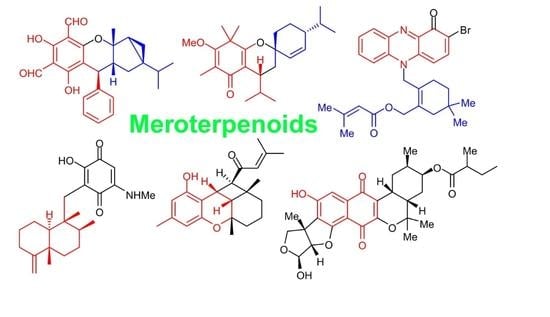
Meroterpenoids: A Comprehensive Update Insight on Structural Diversity and Biology †
Abstract
:1. Introduction
2. Meroterpenoid Classification
3. Phloroglucinol-Based Meroterpenoids
| Compounds | Source | Activities | Ref. |
|---|---|---|---|
| Psiguajavadial A (1) | Psidium guajava | Cytotoxic effects: HCT116 = IC50 7.60 µM; CCRF-CEM = IC50 25.2 µM; DU145 = IC50 20.2 µM; Huh7 = IC50 48.8 µM; A549 = IC50 2.99 µM | [33,34] |
| Psiguajavadial B (2) | Psidium guajava | Cytotoxic effects: HCT116 = IC50 21.6 µM; CCRF-CEM = IC50 9.63 µM; DU145 = IC50 26.3 µM; Huh7 = IC50 13.7 µM; A549 = IC50 0.90 µM | [33,34] |
| Guadial A (3) | Psidium guajava | Cytotoxic effects: HCT116 = IC50 5.74 µM; CCRF-CEM = IC50 2.95 µM; DU145 = IC50 5.35 µM; Huh7 = IC50 28.0 µM; A549 = IC50 9.62 µM; Enzyme Inhibition: PDE4D-4 = IC50 2.70 μM | [33,34] |
| Guajavadial A (4) | Psidium guajava | Cytotoxic effects: HL-60 = IC50 4.73 µM; A-549 = IC50 5.62 µM; SMMC-7721 = IC50 4.37 µM; MCF-7 = IC50 22.28 µM; SW480 = IC50 14.55 µM; Enzyme Inhibition: PDE4D-4 = IC50 2.01 μM | [35] |
| Guajavadial B (5) | Psidium guajava | Cytotoxic effects: HL-60 = IC50 6.49 µM; A-549 = IC50 5.78 µM; SMMC-7721 = IC50 5.05 µM; MCF-7 = IC50 18.02 µM; SW480 = IC50 13.07 µM | [35] |
| Guajavadial C (6) | Psidium guajava | Cytotoxic effects: HL-60 = IC50 3.38 µM; A-549 = IC50 5.66 µM; SMMC-7721 = IC50 3.54 µM; MCF-7 = IC50 14.54 µM; SW480 = IC50 18.97 µM | [35] |
| Eucalrobusone A (7) | Eucalyptus robusta | Cytotoxic effects: HepG2 = IC50 18.52 µM; U2OS = IC50 45.00 μM | [36] |
| Eucalrobusone C (9) | Eucalyptus robusta | Cytotoxic effects: HepG2 = IC50 7.40 µM; U2OS = IC50 8.99 μM; MCF-7 = IC50 8.50 μM | [36] |
| Eucalrobusone D (10) | Eucalyptus robusta | Cytotoxic effects: HepG2 = IC50 26.78 μM | [36] |
| Eucalrobusone H (14) | Eucalyptus robusta | Cytotoxic effects: U2OS = IC50 42.25 μM | [36] |
| Eugenial B (17) | Eugenia umbelliflora | Cytotoxic effects: K562 = IC50 42.8 μM; Nalm-6 = IC50 70.5 μM; B16F10 = IC50 12.0 μM | [37] |
| Eugenial C (18) | Eugenia umbelliflora | Cytotoxic effects: K562 = IC50 0.38 μM; Nalm-6 = IC50 10.5 μM; B16F10 = IC50 6.0 μM | [37] |
| Eugenial D (19) | Eugenia umbelliflora | Cytotoxic effects: K562 = IC50 1.90 μM; Nalm-6 = IC50 7.75 μM; B16F10 = IC50 3.20 μM | [37] |
| Eugenial E (20) | Eugenia umbelliflora | Cytotoxic effects: K562 = IC50 4.97 μM; Nalm-6 = IC50 29.1 μM; B16F10 = IC50 8.80 μM | [37] |
| Eucalteretials C (23) | Eugenia tereticornis | Cytotoxic effects: HCT116 = IC50 4.8 μM | [37] |
| Eucalypglobulusal F (31) | Eugenia globulus | Cytotoxic effects: CCRF-CEM = IC50 3.3 μM | [37] |
| (+)-Japonicol B (37a) | Hypericum japonicum | Antiviral effects: KSHV = EC50 8.75 μM | [40] |
| (+)-Japonicol E (40a) | Hypericum japonicum | Antiviral effects: KSHV = IC50: 8.30 μM | [41] |
| (−)-Japonicol E (40b) | Hypericum japonicum | Antiviral effects: KSHV = IC50: 24.4 μM | [41] |
| (+)-Japonicol G (42a) | Hypericum japonicum | Antiviral effects: KSHV = IC50: 21.3 μM | [41] |
| (−)-Japonicol G (42b) | Hypericum japonicum | Antiviral effects: KSHV = IC50: 6.7 μM | [41] |
| (+)-Japonicol H (43a) | Hypericum japonicum | Antiviral effects: KSHV = IC50: 4.90 μM | [41] |
| (−)-Japonicol H (43b) | Hypericum japonicum | Antiviral effects: KSHV = IC50: 29.4 μM | [41] |
| Psiguajadial A (44) | Psidium guajava | Enzyme Inhibition: PDE4D-4 = IC50 3.11 μM | [42] |
| Psiguajadial B (45) | Psidium guajava | Enzyme Inhibition: PDE4D-4 = IC50 5.03 μM | [42] |
| Psiguajadial C (46) | Psidium guajava | Enzyme Inhibition: PDE4D-4 = IC50 4.50 μM | [42] |
| Psiguajadial D (47) | Psidium guajava | Enzyme Inhibition: PDE4D-4 = IC50 4.14 μM | [42] |
| Psiguajadial E (48) | Psidium guajava | Enzyme Inhibition: PDE4D-4 = IC50 3.25 μM | [42] |
| Psiguajadial F (49) | Psidium guajava | Enzyme Inhibition: PDE4D-4 = IC50 2.63 μM | [42] |
| Psiguajadial G (50) | Psidium guajava | Enzyme Inhibition: PDE4D-4 = IC50 1.34 μM | [42] |
| Psiguajadial H (51) | Psidium guajava | Enzyme Inhibition: PDE4D-4 = IC50 1.81 μM | [42] |
| Psiguajadial I (52) | Psidium guajava | Enzyme Inhibition: PDE4D-4 = IC50 2.51 μM | [42] |
| Psiguajadial J (53) | Psidium guajava | Enzyme Inhibition: PDE4D-4 = IC50 2.53 μM | [42] |
| Psiguajadial K (54) | Psidium guajava | Enzyme Inhibition: PDE4D-4 = IC50 3.68 μM | [42] |
| Psiguadial A (55) | Psidium guajava | Enzyme Inhibition: PDE4D-4 = IC50 7.26 μM | [42] |
| Guapsidial A (56) | Psidium guajava | Enzyme Inhibition: PDE4D-4 = IC50 5.61 μM | [42] |
| Psiguajadial L (57) | Psidium guajava | Enzyme Inhibition: PDE4D-4 = IC50 1.37 μM | [42] |
| Eucarobustol A (63) | Eucalyptus robusta | Enzyme Inhibition: PTP1B = IC50 1.3 μM | [44] |
| Eucarobustol B (64) | Eucalyptus robusta | Enzyme Inhibition: PTP1B = IC50 4.3 μM | [44] |
| Eucarobustol C (65) | Eucalyptus robusta | Enzyme Inhibition: PTP1B = IC50 4.3 μM | [44] |
| Eucarobustol D (66) | Eucalyptus robusta | Enzyme Inhibition: PTP1B = IC50 2.9 μM | [44] |
| Eucarobustol E (67) | Eucalyptus robusta | Enzyme Inhibition: PTP1B = IC50 4.1 μM | [44] |
| Eucarobustol F (68) | Eucalyptus robusta | Enzyme Inhibition: PTP1B = IC50 5.6 μM | [44] |
| Eucarobustol G (69) | Eucalyptus robusta | Enzyme Inhibition: PTP1B = IC50 1.8 μM | [44] |
| Eucarobustol H (70) | Eucalyptus robusta | Enzyme Inhibition: PTP1B = IC50 3.0 μM | [44] |
| Eucarobustol I (71) | Eucalyptus robusta | Enzyme Inhibition: PTP1B = IC50 1.6 μM | [44] |
| Eucalyptusdimers A (73) | Eucalyptus robusta | Enzyme Inhibition: AChE = IC50 17.71 μM | [44] |
4. Syncarpic Acid/β-Triketones-Based Meroterpenes
| Compounds | Source | Anticancer | Ref. |
|---|---|---|---|
| (−)-Baeckfrutone B (125) | Baeckea frutescens | Cytotoxic effects: DU145 = IC50 79.45 μM | [58] |
| (+)-Baeckfrutone C (126) | Baeckea frutescens | Cytotoxic effects: HCT116 = IC50 62.64 μM; Hela = IC50 85.79 μM; DU145 = IC50 17.65 μM; A549 = IC50 86.68 μM | [58] |
| (−)-Baeckfrutone C (126) | Baeckea frutescens | Cytotoxic effects: HCT116 = IC50 49.09 μM; Hela = IC50 91.22 μM; DU145 = IC50 15.85 μM; A549 = IC50 86.62 μM | [58] |
| Baeckfrutone D (127) | Baeckea frutescens | Cytotoxic effects: HCT116 = IC50 38.32 μM; Hela = IC50 83.85 μM; DU145 = IC50 6.46 μM; A549 = IC50 76.47 μM | [58] |
| Baeckfrutone F (129) | Baeckea frutescens | Cytotoxic effects: HCT116 = IC50 39.5 μM; DU145 = IC50 80.72 μM; A549 = IC50 15.61 μM; Anti-inflammatory effects: 74.4% | [58] |
| Baeckfrutone G (130) | Baeckea frutescens | Cytotoxic effects: HCT116 = IC50 49.76 μM; Hela = IC50 31.87 μM; DU145 = IC50 17.40 μM; A549 = IC50 62.64 μM; Anti-inflammatory effects: 75.3% | [58] |
| Baeckfrutone H (131) | Baeckea frutescens | Cytotoxic effects: HCT116 = IC50 19.50 μM; Hela = IC50 30.44 μM; DU145 = IC50 25.14 μM; A549 = IC50 82.75 μM; Anti-inflammatory effects: 55.1% | [58] |
