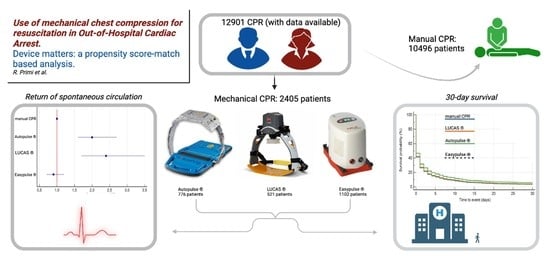
Use of Mechanical Chest Compression for Resuscitation in Out-Of-Hospital Cardiac Arrest—Device Matters: A Propensity-Score-Based Match Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Type of Study and Endpoints
2.2. Study Population and Data Collection
2.3. Territory and EMS Description
- -
- SOREU della Pianura: EMS dispatch center for the provinces of Pavia (PV), Lodi (LO), Cremona (CR), Mantua (MN), and the western part of Milan (MI) province (not yet covered by the Registry); it coordinates 48 ambulances staffed with basic life support and defibrillation (BLS-D)-trained personnel, and 22 advanced life support (ALS)-trained staffed vehicles (a physician and a specialized nurse or a specialized nurse only).
- -
- SOREU dei Laghi: EMS dispatch center for the provinces of Como (CO), Lecco (LC), Monza (MB) (not yet covered by Lombardia CARe), and Varese (VA); it coordinates 56 ambulances staffed with basic life support and defibrillation (BLS-D)-trained personnel and 21 advanced life support (ALS)-trained staffed vehicles (a physician and a specialized nurse or a specialized nurse only).
- -
- SOREU delle Alpi: EMS dispatch center for the provinces of Brescia (BS), Bergamo (BG), and Sondrio (SO) (BG and SOare not yet covered by Lombardia CARe); it coordinates 91 ambulances staffed with basic life support and defibrillation (BLS-D)-trained personnel and 41 advanced life support (ALS)-trained staffed vehicles (a physician and a specialized nurse or a specialized nurse only).
- -
- SOREU Metropolitana: EMS dispatch center for the city of Milan and the eastern part of its province (not involved in the present study).
2.4. Data Management and Statistical Analysis
3. Results
3.1. Study Population
3.2. Primary Endpoint: Use of Mechanical CPR and ROSC
3.3. Secondary Endpoint: Use of Mechanical CPR and 30-Day Survival
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nishiyama, C.; Kiguchi, T.; Okubo, M.; Alihodžić, H.; Al-Araji, R.; Baldi, E.; Beganton, F.; Booth, S.; Bray, J.; Christensen, E.; et al. Three-year trends in out-of-hospital cardiac arrest across the world: Second report from the International Liaison Committee on Resuscitation (ILCOR). Resuscitation 2023, 186, 109757. [Google Scholar] [CrossRef] [PubMed]
- Czapla, M.; Zielińska, M.; Kubica-Cielińska, A.; Diakowska, D.; Quinn, T.; Karniej, P. Factors associated with return of spontaneous circulation after out-of-hospital cardiac arrest in Poland: A one-year retrospective study. BMC Cardiovasc. Disord. 2020, 20, 288. [Google Scholar] [CrossRef] [PubMed]
- Cummins, R.O.; Ornato, J.P.; Thies, W.H.; Pepe, P.E. Improving Survival From Sudden Cardiac Arrest: The “Chain of Survival” Concept. A statement for health professionals from the Advanced Cardiac Life Support Subcommittee and the Emergency Cardiac Care Committee, American Heart Association. Circulation 1991, 83, 1832–1847. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harris, A.W.; Kudenchuk, P.J. Cardiopulmonary resuscitation: The science behind the hands. Heart 2018, 104, 1056–1061. [Google Scholar] [CrossRef]
- Higano, S.T.; Oh, J.K.; Ewy, G.A.; Seward, J.B. The Mechanism of Blood Flow During Closed Chest Cardiac Massage in Humans: Transesophageal Echocardiography Observations. Mayo Clin. Proc. 1990, 65, 1432–1440. [Google Scholar] [CrossRef]
