Furthermore, Jafari-Nasab [
35] reported that
Lactobacilli are susceptible to erythromycin, while
Lactobacillus strains are susceptible to gentamicin and erythromycin [
36]. The susceptibility to such antibiotics is due to nucleic acid synthesis inhibitors, which have a low inhibitory effect on most
Lactobacillus species.
4.2. Phenotypic and Genotypic Identification
The sugar fermentation in LAB strains was determined by employing the API 50 CHL test strips (bioMérieux, Craponne, France). The strips work by utilizing 50 single-carbon sources to determine and identify LAB species, according to the manual provided. The strains of LAB were phenotypically characterized by their cell morphologies, gas productions from glucose, arginine hydrolysis, and lactate configurations. Out of 50 sugars, a few highlighted sugars were tabulated. The positive coloration in the strip was indicated in yellow color, whereas the negative remains blue. However, in some of the results obtained, the coloration was distinctively different from what they were supposed to be. Hence, technical support directly from BioMeriux was sought. The strain code CL8 with the tetrad-forming cocci was presumptively classified as belonging to the genus
Pediococcus spp. In contrast, the rod-forming shape is known as
L. plantarum. Table 5 shows some sugar fermentation, in which all nine isolates were absent of arabinose, whereas the CL8 isolates showed as positive. The commercial identification systems API 50 CHL and Biolog allow only for identification at the species level, which does not allow a clear identification at the strain level.
The patterns of the six strains (TA8, PC4, AFSF2, RB5, CL8, and HML3) (after the results from the total bacterial count) were obtained using PCR (SureCycler 8800A, Agilent, Mississauga, ON, Canada) with two universal primers, 27F (AGAGTTTGATCMTGGCTCAG) and 1492 R (TACGGYTACCTTGTTACGACTT) showed high similarity. Therefore, 16S rRNA gene sequencing of six strains was performed and compared with the 16S rDNA gene sequence of Leuc. lactis DSM 20202 revealed the identities of 97.8% to 100 %. In addition, the DNA of each strain was identified. The results shown were agreed in the study by Adesulu-Dahunsi [
37], where the microbials of fermented Nigerian food revealed that in LABs comprising lactobacilli, pediococci were the predominant micro-organisms present in viable numbers above 107 CFU g
−1. A study that compiled the gene sequence data of
Lactobacillus and
Pediococcus spp. was used to determine the gene evolution and its phylogeny [
38] to identify the strains at the species level.
Lactobacilli spp. are differentiated into obligate homofermentative, facultative heterofermentative, and obligate heterofermentative. The differences in this classification of these
Lactobacilli spp. are based on the carbohydrate metabolism, which is the pentose phosphate pathway [
39]. Thus, a combination of phenotypic properties and molecular techniques 16S rRNA gene sequencing allows for identification at the strain level [
40,
41]. The phylogenetic tree of the
Lactobacilli spp. maybe a cornerstone for future studies on the fermented food industry
4.3. Feeding Samples and Growth Performance
The added probiotic effect was significant on the glutinous rice compared with the traditionally fermented sample using the powder yeast starter culture, as observed for the high acidity content and low pH value. On the other hand, the TSS content was significantly increased for the potential probiotic glutinous rice and fermented glutinous rice. Simultaneously, the highest was observed for the glutinous rice fermented with the traditional starter culture. In a previous study, the fermentation process with different starter cultures increased the TSS content and acidity content of glutinous rice [
42]. The strains present only in the potential probiotic glutinous rice sample indicated the absence of the LAB in the powder starter culture known as (
ragi tapai). Probiotics play an essential role in restoring the gut microbiota, and their bioactive compounds (probioactives) are suggested to contribute to the health functionality of fermented foods [
43]. Therefore, the selected strains (RB5) were added jointly with the starter cultures that produce traditional fermented foods. The fermentation process that includes the added potential probiotics is recommended to increase the cell density of the friendly microorganisms and their bioactive compounds in the final product [
44]. In this study, the correlation between the acid and TSS contents was an indicator of the chemical changes due to the probiotic addition alongside the starter culture.
The overall body weight gain in the TPG group was 4.63 g, followed by the PTPG group (3.69 g) and NG group (2.90 g), which correlated with the TSS content in their diets. The traditional fermented glutinous rice had the highest TSS content (53.7 Brix). However, TSS content alone cannot significantly increase body weight. In a recent study, fermented rice products were reported to increase body weight in mice due to the modification of the intestinal microbiota [
45]. It was reported that diets containing probiotics increased body weight gain in tested animals (New Zealand male rats) [
46]. In another study, a diet containing probiotics belonging to different genera, including
Lactobacillus,
Clostridium sensu stricto,
Prevotella, and
Alloprevotella, was reported to increase the body weight in obese mice compared with the control group. The increased body weight was associated with gut microbiota improvement and the reduction of gut pathogens [
47]. Thus, a probiotic diet was reported to regulate body weight gain in obese mice and reduce the risk of obesity [
48]. There is a high potential that the traditional starter culture contains several probiotics belonging to species other than LAB. Therefore, the microorganisms in the starter cultures may be profiled to determine the important probiotics’ potential presence.