| Baeckfrutone I (132) | Baeckea frutescens | Cytotoxic effects: HCT116 = IC50 19.50 μM; Hela = IC50 53.71 μM; DU145 = IC50 26.11 μM; A549 = IC50 84.13 μM; Anti-inflammatory effects: 75% | [58] |
| Baeckfrutone J (133) | Baeckea frutescens | Cytotoxic effects: HCT116 = IC50 52.93 μM; DU145 = IC50 4.04 μM; A549 = IC50 79.45 μM | [58] |
| Baeckfrutone K (134) | Baeckea frutescens | Cytotoxic effects: HCT116 = IC50 12.89 μM; DU145 = IC50 77.06 μM; A549 = IC50 80.11 μM | [58] |
| Baeckfrutone L (135) | Baeckea frutescens | Cytotoxic effects: HCT116 = IC50 16.48 μM; Hela = IC50 19.81 μM; DU145 = IC50 10.0 μM; A549 = IC50 88.81 μM | [58] |
| Hyperjaponol H (143) | Hypericum japonicum | Antiviral effects: EBV = EC50 25.0 μM | [61] |
| Tomentosenol A (144) | Rhodomyrtus tomentosa | Cytotoxic effects: MCF-7 = IC50 8.66 μM; NCI-H460 = IC50 8.62 μM; SF-268 = IC50 10.01 μM; HepG-2 = IC50 9.44 μM | [61] |
| Myrtucommulone (147) | Myrtus communis | Cytotoxic effects: HepG2 = IC50 4.39 μM; MDA-MB-231 = IC50 19.92 μM | [62] |
| Myrtucommulone (148) | Myrtus communis | Cytotoxic effects: HepG2 = IC50 40.7 μM; MDA-MB-231 = IC50 40.0 μM | [62] |
| Frutescone A (151) | Baeckea frutescens | Cytotoxic effects: Caco-2 = IC50 8.08; A549 = IC50 20.07 µM | [64] |
| Frutescone B (152) | Baeckea frutescens | Cytotoxic effects: Caco-2 = IC50 23.25 µM; A549 = IC50 41.33 µM | [64] |
| Frutescone C (153) | Baeckea frutescens | Cytotoxic effects: Caco-2 = IC50 14.83 µM; A549 = IC50 27.74 µM | [64] |
| Frutescone D (154) | Baeckea frutescens | Cytotoxic effects: Caco-2 = IC50 10.20 µM; A549 = IC50 26.25 µM | [64] |
| Frutescone E (155) | Baeckea frutescens | Cytotoxic effects: Caco-2 = IC50 7.96 µM; A549 = IC50 5.55 µM | [64] |
| Frutescone F (156) | Baeckea frutescens | Cytotoxic effects: Caco-2 = IC50 16.51 µM; A549 = IC50 39.02 µM | [64] |
| (±)-Frutescone G (157) | Baeckea frutescens | Cytotoxic effects: Caco-2 = IC50 14.31 µM; A549 = IC50 25.71 µM | [64] |
| Callisalignene G (174) | Callistemon salignus | Cytotoxic effects: HCT116 = IC50 8.51 μM; A549 = IC50 12.85 μM | [67] |
| Callisalignene H (175) | Callistemon salignus | Cytotoxic effects: HCT116 = IC50 9.12 μM | [67] |
| Callisalignene I (176) | Callistemon salignus | Cytotoxic effects: HCT116 = IC50 16.33 μM; A549 = IC50 10.03 μM | [67] |
| Frutescone I (178) | Baeckea frutescens | Anti-inflammatory effects: NO production = IC50 18.75 μM | [68] |
| Frutescone L (179) | Baeckea frutescens | Anti-inflammatory effects: NO production = IC50 30.54 μM | [68] |
| Frutescone M (180) | Baeckea frutescens | Anti-inflammatory effects: NO production = IC50 15.17 μM | [68] |
| (±)-Compound (181) | Baeckea frutescens | Anti-inflammatory effects: NO production = IC50 1.80 μM | [68] |
| Compound (182) | Baeckea frutescens | Anti-inflammatory effects: NO production = IC50 0.36 μM | [68] |
| Compound (183) | Baeckea frutescens | Anti-inflammatory effects: NO production = IC50 3.70 μM | [68] |
| (±)-Compound (184) | Baeckea frutescens | Anti-inflammatory effects: NO production = IC50 2.07 μM | [68] |
| (±)-Compound (185) | Baeckea frutescens | Anti-inflammatory effects: NO production = IC50 6.50 μM | [68] |
| Baefrutone A (188) | Baeckea frutescens | Anti-inflammatory effects: NO Production = IC50 9.15 μM | [69] |
| Baefrutone B (189) | Baeckea frutescens | Anti-inflammatory effects: NO Production = IC50 17.73 μM | [69] |
| Baefrutone C (190) | Baeckea frutescens | Anti-inflammatory effects: NO Production = IC50 11.62 μM | [69] |
| Baefrutone D (191) | Baeckea frutescens | Anti-inflammatory effects: NO Production = IC50 18.04 μM | [69] |
| hyperjaponols A (194a) | Hypericum japonicum | Antiviral effects: EBV = EC50 10.33 μM | [70] |
| Hyperjaponol B (195a) | Hypericum japonicum | Antiviral effects: EBV = EC50 0.57 μM | [70] |
| Hyperjaponol B (195b) | Hypericum japonicum | Antiviral effects: EBV = EC50 6.60 μM | [70] |
| Hyperjaponol D (197) | Hypericum japonicum | Antiviral effects: EBV = EC50 0.49 μM | [70] |
5. Alklaoid-Based Meroterpenoids
Phenazine- and Phyridine-Based Meroterpenoids
6. Sesquiterpene-Based Meroterpenoids
7. Chromane/Chromene and Flavone Derived Meroterpenoids
8. Quinone-Based Meroterpenoids
9. Miscellaneous
| Compounds | Source | Activities | Ref. |
|---|---|---|---|
| Magterpenoid C (373) | Magnolia officinalis | Enzyme Inhibition: PTP1B = IC50 0.81 μM | [116] |
| Arnebinone B (374) | Arnebia euchroma | Cytotoxic effects: HepG2, SMMC-7721, QGY-7703 and HepG2/ADM IC50 ranging from 9.6 to 18.7 μM | [117] |
| Compound 375 | Arnebia euchroma | Cytotoxic effects: HepG2, SMMC-7721, QGY-7703 and HepG2/ADM IC50 ranging from 3.43 to 11.31 μM | [117] |
| Toluquinol-derivative (378) | Carteriospongia sp. | Cytotoxic effects: Molt 4 = IC50 0.34 μg/mL; HL60 = IC50 0.70 μg/mL; lymphoma U937 = IC50 0.65 μg/mL; Sup-T1 = IC50 0.33 μg/mL; oral Ca9-22 = IC50 0.97 μg/mL; Cal-27 = IC50 0.51 μg/mL; breast T-47D = IC50 1.06 μg/mL | [119] |
| Sargaquinoic acid (379) | Sargassum serratifolium | Enzyme Inhibition: AChE = IC50 69.3 μM; BChE = IC50 10.5 μM; BACE1 = IC50 12.1 μM | [106] |
| Sargahydroquinoic acid (384) | Sargassum serratifolium | Enzyme Inhibition: AChE = IC50 124.3 μM; BChE = IC50 15.2 μM; BACE1 = IC50 4.4 μM | [106] |
| Tetronasin (385) | Streptomyces sp. | Cytotoxic effects: HeLa cells = IC50 0.23 μM | [115] |
| Magterpenoid A (386) | Magnolia officinalis | Enzyme Inhibition: PTP1B = IC50 1.44 μM | [116] |
| Nyingchinoids A (388a,b) | Rhododendron nyingchiense | Enzyme Inhibition: PTP1B = IC50 43.6 μM | [105] |
| Nyingchinoids B (389a,b) | Rhododendron nyingchiense | Enzyme Inhibition: PTP1B = IC50 38.1 μM | [105] |
| Compound 390 | Villorita cyprinoides | Antioxidant effects: DPPH = IC50 0.59 mg/mL; ABTS = IC50 0.65 mg/mL; Enzyme Inhibition: COX-1 = IC50 0.94 mg/mL; COX-2 = IC50 0.70 mg/mL | [122] |
| Compound 391 | Villorita cyprinoides | Antioxidant effects: DPPH = IC50 0.54 mg/mL; ABTS = IC50 0.62 mg/mL; Enzyme Inhibition: COX-1 = IC50 0.86 mg/mL; COX-2 = IC50 0.65 mg/mL | [122] |
| Compound 392 | Villorita cyprinoides | Antioxidant effects: DPPH = IC50 0.69 mg/mL; ABTS = IC50 0.64 mg/mL; Enzyme Inhibition: COX-1 = IC50 0.91 mg/mL; COX-2 = IC50 0.74 mg/mL | [122] |
| {Tetrahydro-3-methoxy-5-((E)-8,12-dimethyloct-8-enyl)-pyran-2-one (393) | Villorita cyprinoides | Antioxidant effects: DPPH = IC50 0.70 mg/mL; ABTS = IC50 0.76 mg/mL; Fe2+ = IC50 0.83 mg/mL; H2O2 = IC50 0.85 mg/mL; COX-1 = IC50 0.99 mg/mL; COX-2 = IC50 0.89 mg/mL | [123] |
| Dihydro-5-(8-(9,12-dihydro-8-methyl-11-propyl-2H-pyran-8-yl)-ethyl)-furan-2(3H)-one (394)} | Villorita cyprinoides | Antioxidant effects: DPPH = IC50 0.63 mg/mL; ABTS = IC50 0.79 mg/mL; Fe2+ = IC50 0.83 mg/mL; H2O2 = IC50 0.84 mg/mL; Enzyme Inhibition: COX-1 = IC50 0.96 mg/mL; COX-2 = IC50 0.84 mg/mL | [123] |
| Hexahydro-iso-chromenyl-meroterpenoid (395) | Villorita cyprinoides | Antioxidant effects: DPPH = IC50 0.76 mg/mL; ABTS = IC50 0.82 mg/mL; Fe2+ = IC50 0.90 mg/mL; H2O2 = IC50 0.86 mg/mL; Enzyme Inhibition: COX-1 = IC50 1.05 mg/mL; COX-2 = IC50 0.90 mg/mL | [123] |