- Cipani, S.; Bartolozzi, C.; Ballo, P.; Sarti, A. Blood flow maintenance by cardiac massage during cardiopulmonary resuscitation: Classical theories, newer hypotheses, and clinical utility of mechanical devices. J. Intensiv. Care Soc. 2019, 20, 2–10. [Google Scholar] [CrossRef] [Green Version]
- Anantharaman, V.; Ng, B.; Ang, S.; Lee, C.; Leong, S.; Ong, M.; Chua, S.; Rabind, A.; Anjali, N.; Hao, Y. Prompt use of mechanical cardiopulmonary resuscitation in out-of-hospital cardiac arrest: The MECCA study report. Singap. Med. J. 2017, 58, 424–431. [Google Scholar] [CrossRef] [Green Version]
- Gao, C.; Chen, Y.; Peng, H.; Chen, Y.; Zhuang, Y.; Zhou, S. Clinical evaluation of the AutoPulse automated chest compression device for out-of-hospital cardiac arrest in the northern district of Shanghai, China. Arch. Med. Sci. 2016, 3, 563–570. [Google Scholar] [CrossRef]
- Hallstrom, A.; Rea, T.D.; Sayre, M.; Christenson, J.; Anton, A.R.; Mosesso, V.N.; Van Ottingham, L.; Olsufka, M.; Pennington, S.; White, L.; et al. Manual Chest Compression vs Use of an Automated Chest Compression Device During Resuscitation Following Out-of-Hospital Cardiac Arrest. JAMA 2006, 295, 2620–2628. [Google Scholar] [CrossRef] [Green Version]
- Perkins, G.D.; Lall, R.; Quinn, T.; Deakin, C.D.; Cooke, M.W.; Horton, J.; Lamb, S.E.; Slowther, A.-M.; Woollard, M.; Carson, A.; et al. Mechanical versus manual chest compression for out-of-hospital cardiac arrest (PARAMEDIC): A pragmatic, cluster randomised controlled trial. Lancet 2015, 385, 947–955. [Google Scholar] [CrossRef] [Green Version]
- Rubertsson, S.; Lindgren, E.; Smekal, D.; Östlund, O.; Silfverstolpe, J.; Lichtveld, R.; Boomars, R.; Ahlstedt, B.; Skoog, G.; Kastberg, R.; et al. Mechanical chest compressions and simultaneous defibrillation vs. conventional cardiopulmonary resuscitation in out-of-hospital cardiac arrest. JAMA 2014, 311, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Wik, L.; Olsen, J.-A.; Persse, D.; Sterz, F.; Lozano, M.; Brouwer, M.; Westfall, M.; Souders, C.M.; Malzer, R.; van Grunsven, P.M.; et al. Manual vs. integrated automatic load-distributing band CPR with equal survival after out of hospital cardiac arrest. The randomized CIRC trial. Resuscitation 2014, 85, 741–748. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Ahn, C.; Kim, I.-Y.; Choi, H.-Y.; Kim, J.-G.; Kim, J.; Shin, H.; Moon, S.; Lee, J.; Lee, J.; et al. Prognostic Impact of In-Hospital Use of Mechanical Cardiopulmonary Resuscitation Devices Compared with Manual Cardiopulmonary Resuscitation: A Nationwide Population-Based Observational Study in South Korea. Medicina 2022, 58, 353. [Google Scholar] [CrossRef]
- Seewald, S.; Obermaier, M.; Lefering, R.; Bohn, A.; Georgieff, M.; Muth, C.-M.; Gräsner, J.-T.; Masterson, S.; Scholz, J.; Wnent, J. Application of mechanical cardiopulmonary resuscitation devices and their value in out-of-hospital cardiac arrest: A retrospective analysis of the German Resuscitation Registry. PLoS ONE 2019, 14, e0208113. [Google Scholar] [CrossRef] [PubMed]
- Ong, M.E.H.; Ornato, J.P.; Edwards, D.P.; Dhindsa, H.S.; Best, A.; Ines, C.S.; Hickey, S.; Clark, B.; Williams, D.C.; Powell, R.G.; et al. Use of an automated, load-distributing band chest compression device for out-of-hospital cardiac arrest resuscitation. JAMA 2006, 295, 2629–2637. [Google Scholar] [CrossRef] [Green Version]
