1. Introduction
Free amino acids (FAAs) play an important role in the freshness and flavor of fish products, ornithine can be used as an indicator of freshness of aquatic products [
1,
2]. Glutamic acid (Glu) and aspartic acid (Asp) are important fresh taste substances, while alanine (Ala) and serine (Ser) contribute to sweetness [
1,
2]. The flavor of blue mussel was the strongest in summer with more umami-enhancing FAAs [
3]. Ala, Ser, and glycine (Gly) were known to be key factors in influencing the sweetness of grass carp [
4]. At present, the study of FAAs and methods for their analysis were mostly concentrated on plants [
5,
6,
7] and terrestrial animal-based foods [
8,
9], while only a few studies focus on aquatic products, especially the different varieties of shrimp. Glutathione (GSH) is a tripeptide composed of cysteine (Cys), Glu, and Gly. It has antioxidation [
10], detoxification, and other physiological functions, and it participates in amino acid transport and absorption, in addition to adding flavor to food. The analysis of GSH has mostly been conducted in fruits [
11] and plasma [
12,
13].
Shrimp, one of the fastest-growing aquaculture species [
14], refers to a wide variety of arthropod crustaceans. These include marine shrimp, such as
Penaeus vannamei (PV) and
Penaeus monodon (PM) [
15,
16], and freshwater shrimp, such as
Exopalaemon modestus (EM) and
Macrobrachium nipponense (MN) [
17,
18]. The market and nutritional values of shrimp [
19] are high because they are rich in protein, FAAs, and minerals; low in fat; and appealing to the taste. Due to different living environments, the contents of FAAs and biologically active peptides in shrimp are also different. Therefore, studying the components and contents of FAAs and GSH in the shrimp from different ecosystems is of great significance.
The currently used analysis methods for FAAs and GSH mainly include capillary electrophoresis [
20], high performance liquid chromatography (HPLC) [
5,
21], liquid chromatography with tandem mass spectrometry (LC-MS/MS) [
6,
8,
22,
23], and automatic amino acid analyzer [
7,
24,
25]. Capillary electrophoresis has the limited stability of electroosmotic flow and the poor reproducibility of the dissociated state of the inner silicon capillary wall. Although HPLC is simple to operate, the sample requires pre-column derivatization, which requires the strict control of the derivatization conditions, and thus the stability of the results is easily affected. LC-MS/MS is another detection method, but it requires stringent analytical conditions and is particularly demanding in terms of sample pretreatment and mobile phase. An automatic amino acid analyzer is commonly used to detect amino acids using a cation exchange resin and acid buffers as the stationary and mobile phases, respectively. After column separation, ninhydrin solution is used to yield amino acid derivatives for their detection by visible light absorption. This method has the advantages of simple sample preparation, high automation, good repeatability, and reliable results and less influence by the stability of derivatives [
25]. However, the currently used automatic amino acid analyzer can only detect the composition of 18 FAAs [
26], and the used methods are mainly limited to the detection of FAAs or GSH. In particular, there is no rapid and effective method for the simultaneous extraction, characterization, and quantification of FAAs and GSH in marine and freshwater shrimp.
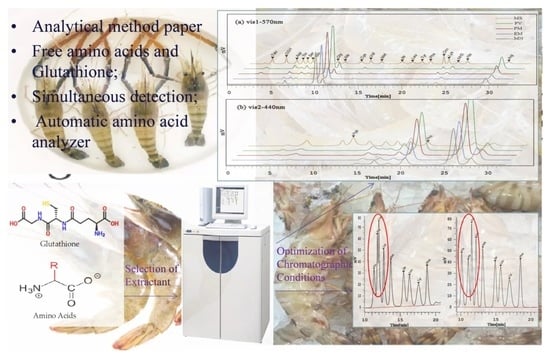
In this study, FAAs and GSH contents were simultaneously measured by a chromatographic method, particularly the selection of an extractant, the optimization of the composition of the buffer solution, and elution procedure. For the first time, this method was applied for the measurement of FAAs and GSH profiles in different shrimp varieties. The measured results were analyzed using analysis of variance (ANOVA) and principal component analysis (PCA).