| Hexahydro-iso-chromenyl-meroterpenoid (396) | Villorita cyprinoides | Antioxidant effects: DPPH = IC50 0.79 mg/mL; ABTS = IC50 0.81 mg/mL; Fe2+ = IC50 0.89 mg/mL; H2O2 = IC50 0.87 mg/mL; Enzyme Inhibition: COX-1 = IC50 1.09 mg/mL; COX-2 = IC50 0.89 mg/mL | [123] |
| 2-(Tetrahydro-5-(4-hydroxyphenyl)-4-pentylfuran-3-yl)-ethyl-4-hydroxybenzoate (397) | Hypnea musciformis | Antioxidant effects: DPPH = IC50 25.05 μM; Fe2+ ion chelating = IC50 350.66 μM | [124] |
| 2-2-[(4-Hydroxybenzoyl)-oxy]-ethyl-4-methoxy-4-2-[(4-methylpentyl)oxy]-3,4-dihydro-2H-6-pyranylbutanoic acid (398) | Hypnea musciformis | Antioxidant effects: DPPH = IC50 322.4 μM; Fe2+ ion chelating = IC50 5115.3 μM | [124] |
| 3-((5-Butyl-3-methyl-5,6-dihydro-2H-pyran-2-yl)-methyl)-4-meth oxy-4-oxobutyl benzoate (399) | Hypnea musciformis | Antioxidant effects: DPPH = IC50 231.2 μM Fe2+ ion chelating = IC50 667.9 μM | [124] |
| 2-((E)-deca-1,8-dien-10-yl)-11,12-dihydro-13-propyl-2H-pyran (412) | Paphia malabarica | Antioxidant effects: DPPH = IC50 0.78 mg/mL; ABTS = IC50 0.92 mg/mL; Enzyme Inhibition: COX-1 = IC50 1.07 mg/mL; COX-2 = IC50 0.95 mg/mL; 5-LOX = IC50 1.02 mg/mL | [126] |
| 1′-((10E)-10-(10-(pentan-4-yl)-cyclohex-4-enyl)-allyloxy)-tetrahydro-2′, 2′-dimethyl-2H-pyran (413) | Paphia malabarica | Antioxidant effects: DPPH = IC50 0.76 mg/mL; ABTS = IC50 0.96 mg/mL; Enzyme Inhibition: COX-1 = IC50 1.05 mg/mL; COX-2 = IC50 0.92 mg/mL; 5-LOX = IC50 1.06 mg/mL | [126] |
| (±)-Rasumatranin B (415) | Radula sumatrana | Cytotoxic effects: MCF-7 = IC50: 38.3 µM | [127] |
| Compound 421 | Radula sumatrana | Cytotoxic effects: MCF-7: IC50: 3.8 µM; PC-3: IC50: 6.6 µM; SMMC-7721 7: IC50: 3.5 µM | [127] |
| (±)-Radulanin I (422) | Radula sumatrana | Cytotoxic effects: MCF-7 = IC50: 24.6 µM | [127] |
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brahmachari, G. Discovery and Development of Neuroprotective Agents from Natural Products: An Overview. In Discovery and Development of Neuroprotective Agents from Natural Products, 1st ed.; Brahmachari, G., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1–7. [Google Scholar]
- Swargiary, A. Recent trends in traditionally used medicinal plants and drug discovery. Asian J. Pharm. Pharmacol. 2017, 3, 111–120. [Google Scholar]
- Annang, F.; Genilloud, O.; Vicente, F. Contribution of Natural Products to Drug Discovery in Tropical Diseases. In Comprehensive Analysis of Parasite Biology, From Metabolism to Drug Discovery; Müller, S., Cerdan, R., Radulescu, O., Eds.; Wiley-VCH: Weheim, Germany, 2016; pp. 75–104. [Google Scholar]
- Shen, B. A new golden age of natural products drug discovery. Cell 2015, 163, 1297–1300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- David, B.; Wolfender, J.L.; Dias, D.A. The pharmaceutical industry and natural products, historical status and new trends. Phytochem. Rev. 2015, 14, 299–315. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M.; Snader, K.M. Natural products as sources of new drugs over the period 1981–2002. J. Nat. Prod. 2003, 66, 1022–1037. [Google Scholar] [CrossRef] [PubMed]
- Koehn, F.E.; Carter, G.T. The evolving role of natural products in drug discovery. Nat. Rev. Drug Discov. 2005, 4, 206. [Google Scholar] [CrossRef]
- Wolfender, J.L.; Queiroz, E.F. Chemical diversity of natural resources and the bioactivity of their constituents. CHIMIA Int. J. Chem. 2012, 66, 324–329. [Google Scholar] [CrossRef]
- Kikuchi, H.; Kawai, K.; Nakashiro, Y.; Yonezawa, T.; Kawaji, K.; Kodama, E.N.; Oshima, Y. Construction of a meroterpenoid-like compounds library based on diversity-enhanced extracts. Chem. Eur. J. 2019, 25, 1106–1112. [Google Scholar] [CrossRef]
- Cornforth, J.W. Terpenoid biosynthesis. Chem. Br. 1968, 4, 102–106. [Google Scholar]
- Matsuda, Y.; Abe, I. Biosynthesis of fungal meroterpenoids. Nat. Prod. Rep. 2016, 33, 26–53. [Google Scholar] [CrossRef] [Green Version]
- Geris, R.; Simpson, T.J. Meroterpenoids produced by fungi. Nat. Prod. Rep. 2009, 26, 1063–1094. [Google Scholar] [CrossRef] [Green Version]
- Peng, X.; Qiu, M. Meroterpenoids from Ganoderma Species: A review of last five years. Nat. Prod. Bioprospect. 2018, 8, 137–149. [Google Scholar] [CrossRef] [Green Version]
- Matsuda, Y.; Awakawa, T.; Abe, I. Reconstituted biosynthesis of fungal meroterpenoid andrastin A. Tetrahedron 2013, 69, 8199–8204. [Google Scholar] [CrossRef]
- El-Elimat, T.; Raja, H.A.; Ayers, S.; Kurina, S.J.; Burdette, J.E.; Mattes, Z.; Sabatelle, R.; Bacon, J.W.; Colby, A.H.; Grinstaff, M.W. Meroterpenoids from Neosetophoma sp., A dioxa [4.3. 3] ropellane ring system, potent cytotoxicity, and prolific expression. Org. Lett. 2019, 21, 529–534. [Google Scholar] [CrossRef] [Green Version]
- Joung, E.-J.; Gwon, W.-G.; Shin, T.; Jung, B.-M.; Choi, J.; Kim, H.-R. Anti-inflammatory action of the ethanolic extract from Sargassum serratifolium on lipopolysaccharide-stimulated mouse peritoneal macrophages and identification of active components. J. Appl. Phycol. 2017, 29, 563–573. [Google Scholar] [CrossRef]
- Seo, Y.; Park, K.E.; Kim, Y.A.; Lee, H.-J.; Yoo, J.-S.; Ahn, J.-W.; Lee, B.-J. Isolation of tetraprenyltoluquinols from the brown alga Sargassum thunbergii. Chem. Pharm. Bull. 2006, 54, 1730–1733. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.A.; Ahn, B.N.; Kong, C.S.; Kim, S.K. Protective effect of chromene isolated from Sargassum horneri against UV-A-induced damage in skin dermal fibroblasts. Exp. Dermatol. 2012, 21, 630–631. [Google Scholar] [CrossRef]
- Kim, S.; Lee, M.-S.; Lee, B.; Gwon, W.-G.; Joung, E.-J.; Yoon, N.-Y.; Kim, H.-R. Anti-inflammatory effects of sargachromenol-rich ethanolic extract of Myagropsis myagroides on lipopolysaccharide-stimulated BV-2 cells. BMC Complement. Altern. Med. 2014, 14, 231. [Google Scholar] [CrossRef] [Green Version]
- Jeon, Y.; Jung, Y.; Kim, M.C.; Kwon, H.C.; Kang, K.S.; Kim, Y.K.; Kim, S.-N. Sargahydroquinoic acid inhibits TNFα-induced AP-1 and NF-κB signaling in HaCaT cells through PPARα activation. Biochem. Biophys. Res. Commun. 2014, 450, 1553–1559. [Google Scholar] [CrossRef]
- Joung, E.-J.; Lee, B.; Gwon, W.-G.; Shin, T.; Jung, B.-M.; Yoon, N.-Y.; Choi, J.-S.; Oh, C.W.; Kim, H.-R. Sargaquinoic acid attenuates inflammatory responses by regulating NF-κB and Nrf2 Pathways in lipopolysaccharide-stimulated RAW 264.7 cells. Int. Immunopharmacol. 2015, 29, 693–700. [Google Scholar] [CrossRef]
- Oh, S.-J.; Joung, E.-J.; Kwon, M.-S.; Lee, B.; Utsuki, T.; Oh, C.-W.; Kim, H.-R. Anti-inflammatory effect of ethanolic extract of Sargassum serratifolium in lipopolysaccharide-stimulated BV2 microglial cells. J. Med. Food 2016, 19, 1023–1031. [Google Scholar] [CrossRef]
- Gwon, W.-G.; Joung, E.-J.; Kwon, M.-S.; Lim, S.-J.; Utsuki, T.; Kim, H.-R. Sargachromenol protects against vascular inflammation by preventing TNF-α-induced monocyte adhesion to primary endothelial cells via inhibition of NF-κB activation. Int. Immunopharmacol. 2017, 42, 81–89. [Google Scholar] [CrossRef]