- Axelsson, C.; Nestin, J.; Svensson, L.; Axelsson, Å.B.; Herlitz, J. Clinical consequences of the introduction of mechanical chest compression in the EMS system for treatment of out-of-hospital cardiac arrest—A pilot study. Resuscitation 2006, 71, 47–55. [Google Scholar] [CrossRef]
- Casner, M.; Andersen, D.; Isaacs, S.M. The impact of a new cpr assist device on rate of return of spontaneous circulation in out-of-hospital cardiac arrest. Prehospital Emerg. Care 2005, 9, 61–67. [Google Scholar] [CrossRef]
- Jennings, P.A.; Harriss, L.; Bernard, S.; Bray, J.; Walker, T.; Spelman, T.; Smith, K.; Cameron, P. An automated CPR device compared with standard chest compressions for out-of-hospital resuscitation. BMC Emerg. Med. 2012, 12, 8. [Google Scholar] [CrossRef] [Green Version]
- Mistraletti, G.; Lancioni, A.; Bassi, G.; Nespoli, F.; Umbrello, M.; Salini, S.; Zangrillo, A.; Pappalardo, F.; Scandroglio, A.M.; Foti, G.; et al. Mechanical chest compression and extracorporeal life support for out-of-hospital cardiac arrest. A 30-month observational study in the metropolitan area of Milan, Italy. Resuscitation 2023, 182, 109659. [Google Scholar] [CrossRef]
- Zeiner, S.; Sulzgruber, P.; Datler, P.; Keferböck, M.; Poppe, M.; Lobmeyr, E.; van Tulder, R.; Zajicek, A.; Buchinger, A.; Polz, K.; et al. Mechanical chest compression does not seem to improve outcome after out-of hospital cardiac arrest. A single center observational trial. Resuscitation 2015, 96, 220–225. [Google Scholar] [CrossRef]
- Jung, E.; Park, J.H.; Lee, S.Y.; Ro, Y.S.; Hong, K.J.; Song, K.J.; Ryu, H.H.; Shin, S.D. Mechanical Chest Compression Device for Out-Of-Hospital Cardiac Arrest: A Nationwide Observational Study. J. Emerg. Med. 2020, 58, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Ujvárosy, D.; Sebestyén, V.; Pataki, T.; Ötvös, T.; Lőrincz, I.; Paragh, G.; Szabó, Z. Cardiovascular risk factors differently affect the survival of patients undergoing manual or mechanical resuscitation. BMC Cardiovasc. Disord. 2018, 18, 227. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.T.; Kim, J.G.; Jang, Y.S.; Kang, G.H.; Kim, W.; Choi, H.Y.; Jun, G.S. Comparison of in-hospital use of mechanical chest compression devices for out-of-hospital cardiac arrest patients. Medicine 2019, 98, e17881. [Google Scholar] [CrossRef] [PubMed]
- Schmidbauer, S.; Herlitz, J.; Karlsson, T.; Axelsson, C.; Friberg, H. Use of automated chest compression devices after out-of-hospital cardiac arrest in Sweden. Resuscitation 2017, 120, 95–102. [Google Scholar] [CrossRef]
- Savastano, S.; Baldi, E.; Palo, A.; Raimondi, M.; Belliato, M.; Compagnoni, S.; Buratti, S.; Cacciatore, E.; Canevari, F.; Iotti, G.; et al. Load distributing band device for mechanical chest compressions: An Utstein-categories based analysis of survival to hospital discharge. Int. J. Cardiol. 2019, 287, 81–85. [Google Scholar] [CrossRef]
- Tantarattanapong, S.; Chantaramanee, K. Comparison of Sustained Return of Spontaneous Circulation Rate Between Manual and Mechanical Chest Compression in Adult Cardiac Arrest. Open Access Emerg. Med. 2022, 14, 599–608. [Google Scholar] [CrossRef]
- Newberry, R.; Redman, T.; Ross, E.; Ely, R.; Saidler, C.; Arana, A.; Wampler, D.; Miramontes, D. No Benefit in Neurologic Outcomes of Survivors of Out-of-Hospital Cardiac Arrest with Mechanical Compression Device. Prehospital Emerg. Care 2018, 22, 338–344. [Google Scholar] [CrossRef]
