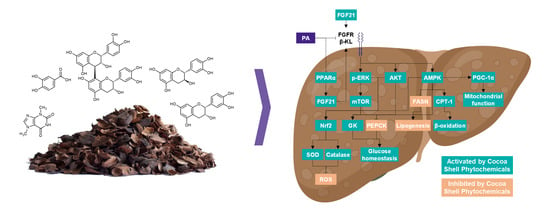
Phytochemicals from the Cocoa Shell Modulate Mitochondrial Function, Lipid and Glucose Metabolism in Hepatocytes via Activation of FGF21/ERK, AKT, and mTOR Pathways
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cocoa Shell Aqueous Extract (CAE) Preparation and Phytochemical Characterization by UPLC–MS/MS
2.3. Cell Culture
2.4. Experimental Design
2.5. Cell Viability
2.6. Assessment of the Effect of the Cocoa Shell Phytochemicals on FGF21 Signaling Activation
2.6.1. In Silico Molecular Docking of the Interaction of Phytochemicals with the FGF21 Receptor
2.6.2. ERK1/2 Phosphorylation Evaluation
2.6.3. FGF21 Release Quantification
2.7. Evaluation of the Effect of the Phytochemicals from the Cocoa Shell on Hepatic Lipotoxicity
2.7.1. Lactate Dehydrogenase (LDH) Cytotoxicity Assay
2.7.2. Quantification of Cytokines Release
2.7.3. Determination of Nitric Oxide Synthase (NOS) Activity
2.8. Evaluation of the Effect of the Phytochemicals from the Cocoa Shell on Oxidative Stress and Mitochondrial Function
2.8.1. Detection of Intracellular ROS, Mitochondrial Superoxide, Mitochondrial Membrane Potential (ΔΨm), and Enzymatic Antioxidant Activity
2.8.2. Assessment of Mitochondrial Content and Function
2.9. Evaluation of the Effect of the Phytochemicals from the Cocoa Shell on Lipid Metabolism
2.9.1. Determination of Cellular Lipid Accumulation
2.9.2. Assessment of Lipolysis
2.9.3. Measurement of Fatty Acid Synthase (FASN) Activity
2.9.4. Measurement of Carnitine Palmitoyltransferase 1 (CPT-1) Activity
2.10. Evaluation of the Effect of the Phytochemicals from the Cocoa Shell on Glucose Metabolism
2.10.1. Determination of Glucose Uptake
2.10.2. Measurement of Glucokinase (GK) Activity
2.10.3. Assessment of Gluconeogenesis
2.10.4. Measurement of Phosphoenolpyruvate Carboxykinase (PEPCK) Activity
2.11. Evaluation of Protein Expression and Phosphorylation by Western Blot
2.12. Evaluation of the Effect of the Cocoa Shell Phytochemicals on Metabolic Related Signaling Pathways Phosphorylation Pattern
2.13. Bioinformatic Analysis
2.14. Statistical Analysis
3. Results and Discussion
3.1. Theobromine and Protocatechuic Acid Are the Major Compounds in the Cocoa Shell Extract
3.2. Protocatechuic Acid and Procyanidin B2 Mimicked FGF21 and Stimulated ERK Signaling
3.3. Cocoa Shell Phytochemicals, Mainly Flavanols, Preserved Cell Viability and Reduced Inflammation
3.4. Cocoa Shell Phytochemicals, Mainly Protocatechuic Acid, Diminished Oxidative Stress via Regulation of Antioxidative Systems
3.5. Protocatechuic Acid and Catechin, among Other Cocoa Shell Phytochemicals, Mimicked FGF21 Protecting Hepatocytes from Mitochondrial Dysfunction
3.6. Cocoa Shell Phytochemicals, Primarily Protocatechuic Acid, Reduced the Hepatic Lipid Load by Diminishing De Novo Fatty Acid Synthesis and Stimulation Fatty Acid Oxidation
3.7. Protocatechuic Acid and Epicatechin, among Cocoa Shell Phytochemicals, Regulated Glucose Metabolism in Palmitic Acid-Challenged Hepatocytes
3.8. Cocoa Shell Phytochemicals Differentially Modulate Protein Phosphorylation Thereby Regulating mTOR, AKT, and ERK Signaling Pathways
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Noncommunicable Diseases. Available online: https://www.who.int/news-room/fact-sheets/detail/noncommunicable-diseases (accessed on 20 May 2019).
