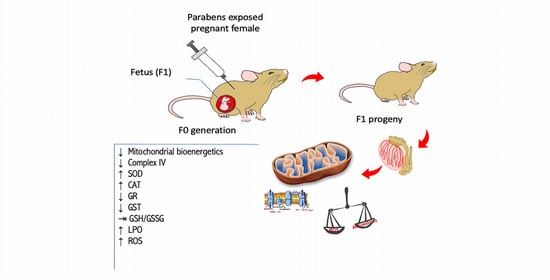
Use of Parabens (Methyl and Butyl) during the Gestation Period: Mitochondrial Bioenergetics of the Testes and Antioxidant Capacity Alterations in Testes and Other Vital Organs of the F1 Generation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemical Reagents
2.2. Animals and Treatments
2.3. Testis Mitochondria Isolation
2.4. Mitochondria Respiration
2.5. Mitochondrial Membrane Potential (ΔΨ)
2.6. Determination of Mitochondrial Enzyme Activity
2.7. Mitochondrial ROS Production
2.8. Determination of Antioxidant Enzymes
2.9. Statistical Analysis
3. Results
3.1. Animal Data
3.2. Respiration and Oxidative Phosphorylation of Isolated Mitochondria
3.3. Oxidative Stress
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Routledge, E.J.; Sumpter, J.P. Structural features of alkylphenolic chemicals associated with estrogenic activity. J. Biol. Chem. 1997, 272, 3280–3288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Routledge, E.J.; Parker, J.; Odum, J.; Ashby, J.; Sumpter, J.P. Some alkyl hydroxy benzoate preservatives (parabens) are estrogenic. Toxicol. Appl. Pharm. 1998, 153, 12–19. [Google Scholar] [CrossRef]
- Byford, J.R.; Shaw, L.E.; Drew, M.G.; Pope, G.S.; Sauer, M.J.; Darbre, P.D. Oestrogenic activity of parabens in MCF7 human breast cancer cells. J. Steroid Biochem. Mol. Biol. 2002, 80, 49–60. [Google Scholar] [CrossRef]
- Xue, X.H.; Xue, J.C.; Liu, W.B.; Adams, D.H.; Kannan, K. Trophic Magnification of Parabens and Their Metabolites in a Subtropical Marine Food Web. Environ. Sci. Technol. 2017, 51, 780–789. [Google Scholar] [CrossRef]
- Wen, Q.P.; Zhou, Y.Q.; Wang, Y.J.; Li, J.F.; Zhao, H.Z.; Liao, J.Q.; Liu, H.X.; Li, Y.Y.; Cai, Z.W.; Xia, W. Association between urinary paraben concentrations and gestational weight gain during pregnancy. J. Expo. Sci. Environ. Epidemiol. 2020, 30, 845–855. [Google Scholar] [CrossRef]
- Oishi, S. Effects of butylparaben on the male reproductive system in rats. Toxicol. Ind. Health 2001, 17, 31–39. [Google Scholar] [CrossRef]
- Oishi, S. Effects of butyl paraben on the male reproductive system in mice. Arch. Toxicol. 2002, 76, 423–429. [Google Scholar] [CrossRef]
- Kang, K.S.; Che, J.H.; Ryu, D.Y.; Kim, T.W.; Li, G.X.; Lee, Y.S. Decreased sperm number and motile activity on the F1 offspring maternally exposed to butyl p-hydroxybenzoic acid (butyl paraben). J. Vet. Med. Sci 2002, 64, 227–235. [Google Scholar] [CrossRef] [Green Version]
- Maske, P.; Dighe, V.; Vanage, G. n-butylparaben exposure during perinatal period impairs fertility of the F1 generation female rats. Chemosphere 2018, 213, 114–123. [Google Scholar] [CrossRef]
- Shah, K.H.; Verma, R.J. Butyl p-hydroxybenzoic acid induces oxidative stress in mice liver—An in vivo study. Acta Pol. Pharm. 2011, 68, 875–879. [Google Scholar]
- Nishizawa, C.; Takeshita, K.; Ueda, J.; Nakanishi, I.; Suzuki, K.T.; Ozawa, T. Reaction of para-hydroxybenzoic acid esters with singlet oxygen in the presence of glutathione produces glutathione conjugates of hydroquinone, potent inducers of oxidative stress. Free Radic. Res. 2006, 40, 233–240. [Google Scholar] [CrossRef]
- Tavares, R.S.; Martins, F.C.; Oliveira, P.J.; Ramalho-Santos, J.; Peixoto, F.P. Parabens in male infertility-Is there a mitochondrial connection? Reprod. Toxicol. 2009, 27, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Handa, O.; Kokura, S.; Adachi, S.; Takagi, T.; Naito, Y.; Tanigawa, T.; Yoshida, N.; Yoshikawa, T. Methylparaben potentiates UV-induced damage of skin keratinocytes. Toxicology 2006, 227, 62–72. [Google Scholar] [CrossRef]