- Satterlee, P.A.; Boland, L.L.; Johnson, P.J.; Hagstrom, S.G.; Page, D.I.; Lick, C.J. Implementation of a Mechanical Chest Compression Device as Standard Equipment in a Large Metropolitan Ambulance Service. J. Emerg. Med. 2013, 45, 562–569. [Google Scholar] [CrossRef]
- Axelsson, C.; Herrera, M.J.; Fredriksson, M.; Lindqvist, J.; Herlitz, J. Implementation of mechanical chest compression in out-of-hospital cardiac arrest in an emergency medical service system. Am. J. Emerg. Med. 2013, 31, 1196–1200. [Google Scholar] [CrossRef]
- Chen, Y.-R.; Liao, C.-J.; Huang, H.-C.; Tsai, C.-H.; Su, Y.-S.; Liu, C.-H.; Hsu, C.-F.; Tsai, M.-J. The Effect of implementing mechanical cardiopulmonary resuscitation devices on out-of-hospital cardiac arrest patients in an urban city of Taiwan. Int. J. Environ. Res. Public Health 2021, 18, 3636. [Google Scholar] [CrossRef]
- Alexander, E.; Katharina, T.; Verena, F.; Jürgen, G.; Maximilian, N.; Calvin, K.; Andreas, S.; Wolfgang, S.; Harald, H.; Dominik, R. Comparison of different mechanical chest compression devices in the alpine rescue setting: A randomized triple crossover experiment. Scand. J. Trauma Resusc. Emerg. Med. 2021, 29, 84. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.H.; Min, M.K.; Lee, D.S.; Lee, M.J.; Chun, M.S.; Hyun, T.; Shon, S.W. Comparison of blood flow between two mechanical compression devices using ultrasound: Animal trial. Am. J. Emerg. Med. 2022, 60, 116–120. [Google Scholar] [CrossRef] [PubMed]
- Brooks, S.C.; Bigham, B.L.; Morrison, L.J. Mechanical chest compressions versus manual chest compressions for cardiac arrest. Emergencias 2020, 32, 365–366. [Google Scholar] [CrossRef]
- Sheraton, M.; Columbus, J.; Surani, S.; Chopra, R.; Kashyap, R. Effectiveness of mechanical chest compression devices over manual cardiopulmonary resuscitation: A systematic review with meta-analysis and trial sequential analysis. West. J. Emerg. Med. 2021, 22, 810–819. [Google Scholar] [CrossRef] [PubMed]
- Ni Zhu, N.; Chen, Q.; Jiang, Z.; Liao, F.; Kou, B.; Tang, H.; Zhou, M. A meta-analysis of the resuscitative effects of mechanical and manual chest compression in out-of-hospital cardiac arrest patients. Crit. Care 2019, 23, 100. [Google Scholar] [CrossRef] [Green Version]
- Liu, M.; Shuai, Z.; Ai, J.; Tang, K.; Liu, H.; Zheng, J.; Gou, J.; Lv, Z. Mechanical chest compression with LUCAS device does not improve clinical outcome in out-of-hospital cardiac arrest patients: A systematic review and meta-analysis. Medicine 2019, 98, e17550. [Google Scholar] [CrossRef]
- Wang, P.; Brooks, S. Cochrane corner: Are mechanical compressions better than manual compressions in cardiac arrest? Heart 2020, 106, 559–561. [Google Scholar] [CrossRef]
- Ong, M.E.; Mackey, K.E.; Zhang, Z.C.; Tanaka, H.; Ma, M.H.; Swor, R.; Shin, S.D. Mechanical CPR devices compared to manual CPR during out-of-hospital cardiac arrest and ambulance transport: A systematic review. Scand. J. Trauma Resusc. Emerg. Med. 2012, 20, 39. [Google Scholar] [CrossRef] [Green Version]
- Khan, S.U.; Lone, A.N.; Talluri, S.; Khan, M.Z.; Kaluski, E. Efficacy and safety of mechanical versus manual compression in cardiac arrest—A Bayesian network meta-analysis. Resuscitation 2018, 130, 182–188. [Google Scholar] [CrossRef]