- Rani, V.; Deep, G.; Singh, R.K.; Palle, K.; Yadav, U.C.S. Oxidative stress and metabolic disorders: Pathogenesis and therapeutic strategies. Life Sci. 2016, 148, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Kader, S.M.; El-Den Ashmawy, E.M.S. Non-alcoholic fatty liver disease: The diagnosis and management. World J. Hepatol. 2015, 7, 846–858. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.; Anstee, Q.M.; Marietti, M.; Hardy, T.; Henry, L.; Eslam, M.; George, J.; Bugianesi, E. Global burden of NAFLD and NASH: Trends, predictions, risk factors and prevention. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 11–20. [Google Scholar] [CrossRef]
- Schulze, M.B.; Martínez-González, M.A.; Fung, T.T.; Lichtenstein, A.H.; Forouhi, N.G. Food based dietary patterns and chronic disease prevention. BMJ 2018, 361, k2396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tezze, C.; Romanello, V.; Sandri, M. FGF21 as Modulator of Metabolism in Health and Disease. Front. Physiol. 2019, 10, 419. [Google Scholar] [CrossRef]
- So, W.Y.; Leung, P.S. Fibroblast Growth Factor 21 As an Emerging Therapeutic Target for Type 2 Diabetes Mellitus. Med. Res. Rev. 2016, 36, 672–704. [Google Scholar] [CrossRef]
- Fisher, M.F.; Chui, P.C.; Antonellis, P.J.; Bina, H.A.; Kharitonenkov, A.; Flier, J.S.; Maratos-Flier, E. Obesity Is a Fibroblast Growth Factor 21 (FGF21)-Resistant State. Diabetes 2010, 59, 2781–2789. [Google Scholar] [CrossRef] [Green Version]
- Montagner, A.; Polizzi, A.; Fouché, E.; Ducheix, S.; Lippi, Y.; Lasserre, F.; Barquissau, V.; Régnier, M.; Lukowicz, C.; Benhamed, F.; et al. Liver PPARα is crucial for whole-body fatty acid homeostasis and is protective against NAFLD. Gut 2016, 65, 1202–1214. [Google Scholar] [CrossRef] [Green Version]
- Yie, J.; Wang, W.; Deng, L.; Tam, L.-T.; Stevens, J.; Chen, M.M.; Li, Y.; Xu, J.; Lindberg, R.; Hecht, R.; et al. Understanding the Physical Interactions in the FGF21/FGFR/β-Klotho Complex: Structural Requirements and Implications in FGF21 Signaling. Chem. Biol. Drug Des. 2012, 79, 398–410. [Google Scholar] [CrossRef]
- Nies, V.J.M.; Sancar, G.; Liu, W.; van Zutphen, T.; Struik, D.; Yu, R.T.; Atkins, A.R.; Evans, R.M.; Jonker, J.W.; Downes, M.R. Fibroblast Growth Factor Signaling in Metabolic Regulation. Front. Endocrinol. 2016, 6, 193. [Google Scholar] [CrossRef] [Green Version]
- Zarei, M.; Pizarro-Delgado, J.; Barroso, E.; Palomer, X.; Vázquez-Carrera, M. Targeting FGF21 for the Treatment of Nonalcoholic Steatohepatitis. Trends Pharmacol. Sci. 2020, 41, 199–208. [Google Scholar] [CrossRef]
- Panak Balentić, J.; Ačkar, Đ.; Jokić, S.; Jozinović, A.; Babić, J.; Miličević, B.; Šubarić, D.; Pavlović, N. Cocoa Shell: A By-Product with Great Potential for Wide Application. Molecules 2018, 23, 1404. [Google Scholar] [CrossRef] [Green Version]
- Rebollo-Hernanz, M.; Zhang, Q.; Aguilera, Y.; Martín-Cabrejas, M.A.; de Mejia, E.G. Cocoa Shell Aqueous Phenolic Extract Preserves Mitochondrial Function and Insulin Sensitivity by Attenuating Inflammation between Macrophages and Adipocytes In Vitro. Mol. Nutr. Food Res. 2019, 63, 1801413. [Google Scholar] [CrossRef] [PubMed]
- Rebollo-Hernanz, M.; Zhang, Q.; Aguilera, Y.; Martín-Cabrejas, M.A.; de Mejia, E.G. Relationship of the phytochemicals from coffee and cocoa by-products with their potential to modulate biomarkers of metabolic syndrome in vitro. Antioxidants 2019, 8, 279. [Google Scholar] [CrossRef] [Green Version]
- Yang, W.; Chen, X.; Liu, Y.; Chen, M.; Jiang, X.; Shen, T.; Li, Q.; Yang, Y.; Ling, W. N-3 polyunsaturated fatty acids increase hepatic fibroblast growth factor 21 sensitivity via a PPAR-γ-β-klotho pathway. Mol. Nutr. Food Res. 2017, 61, 1601075. [Google Scholar] [CrossRef]
- Ejaz, A.; Martinez-Guino, L.; Goldfine, A.B.; Ribas-Aulinas, F.; De Nigris, V.; Ribó, S.; Gonzalez-Franquesa, A.; Garcia-Roves, P.M.; Li, E.; Dreyfuss, J.M.; et al. Dietary Betaine Supplementation Increases Fgf21 Levels to Improve Glucose Homeostasis and Reduce Hepatic Lipid Accumulation in Mice. Diabetes 2016, 65, 902–912. [Google Scholar] [CrossRef] [Green Version]
- Zeng, K.; Tian, L.; Patel, R.; Shao, W.; Song, Z.; Liu, L.; Manuel, J.; Ma, X.; McGilvray, I.; Cummins, C.L.; et al. Diet polyphenol curcumin stimulates hepatic Fgf21 production and restores its sensitivity in high fat diet fed male mice. Endocrinology 2016, 158, 277–292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rebollo-Hernanz, M.; Cañas, S.; Taladrid, D.; Bartolomeé, B.; Aguilera, Y.; Martin-Cabrejas, M.A. Extraction of phenolic compounds from cocoa shell: Modeling using response surface methodology and artificial neural networks. Sep. Purif. Technol. 2021, 270, 118779. [Google Scholar] [CrossRef]
- Aguilera, Y.; Rebollo-Hernanz, M.; Cañas, S.; Taladrid, D.; Martín-Cabrejas, M.A. Response surface methodology to optimise the heat-assisted aqueous extraction of phenolic compounds from coffee parchment and their comprehensive analysis. Food Funct. 2019, 10, 4739–4750. [Google Scholar] [CrossRef] [PubMed]