- Lakeram, M.; Lockley, D.J.; Sanders, D.J.; Pendlington, R.; Forbes, B. Paraben transport and metabolism in the biomimetic artificial membrane permeability assay (BAMPA) and 3-day and 21-day Caco-2 cell systems. J. Biomol. Screen 2007, 12, 84–91. [Google Scholar] [CrossRef] [Green Version]
- Samarasinghe, S.; Krishnan, K.; Naidu, R.; Megharaj, M.; Miller, K.; Fraser, B.; Aitken, R.J. Parabens generate reactive oxygen species in human spermatozoa. Andrology 2018, 6, 532–541. [Google Scholar] [CrossRef] [Green Version]
- Martins, F.; Videira, R.A.; Oliveira, M.M.; Silva-Maia, D.; Ferreira, F.M.; Peixoto, F.P. Parabens enhance the calcium-dependent testicular mitochondrial permeability transition: Their relevance on the reproductive capacity in male animals. J. Biochem. Mol. Toxicol. 2020, e22661. [Google Scholar] [CrossRef]
- Soni, M.G.; Carabin, I.G.; Burdock, G.A. Safety assessment of esters of p-hydroxybenzoic acid (parabens). Food Chem. Toxicol. 2005, 43, 985–1015. [Google Scholar] [CrossRef]
- Martins, F.; Margarida, A.; Oliveira, M.M.; Oliveira, P.J.; Peixoto, F.P. Toxicity of parabens in testis mitochondria; a possible role on male infertility. BBA Bioenerg. 2008, 1777, S60–S61. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.Y.; Dong, L.; Ding, S.J.; Qiao, P.H.; Wang, C.; Zhang, M.; Zhang, L.X.; Du, Q.C.; Li, Y.M.; Tang, N.; et al. Effects of n-butylparaben on steroidogenesis and spermatogenesis through changed E-2 levels in male rat offspring. Environ. Toxicol. Pharm. 2014, 37, 705–717. [Google Scholar] [CrossRef]
- Aubert, N.; Ameller, T.; Legrand, J.J. Systemic exposure to parabens: Pharmacokinetics, tissue distribution, excretion balance and plasma metabolites of C-14 -methyl-, propyl- and butylparaben in rats after oral, topical or subcutaneous administration. Food Chem. Toxicol. 2012, 50, 445–454. [Google Scholar] [CrossRef]
- Vo, T.T.B.; Yoo, Y.M.; Choi, K.C.; Jeung, E.B. Potential estrogenic effect(s) of parabens at the prepubertal stage of a postnatal female rat model. Reprod. Toxicol. 2010, 29, 306–316. [Google Scholar] [CrossRef] [PubMed]
- Gornall, A.G.; Bardawill, C.J.; David, M.M. Determination of Serum Proteins by Means of the Biuret Reaction. J. Biol. Chem. 1949, 177, 751–766. [Google Scholar] [PubMed]
- Monteiro-Cardoso, V.F.; Silva, A.M.; Oliveira, M.M.; Peixoto, F.; Videira, R.A. Membrane lipid profile alterations are associated with the metabolic adaptation of the Caco-2 cells to aglycemic nutritional condition. J. Bioenerg. Biomembr. 2014, 46, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Akerman, K.E.; Wikstrom, M.K. Safranine as a probe of the mitochondrial membrane potential. Febs Lett. 1976, 68, 191–197. [Google Scholar] [CrossRef] [Green Version]
- Cassina, A.; Radi, R. Differential inhibitory action of nitric oxide and peroxynitrite on mitochondrial electron transport. Arch. Biochem. Biophys. 1996, 328, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Monteiro-Cardoso, V.F.; Oliveira, M.M.; Melo, T.; Domingues, M.R.M.; Moreira, P.I.; Ferreiro, E.; Peixoto, F.; Videira, R.A. Cardiolipin Profile Changes are Associated to the Early Synaptic Mitochondrial Dysfunction in Alzheimer’s Disease. J. Alzheimers Dis. 2015, 43, 1375–1392. [Google Scholar] [CrossRef] [Green Version]
- Paya, M.; Halliwell, B.; Hoult, J.R.S. Interactions of a Series of Coumarins with Reactive Oxygen Species—Scavenging of Superoxide, Hypochlorous Acid and Hydroxyl Radicals. Biochem. Pharm. 1992, 44, 205–214. [Google Scholar] [CrossRef]