4.4. Haematology Analysis
In this study, we demonstrated that the levels of immune cells in the blood, including WBCs, neutros, and lymphs, were significantly (
p < 0.05) increased in mice treated with the probiotic glutinous rice. Probiotic foods and beverages are well known for elevating the immune system cell populations [
49,
50,
51]. Probiotics colonizing in the epithelial cells will boost immune system function by excluding or reducing pathogenic bacteria colonization and reducing the immune cells’ stress [
52]. On the other hand, the antimicrobial compounds (organic acids, hydrogen peroxide, bacteriocins, and bioactive peptides) produced by the probiotic strains will enhance intestinal barriers and improve immunomodulation [
53]. Probiotic soy milk was observed to increase the white blood cells, including lymphocytes, in rats and improve immune system function [
54]. Furthermore, the probiotic and prebiotic combination was reported to significantly increase the neutrophil levels in mice, indicating potential benefits for immune system function [
55]. In this study, the increased WBC levels, including neutrophils, demonstrated the probiotic glutinous rice’s immunomodulation effects on the innate immune system [
56].
4.5. T-Cell and B-Cell Analysis
The results show a significantly increased glucose level in the TPTG group, followed by PTG and NG (
Table 2). The results were correlated with the TSS content in the un-fermented (37.6 Brix), traditionally fermented (53.7 Brix), and probiotic glutinous rice samples (42.8 Brix). On the other hand, this study demonstrated increased triglycerides and LDL contents in the PTPG group. The increase in glucose and lipid levels may be due to the high TSS content. According to Frayn & Kingman [
57], high sugar content diets can alter the plasma lipid profile and cause elevation of the triglycerides and low-density lipoprotein due to the metabolism of sugars stored as fats. Diets with high carbohydrate content were reported as high-risk foods for consumers due to the negative impact on sugar and lipid levels in the blood [
58]. In this study, the serum AST and ALT enzymes were significantly increased for the TPG and PTPG groups. However, the PTG group showed a significantly higher level than the PTPG. The diet supplemented with RB5 strains results showed that excessive consumption of traditional fermented glutinous rice might have a risk of causing slight liver damage. This can be justified because of the high carbohydrate content in the traditional fermented glutinous rice, as indicated by the high TSS.
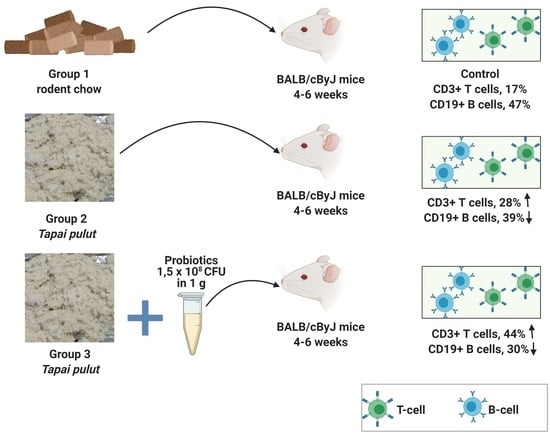
The effects of probiotic, fermented, and non-fermented glutinous rice (
tapai pulut) diets on the BALB/cByJ mice’s immune cells, specifically B-cell and T-cells derived from splenocytes, were investigated. The PTPG mice showed the most significant decrease in B-cells and an increase in T-cells after four weeks of treatment, followed by the TPG mice group, which showed a similar trend but a weaker response. Similar observations were seen when the supplementation of
Lp. plantarum and
Lacticaseibacillus casei in mice showed similar trends in the B-cell and T-cell distribution in the Peyer’s patches and mesenteric lymph nodes. The study found that the CD19
+ B-cell population was reduced, while the CD3
+ T-cell population was increased [
59].
In contrast, the increase in regulatory T-cells (Treg) was specifically highlighted [
60]. Recent research has shown that B-cells play an important role in shaping the microbiome by producing an extensive array of secretory IgA antibodies in response to commensals. The decrease or absence of B-cells or IgA could lead to the upregulation of epithelium-inherent immune defense mechanisms conferred by the intestinal epithelia, which eventually causes the microbiome’s changes [
61]. On the other hand, the
Lactobacillus strains have been reported to have anti-inflammatory properties by promoting T-cell responses, particularly Treg cells, in an IL-10-dependent manner [
62]. A previous study in a swine model fed with a high dose of
Lactobacillus rhamnosus observed an increased and higher CD3
+ T-cells percentage among Peyer’s patch and small intestinal lamina propria lymphocytes [
63]. The probiotic diet exhibited an ameliorative effect against potential enteric pathogens.
Furthermore, the administration of probiotic bacteria leads to the increase of CD4 T-cells and CD8 T-cells in the small intestinal lamina propria, which shifts the Th1/Th2 ratio towards a Th2 bias response [
64]. Furthermore, a high treatment dose of
L.plantarum has been linked to the immunomodulating activity by the significant increase in IL-17 and IL-19 cytokines from the Th17-type immune response. The regulation of Th17 further highlights probiotics’ potential for improving intestinal mucosa immunity [
65]. Hence, this study’s results hypothesize the immunomodulatory effect of
Lp. plantarum as a probiotic in fermented glutinous rice by reducing the CD19+ B = cell population and stimulating the CD3+ T-cell population in mice splenocytes. Further research can be conducted using T-cell- and B-cell-specific assays to identify the specific cytokines and antibodies released to confirm the findings.