- Vangone, A.; Schaarschmidt, J.; Koukos, P.; Geng, C.; Citro, N.; Trellet, M.E.; Xue, L.C.; Bonvin, A.M.J.J. Large-scale prediction of binding affinity in protein–small ligand complexes: The PRODIGY-LIG web server. Bioinformatics 2019, 35, 1585–1587. [Google Scholar] [CrossRef] [Green Version]
- Xue, L.C.; Rodrigues, J.P.; Kastritis, P.L.; Bonvin, A.M.; Vangone, A. PRODIGY: A web server for predicting the binding affinity of protein–protein complexes. Bioinformatics 2016, 32, 3676–3678. [Google Scholar] [CrossRef]
- Herrera, B.; Murillo, M.M.; Álvarez-Barrientos, A.; Beltrán, J.; Fernández, M.; Fabregat, I. Source of early reactive oxygen species in the apoptosis induced by transforming growth factor-β in fetal rat hepatocytes. Free Radic. Biol. Med. 2004, 36, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.Y.; Prognon, P. Raw material enzymatic activity determination: A specific case for validation and comparison of analytical methods—The example of superoxide dismutase (SOD). J. Pharm. Biomed. Anal. 2006, 40, 1143–1148. [Google Scholar] [CrossRef]
- Wang, Z.J.; Liang, C.L.; Li, G.M.; Yu, C.Y.; Yin, M. Neuroprotective effects of arachidonic acid against oxidative stress on rat hippocampal slices. Chem. Biol. Interact. 2006, 163, 207–217. [Google Scholar] [CrossRef]
- Desquiret-Dumas, V.; Gueguen, N.; Leman, G.; Baron, S.; Nivet-Antoine, V.; Chupin, S.; Chevrollier, A.; Vessières, E.; Ayer, A.; Ferré, M.; et al. Resveratrol induces a mitochondrial complex i-dependent increase in nadh oxidation responsible for sirtuin activation in liver cells. J. Biol. Chem. 2013, 288, 36662–36675. [Google Scholar] [CrossRef] [Green Version]
- Rebollo-Hernanz, M.; Zhang, Q.; Aguilera, Y.; Martín-Cabrejas, M.A.; Gonzalez de Mejia, E. Phenolic compounds from coffee by-products modulate adipogenesis-related inflammation, mitochondrial dysfunction, and insulin resistance in adipocytes, via insulin/PI3K/AKT signaling pathways. Food Chem. Toxicol. 2019, 132, 110672. [Google Scholar] [CrossRef] [PubMed]
- Luna-Vital, D.; Weiss, M.; Gonzalez de Mejia, E. Anthocyanins from Purple Corn Ameliorated Tumor Necrosis Factor-α-Induced Inflammation and Insulin Resistance in 3T3-L1 Adipocytes via Activation of Insulin Signaling and Enhanced GLUT4 Translocation. Mol. Nutr. Food Res. 2017, 61, 1700362. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.J.; Liu, Y.C.; Chang, C.J.; Pan, M.H.; Lee, M.F.; Pan, B.S. Hepatoprotective mechanism of freshwater clam extract alleviates non-alcoholic fatty liver disease: Elucidated: In vitro and in vivo models. Food Funct. 2018, 9, 6315–6325. [Google Scholar] [CrossRef]
- Kololziej, M.P.; Crilly, P.J.; Corstorphine, C.G.; Zammit, V.A. Development and characterization of a polyclonal antibody against rat liver mitochondrial overt carnitine palmitoyltransferase (CPT I). Distinction of CPT I from CPT II and of isoforms of CPT I in different tissues. Biochem. J. 1992, 282, 415–421. [Google Scholar] [CrossRef] [Green Version]
- Dhanesha, N.; Joharapurkar, A.; Shah, G.; Dhote, V.; Kshirsagar, S.; Bahekar, R.; Jain, M. Exendin-4 reduces glycemia by increasing liver glucokinase activity: An insulin independent effect. Pharmacol. Rep. 2012, 64, 140–149. [Google Scholar] [CrossRef]
- Teng, H.; Chen, L.; Song, H. The potential beneficial effects of phenolic compounds isolated from: A. pilosa Ledeb on insulin-resistant hepatic HepG2 cells. Food Funct. 2016, 7, 4400–4409. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhou, B.; Pache, L.; Chang, M.; Khodabakhshi, A.H.; Tanaseichuk, O.; Benner, C.; Chanda, S.K. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat. Commun. 2019, 10, 1523. [Google Scholar] [CrossRef]
- Kanehisa, M.; Furumichi, M.; Sato, Y.; Ishiguro-Watanabe, M.; Tanabe, M. KEGG: Integrating viruses and cellular organisms. Nucleic Acids Res. 2021, 49, D545–D551. [Google Scholar] [CrossRef] [PubMed]
- Mazzutti, S.; Rodrigues, L.G.G.; Mezzomo, N.; Venturi, V.; Ferreira, S.R.S. Integrated green-based processes using supercritical CO2 and pressurized ethanol applied to recover antioxidant compouds from cocoa (Theobroma cacao) bean hulls. J. Supercrit. Fluids 2018, 135, 52–59. [Google Scholar] [CrossRef]
- Jokić, S.; Gagić, T.; Knez, Ž.; Šubarić, D.; Škerget, M. Separation of Active Compounds from Food by-Product (Cocoa Shell) Using Subcritical Water Extraction. Molecules 2018, 23, 1408. [Google Scholar] [CrossRef] [Green Version]
- Mellinas, A.C.; Jiménez, A.; Garrigós, M.C. Optimization of microwave-assisted extraction of cocoa bean shell waste and evaluation of its antioxidant, physicochemical and functional properties. LWT 2020, 127, 109361. [Google Scholar] [CrossRef]
- Pavlović, N.; Jokić, S.; Jakovljević, M.; Blažić, M.; Molnar, M. Green Extraction Methods for Active Compounds from Food Waste—Cocoa Bean Shell. Foods 2020, 9, 140. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.Y.; Yu, L.; Chen, J.H.; Yang, L.N.; Lin, C.; Shi, X.Q.; Qin, H. Sesamol alleviates obesity-related hepatic steatosis via activating hepatic PKA pathway. Nutrients 2020, 12, 329. [Google Scholar] [CrossRef] [Green Version]