- Kwon, M.; Lim, S.-J.; Lee, B.; Shin, T.; Kim, H.-R. Ethanolic extract of Sargassum serratifolium inhibits adipogenesis in 3T3-L1 preadipocytes by cell cycle arrest. J. Appl. Phycol. 2018, 30, 559–568. [Google Scholar] [CrossRef]
- Kim, S.-N.; Lee, W.; Bae, G.-U.; Kim, Y.K. Anti-diabetic and hypolipidemic effects of Sargassum yezoense in db/db mice. Biochem. Biophys. Res. Commun. 2012, 424, 675–680. [Google Scholar] [CrossRef]
- De la Mare, J.-A.; Lawson, J.C.; Chiwakata, M.T.; Beukes, D.R.; Edkins, A.L.; Blatch, G.L. Quinones and halogenated monoterpenes of algal origin show anti-proliferative effects against breast cancer cells in vitro. Investig. N. Drugs 2012, 30, 2187–2200. [Google Scholar] [CrossRef]
- Kang, C.-W.; Park, M.-S.; Kim, N.-H.; Lee, J.-H.; Oh, C.-W.; Kim, H.-R.; Kim, G.-D. Hexane extract from Sargassum serratifolium inhibits the cell proliferation and metastatic ability of human glioblastoma U87MG cells. Oncol. Rep. 2015, 34, 2602–2608. [Google Scholar] [CrossRef] [Green Version]
- Tsang, C.K.; Kamei, Y. Sargaquinoic acid supports the survival of neuronal PC12D cells in a nerve growth factor-independent manner. Eur. J. Pharmacol. 2004, 488, 11–18. [Google Scholar] [CrossRef]
- El-Demerdash, A.; Kumla, D.; Kijjoa, A. Chemical diversity and biological activities of meroterpenoids from marine derived-fungi: A comprehensive update. Mar. Drugs 2020, 18, 317. [Google Scholar] [CrossRef]
- Zhao, M.; Tang, Y.; Xie, J.; Zhao, Z.; Cui, H. Meroterpenoids produced by fungi: Occurrence, structural diversity, biological activities, and their molecular targets. Eur. J. Med. Chem. 2021, 209, 112860. [Google Scholar] [CrossRef]
- Gozari, M.; Alborz, M.; El-Seedi, R.H.; Reza, R.J. Chemistry, biosynthesis and biological activity of terpenoids and meroterpenoids in bacteria and fungi isolated from different marine habitats. Eur. J. Med. Chem. 2021, 210, 112957. [Google Scholar] [CrossRef]
- Jiang, M.; Wu, Z.; Liu, L.; Chen, S. The chemistry and biology of fungal meroterpenoids (2009–2019). Org. Biomol. Chem. 2021, 19, 1644–1704. [Google Scholar] [CrossRef]
- Qin, X.-J.; Yu, Q.; Yan, H.; Khan, A.; Feng, M.-Y.; Li, P.-P.; Hao, X.-J.; An, L.-K.; Liu, H.-Y. Meroterpenoids with antitumor activities from guava (Psidium guajava). J. Agric. Food Chem. 2017, 65, 4993–4999. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Duan, M.; Zhao, L.; Ma, P. Guajadial inhibits NSCLC growth and migration following activation of the VEGF receptor-2. Fitoterapia 2018, 129, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.-J.; Yan, H.; Ni, W.; Yu, M.-Y.; Khan, A.; Liu, H.; Zhang, H.-X.; He, L.; Hao, X.-J.; Di, Y.-T. Cytotoxic meroterpenoids with rare skeletons from Psidium guajava cultivated in temperate zone. Sci. Rep. 2016, 6, 32748. [Google Scholar] [CrossRef] [PubMed]
- Shang, Z.C.; Yang, M.H.; Jian, K.L.; Wang, X.B.; Kong, L.Y. 1H NMR-guided isolation of formyl-phloroglucinol meroterpenoids from the leaves of Eucalyptus robusta. Chem. Eur. J. 2016, 22, 11778–11784. [Google Scholar] [CrossRef]
- Farias, I.V.; Faqueti, L.G.; Noldin, V.F.; Junior, G.F.; Nowil, A.E.; Schuquel, I.T.; DelleMonache, F.; García, P.A.; López-Pérez, J.L.; SanFeliciano, A. Cytotoxic phloroglucinol meroterpenoid from Eugenia umbelliflora fruits. Phytochem. Lett. 2018, 27, 187–192. [Google Scholar] [CrossRef]
- Liu, H.; Feng, M.-Y.; Yu, Q.; Yan, H.; Zeng, Y.; Qin, X.-J.; He, L.; Liu, H.-Y. Formyl phloroglucinol meroterpenoids from Eucalyptus tereticornis and their bioactivities. Tetrahedron 2018, 74, 1540–1545. [Google Scholar] [CrossRef]
- Qin, X.-J.; Jin, L.-Y.; Yu, Q.; Liu, H.; Khan, A.; Yan, H.; Hao, X.-J.; An, L.-K.; Liu, H.-Y. Eucalypglobulusals A–J: Formyl-phloroglucinol–terpene meroterpenoids from Eucalyptus globulus fruits. J. Nat. Prod. 2018, 81, 2638–2646. [Google Scholar] [CrossRef]
- Hu, L.; Xue, Y.; Zhang, J.; Zhu, H.; Chen, C.; Li, X.-N.; Liu, J.; Wang, Z.; Zhang, Y.; Zhang, Y. (±)-Japonicols A–D: Acylphloroglucinol-based meroterpenoid enantiomers with anti-KSHV activities from Hypericum japonicum. J. Nat. Prod. 2016, 79, 1322–1328. [Google Scholar] [CrossRef]
- Hu, L.; Liu, Y.; Wang, Y.; Wang, Z.; Huang, J.; Xue, Y.; Liu, J.; Liu, Z.; Chen, Y.; Zhang, Y. Discovery of acylphloroglucinol-based meroterpenoid enantiomers as KSHV inhibitors from Hypericum japonicum. RSC Adv. 2018, 8, 24101–24109. [Google Scholar] [CrossRef] [Green Version]
- Tang, G.-H.; Dong, Z.; Guo, Y.-Q.; Cheng, Z.-B.; Zhou, C.-J.; Yin, S. Psiguajadials A-K: Unusual Psidium meroterpenoids as phosphodiesterase-4 inhibitors from the leaves of Psidium guajava. Sci. Rep. 2017, 7, 1047. [Google Scholar] [CrossRef] [Green Version]
- Shang, Z.-C.; Yang, M.-H.; Liu, R.-H.; Wang, X.-B.; Kong, L.-Y. New formyl phloroglucinol meroterpenoids from the leaves of Eucalyptus robusta. Sci. Rep. 2016, 6, 39815. [Google Scholar] [CrossRef]
- Yu, Y.; Gan, L.-S.; Yang, S.-P.; Sheng, L.; Liu, Q.-F.; Chen, S.-N.; Li, J.; Yue, J.-M. Eucarobustols A-I: Conjugates of sesquiterpenoids and acylphloroglucinols from Eucalyptus robusta. J. Nat. Prod. 2016, 79, 1365–1372. [Google Scholar] [CrossRef]
- Liu, J.; Jiang, L.-R.; Liu, M.-F. Unusual meroterpenoids from leaves of Psidium guajava. Chem. Nat. Compd. 2016, 52, 67–70. [Google Scholar] [CrossRef]
- Qin, X.-J.; Feng, M.-Y.; Liu, H.; Ni, W.; Rauwolf, T.; Porco, J.A., Jr.; Yan, H.; He, L.; Liu, H.-Y. Eucalyptusdimers A–C: Dimeric phloroglucinol–phellandrene meroterpenoids from Eucalyptus robusta. Org. Lett. 2018, 20, 5066–5070. [Google Scholar] [CrossRef] [Green Version]
- Qin, X.-J.; Liu, H.; Yu, Q.; Yan, H.; Tang, J.-F.; An, L.-K.; Khan, A.; Chen, Q.-R.; Hao, X.-J.; Liu, H.-Y. Acylphloroglucinol derivatives from the twigs and leaves of Callistemon salignus. Tetrahedron 2017, 73, 1803–1811. [Google Scholar] [CrossRef]
- Chen, N.-H.; Zhang, Y.-B.; Huang, X.-J.; Jiang, L.; Jiang, S.-Q.; Li, G.-Q.; Li, Y.-L.; Wang, G.-C. Drychampones A−C: Three meroterpenoids from Dryopteris championii. J. Org. Chem. 2016, 81, 9443–9448. [Google Scholar] [CrossRef]
- Li, C.-J.; Ma, J.; Sun, H.; Zhang, D.; Zhang, D.-M. Guajavadimer A, a dimeric caryophyllene-derived meroterpenoid with a new carbon skeleton from the leaves of Psidium guajava. Org. Lett. 2016, 18, 168–171. [Google Scholar] [CrossRef]
- Chen, N.; Wu, Z.; Li, W.; Li, Y.; Luo, D.; Chen, L.; Zhang, X.; Zhang, Y.; Wang, G.; Li, Y. Acylphloroglucinols-based meroterpenoid enantiomers with antiviral activities from Dryopteris crassirhizoma. Ind. Crops Prod. 2020, 150, 112415. [Google Scholar] [CrossRef]
- Hou, J.-Q.; Fan, C.-L.; Pei, X.; Hang, P.-L.; Deng, F.; Jiang, W.-Q.; Wang, G.-C.; Zhang, X.-Q.; Ye, W.-C.; Wang, H. Psiguadiols A−J, rearranged meroterpenoids as potent PTP1B inhibitors from Psidium guajava. J. Nat. Prod. 2019, 82, 3267–3278. [Google Scholar] [CrossRef]