- Li, H.; Wang, D.; Yu, Y.; Zhao, X.; Jing, X. Mechanical versus manual chest compressions for cardiac arrest: A systematic review and meta-analysis. Scand. J. Trauma Resusc. Emerg. Med. 2016, 24, 10. [Google Scholar] [CrossRef] [Green Version]
- Couper, K.; Yeung, J.; Nicholson, T.; Quinn, T.; Lall, R.; Perkins, G.D. Mechanical chest compression devices at in-hospital cardiac arrest: A systematic review and meta-analysis. Resuscitation 2016, 103, 24–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bonnes, J.L.; Brouwer, M.; Navarese, E.; Verhaert, D.; Verheugt, F.W.; Smeets, J.L.; de Boer, M.-J. Manual Cardiopulmonary Resuscitation Versus CPR Including a Mechanical Chest Compression Device in Out-of-Hospital Cardiac Arrest: A Comprehensive Meta-analysis From Randomized and Observational Studies. Ann. Emerg. Med. 2016, 67, 349–360.e3. [Google Scholar] [CrossRef] [PubMed]
- Gates, S.; Quinn, T.; Deakin, C.D.; Blair, L.; Couper, K.; Perkins, G.D. Mechanical chest compression for out of hospital cardiac arrest: Systematic review and meta-analysis. Resuscitation 2015, 94, 91–97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiang, C.-Y.; Lim, K.-C.; Lai, P.C.; Tsai, T.-Y.; Huang, Y.T.; Tsai, M.-J. Comparison between Prehospital Mechanical Cardiopulmonary Resuscitation (CPR) Devices and Manual CPR for Out-of-Hospital Cardiac Arrest: A Systematic Review, Meta-Analysis, and Trial Sequential Analysis. J. Clin. Med. 2022, 11, 1448. [Google Scholar] [CrossRef]
- Soar, J.; Böttiger, B.W.; Carli, P.; Couper, K.; Deakin, C.D.; Djärv, T.; Lott, C.; Olasveengen, T.; Paal, P.; Pellis, T.; et al. European Resuscitation Council Guidelines 2021: Adult advanced life support. Resuscitation 2021, 161, 115–151. [Google Scholar] [CrossRef]
- Panchal, A.R.; Bartos, J.A.; Cabañas, J.G.; Donnino, M.W.; Drennan, I.R.; Hirsch, K.G.; Kudenchuk, P.J.; Kurz, M.C.; Lavonas, E.J.; Morley, P.T.; et al. Part 3: Adult Basic and Advanced Life Support: 2020 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2020, 142, S366–S468. [Google Scholar] [CrossRef] [PubMed]
- Perkins, G.D.; Jacobs, I.G.; Nadkarni, V.M.; Berg, R.A.; Bhanji, F.; Biarent, D.; Bossaert, L.L.; Brett, S.J.; Chamberlain, D.; de Caen, A.R.; et al. Cardiac Arrest and Cardiopulmonary Resuscitation Outcome Reports: Update of the Utstein Resuscitation Registry Templates for Out-of-Hospital Cardiac Arrest. Resuscitation 2015, 96, 328–340. [Google Scholar] [CrossRef] [Green Version]
- Harris, P.A.; Taylor, R.; Minor, B.L.; Elliott, V.; Fernandez, M.; O’Neal, L.; McLeod, L.; Delacqua, G.; Delacqua, F.; Kirby, J.; et al. The REDCap consortium: Building an international community of software platform partners. J. Biomed. Inform. 2019, 95, 103208. [Google Scholar] [CrossRef]
- Baldi, E.; Caputo, M.L.; Savastano, S.; Burkart, R.; Klersy, C.; Benvenuti, C.; Sgromo, V.; Palo, A.; Cianella, R.; Cacciatore, E.; et al. An Utstein-based model score to predict survival to hospital admission: The UB-ROSC score. Int. J. Cardiol. 2020, 308, 84–89. [Google Scholar] [CrossRef] [Green Version]
- Ogawa, Y.; Shiozaki, T.; Hirose, T.; Ohnishi, M.; Nakamori, Y.; Ogura, H.; Shimazu, T. Load-distributing-band cardiopulmonary resuscitation for out-of-hospital cardiac arrest increases regional cerebral oxygenation: A single-center prospective pilot study. Scand. J. Trauma Resusc. Emerg. Med. 2015, 23, 99. [Google Scholar] [CrossRef] [Green Version]