2. Materials and Methods
2.1. Materials and Instruments
Fished from the sea, live Penaeus vannamei (PV) and Penaeus monodon (PM) (with an average weight of 15–20 g) were purchased from Luchaogang Market (Shanghai, China) and placed in an oxygenated sampling vessel. Farmed in freshwater, live Exopalaemon modestus (EM) and Macrobrachium nipponense (MN) (average weight 10–15 g) were obtained from Henanfei Food Store (Shanghai, China). Among them were a total of 95 PV; a total of 5 PM; a total of 5 MN; and a total of 5 EM. Five parallel experiments per shrimp species were carried out.
For sample preparation, all shrimp were quickly transported to the lab within 30 min, and upon arrival, the shrimp were immediately placed on ice, washed with cold water, and blotted dry. The experiment was approved by the Animal Care and Use Committee of Shanghai Ocean University (SHOU-DW-2021-096).
Seventeen types of mixed amino acid standard solution were purchased from Wako Pure Chemical Corporation (Osaka, Japan), taurine (Tau), and GSH from Sigma-Aldrich China (Shanghai, China); L-hydroxyproline (Hyp) from Yuanye Biological Technology (Shanghai, China); and ornithine (Orn) and trichloroacetic acid (TCA) from Maclean Biochemical Technology (Shanghai, China). Analytical grade sodium citrate dihydrate, sodium hydroxide, sodium chloride, citric acid, ethanol, hydrochloric acid (HCl), octoic acid, and polyoxyethylene lauryl ether (Brij-35) were obtained from Sinopharm Chemical Reagent (Shanghai, China).
A DS-1 high-speed tissue masher was purchased from Shanghai Specimen Model Factory (Shanghai, China), a Sartorius BSA124S electronic balance from Sartorius Scientific Instruments (Beijing, China), a H1850R desktop high-speed refrigerated centrifuge from Xiangyi Centrifuge Instrument Co., Ltd. (Shanghai, China), and a T10 basic ULTRA-TURRAX homogenizer from IKA (Staufen, Germany). These instruments were used for sample preparation. A LA8080 amino acid automatic analyzer (Hitachi High-Technologies Corporation, Tokyo, Japan) was used to separate and detect FAAs and GSH in the samples. The water used in the experiment was obtained from a Milli-Q IQ7000 ultra-pure water purification system (Merck Chemical Technology Co., Ltd., Shanghai China). A Mettler-Toledo S2 pH meter (Mettler-Toledo Instruments Co., Ltd., Shanghai China) was used to determine the pH of the sample solution.
2.2. Chromatographic Conditions
The parameters of the automatic amino acid analyzer are as follows:
The LA8080 automatic amino acid analyzer (Hitachi, Japan) used ion exchange chromatography separation and post-column derivatization of ninhydrin. The amino acids of the sample were separated in the separation column and then transported by the buffer to the reaction unit for derivatization with the ninhydrin solution. Stationary phase and separation column: cation exchange resin column with a particle size of 3 μm (i.d. 4.6 mm × 60 mm); Separation column temperature 57 °C. Detection wavelength: 570 nm and 440 nm; Mobile phase: sodium citrate buffer B1, B2, B3, B4, and B5 (as shown in
Table 1). The total flow rate of sodium citrate buffer B1, B2, B3, B4, and B5 was kept constant at 0.4 mL·min
−1 (as shown in
Table 2); Injection volume: 20 µL.
The reaction unit: ninhydrin reaction solutions, R1, R2, and R3, were prepared with compositions as follows: R1 contained 39 g of ninhydrin, 81 g of sodium borohydride, and 979 mL of propylene glycol monomethyl. R2 consisted of 204 g sodium acetate, 123 g glacial acetic acid, 401 mL propylene glycol monomethyl ether, and 336 mL of ultra-pure water. R3 comprised 50 mL ethanol and 950 mL of ultra-pure water. Ninhydrin reaction solution flow rate was kept constant at 0.35 mL·min
−1 (as shown in
Table 2), reaction unit temperature 135 °C.
The optimization of the elution procedure and heating process are shown in
Table 2.