- Xu, J.; Zhu, H.-L.; Zhang, J.; Liu, W.-Y.; Luo, J.-G.; Pan, K.; Cao, W.-Y.; Bi, Q.-R.; Feng, F.; Qu, W. Littordials A–E, novel formyl-phloroglucinol-β-caryophyllene meroterpenoids from the leaves of Psidium littorale. Org. Chem. Front. 2019, 6, 1667–1673. [Google Scholar] [CrossRef]
- Shang, Z.-C.; Han, C.; Xu, J.-L.; Liu, R.-H.; Yin, Y.; Wang, X.-B.; Yang, M.-H.; Kong, L.-Y. Twelve formyl phloroglucinol meroterpenoids from the leaves of Eucalyptus robusta. Phytochemistry 2019, 163, 111–117. [Google Scholar] [CrossRef]
- Xie, X.; Wu, L.; Cui, Z.; Yang, M.; Yin, Y.; Luo, J.; Kong, L. Melaleucadines A and B: Two rare benzylic phloroglucinol-terpene hybrids from Melaleuca leucadendron. Tetrahedron Lett. 2019, 60, 1011–1013. [Google Scholar] [CrossRef]
- Zhu, H.-L.; Hu, Y.-W.; Qu, W.; Zhang, J.; Guo, E.-Y.; Jiang, X.-Y.; Liu, W.-Y.; Feng, F.; Xu, J. Littordial F, a novel phloroglucinol meroterpenoid from the Leaves of Psidium littorale. Tetrahedron Lett. 2019, 60, 1868–1870. [Google Scholar] [CrossRef]
- Zhang, J.; He, J.; Cheng, Y.-C.; Zhang, P.-C.; Yan, Y.; Zhang, H.-J.; Zhang, W.-K.; Xu, J.-K. Fischernolides A–D, four novel diterpene-based eroterpenoid scaffolds with antitumor activities from Euphorbia fischeriana. Org. Chem. Front. 2019, 6, 2312–2318. [Google Scholar] [CrossRef]
- Xie, R.; Li, L.; Fan, X.; Zi, J. Euphoractone, a cytotoxic meroterpenoid with an unusual ent-abietane-phloroglucinol skeleton, from Euphorbia fischeriana Steud. Chin. Chem. Lett. 2020, 31, 431–433. [Google Scholar] [CrossRef]
- Qin, X.-J.; Zhi, Y.-E.; Yan, H.; Zhang, Y.; Liu, H.; Yu, Q.; Wang, S.; Zhao, Q.; He, L.; Ma, X. Baeckfrutones A-L: Polymethylated phloroglucinol meroterpenoids from the twigs and leaves of Baeckea frutescens. Tetrahedron 2018, 74, 6658–6666. [Google Scholar] [CrossRef]
- Zhi, Y.-E.; Qi, X.-J.; Liu, H.; Zeng, Y.; Ni, W.; He, L.; Wang, Z.-D.; Liu, H.-Y. Structurally diverse polymethylated phloroglucinol meroterpenoids from Baeckea frutescens. Nat. Products Bioprospect. 2018, 8, 431–439. [Google Scholar] [CrossRef] [Green Version]
- Wu, R.; Le, Z.; Wang, Z.; Tian, S.; Xue, Y.; Chen, Y.; Hu, L.; Zhang, Y. Hyperjaponol H: A new bioactive filicinic acid-based meroterpenoid from Hypericum japonicum Thunb. ex Murray. Molecules 2018, 23, 683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, H.-X.; Zhang, W.-M.; Xu, Z.-F.; Chen, Y.-C.; Tan, H.-B.; Qiu, S.-X. Isolation, synthesis, and biological activity of tomentosenol A from the leaves of Rhodomyrtus tomentosa. RSC Adv. 2016, 6, 25882–25886. [Google Scholar] [CrossRef]
- Liu, C.; Ang, S.; Huang, X.-J.; Tian, H.-Y.; Deng, Y.-Y.; Zhang, D.-M.; Wang, Y.; Ye, W.-C.; Wang, L. Meroterpenoids with new skeletons from Myrtus communis and structure revision of myrtucommulone K. Org. Lett. 2016, 18, 4004–4007. [Google Scholar] [CrossRef] [PubMed]
- Cottiglia, F.; Casu, L.; Leonti, M.; Caboni, P.; Floris, C.; Busonera, B.; Farci, P.; Ouhtit, A.; Sanna, G. Cytotoxic phloroglucinols from the leaves of Myrtus communis. J. Nat. Prod. 2012, 75, 225–229. [Google Scholar] [CrossRef]
- Hou, J.-Q.; Guo, C.; Zhao, J.-J.; He, Q.-W.; Zhang, B.-B.; Wang, H. Frutescone A–G: Tasmanone-based meroterpenoids from the aerial parts of Baeckea frutescens. J. Org. Chem. 2017, 82, 1448–1457. [Google Scholar] [CrossRef]
- Zhang, Y.-L.; Chen, C.; Wang, X.-B.; Wu, L.; Yang, M.-H.; Luo, J.; Zhang, C.; Sun, H.-B.; Luo, J.-G.; Kong, L.-Y. Rhodomyrtials A and B: Two meroterpenoids with a triketone-sesquiterpene-triketone skeleton from Rhodomyrtus tomentosa, structural elucidation and biomimetic synthesis. Org. Lett. 2016, 18, 4068–4071. [Google Scholar] [CrossRef]
- Zhang, Y.-L.; Zhou, X.-W.; Wu, L.; Wang, X.-B.; Yang, M.-H.; Luo, J.; Luo, J.-G.; Kong, L.-Y. Isolation, structure elucidation, and absolute configuration of syncarpic acid-conjugated terpenoids from Rhodomyrtus tomentosa. J. Nat. Prod. 2017, 80, 989–998. [Google Scholar] [CrossRef]
- Qin, X.-J.; Shu, T.; Yu, Q.; Yan, H.; Ni, W.; An, L.-K.; Li, P.-P.; Zhi, Y.-E.; Khan, A.; Liu, H.-Y. Cytotoxic acylphloroglucinol derivatives from Callistemon salignus. Nat. Prod. Bioprospect. 2017, 7, 315–321. [Google Scholar] [CrossRef] [Green Version]
- Hou, J.-Q.; Guo, C.; Zhao, J.-J.; Dong, Y.-Y.; Hu, X.-L.; He, Q.-W.; Zhang, B.-B.; Yan, M.; Wang, H. Anti-inflammatory meroterpenoids from Baeckea frutescens. J. Nat. Prod. 2017, 80, 2204–2214. [Google Scholar] [CrossRef]
- Hou, J.-Q.; Wang, B.-L.; Han, C.; Xu, J.; Wang, Z.; He, Q.-W.; Zhang, P.-L.; Zhao, S.-M.; Pei, X.; Wang, H. Atropisomeric meroterpenoids with rare triketone-phloroglucinol-terpene hybrids from Baeckea frutescens. Org. Biomol. Chem. 2018, 16, 8513–8524. [Google Scholar] [CrossRef]
- Hu, L.; Zhang, Y.; Zhu, H.; Liu, J.; Li, H.; Li, X.-N.; Sun, W.; Zeng, J.; Xue, Y.; Zhang, Y. Filicinic acid based meroterpenoids with anti-epstein-barr virus activities from Hypericum japonicum. Org. Lett. 2016, 18, 2272–2275. [Google Scholar] [CrossRef]
- Zhang, H.-X.; Lunga, P.K.; Li, Z.J.; Dai, Q.; Du, Z.-Z. Flavonoids and stilbenoids from Derris eriocarpa. Fitoterapia 2014, 95, 147–153. [Google Scholar] [CrossRef]
- Chen, M.; Chen, L.-F.; Li, M.-M.; Li, N.-P.; Cao, J.-Q.; Wang, Y.; Li, Y.-L.; Wang, L.; Ye, W.-C. Myrtucomvalones A–C, three unusual triketone—Sesquiterpene adducts from the leaves of Myrtus communis ‘Variegata’. RSC Adv. 2017, 7, 22735–22740. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.-X.; Chen, K.; Yuan, Y.; Xu, Z.-F.; Tan, H.-B.; Qiu, S.-X. Rhodomentones A and B, novel meroterpenoids with unique NMR characteristics from Rhodomyrtus tomentosa. Org. Biomol. Chem. 2016, 14, 7354–7360. [Google Scholar] [CrossRef] [PubMed]
- Senadeera, S.P.D.; Robertson, L.P.; Duffy, S.; Wang, Y.; Avery, V.M.; Carroll, A.R. β-Triketone–monoterpene mybrids from the flowers of the australian tree Corymbia intermedia. J. Nat. Prod. 2018, 81, 2455–2461. [Google Scholar] [CrossRef]
- Li, Q.-J.; Tang, P.-F.; Zhou, X.; Lu, W.-J.; Xu, W.-J.; Luo, J.; Kong, L.-Y. Dimethylated acylphloroglucinol meroterpenoids with anti-oral-bacterialand anti-inflammatory activities from Hypericum elodeoides. Bioorgan. Chem. 2020, 104, 104275. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Li, P.; Bi, L.-S.; Wu, W.-J.; Yan, H.; He, L.; Qin, X.-J.; Liu, H.-Y. Polymethylated phloroglucinol meroterpenoids from Rhodomyrtus tomentosa and their antibacterial and acetylcholinesterase inhibitory effects. Chem. Biodivers. 2020, 17, e2000489. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.-Q.; Zhao, H.; Yu, J.-H.; Chen, L.-F.; Hao, W. New meroterpenoids and C-methylated flavonoid isolated from Baeckea frutescens. Chin. J. Nat. Med. 2020, 18, 379–384. [Google Scholar] [CrossRef]
- Deng, X.; Wang, X.; Wu, L. Triketone-terpene meroterpenoids from the leaves of Rhodomyrtus tomentosa. Fitoterapia 2020, 143, 104585. [Google Scholar] [CrossRef] [PubMed]