- Del Rio, L.A.; Ortega, M.G.; Lopez, A.L.; Gorge, J.L. A more sensitive modification of the catalase assay with the Clark oxygen electrode. Application to the kinetic study of the pea leaf enzyme. Anal. Biochem. 1977, 80, 409–415. [Google Scholar] [CrossRef]
- Carlberg, I.; Mannervik, B. Purification and Characterization of Flavoenzyme Glutathione Reductase from Rat-Liver. J. Biol. Chem. 1975, 250, 5475–5480. [Google Scholar]
- Hatton, P.J.; Dixon, D.; Cole, D.J.; Edwards, R. Glutathione transferase activities and herbicide selectivity in maize and associated weed species. Pestic. Sci. 1996, 46, 267–275. [Google Scholar] [CrossRef]
- Hair, J.F.; Black, W.C.; Babin, B.J.; Anderson, R.E. Multivariate Data Analysis; Printice-Hall: Upper Saddle River, NJ, USA, 2014. [Google Scholar]
- Pollock, T.; Weaver, R.E.; Ghasemi, R.; de Catanzaro, D. Butyl paraben and propyl paraben modulate bisphenol A and estradiol concentrations in female and male mice. Toxicol. Appl. Pharm. 2017, 325, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.S.; Kurohmaru, M. Disruption of Sertoli cell vimentin filaments in prepubertal rats: An acute effect of butylparaben in vivo and in vitro. Acta Histochem. 2014, 116, 682–687. [Google Scholar] [CrossRef] [PubMed]
- Daston, G.P. Developmental toxicity evaluation of butylparaben in Sprague-Dawley rats. Birth Defects Res. B Dev. Reprod. Toxicol. 2004, 71, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Taxvig, C.; Vinggaard, A.M.; Hass, U.; Axelstad, M.; Boberg, J.; Hansen, P.R.; Frederiksen, H.; Nellemann, C. Do parabens have the ability to interfere with steroidogenesis? Toxicol. Sci. 2008, 106, 206–213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De la Vega, A.C.S.; Molins-Delgado, D.; Barcelo, D.; Diaz-Cruz, M.S. Nanosized titanium dioxide UV filter increases mixture toxicity when combined with parabens. Ecotox Environ. Safe 2019, 184, 109565–109573. [Google Scholar] [CrossRef]
- Leppert, B.; Strunz, S.; Seiwert, B.; Schlittenbauer, L.; Schlichting, R.; Pfeiffer, C.; Roder, S.; Bauer, M.; Borte, M.; Stangl, G.I.; et al. Maternal paraben exposure triggers childhood overweight development. Nat. Commun. 2020, 11, 561. [Google Scholar] [CrossRef]
- Smarr, M.M.; Honda, M.; Kannan, K.; Chen, Z.; Kim, S.; Louis, G.M.B. Male urinary biomarkers of antimicrobial exposure and bi-directional associations with semen quality parameters. Reprod. Toxicol. 2018, 77, 103–108. [Google Scholar] [CrossRef]
- Jimenez-Diaz, I.; Vela-Soria, F.; Zafra-Gomez, A.; Navalon, A.; Ballesteros, O.; Navea, N.; Fernandez, M.F.; Olea, N.; Vilchez, J.L. A new liquid chromatography-tandem mass spectrometry method for determination of parabens in human placental tissue samples. Talanta 2011, 84, 702–709. [Google Scholar] [CrossRef]
- Faniband, M.; Lindh, C.H.; Jonsson, B.A.G. Human biological monitoring of suspected endocrine-disrupting compounds. Asian J. 2014, 16, 5–16. [Google Scholar] [CrossRef]