- Pace, E.; Jiang, Y.; Clemens, A.; Crossman, T.; Rupasinghe, H.P.V. Impact of thermal degradation of cyanidin-3-O-glucoside of haskap berry on cytotoxicity of hepatocellular carcinoma HepG2 and breast cancer MDA-MB-231 cells. Antioxidants 2018, 7, 24. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Garza, Ú.; Torres-Oteros, D.; Yarritu-Gallego, A.; Marrero, P.F.; Haro, D.; Relat, J. Fibroblast Growth Factor 21 and the Adaptive Response to Nutritional Challenges. Int. J. Mol. Sci. 2019, 20, 4692. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.; Choi, J.; Mohanty, J.; Sousa, L.P.; Tome, F.; Pardon, E.; Steyaert, J.; Lemmon, M.A.; Lax, I.; Schlessinger, J. Structures of β-klotho reveal a â € zip code’-like mechanism for endocrine FGF signalling. Nature 2018, 553, 501–505. [Google Scholar] [CrossRef]
- Asrih, M.; Montessuit, C.; Philippe, J.; Jornayvaz, F.R. Free Fatty Acids Impair FGF21 Action in HepG2 Cells. Cell. Physiol. Biochem. 2015, 37, 1767–1778. [Google Scholar] [CrossRef]
- Sonoda, J.; Chen, M.Z.; Baruch, A. FGF21-receptor agonists: An emerging therapeutic class for obesity-related diseases. Horm. Mol. Biol. Clin. Investig. 2017, 30, 20170002. [Google Scholar] [CrossRef]
- Minard, A.Y.; Tan, S.-X.; Yang, P.; Fazakerley, D.J.; Domanova, W.; Parker, B.L.; Humphrey, S.J.; Jothi, R.; Stöckli, J.; James, D.E. mTORC1 Is a Major Regulatory Node in the FGF21 Signaling Network in Adipocytes. Cell Rep. 2016, 17, 29–36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ge, X.; Wang, Y.; Lam, K.S.; Xu, A. Metabolic actions of FGF21: Molecular mechanisms and therapeutic implications. Acta Pharm. Sin. B 2012, 2, 350–357. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Fernández, L.; González-Muniesa, P.; Sáinz, N.; Laiglesia, L.M.; Escoté, X.; Martínez, J.A.; Moreno-Aliaga, M.J. Maresin 1 Regulates Hepatic FGF21 in Diet-Induced Obese Mice and in Cultured Hepatocytes. Mol. Nutr. Food Res. 2019, 63, 1900358. [Google Scholar] [CrossRef]
- Zang, Y.; Fan, L.; Chen, J.; Huang, R.; Qin, H. Improvement of Lipid and Glucose Metabolism by Capsiate in Palmitic Acid-Treated HepG2 Cells via Activation of the AMPK/SIRT1 Signaling Pathway. J. Agric. Food Chem. 2018, 66, 6772–6781. [Google Scholar] [CrossRef] [PubMed]
- Dongiovanni, P.; Crudele, A.; Panera, N.; Romito, I.; Meroni, M.; De Stefanis, C.; Palma, A.; Comparcola, D.; Fracanzani, A.L.; Miele, L.; et al. β-Klotho gene variation is associated with liver damage in children with NAFLD. J. Hepatol. 2020, 72, 411–419. [Google Scholar] [CrossRef] [Green Version]
- García-Ruiz, I.; Solís-Muñoz, P.; Fernández-Moreira, D.; Muñoz-Yagüe, T.; Solís-Herruzo, J.A. In vitro treatment of HepG2 cells with saturated fatty acids reproduces mitochondrial dysfunction found in nonalcoholic steatohepatitis. Dis. Model. Mech. 2015, 8, 183–191. [Google Scholar] [CrossRef] [Green Version]
- Hsu, J.-Y.; Lin, H.-H.; Chyau, C.-C.; Wang, Z.-H.; Chen, J.-H. Aqueous Extract of Pepino Leaves Ameliorates Palmitic Acid-Induced Hepatocellular Lipotoxicity via Inhibition of Endoplasmic Reticulum Stress and Apoptosis. Antioxidants 2021, 10, 903. [Google Scholar] [CrossRef]
- Rada, P.; González-Rodríguez, Á.; García-Monzón, C.; Valverde, Á.M. Understanding lipotoxicity in NAFLD pathogenesis: Is CD36 a key driver? Cell Death Dis. 2020, 11, 802. [Google Scholar] [CrossRef]
- Diehl, A.M.; Day, C. Cause, Pathogenesis, and Treatment of Nonalcoholic Steatohepatitis. N. Engl. J. Med. 2017, 377, 2063–2072. [Google Scholar] [CrossRef]
- Xiao, Q.; Zhang, S.; Yang, C.; Du, R.; Zhao, J.; Li, J.; Xu, Y.; Qin, Y.; Gao, Y.; Huang, W. Ginsenoside Rg1 Ameliorates Palmitic Acid-Induced Hepatic Steatosis and Inflammation in HepG2 Cells via the AMPK/NF- B Pathway. Int. J. Endocrinol. 2019, 2019, 7514802. [Google Scholar] [CrossRef] [Green Version]
- Delli Bovi, A.P.; Marciano, F.; Mandato, C.; Siano, M.A.; Savoia, M.; Vajro, P. Oxidative Stress in Non-alcoholic Fatty Liver Disease. An Updated Mini Review. Front. Med. 2021, 8, 165. [Google Scholar] [CrossRef]
- Tillman, E.J.; Rolph, T. FGF21: An Emerging Therapeutic Target for Non-Alcoholic Steatohepatitis and Related Metabolic Diseases. Front. Endocrinol. 2020, 11, 976. [Google Scholar] [CrossRef]
- Gómez-Sámano, M.Á.; Grajales-Gómez, M.; Zuarth-Vázquez, J.M.; Navarro-Flores, M.F.; Martínez-Saavedra, M.; Juárez-León, Ó.A.; Morales-García, M.G.; Enríquez-Estrada, V.M.; Gómez-Pérez, F.J.; Cuevas-Ramos, D. Fibroblast growth factor 21 and its novel association with oxidative stress. Redox Biol. 2017, 11, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Varì, R.; D’Archivio, M.; Filesi, C.; Carotenuto, S.; Scazzocchio, B.; Santangelo, C.; Giovannini, C.; Masella, R. Protocatechuic acid induces antioxidant/detoxifying enzyme expression through JNK-mediated Nrf2 activation in murine macrophages. J. Nutr. Biochem. 2011, 22, 409–417. [Google Scholar] [CrossRef]
- Granado-Serrano, A.B.; Martín, M.A.; Haegeman, G.; Goya, L.; Bravo, L.; Ramos, S. Epicatechin induces NF-κB, activator protein-1 (AP-1) and nuclear transcription factor erythroid 2p45-related factor-2 (Nrf2) via phosphatidylinositol-3-kinase/protein kinase B (PI3K/AKT) and extracellular regulated kinase (ERK) signalling in HepG2 cells. Br. J. Nutr. 2010, 103, 168–179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Talebi, M.; Talebi, M.; Farkhondeh, T.; Mishra, G.; İlgün, S.; Samarghandian, S. New insights into the role of the Nrf2 signaling pathway in green tea catechin applications. Phyther. Res. 2021, 35, 3078–3112. [Google Scholar] [CrossRef]