- Asolkar, R.N.; Singh, A.; Jensen, P.R.; Aalbersberg, W.; Carte, B.K.; Feussner, K.-D.; Subramani, R.; DiPasquale, A.; Rheingold, A.L.; Fenical, W. Marinocyanins: Cytotoxic bromo-phenazinone meroterpenoids from a marine bacterium from the Streptomycete clade MAR4. Tetrahedron 2017, 73, 2234–2241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kohatsu, H.; Kamo, S.; Tomoshige, S.; Kuramochi, K. Total syntheses of pyocyanin, lavanducyanin, and marinocyanins A and B. Org. Lett. 2019, 21, 7311–7314. [Google Scholar] [CrossRef]
- Zhang, X.; Li, P.-L.; Qin, G.-F.; Li, S.; de Voogd, N.J.; Tang, X.-L.; Li, G.-Q. Isolation and absolute configurations of diversiform C17, C21 and C25 terpenoids from the marine Sponge Cacospongia sp. Mar. Drugs 2019, 17, 14. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, H.M.; Ito, T.; Win, N.N.; Kodama, T.; Hung, V.Q.; Nguyen, H.T.; Morita, H. New antibacterial sesquiterpene aminoquinones from a vietnamese marine sponge of Spongia sp. Phytochem. Lett. 2016, 17, 288–292. [Google Scholar] [CrossRef]
- Nguyen, H.M.; Ito, T.; Kurimoto, S.-I.; Ogawa, M.; Win, N.N.; Hung, V.Q.; Nguyen, H.T.; Kubota, T.; Kobayashi, J.I.; Morita, H. New merosesquiterpenes from a Vietnamese marine sponge of Spongia sp. and their biological activities. Bioorg. Med. Chem. Lett. 2017, 27, 3043–3047. [Google Scholar] [CrossRef]
- Zhang, X.; Xu, H.-Y.; Huang, A.-M.; Wang, L.; Wang, Q.; Cao, P.-Y.; Yang, P.-M. Antibacterial meroterpenoids from the south China sea sponge Dysidea sp. Chem. Pharm. Bull. 2016, 64, 1036–1042. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Yang, F.; Wang, Z.; Wu, W.; Liu, L.; Wang, S.-P.; Zhao, B.-X.; Jiao, W.-H.; Xu, S.-H.; Lin, H.-W. Unusual anti-inflammatory meroterpenoids from the marine sponge Dactylospongia sp. Org. Biomol. Chem. 2018, 16, 6773–6782. [Google Scholar] [CrossRef]
- Oba, Y.; Yoshida, N.; Kanie, S.; Ojika, M.; Inouye, S. Biosynthesis of firefly luciferin in adult lantern, decarboxylation of L-cysteine is a key step for benzothiazole ring formation in firefly luciferin synthesis. PLoS ONE 2013, 8, e84023. [Google Scholar] [CrossRef]
- Wang, J.; Mu, F.-R.; Jiao, W.-H.; Huang, J.; Hong, L.-L.; Yang, F.; Xu, Y.; Wang, S.-P.; Sun, F.; Lin, H.-W. Meroterpenoids with protein tyrosine phosphatase 1b inhibitory activity from a Hyrtios sp. marine sponge. J. Nat. Prod. 2017, 80, 2509–2514. [Google Scholar] [CrossRef]
- Jiao, W.-H.; Cheng, B.-H.; Shi, G.-H.; Chen, G.-D.; Gu, B.-B.; Zhou, Y.-J.; Hong, L.-L.; Yang, F.; Liu, Z.-Q.; Qiu, S.-Q. Dysivillosins A–D: Unusual anti-allergic meroterpenoids from the marine sponge Dysidea villosa. Sci. Rep. 2017, 7, 8947. [Google Scholar] [CrossRef]
- Salame, R.; Gravel, E.; Leblanc, K.; Poupon, E. Biomimetic synthesis of tangutorine following new biogenetic proposals. Org. Lett. 2009, 11, 1891–1894. [Google Scholar] [CrossRef]
- Liu, D.; Li, Y.; Li, X.; Cheng, Z.; Huang, J.; Proksch, P.; Lin, W. Chartarolides A–C: Novel meroterpenoids with antitumor activities. Tetrahedron Lett. 2017, 58, 1826–1829. [Google Scholar] [CrossRef]
- Jiao, W.-H.; Cheng, B.-H.; Chen, G.-D.; Shi, G.-H.; Li, J.; Hu, T.-Y.; Lin, H.-W. Dysiarenone, a dimeric C21 meroterpenoid with inhibition of COX-2 expression from the marine sponge Dysidea arenaria. Org. Lett. 2018, 20, 3092–3095. [Google Scholar] [CrossRef]
- Rehman, N.; Hussain, H.; Al-Shidhani, S.; Avual, S.K.; Abbas, G.; Anwar, M.U.; Górecki, M.; Pescitelli, G.; Al-Harrasi, A. Incensfuran: Isolation, X-ray crystal structure and absolute configuration by means of chiroptical studies in solution and solid state. RSC Adv. 2017, 7, 42357–42362. [Google Scholar] [CrossRef] [Green Version]
- Jiao, W.-H.; Shi, G.-H.; Xu, T.-T.; Chen, G.-D.; Gu, B.-B.; Wang, Z.; Peng, S.; Wang, S.-P.; Li, J.; Han, B.-N.; et al. Dysiherbols A−C and dysideanone E, cytotoxic and NF-κB inhibitory tetracyclic meroterpenes from a Dysidea sp. marine sponge. J. Nat. Prod. 2016, 79, 406–411. [Google Scholar] [CrossRef]
- Jiao, W.-H.; Li, J.; Wang, D.; Zhang, M.-M.; Liu, L.-Y.; Sun, F.; Li, J.Y.; Capon, R.J.; Lin, H.-W. Cinerols, nitrogenous meroterpenoids from the marine sponge Dysidea cinerea. J. Nat. Prod. 2019, 82, 2586–2593. [Google Scholar] [CrossRef]
- Le, T.C.; Lee, E.J.; Lee, J.; Hong, A.; Yim, C.-Y.; Yang, I.; Choi, H.; Chin, J.; Cho, S.J.; Ko, J.; et al. Saccharoquinoline, a cytotoxic alkaloidal meroterpenoid from marine-derived bacterium Saccharomonospora sp. Mar. Drugs 2019, 17, 98. [Google Scholar] [CrossRef] [Green Version]
- Yu, H.-B.; Yin, Z.-F.; Gu, B.-N.; Zhang, J.-P.; Wang, S.-P.; Yang, F.; Lin, H.-W. Cytotoxic meroterpenoids from the marine sponge Dactylospongia elegans. Nat. Prod. Res. 2021. in print. [Google Scholar] [CrossRef]
- Gui, Y.-H.; Jiao, W.-H.; Zhou, M.; Zhang, Y.; Zeng, D.-Q.; Zhu, H.-R.; Liu, K.-C.; Sun, F.; Chen, H.-F.; Lin, H.W. Septosones A−C, in vivo anti-inflammatory meroterpenoids with rearranged carbon skeletons from the marine sponge Dysidea septosa. Org. Lett. 2019, 21, 767–770. [Google Scholar] [CrossRef]
- Hamed, A.; Abdel-Razek, A.; Frese, M.; Stammler, H.; El-Haddad, A.; Ibrahim, T.; Sewald, N.; Shaaban, M.; Terretonin, N. A new meroterpenoid from Nocardiopsis sp. Molecules 2018, 23, 299. [Google Scholar] [CrossRef] [Green Version]
- Choi, H.; Hwang, H.; Chin, J.; Kim, E.; Lee, J.; Nam, S.-J.; Lee, B.C.; Rho, B.J.; Kang, H. Tuberatolides, potent fxr antagonists from the korean marine tunicate Botryllus tuberatus. J. Nat. Prod. 2011, 74, 90–94. [Google Scholar] [CrossRef]
- Choi, Y.; Kim, J.; Lee, K.; Choi, Y.-J.; Ye, B.-R.; Kim, M.-S.; Ko, S.-G.; Lee, S.-H.; Kang, D.-H.; Heo, S.-J. Tuberatolide B suppresses cancer progression by promoting ROS-mediated inhibition of STAT3 signaling. Mar. Drugs 2017, 15, 55. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.-X.; Wang, J.-X.; Wang, Q.; Li, H.-I.; Tao, M.; Luo, Q.; Liu, H. New chromane and chromene meroterpenoids from flowers of Rhododendron rubiginosum Franch. var. rubiginosum. Fitoterapia 2018, 127, 396–401. [Google Scholar] [CrossRef]
- Hanuš, L.O.; Meyer, S.M.; Muñoz, E.; Taglialatela-Scafati, O.; Appendino, G. Phytocannabinoids, a unified critical inventory. Nat. Prod. Rep. 2016, 33, 1357–1392. [Google Scholar] [CrossRef] [Green Version]
- Kang, H.-S.; Kim, J.-P. New chromene derivatives with radical scavenging activities from the brown alga Sargassum siliquastrum. J. Chem. Res. 2017, 41, 116–119. [Google Scholar] [CrossRef]
- Liao, H.-B.; Huang, G.-H.; Yu, M.-H.; Lei, C.; Hou, A.-J. Five pairs of meroterpenoid enantiomers from Rhododendron capitatum. J. Org. Chem. 2017, 82, 1632–1637. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.-H.; Hu, Z.; Lei, C.; Wang, P.-P.; Yang, J.; Li, J.-Y.; Li, J.; Hou, A.-J. Enantiomeric pairs of meroterpenoids with diverse heterocyclic systems from Rhododendron nyingchiense. J. Nat. Prod. 2018, 81, 1810–1818. [Google Scholar] [CrossRef] [PubMed]