- Duchateau, F.-X.; Gueye, P.; Curac, S.; Tubach, F.; Broche, C.; Plaisance, P.; Payen, D.; Mantz, J.; Ricard-Hibon, A. Effect of the AutoPulse™ automated band chest compression device on hemodynamics in out-of-hospital cardiac arrest resuscitation. Intensiv. Care Med. 2010, 36, 1256–1260. [Google Scholar] [CrossRef] [Green Version]
- Colombo, R.; Fossali, T.; Ottolina, D.; Borghi, B.; Bergomi, P.; Ballone, E.; Rech, R.; Castelli, A.; Catena, E. Kinetics of manual and automated mechanical chest compressions. Resuscitation 2019, 145, 70–74. [Google Scholar] [CrossRef] [PubMed]
- Giorgetti, R.; Chiricolo, G.; Melniker, L.; Calaf, C.; Gaeta, T. RESCUE transesophageal echocardiography for monitoring of mechanical chest compressions and guidance for extracorporeal cardiopulmonary resuscitation cannulation in refractory cardiac arrest. J. Clin. Ultrasound 2020, 48, 184–187. [Google Scholar] [CrossRef] [PubMed]
- LA Via, L.; Astuto, M.; Bignami, E.G.; Busalacchi, D.; Dezio, V.; Girardis, M.; Lanzafame, B.; Ristagno, G.; Pelosi, P.; Sanfilippo, F. The effects of exposure to severe hyperoxemia on neurological outcome and mortality after cardiac arrest. Minerva Anestesiol. 2022, 88, 853–863. [Google Scholar] [CrossRef] [PubMed]
- Bernard, S.A.; Bray, J.E.; Smith, K.; Stephenson, M.; Finn, J.; Grantham, H.; Hein, C.; Masters, S.; Stub, D.; Perkins, G.D.; et al. Effect of Lower vs Higher Oxygen Saturation Targets on Survival to Hospital Discharge Among Patients Resuscitated After Out-of-Hospital Cardiac Arrest: The EXACT Randomized Clinical Trial. JAMA 2022, 328, 1818–1826. [Google Scholar] [CrossRef] [PubMed]
- Robba, C.; Badenes, R.; Battaglini, D.; Ball, L.; Sanfilippo, F.; Brunetti, I.; Jakobsen, J.C.; Lilja, G.; Friberg, H.; Wendel-Garcia, P.D.; et al. Oxygen targets and 6-month outcome after out of hospital cardiac arrest: A pre-planned sub-analysis of the targeted hypothermia versus targeted normothermia after Out-of-Hospital Cardiac Arrest (TTM2) trial. Crit. Care 2022, 26, 323. [Google Scholar] [CrossRef]



| Variable | Manual CPR | Mechanical CPR | General Population | p-Value |
|---|---|---|---|---|
| n = 10,496 | n = 2405 | n = 12,901 | ||
| Male gender, n (%) | 5960 (56.8%) | 1745 (72.6%) | 7705 (59.7%) | <0.001 |
| Age, years (IQR) | 80.0 (69.0–87.0) | 66.0 (55.0–76.0) | 78.0 (65.0–86.0) | <0.001 |
| EMS arrival time, min (IQR) | 13.0 (10.0–16.0) | 12.0 (10.0–15.6) | 12.8 (10.0–16.0) | 0.002 |
| Medical etiology, n (%) | 9735 (92.7%) | 2213 (92.0%) | 11,948 (92.6%) | 0.22 |
| Home Location, n (%) | 8792 (83.8%) | 1809 (75.2%) | 10,601 (82.2%) | <0.001 |
| Witnessed status, n (%) | <0.001 | |||
| Yes—bystander | 5095 (48.5%) | 1498 (62.3%) | 6593 (51.1%) | |
| No | 3750 (35.7%) | 604 (25.1%) | 4354 (33.7%) | |
| Yes—EMS | 1371 (13.1%) | 268 (11.1%) | 1639 (12.7%) | |
| Unknown | 280 (2.7%) | 35 (1.5%) | 315 (2.4%) | |
| Bystander CPR, n (%) * | 3413 (37.4%) | 1283 (60.0%) | 4696 (41.7%) | <0.001 |
| Shockable-presenting rhythm, n (%) | 1176 (11.2%) | 724 (30.1%) | 1900 (14.7%) | <0.001 |
| PAD Shock, n (%) ** | 76 (25.3%) | 42 (40.4%) | 123 (29.5%) | <0.001 |
| Resuscitation duration, min (IQR) | 25.0 (15.0–37.2) | 45.0 (32.1–61.5) | 28.0 (16.7–42.0) | <0.001 |