- Li, S.; Hong, M.; Tan, H.Y.; Wang, N.; Feng, Y. Insights into the Role and Interdependence of Oxidative Stress and Inflammation in Liver Diseases. Oxid. Med. Cell. Longev. 2016, 2016, 4234061. [Google Scholar] [CrossRef]
- Begriche, K.; Massart, J.; Robin, M.-A.; Bonnet, F.; Fromenty, B. Mitochondrial adaptations and dysfunctions in nonalcoholic fatty liver disease. Hepatology 2013, 58, 1497–1507. [Google Scholar] [CrossRef]
- Zhang, Q.; Gonzalez de Mejia, E. Protocatechuic acid attenuates adipogenesis-induced inflammation and mitochondrial dysfunction in 3T3-L1 adipocytes by regulation of AMPK pathway. J. Funct. Foods 2020, 69, 103972. [Google Scholar] [CrossRef]
- Rafiei, H.; Omidian, K.; Bandy, B. Protection by different classes of dietary polyphenols against palmitic acid-induced steatosis, nitro-oxidative stress and endoplasmic reticulum stress in HepG2 hepatocytes. J. Funct. Foods 2018, 44, 173–182. [Google Scholar] [CrossRef]
- Rafiei, H.; Omidian, K.; Bandy, B. Dietary Polyphenols Protect Against Oleic Acid-Induced Steatosis in an in Vitro Model of NAFLD by Modulating Lipid Metabolism and Improving Mitochondrial Function. Nutrients 2019, 11, 541. [Google Scholar] [CrossRef] [Green Version]
- Chau, M.D.L.; Gao, J.; Yang, Q.; Wu, Z.; Gromada, J. Fibroblast growth factor 21 regulates energy metabolism by activating the AMPK–SIRT1–PGC-1α pathway. Proc. Natl. Acad. Sci. USA 2010, 107, 12553. [Google Scholar] [CrossRef] [Green Version]
- Sun, M.; Gu, Y.; Glisan, S.L.; Lambert, J.D. Dietary cocoa ameliorates non-alcoholic fatty liver disease and increases markers of antioxidant response and mitochondrial biogenesis in high fat-fed mice. J. Nutr. Biochem. 2021, 92, 108618. [Google Scholar] [CrossRef]
- Wu, L.; Mo, W.; Feng, J.; Li, J.; Yu, Q.; Li, S.; Zhang, J.; Chen, K.; Ji, J.; Dai, W.; et al. Astaxanthin attenuates hepatic damage and mitochondrial dysfunction in non-alcoholic fatty liver disease by up-regulating the FGF21/PGC-1α pathway. Br. J. Pharmacol. 2020, 177, 3760–3777. [Google Scholar] [CrossRef]
- Lee, J.H.; Kang, Y.E.; Chang, J.Y.; Park, K.C.; Kim, H.-W.; Kim, J.T.; Kim, H.J.; Yi, H.-S.; Shong, M.; Chung, H.K.; et al. An engineered FGF21 variant, LY2405319, can prevent non-alcoholic steatohepatitis by enhancing hepatic mitochondrial function. Am. J. Transl. Res. 2016, 8, 4750. [Google Scholar]
- Ipsen, D.H.; Lykkesfeldt, J.; Tveden-Nyborg, P. Molecular mechanisms of hepatic lipid accumulation in non-alcoholic fatty liver disease. Cell. Mol. Life Sci. 2018, 75, 3313–3327. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.-H.; Lin, C.-C.; Wang, Z.-H.; Mong, M.-C.; Yin, M.-C. Effects of Protocatechuic Acid on Trans Fat Induced Hepatic Steatosis in Mice. J. Agric. Food Chem. 2010, 58, 10247–10252. [Google Scholar] [CrossRef]
- Sun, R.; Kang, X.; Zhao, Y.; Wang, Z.; Wang, R.; Fu, R.; Li, Y.; Hu, Y.; Wang, Z.; Shan, W.; et al. Sirtuin 3-mediated deacetylation of acyl-CoA synthetase family member 3 by protocatechuic acid attenuates non-alcoholic fatty liver disease. Br. J. Pharmacol. 2020, 177, 4166–4180. [Google Scholar] [CrossRef]
- Loomba, R.; Friedman, S.L.; Shulman, G.I. Mechanisms and disease consequences of nonalcoholic fatty liver disease. Cell 2021, 184, 2537–2564. [Google Scholar] [CrossRef] [PubMed]
- Rui, L. Energy Metabolism in the Liver. Compr. Physiol. 2014, 4, 177. [Google Scholar] [CrossRef] [Green Version]
- Cordero-Herrera, I.; Martín, M.A.; Bravo, L.; Goya, L.; Ramos, S. Cocoa flavonoids improve insulin signalling and modulate glucose production via AKT and AMPK in HepG2 cells. Mol. Nutr. Food Res. 2013, 57, 974–985. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blanco, A.M.; Bertucci, J.I.; Unniappan, S. FGF21 Mimics a Fasting-Induced Metabolic State and Increases Appetite in Zebrafish. Sci. Rep. 2020, 10, 6993. [Google Scholar] [CrossRef]
- El-Sonbaty, Y.A.; Suddek, G.M.; Megahed, N.; Gameil, N.M. Protocatechuic acid exhibits hepatoprotective, vasculoprotective, antioxidant and insulin-like effects in dexamethasone-induced insulin-resistant rats. Biochimie 2019, 167, 119–134. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yang, K.; Yang, J.; Xiao, W.; Le, Y.; Yu, F.; Gu, L.; Lang, S.; Tian, Q.; Jin, T.; et al. Liver-derived fibroblast growth factor 21 mediates effects of glucagon-like peptide-1 in attenuating hepatic glucose output. EBioMedicine 2019, 41, 73–84. [Google Scholar] [CrossRef] [Green Version]
- Chao, H.-W.; Chao, S.-W.; Lin, H.; Ku, H.-C.; Cheng, C.-F. Homeostasis of Glucose and Lipid in Non-Alcoholic Fatty Liver Disease. Int. J. Mol. Sci. 2019, 20, 298. [Google Scholar] [CrossRef] [Green Version]
- Matsuda, S.; Kobayashi, M.; Kitagishi, Y. Roles for PI3K/AKT/PTEN Pathway in Cell Signaling of Nonalcoholic Fatty Liver Disease. ISRN Endocrinol. 2013, 2013, 472432. [Google Scholar] [CrossRef]
- Foretz, M.; Even, P.C.; Viollet, B. AMPK activation reduces hepatic lipid content by increasing fat oxidation in vivo. Int. J. Mol. Sci. 2018, 19, 2826. [Google Scholar] [CrossRef] [Green Version]