- Seong, S.H.; Ali, M.Y.; Kim, H.-R.; Jung, H.A.; Choi, J.S. BACE1 inhibitory activity and molecular docking analysis of meroterpenoids from Sargassum serratifolium. Bioorg. Med. Chem. 2017, 25, 3964–3970. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, C.B.; Gangadhar, K.N.; Morais, T.R.; Conserva, G.A.; Vizetto-Duarte, C.; Pereira, H.; Laurenti, M.D.; Campino, L.; Levy, D.; Uemi, M. Antileishmanial activity of meroditerpenoids from the macroalgae Cystoseira baccata. Exp. Parasitol. 2017, 174, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Chiou, C.-T.; Shen, C.-C.; Tsai, T.-H.; Chen, Y.-J.; Lin, L.-C. Meroterpenoids and chalcone-lignoids from the roots of Mimosa diplotricha. J. Nat. Prod. 2016, 79, 2439–2445. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.-Q.; Hai, P.; Xiao, H.; Gao, Y.; Tao, Y.; Miao, D.; Wang, F. Glabralides A–C, three novel meroterpenoids from Sarcandra glabra. Tetrahedron 2018, 74, 341–347. [Google Scholar] [CrossRef]
- Huang, G.-H.; Lei, C.; Zhu, K.-X.; Li, J.-Y.; Li, J.; Hou, A.-J. Enantiomeric pairs of meroterpenoids from Rhododendron fastigiatum. Chin. J. Nat. Med. 2019, 17, 963–969. [Google Scholar] [CrossRef]
- Shi, Q.; Li, F.-T.; Wu, Y.-M.; Sun, X.-Y.; Lei, C.; Li, J.-Y.; Hou, A.-J. Meroterpenoids with diverse structures and anti-inflammatory activities from Rhododendron anthopogonoides. Phytochemistry 2020, 180, 112524. [Google Scholar] [CrossRef]
- Xu, Q.-X.; Zhang, Y.-B.; Liu, X.-Y.; Xu, W.; Yang, X.-W. Cytotoxic heterodimers of meroterpene phenol from the fruits of Psoralea Corylifolia. Phytochemistry 2020, 176, 112394. [Google Scholar] [CrossRef]
- Ni, G.; Shi, G.-R.; Li, J.-Y.; Yu, D.-Q. The unprecedented iridal lactone and adducts of spiroiridal and isoflavonoid from Belamcanda chinensis. RSC Adv. 2017, 7, 20160–20166. [Google Scholar] [CrossRef] [Green Version]
- Zhu, Y.-D.; Chen, R.-C.; Wang, H.; Jiang, H.; Huang, X.-L.; Zhang, M.-L.; Li, L.-Y.; Hu, Z.; Xu, X.-D.; Wang, C.-J. Two new flavonoid–triterpene saponin meroterpenoids from Clinopodium chinense and their protective effects against anoxia/reoxygenation-induced apoptosis in H9c2 cells. Fitoterapia 2018, 128, 180–186. [Google Scholar] [CrossRef]
- Martucci, H.; Campit, S.E.; Gee, S.R.; Bray, W.M.; Gokey, T.; Cada, A.K.; Yen, T.-Y.; Minoura, K.; Guliaev, A.B.; Lokey, R.S. Naphthablins B and C: Meroterpenoids identified from the marine sediment-derived Streptomyces sp. CP26-58 using HeLa cell-based cytological profiling. J. Nat. Prod. 2017, 80, 684–691. [Google Scholar] [CrossRef]
- Li, C.; Li, C.-J.; Ma, J.; Chen, F.-Y.; Li, L.; Wang, X.-L.; Ye, F.; Zhang, D.-M. Magterpenoids A–C: Three polycyclic meroterpenoids with PTP1B inhibitory activity from the bark of Magnolia officinalis var. biloba. Org. Lett. 2018, 20, 3682–3686. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, Y.; Xiao, L.; Ge, L.; Wu, X.; Wu, W.; Wan, H.; Zhang, K.; Li, J.; Zhou, B. Meroterpenoids isolated from Arnebia euchroma (Royle) Johnst. and their cytotoxic activity in human hepatocellular carcinoma cells. Fitoterapia 2018, 131, 236–244. [Google Scholar] [CrossRef]
- Wan, H.; Li, J.; Zhang, K.; Zou, X.; Ge, L.; Zhu, F.; Zhou, H.; Gong, M.; Wang, T.; Chen, D. A new meroterpenoid functions as an anti-tumor agent in hepatoma cells by downregulating mTOR activation and inhibiting EMT. Sci. Rep. 2018, 8, 13152. [Google Scholar] [CrossRef]
- Lai, K.-H.; Liu, Y.-C.; Su, J.-H.; El-Shazly, M.; Wu, C.-F.; Du, Y.-C.; Hsu, Y.-M.; Yang, J.-C.; Weng, M.-K.; Chou, C.-H. Antileukemic scalarane sesterterpenoids and meroditerpenoid from Carteriospongia (Phyllospongia) sp., induce apoptosis via dual inhibitory effects on topoisomerase II and Hsp90. Sci. Rep. 2016, 6, 36170. [Google Scholar] [CrossRef]
- Shen, X.; Wang, X.; Huang, T.; Deng, Z.; Lin, S. Naphthoquinone-based meroterpenoids from marine-derived streptomyces sp. B9173. Biomolecules 2020, 10, 1187. [Google Scholar] [CrossRef]
- Ryu, M.-J.; Hwang, S.; Kim, S.; Yang, I.; Oh, D.-C.; Nam, S.-J.; Fenical, W. Meroindenon and merochlorins E and F, antibacterial meroterpenoids from a marine-derived sediment bacterium of the Genus Streptomyces. Org. Lett. 2019, 21, 5779–5783. [Google Scholar] [CrossRef]
- Joy, M.; Chakraborty, K. Specialized oxygenated heterocyclics from Villorita cyprinoides with cyclooxygenase-2 and 5-lipoxygenase inhibitory properties. Food Res. Int. 2018, 106, 164–172. [Google Scholar] [CrossRef]
- Joy, M.; Chakraborty, K. Antioxidative and anti-inflammatory pyranoids and isochromenyl analogues from Corbiculid bivalve clam, Villorita cyprinoides. Food Chem. 2018, 251, 125–134. [Google Scholar] [CrossRef]
- Chakraborty, K.; Joseph, D.; Joy, M.; Raola, V.K. Characterization of substituted aryl meroterpenoids from red seaweed Hypnea musciformis as potential antioxidants. Food Chem. 2016, 212, 778–788. [Google Scholar] [CrossRef]
- De los Reyes, C.; Ortega, M.J.; Zbakh, H.; Motilva, V.; Zubía, E. Cystoseira usneoides, A brown alga rich in antioxidant and anti-inflammatory meroditerpenoids. J. Nat. Prod. 2016, 79, 395–405. [Google Scholar] [CrossRef]
- Joy, M.; Chakraborty, K. First report of two new antioxidative meroterpeno 2 H-pyranoids from short-necked yellow-foot clam Paphia malabarica (family, Veneridae) with bioactivity against pro-inflammatory cyclooxygenases and lipoxygenase. Nat. Prod. Res. 2016, 31, 615–625. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Li, L.; Zhu, R.; Zhang, J.; Zhou, J.; Lou, H. Bibenzyl-based meroterpenoid enantiomers from the Chinese liverwort Radula sumatrana. J. Nat. Prod. 2017, 80, 3143–3150. [Google Scholar] [CrossRef]
- Jiaa, X.; Wu, Y.; Lei, C.; Yu, Y.; Li, J.; Li, J.; Hou, A. Hyperinoids A and B, two polycyclic meroterpenoids from Hypericum patulum. Chin. Chem. Lett. 2020, 31, 1263–1266. [Google Scholar] [CrossRef]
- Wang, S.-X.; Zhao, R.-L.; Guo, C.; Chen, B.-S.; Dai, H.-Q.; Liu, G.-Q.; Liu, H.-W. New meroterpenoid compounds from the culture of mushroom Panus lecomtei. Chin. J. Nat. Med. 2020, 18, 268–272. [Google Scholar] [CrossRef]
- Day, A.J.; George, J.H. Isolation and biomimetic oxidation of prenylbruceol A, an anticipated meroterpenoid natural product from Philotheca myoporoides. J. Nat. Prod. 2020, 83, 2305–2309. [Google Scholar] [CrossRef]
- Gurr, J.R.; O’Donnell, T.J.; Luo, Y.; Yoshida, W.Y.; Hall, M.L.; Mayer, A.M.S.; Sun, R.; Williams, P.G. 6-Deoxy- and 11-hydroxytolypodiols: Meroterpenoids from the cyanobacterium HT-58-2. J. Nat. Prod. 2020, 83, 1691–1695. [Google Scholar] [CrossRef]
- Zong, J.-F.; Hu, Z.; Shao, Y.-Y.; Shi, Q.; Zhang, M.-M.; Zhou, Y.-B.; Li, J.; Hou, A.J. Hyperprins A and B, two complex meroterpenoids from Hypericum przewalskii. Org. Lett. 2020, 22, 2797–2800. [Google Scholar] [CrossRef]
- Ye, Y.-S.; Du, S.-Z.; Jiang, N.-N.; Xu, H.-X.; Yang, J.; Fu, W.-W.; Nian, Y.; Xu, G. Novel meroterpenoids from Hypericum patulum: Highly potent late Nav1.5 sodium current inhibitors. Org. Lett. 2020, 22, 6339–6343. [Google Scholar] [CrossRef]