| Variable | Multivariable Logistic Regression Analysis for Mechanical Chest Compressor Use | |
|---|---|---|
| Coefficient | p-Value | |
| Male gender | 0.35 | <0.0001 |
| Province | ||
| Brescia | 0 | ref |
| Como | −0.28 | 00036 |
| Cremona | −1.33 | <0.0001 |
| Lodi | −1.77 | <0.0001 |
| Mantova | −0.54 | <0.0001 |
| Pavia | −0.93 | <0.0001 |
| Varese | −1.31 | <0.0001 |
| Age (years) | −0.04 | <0.0001 |
| EMS arrival time (min) | −0.01 | 0.024 |
| Home Location | −0.19 | 0.003 |
| Witnessed event and bystanders CPR (BCPR) | ||
| No Witnessed/No BCPR | 0 | ref |
| No Witnessed/Yes BCPR | 0.44 | <0.0001 |
| EMS witnessed | 0.38 | 0.0001 |
| Bystander witnessed/No BCPR | 0.47 | <0.0001 |
| Bystander witnessed/Yes BCPR | 0.94 | <0.0001 |
| Shockable-presenting rhythm | 0.75 | <0.0001 |
| PAD Shock | −0.54 | 0.04 |
| Medical Etiology | 0.85 | <0.001 |
| Propensity score performance | AUC = 0.79; 95%CI (0.78–0.79), p < 0.0001 | |
| Variable | Manual CPR | Mechanical CPR | p-Value | Autopulse® | LUCAS® | EasyPulse® | p-Value |
|---|---|---|---|---|---|---|---|
| n = 2146 | n = 2146 | n = 716 | n = 425 | n = 1001 | |||
| Male gender, n (%) | 1584 (74) | 1533 (71) | 0.08 | 560 (78) | 305 (72) | 665 (66) | <0.001 |
| Age, years (IQR) | 69 (56–79) | 67 (57–77) | 0.04 | 64 (54–72) | 69 (58–78) | 70 (59–80) | <0.001 |
| EMS arrival time, min (IQR) | 12 (9.6–15) | 12.2 (10–16) | 0.17 | 11.7 (8.4–15) | 12 (10–15) | 13 (10–16) | <0.001 |
| Medical etiology, n (%) | 1962 (91.4) | 1981 (92.3) | 0.29 | 665 (93) | 382 (90) | 930 (93) | 0.11 |
| Home Location, n (%) | 1661 (77.4) | 1644 (76.6) | 0.54 | 528 (74) | 320 (75) | 793 (79) | 0.02 |
| Witnessed status, n (%) | 0.26 | <0.001 | |||||
| Yes—bystander | 1325 (62) | 1322 (61) | 459 (64) | 250 (59) | 612 (61) | ||
| No | 537 (25) | 570 (27) | 140 (20) | 135 (32) | 293 (29) | ||
| Yes—EMS | 284 (13) | 254 (12) | 117 (16) | 40 (9) | 96 (10) | ||
| Bystander CPR, n (%) * | 1087 (58) | 1107 (58) | 0.93 | 364 (61) | 209 (54) | 531 (59) | 0.53 |
| Shockable-presenting rhythm, n (%) | 566 (26) | 585 (27) | 0.51 | 236 (39) | 91 (24) | 213 (24) | <0.001 |
| PAD Shock, n (%) ** | 32 (33) | 34 (40) | 0.33 | 14 (56) | 5 (28) | 15 (36) | 0.14 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Primi, R.; Bendotti, S.; Currao, A.; Sechi, G.M.; Marconi, G.; Pamploni, G.; Panni, G.; Sgotti, D.; Zorzi, E.; Cazzaniga, M.; et al. Use of Mechanical Chest Compression for Resuscitation in Out-Of-Hospital Cardiac Arrest—Device Matters: A Propensity-Score-Based Match Analysis. J. Clin. Med. 2023, 12, 4429. https://doi.org/10.3390/jcm12134429
Primi R, Bendotti S, Currao A, Sechi GM, Marconi G, Pamploni G, Panni G, Sgotti D, Zorzi E, Cazzaniga M, et al. Use of Mechanical Chest Compression for Resuscitation in Out-Of-Hospital Cardiac Arrest—Device Matters: A Propensity-Score-Based Match Analysis. Journal of Clinical Medicine. 2023; 12(13):4429. https://doi.org/10.3390/jcm12134429
Chicago/Turabian StylePrimi, Roberto, Sara Bendotti, Alessia Currao, Giuseppe Maria Sechi, Gianluca Marconi, Greta Pamploni, Gianluca Panni, Davide Sgotti, Ettore Zorzi, Marco Cazzaniga, and et al. 2023. "Use of Mechanical Chest Compression for Resuscitation in Out-Of-Hospital Cardiac Arrest—Device Matters: A Propensity-Score-Based Match Analysis" Journal of Clinical Medicine 12, no. 13: 4429. https://doi.org/10.3390/jcm12134429