- Lv, H.; Ren, H.; Wang, L.; Chen, W.; Ci, X. Lico A Enhances Nrf2-Mediated Defense Mechanisms against t -BHP-Induced Oxidative Stress and Cell Death via Akt and ERK Activation in RAW 264.7 Cells. Oxid. Med. Cell. Longev. 2015, 2015, 9845. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jasek-Gajda, E.; Jurkowska, H.; Jasińska, M.; Lis, G.J. Targeting the MAPK/ERK and PI3K/AKT Signaling Pathways Affects NRF2, Trx and GSH Antioxidant Systems in Leukemia Cells. Antioxidants 2020, 9, 633. [Google Scholar] [CrossRef]
- Li, Y.; Ding, H.; Liu, L.; Song, Y.; Du, X.; Feng, S.; Wang, X.; Li, X.; Wang, Z.; Li, X.; et al. Non-esterified Fatty Acid Induce Dairy Cow Hepatocytes Apoptosis via the Mitochondria-Mediated ROS-JNK/ERK Signaling Pathway. Front. Cell Dev. Biol. 2020, 8, 245. [Google Scholar] [CrossRef] [PubMed]
- Dou, X.; Ding, Q.; Lai, S.; Jiang, F.; Song, Q.; Zhao, X.; Fu, A.; Moustaid-Moussa, N.; Su, D.; Li, S. Salidroside alleviates lipotoxicity-induced cell death through inhibition of TLR4/MAPKs pathway, and independently of AMPK and autophagy in AML-12 mouse hepatocytes. J. Funct. Foods 2020, 65, 103691. [Google Scholar] [CrossRef]
- Hagiwara, A.; Cornu, M.; Cybulski, N.; Polak, P.; Betz, C.; Trapani, F.; Terracciano, L.; Heim, M.H.; Rüegg, M.A.; Hall, M.N. Hepatic mTORC2 activates glycolysis and lipogenesis through Akt, glucokinase, and SREBP1c. Cell Metab. 2012, 15, 725–738. [Google Scholar] [CrossRef] [Green Version]
- Smith, B.K.; Marcinko, K.; Desjardins, E.M.; Lally, J.S.; Ford, R.J.; Steinberg, G.R. Treatment of nonalcoholic fatty liver disease: Role of AMPK. Am. J. Physiol. Endocrinol. Metab. 2016, 311, E730–E740. [Google Scholar] [CrossRef] [Green Version]
- Xie, T.; Leung, P.S. Fibroblast growth factor 21: A regulator of metabolic disease and health span. Am. J. Physiol. Endocrinol. Metab. 2017, 313, E292. [Google Scholar] [CrossRef]
- Scazzocchio, B.; Varì, R.; Filesi, C.; Del Gaudio, I.; D’Archivio, M.; Santangelo, C.; Iacovelli, A.; Galvano, F.; Pluchinotta, F.R.; Giovannini, C.; et al. Protocatechuic acid activates key components of insulin signaling pathway mimicking insulin activity. Mol. Nutr. Food Res. 2015, 59, 1472–1481. [Google Scholar] [CrossRef]
- Kim, J.; Kim, J.; Shim, J.; Lee, C.Y.; Lee, K.W.; Lee, H.J. Cocoa Phytochemicals: Recent Advances in Molecular Mechanisms on Health. Crit. Rev. Food Sci. Nutr. 2014, 54, 1458–1472. [Google Scholar] [CrossRef]
- Bohn, T.; Mcdougall, G.J.; Alegría, A.; Alminger, M.; Arrigoni, E.; Aura, A.M.; Brito, C.; Cilla, A.; El, S.N.; Karakaya, S.; et al. Mind the gap-deficits in our knowledge of aspects impacting the bioavailability of phytochemicals and their metabolites-a position paper focusing on carotenoids and polyphenols. Mol. Nutr. Food Res. 2015, 59, 1307–1323. [Google Scholar] [CrossRef] [Green Version]
- Rebollo-Hernanz, M.; Cañas, S.; Aguilera, Y.; Benitez, V.; Gila-Díaz, A.; Rodriguez-Rodriguez, P.; Cobeta, I.M.; de Pablo, A.L.L.; Gonzalez, M.C.; Arribas, S.M. Validation of Cocoa Shell as a Novel Antioxidant Dietary Fiber Food Ingredient: Nutritional Value, Functional Properties, and Safety. Curr. Dev. Nutr. 2020, 4, 773. [Google Scholar] [CrossRef]


 ) to the highest (
) to the highest (  ) value for each parameter) (G) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on lipo/cytotoxicity and inflammation (H). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–f) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
) value for each parameter) (G) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on lipo/cytotoxicity and inflammation (H). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–f) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
 ) to the highest (
) to the highest (  ) value for each parameter) (G) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on lipo/cytotoxicity and inflammation (H). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–f) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
) value for each parameter) (G) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on lipo/cytotoxicity and inflammation (H). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–f) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
 ) to the highest (
) to the highest (  ) value for each parameter) (H) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on oxidative stress (I). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–f) significantly (p < 0.05) differ according to ANOVA and Tukey’s test. NT: non-treated cells; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
) value for each parameter) (H) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on oxidative stress (I). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–f) significantly (p < 0.05) differ according to ANOVA and Tukey’s test. NT: non-treated cells; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
 ) to the highest (
) to the highest (  ) value for each parameter) (H) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on oxidative stress (I). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–f) significantly (p < 0.05) differ according to ANOVA and Tukey’s test. NT: non-treated cells; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