- Alam, M.S.; Ohsako, S.; Kanai, Y.; Kurohmaru, M. Single administration of butylparaben induces spermatogenic cell apoptosis in prepubertal rats. Acta Histochem. 2014, 116, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Mishra, D.P.; Shaha, C. Estrogen-induced spermatogenic cell apoptosis occurs via the mitochondrial pathway—Role of superoxide and nitric oxide. J. Biol. Chem. 2005, 280, 6181–6196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watkins, D.J.; Ferguson, K.K.; Del Toro, L.V.A.; Alshawabkeh, A.N.; Cordero, J.F.; Meeker, J.D. Associations between urinary phenol and paraben concentrations and markers of oxidative stress and inflammation among pregnant women in Puerto Rico. Int. J. Hyg. Environ. Health 2015, 218, 212–219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hegazy, H.G.; Ali, E.H.A.; Elgoly, A.H.M. Interplay between pro-inflammatory cytokines and brain oxidative stress biomarkers: Evidence of parallels between butyl paraben intoxication and the valproic acid brain physiopathology in autism rat model. Cytokine 2015, 71, 173–180. [Google Scholar] [CrossRef]
- Aung, M.T.; Ferguson, K.K.; Cantonwine, D.E.; Bakulski, K.M.; Mukherjee, B.; Loch-Caruso, R.; McElrath, T.F.; Meeker, J.D. Associations between maternal plasma measurements of inflammatory markers and urinary levels of phenols and parabens during pregnancy: A repeated measures study. Sci. Total Environ. 2019, 650, 1131–1140. [Google Scholar] [CrossRef]
- Nicholls, D.G.; Ferguson, S.J. Bioenergetics 3. Biochemistry 2002, 69, 818–819. [Google Scholar] [CrossRef]
- Park, C.J.; Nah, W.H.; Lee, J.E.; Oh, Y.S.; Gye, M.C. Butyl paraben-induced changes in DNA methylation in rat epididymal spermatozoa. Andrologia 2012, 44, 187–193. [Google Scholar] [CrossRef]
- Garcia-Espineira, M.C.; Tejeda-Benitez, L.P.; Olivero-Verbel, J. Toxic Effects of Bisphenol A, Propyl Paraben, and Triclosan on Caenorhabditis elegans. Int. J. Environ. Res. Public Health 2018, 15, 684. [Google Scholar] [CrossRef] [PubMed] [Green Version]







| Groups | Heart | Liver | Right Kidney | Left Kidney | Right Testicle | Left Testicle | Seminal Vesicles |
|---|---|---|---|---|---|---|---|
| Control | 0.34 ± 0.04 a | 3.56 ± 0.33 a | 0.34 ± 0.05 a | 0.36 ± 0.09 a | 0.74 ± 0.01 a | 0.75 ± 0.09 a | 0.33 ± 0.11 a |
| Methyl 100 | 0.40 ± 0.08 a | 3.79 ± 0.49 a | 0.44 ± 0.12 a | 0.42 ± 0.06 a | 0.63 ± 0.06 a.b | 0.67 ± 0.08 a.b | 0.29 ± 0.10 a |
| Methyl 200 | 0.43 ± 0.11 a | 3.89 ± 0.34 a | 0.40 ± 0.06 a | 0.41 ± 0.09 a | 0.60 ±0.01 b | 0.59 ± 0.14 b.c | 0.22 ± 0.06 b |
| Butyl 100 | 0.43 ± 0.11 a | 3.69 ± 0.33 a | 0.43 ± 0.07 a | 0.42 ± 0.06 a | 0.58 ± 0.15 b | 0.56 ± 0.1 b.c | 0.20 ± 0.07 b |
| Butyl 200 | 0.39 ± 0.07 a | 3.65 ± 0.46 a | 0.39 ± 0.05 a | 0.38 ± 0.05 a | 0.52 ± 0.12 b | 0.54 ± 0.09 c | 0.20 ± 0.06 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira, M.M.; Martins, F.; Silva, M.G.; Correia, E.; Videira, R.; Peixoto, F. Use of Parabens (Methyl and Butyl) during the Gestation Period: Mitochondrial Bioenergetics of the Testes and Antioxidant Capacity Alterations in Testes and Other Vital Organs of the F1 Generation. Antioxidants 2020, 9, 1302. https://doi.org/10.3390/antiox9121302
Oliveira MM, Martins F, Silva MG, Correia E, Videira R, Peixoto F. Use of Parabens (Methyl and Butyl) during the Gestation Period: Mitochondrial Bioenergetics of the Testes and Antioxidant Capacity Alterations in Testes and Other Vital Organs of the F1 Generation. Antioxidants. 2020; 9(12):1302. https://doi.org/10.3390/antiox9121302
Chicago/Turabian StyleOliveira, Maria Manuel, Fátima Martins, Mónica G. Silva, Elisete Correia, Romeu Videira, and Francisco Peixoto. 2020. "Use of Parabens (Methyl and Butyl) during the Gestation Period: Mitochondrial Bioenergetics of the Testes and Antioxidant Capacity Alterations in Testes and Other Vital Organs of the F1 Generation" Antioxidants 9, no. 12: 1302. https://doi.org/10.3390/antiox9121302