2.3. Preparation of Standard Stock Solution
GSH (2.5 µmol·mL−1) standard solution was prepared by dissolving 76.8 mg GSH in 80 mL of 0.2 mol·L−1 HCl, and then the solution was made up to 100 mL using HCl. By dissolving 16.5 mg, 16.4 mg, and 15.6 mg of Orn, Hyp, and Tau, respectively, in 0.2 mol·L−1 HCl, 2.5 µmol·mL−1 standard solutions (50 mL) were prepared. A mixed amino acid stock solution (0.1 µmol·mL−1) was prepared from 2 mL of 2.5 µmol·mL−1 GSH, Orn, Hyp, Tau, and 17 amino acids standard solutions, made up to 50 mL with 0.2 mol·L−1 HCl and stored at −20 °C.
2.4. Sample Preparation
A mashed shrimp muscle sample (2.0 g) was weighed into a 50 mL centrifuge tube, and 15 mL of the extractant (12% TCA) was added. After sufficient homogenization (3 min), the sample was centrifuged at 10,610× g for 20 min with the temperature maintained at 4 °C. The supernatant was collected and diluted to 50 mL with the same extractant. A 5 mL aliquot was transferred into a centrifuge tube, neutralized with sodium hydroxide (1 mol·L−1) to a pH 2.2 ± 0.02 and diluted to 15 mL with ultra-pure water. The resulting solution was filtered through a 0.22 µm aqueous phase filtration membrane, and the filtrate was injected into an amino acid automatic analyzer for testing. The remaining sample was stored at −20 °C and was frozen in time for the next injection. Extraction experiments were all carried out with five parallel.
2.5. Method Validation
Linearity range, the limit of detection (LOD), the limit of quantitation (LOQ), intra-day and inter-day precision, spike recovery, and accuracy were considered in method validation. The linearity range, LOD, and LOQ were detected by diluting the mixed standard stock solution (0.1 µmol·mL
−1) with 0.02 mol·L
−1 HCl to a concentration range of 1–250 µg·mL
−1. The intra-day and inter-day precisions were obtained by adding the mixed standard stock solution to the shrimp sample and detecting five replicates, while the spike recovery was calculated by detecting high, medium, and low concentrations of the mixed standard stock solution (1–50 µmol·mL
−1) added to shrimp samples.
2.6. Statistical Analysis
ANOVA is a statistical tool used to evaluate the statistical significance of the survey or experimental data for different classes or groups. PCA is a multivariate statistical technique that is most widely used for data exploration, pattern recognition, dimensionality reduction, and data visualization. In this study, ANOVA and PCA were employed using SPSS version 18.0 software (US) to identify the presence of any significant differences (p < 0.05) and account for the correlations that exist between the FAAs and GSH data of the four types of shrimp. The different shrimp varieties were comprehensively evaluated using PCA. The FAAs and GSH contents were expressed as mean ± standard deviation.
4. Conclusions
In this study, a simple and fast amino acid analysis method for the simultaneous extraction and determination of FAAs and GSH in shrimp with 12% TCA was established. This method was shown to have a good linear range, precision, recovery, LOD, and LOQ, and thus can be used to effectively measure 20 FAAs and GSH in shrimp.
All the four species of shrimp investigated were rich in FAAs, and their main FAAs were Gly, Pro, Arg, and Ala. The content of TFAAs and EAAs followed the order: PV > PV > MN > EM. Only a small amount of GSH was detected in marine shrimp. GSH and Cys were used to identify marine shrimp and freshwater shrimp. Hyp could be used to distinguish PV from other shrimp, and Ser could be used to identify MN from other shrimp. Sweet amino acids had a significant impact on the taste of shrimp muscle, particularly Glu, Arg, Gly, Pro, and Ala. Arg was found to have the greatest influence on the flavor of shrimp.
PCA extracted two principal components from 20 types of FAAs and GSH, with a cumulative contribution rate of 98.68%, which can better reflect the comprehensive information on the quality of shrimp FAAs and GSH. A comprehensive evaluation model, F = 0.873F1 + 0.117F2, was developed using PCA. According to the comprehensive analysis, the comprehensive quality of FAAs and GSH followed the order: PV > PM > MN > EM.