) value for each parameter) (H) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on oxidative stress (I). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–f) significantly (p < 0.05) differ according to ANOVA and Tukey’s test. NT: non-treated cells; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
 ) to the highest (
) to the highest (  ) value for each parameter) (J) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on mitochondrial function (K). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–e) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; PA: palmitic acid; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
) value for each parameter) (J) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on mitochondrial function (K). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–e) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; PA: palmitic acid; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
 ) to the highest (
) to the highest (  ) value for each parameter) (J) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on mitochondrial function (K). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–e) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; PA: palmitic acid; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
) value for each parameter) (J) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on mitochondrial function (K). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–e) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; PA: palmitic acid; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
 ) to the highest (
) to the highest (  ) value for each parameter) (G) and an integrative diagram illustrating the effects of phytochemicals from the cocoa shell on lipid metabolism (H). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–e) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; PA: palmitic acid; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21; FFAs: free fatty acids.
) value for each parameter) (G) and an integrative diagram illustrating the effects of phytochemicals from the cocoa shell on lipid metabolism (H). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–e) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; PA: palmitic acid; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21; FFAs: free fatty acids.
 ) to the highest (
) to the highest (  ) value for each parameter) (G) and an integrative diagram illustrating the effects of phytochemicals from the cocoa shell on lipid metabolism (H). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–e) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; PA: palmitic acid; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21; FFAs: free fatty acids.
) value for each parameter) (G) and an integrative diagram illustrating the effects of phytochemicals from the cocoa shell on lipid metabolism (H). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–e) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; PA: palmitic acid; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21; FFAs: free fatty acids.
 ) to the highest (
) to the highest (  ) value for each parameter) (H) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on glucose metabolism (I). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–e) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; PA: palmitic acid; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
) value for each parameter) (H) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on glucose metabolism (I). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–e) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; PA: palmitic acid; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
 ) to the highest (
) to the highest (  ) value for each parameter) (H) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on glucose metabolism (I). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–e) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; PA: palmitic acid; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
) value for each parameter) (H) and an integrative diagram illustrating the effects of the phytochemicals from the cocoa shell on glucose metabolism (I). The results are expressed as mean ± SD (n = 3). Bars with different letters (a–e) significantly (p < 0.05) differ according to ANOVA and Tukey’s multiple range test. NT: non-treated cells; PA: palmitic acid; TH: theobromine; PCA: protocatechuic acid; PB2: procyanidin B2; EPI: epicatechin; CAT: catechin; FGF21: fibroblast growth factor 21.
 ) to the highest (
) to the highest (  ) value for each parameter) classifying all the treatments studies according to the effects observed (C).
) value for each parameter) classifying all the treatments studies according to the effects observed (C).
 ) to the highest (
) to the highest (  ) value for each parameter) classifying all the treatments studies according to the effects observed (C).
) value for each parameter) classifying all the treatments studies according to the effects observed (C).