- Nguyen, N.Y.T.; Pham, N.S.L.; Dang, P.H.; Huu, D.M.N.; Dang, H.P.; Tran, Q.L. Two new meroterpenoids from the aerial parts of Ampelopsis cantoniensis (Vitaceae). J. Asian Nat. Prod. Res. 2020, 22, 1152–1158. [Google Scholar] [CrossRef]
- Wang, D.; Xiu, M.-X.; Li, H.-Z.; Xiong, D.-X.; Lee, H.-X.; Sun, Y.-N.; Cui, L. Two new meroterpenes with activity against diacylglycerol acyltransferase from seeds of Psoralea corylifolia. Phytochem. Lett. 2020, 40, 171–175. [Google Scholar] [CrossRef]
- Xu, Q.-X.; Xu, W.; Yang, X.-W. Meroterpenoids from the fruits of Psoralea corylifolia. Tetrahedron 2020, 76, 131343. [Google Scholar] [CrossRef]


















































| Compounds | Source | Anticancer | Ref. |
|---|---|---|---|
| Marinocyanin A (240) | Actinomycete strains | Cytotoxic effects: HCT-116 = IC50 0.049 μM; Antimicrobial effects: Candida albicans = MIC 0.95 μM; Staphylococcus aureus = MIC 2.3 μM | [79] |
| Marinocyanin B (241) | Actinomycete strains | Cytotoxic effects: HCT-116 = IC50 0.029 μM; Antimicrobial effects: Candida albicans = MIC 5.79 μM; Staphylococcus aureus = MIC 33.92 μM | [79] |
| Marinocyanin C (242) | Actinomycete strains | Antimicrobial effects: Candida albicans = MIC 3.90 μM; Staphylococcus aureus = MIC 30.71 μM | [79] |
| Marinocyanin D (243) | Actinomycete strains | Antimicrobial effects: Candida albicans = MIC 14.65 μM; Staphylococcus aureus = MIC 36.62 μM | [79] |
| Marinocyanin E (244) | Actinomycete strains | Antimicrobial effects: Candida albicans = MIC 14.65 μM; Staphylococcus aureus = MIC 36.62 μM | [79] |
| Marinocyanin F (245) | Actinomycete strains | Antimicrobial effects: Candida albicans = MIC 14.65 μM; Staphylococcus aureus = MIC 36.62 μM | [79] |
| Lavanducyanin (246) | Streptomyces sp. | Antimicrobial effects: Candida albicans = MIC 114.67 μM; Staphylococcus aureus = MIC 56.93 μM | [79] |
| Compounds | Source | Activities | Ref. |
|---|---|---|---|
| Langcoquinone A (248) | Spongia sp. | Antimicrobial effects: Staphylococcus aureus = MIC 12.5 μM; Bacillus subtilis = MIC 12.5 μM | [82] |
| Langcoquinone B (249) | Spongia sp. | Antimicrobial effects: Staphylococcus aureus = MIC 12.5 μM; Bacillus subtilis = MIC 12.5 μM | [82] |
| Langconol A (250) | Antimicrobial effects: B. subtilis MIC 12.5 μM | [83] | |
| Langconol C (252) | Antimicrobial effects: B. subtilis = MIC 25.0 μM | [83] | |
| Langcoquinone C (253) | Antimicrobial effects: Staphylococcus aureus = MIC 12.50 μM; Bacillus subtilis = MIC 6.25 μM | [83] | |
| Aminoquinone (257) | Dysidea sp. | Antimicrobial effects: B. subtilis = MIC 50.0 μg/mL; S. aureus = MIC 50.0 μg/mL; E. coli = MIC 50.0 μg/mL | [84] |
| Nakijinol G (267) | Hyrtios sp. | Enzyme Inhibition: PTP1B = IC50 4.8 μM | [87] |
| Dysivillosin A (270) | Dysidea villosa | Enzyme Inhibition: β-hexosaminidase = IC50 8.2 μM | [88] |
| Dysivillosin B (271) | Enzyme Inhibition: β-hexosaminidase = IC50 10.2 μM | [88] | |
| Dysivillosin C (272) | Enzyme Inhibition: β-hexosaminidase = IC50 19.9 μM | [88] | |
| Dysivillosin D (273) | Enzyme Inhibition: β-hexosaminidase = IC50 16.2 μM | [88] | |
| Chartarolide A (274) | Niphates recondite | Cytotoxic effects: HCT-116 = IC50 1.9 μM; HepG2 = IC50 1.8 μM; BGC-823 = IC50 1.3 μM; NCI-H1650 = IC50 5.5 μM; A2780 = IC50 1.5 μM; MCF7 = IC50 1.4 μM | [90] |
| Chartarolide B (275) | Niphates recondite | Cytotoxic effects: HCT-116 = IC50 2.3 μM; HepG2 = IC50 2.8 μM; BGC-823 = IC50 1.6 μM; NCI-H1650 = IC50 4.8 μM; A2780 = IC50 3.2 μM; MCF7 = IC50 3.8 μM | [90] |
| Chartarolide C (276) | Niphates recondite | Cytotoxic effects: HCT-116 = IC50 7.8 μM; HepG2 = IC50 8.9 μM; BGC-823 = IC50 5.4 μM; NCI-H1650 = IC50 11.3 μM; A2780 = IC50 12.5 μM; MCF7 = IC50 8.7 μM | [90] |
| Terretonin N (304) | Nocardiopsis sp. | Antimicrobial effects: S. warneri = IZ 14 mm E. coli = IZ 8 mm | [98] |
| Rubiginosin A (306) | Rhododendron rubiginosum | Cytotoxic effects: A549 = IC50 16.15 μM; HCT116 = IC50 15.56 μM; SK-HEP-1 = IC50 13.80 μM; HL-60 = IC50 12.84 μM | [101] |
| Rubiginosin B (307) | Rhododendron rubiginosum | Cytotoxic effects: HCT116 = IC50 65.72 μM; SK-HEP-1 = IC50 84.66 μM | [101] |
| Rubiginosin C (308) | Rhododendron rubiginosum | Cytotoxic effects: A549 = IC50 40.45 μM; HCT116 = IC50 17.43 μM; SK-HEP-1 = IC50 26.26 μM; HL-60 = IC50 16.44 μM | [101] |
| Rubiginosin D (309) | Rhododendron rubiginosum | Cytotoxic effects: A549 = IC50 49.18 μM; HCT116 = IC50 32.17 μM; SK-HEP-1 = IC50 13.66 μM; HL-60 = IC50 40.07 μM | [101] |
| Rubiginosin E (310) | Rhododendron rubiginosum | Cytotoxic effects: A549 = IC50 38.90 μM; HCT116 = IC50 38.90 μM; SK-HEP-1 = IC50 38.90 μM; HL-60 = IC50 38.90 μM | [101] |
| Rubiginosin F (311) | Rhododendron rubiginosum | Cytotoxic effects: A549 = IC50 38.90 μM; HCT116 = IC50 38.90 μM; SK-HEP-1 = IC50 38.90 μM; HL-60 = IC50 38.90 μM | [101] |
| Rubiginosins G (312) | Rhododendron rubiginosum | Cytotoxic effects: A549 = IC50 38.90 μM; HCT116 = IC50 38.90 μM; SK-HEP-1 = IC50 38.90 μM; HL-60 = IC50 38.90 μM | [101] |
| Anthopogochromene A (313) | Rhododendron rubiginosum | Cytotoxic effects: A549 = IC50 38.90 μM; HCT116 = IC50 38.90 μM; SK-HEP-1 = IC50 38.90 μM; HL-60 = IC50 38.90 μM | [101] |
| Anthopogochromene B (314) | Rhododendron rubiginosum | Cytotoxic effects: A549 = IC50 38.90 μM; HCT116 = IC50 38.90 μM; SK-HEP-1 = IC50 38.90 μM; HL-60 = IC50 38.90 μM | [101] |
| Isopolycerasoidol (315) | Sargassum siliquastrum | Antioxidant effects: DPPH = EC50 8.23 μM; ABTS = EC50 2.33 μM | [103] |
| Sargachromanol D (316) | Sargassum siliquastrum | Antioxidant effects: DPPH = EC50 26.35 μM; ABTS = EC50 4.84 μM | [103] |
| Sargachromanol E (317) | Sargassum siliquastrum | Antioxidant effects: DPPH = EC50 23.84 μM; ABTS = EC50 4.57 μM | [103] |
| Sargachromanol G (318) | Sargassum siliquastrum | Antioxidant effects: DPPH = EC50 33.43 μM; ABTS = EC50 4.05 μM | [103] |
| Sargachromanol I (319) | Sargassum siliquastrum | Antioxidant effects: DPPH = EC50 32.83 μM; ABTS = EC50 6.86 μM | [103] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nazir, M.; Saleem, M.; Tousif, M.I.; Anwar, M.A.; Surup, F.; Ali, I.; Wang, D.; Mamadalieva, N.Z.; Alshammari, E.; Ashour, M.L.; et al. Meroterpenoids: A Comprehensive Update Insight on Structural Diversity and Biology. Biomolecules 2021, 11, 957. https://doi.org/10.3390/biom11070957
Nazir M, Saleem M, Tousif MI, Anwar MA, Surup F, Ali I, Wang D, Mamadalieva NZ, Alshammari E, Ashour ML, et al. Meroterpenoids: A Comprehensive Update Insight on Structural Diversity and Biology. Biomolecules. 2021; 11(7):957. https://doi.org/10.3390/biom11070957
Chicago/Turabian StyleNazir, Mamona, Muhammad Saleem, Muhammad Imran Tousif, Muhammad Aijaz Anwar, Frank Surup, Iftikhar Ali, Daijie Wang, Nilufar Z. Mamadalieva, Elham Alshammari, Mohamed L. Ashour, and et al. 2021. "Meroterpenoids: A Comprehensive Update Insight on Structural Diversity and Biology" Biomolecules 11, no. 7: 957. https://doi.org/10.3390/biom11070957