| Target Protein | Phosphosite | Effect of Phosphorylation | Relative Phosphorylation | Fold Change | ||
|---|---|---|---|---|---|---|
| PA | CAE | CAE/PA | p-Value | |||
| Insulin signaling | ||||||
| IGF1R | Y1165 | Induces activity | 0.23 ± 0.02 | 0.45 ± 0.04 | 1.99 ± 0.22 | 0.033 * |
| INSR | Y1189 | Induces activity | 0.68 ± 0.05 | 1.96 ± 0.11 | 2.87 ± 0.21 | 0.009 ** |
| IRS-1 | S318 | Inhibits molecular association | 0.06 ± 0.01 | 0.26 ± 0.03 | 4.28 ± 0.48 | 0.020 * |
| SHC-1 | Y427 | Induces activity | 0.18 ± 0.02 | 0.32 ± 0.05 | 1.77 ± 0.35 | 0.117 |
| SHIP-1 | Y1020 | Induces activity | 0.13 ± 0.01 | 0.30 ± 0.05 | 2.21 ± 0.39 | 0.060 |
| SHP-2 | T542 | Induces molecular association | 0.34 ± 0.03 | 0.56 ± 0.04 | 1.65 ± 0.21 | 0.081 |
| PI3K-AKT-PKB signaling | ||||||
| AKT | S473 | Induces activity | 0.57 ± 0.04 | 1.54 ± 0.09 | 2.71 ± 0.21 | 0.011 * |
| BAD | S112 | Inhibits molecular association | 0.29 ± 0.02 | 1.08 ± 0.11 | 3.65 ± 0.32 | 0.008 ** |
| GSK3α | S21 | Inhibits activity | 0.46 ± 0.03 | 1.31 ± 0.08 | 2.85 ± 0.20 | 0.008 ** |
| GSK3β | S9 | Inhibits activity | 0.67 ± 0.02 | 1.44 ± 0.12 | 2.15 ± 0.17 | 0.012 * |
| PDK1 | S241 | Induces activity | 0.63 ± 0.02 | 1.24 ± 0.08 | 1.97 ± 0.14 | 0.013 * |
| PTEN | S370 | Inhibits activity | 0.40 ± 0.05 | 1.85 ± 0.08 | 4.65 ± 0.39 | 0.008 ** |
| mTOR/S6K signaling | ||||||
| 4E-BP1 | T36 | Inhibits activity | 0.95 ± 0.03 | 1.77 ± 0.39 | 1.86 ± 0.35 | 0.075 |
| EIF4E | S209 | Inhibits molecular interaction | 0.10 ± 0.01 | 0.22 ± 0.01 | 2.12 ± 0.24 | 0.038 * |
| mTOR | T2448 | Induces activity | 0.73 ± 0.07 | 1.64 ± 0.17 | 2.23 ± 0.29 | 0.038 * |
| p70S6K | T421/S424 | Induces activity | 0.61 ± 0.02 | 0.94 ± 0.09 | 1.54 ± 0.17 | 0.055 |
| PRAS40 | T246 | Inhibits activity | 0.63 ± 0.04 | 1.38 ± 0.16 | 2.19 ± 0.26 | 0.026 * |
| rpS6 | S235/236 | Induces activity | 0.87 ± 0.08 | 2.21 ± 0.06 | 2.55 ± 0.19 | 0.014 * |
| FoxO signaling | ||||||
| FOXO3 | S413 | Induces activity | 0.02 ± 0.00 | 0.04 ± 0.01 | 2.12 ± 0.39 | 0.074 |
| p27 | T198 | Inhibits molecular interaction | 0.38 ± 0.05 | 1.00 ± 0.10 | 2.61 ± 0.38 | 0.038 * |
| AMPK signaling | ||||||
| AMPKα | T172 | Induces activity | 0.65 ± 0.03 | 1.55 ± 0.09 | 2.38 ± 0.16 | 0.009 ** |
| LKB1 | S428 | Induces activity | 0.44 ± 0.05 | 1.00 ± 0.05 | 2.24 ± 0.25 | 0.036 * |
| p53 | S15 | Induces activity | 0.60 ± 0.03 | 1.70 ± 0.13 | 2.85 ± 0.20 | 0.007 * |
| MAPK signaling | ||||||
| ERK1/2 | T202/Y204 Y185/187 | Induces activity | 0.50 ± 0.02 | 1.51 ± 0.10 | 2.99 ± 0.19 | 0.006 ** |
| Raf-1 | S301 | Inhibits activity | 0.51 ± 0.06 | 1.48 ± 0.11 | 2.88 ± 0.33 | 0.022 * |
| RSK1 | S380 | Induces protein degradation | 0.49 ± 0.03 | 1.19 ± 0.05 | 2.45 ± 0.15 | 0.008 ** |
| RSK2 | S386 | Induces activity | 0.71 ± 0.03 | 1.38 ± 0.05 | 1.96 ± 0.11 | 0.009 ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rebollo-Hernanz, M.; Aguilera, Y.; Martin-Cabrejas, M.A.; Gonzalez de Mejia, E. Phytochemicals from the Cocoa Shell Modulate Mitochondrial Function, Lipid and Glucose Metabolism in Hepatocytes via Activation of FGF21/ERK, AKT, and mTOR Pathways. Antioxidants 2022, 11, 136. https://doi.org/10.3390/antiox11010136
Rebollo-Hernanz M, Aguilera Y, Martin-Cabrejas MA, Gonzalez de Mejia E. Phytochemicals from the Cocoa Shell Modulate Mitochondrial Function, Lipid and Glucose Metabolism in Hepatocytes via Activation of FGF21/ERK, AKT, and mTOR Pathways. Antioxidants. 2022; 11(1):136. https://doi.org/10.3390/antiox11010136
Chicago/Turabian StyleRebollo-Hernanz, Miguel, Yolanda Aguilera, Maria A. Martin-Cabrejas, and Elvira Gonzalez de Mejia. 2022. "Phytochemicals from the Cocoa Shell Modulate Mitochondrial Function, Lipid and Glucose Metabolism in Hepatocytes via Activation of FGF21/ERK, AKT, and mTOR Pathways" Antioxidants 11, no. 1: 136. https://doi.org/10.3390/antiox11010136