1. Introduction
Titanium alloys were developed in the mid-1940s for the aviation industry. Two post-World War II alloys, commercially pure titanium (CPTi) and Ti-6Al-4V, remain the two dominant titanium alloys used in applications in aerospace, aeronautics, biomedical and automotive industries due to the density and mechanical and corrosion resistance properties being higher in comparison with competing materials such as aluminum, steels, and superalloys [
1,
2]. The use of these alloys increased significantly in the 1980s, particularly in aircraft combat construction as opposed to transport aircraft. This increase continued in the 1990s to the stage when, for combat aircraft, the percentage of titanium alloy as a fraction of the structural weight was the same order as that of aluminum alloy. The aviation industry demands improvements in the characteristics of the structural and functional materials components of aircraft based on scientific research conducted on new materials [
1,
2,
3,
4].
The aeronautical industry has an important role in the development and application of new materials and technologies because damage tolerance is especially low in this industry. For this reason, materials should present excellent properties for service conditions, and constants optimization processes should be carried out to increases mechanical, fatigue, corrosion, and oxidation resistance, which should be certificated and satisfy security standards [
3,
4].
Titanium alloys (Ti-alloys) can be divided into four types, Ti α, near to α, α + β, and metastable β, where the microstructure depends on the β stabilizer (Mo, V, Cr, Ni, Fe, Ta) [
5]. α-Ti alloys are denominated as commercially pure (CP) and highly pure titanium, and these alloys could have interstitial elements such as oxygen and nitrogen, which increase the mechanical resistance of titanium but decrease the ductility; for some cryogenic or high-temperature applications, α alloys add Al, Zr, or Sn as α stabilizers [
6]. If near α alloys combine α and α + β alloys properties, high temperature resistance, and high mechanical resistance, they would have a 2% β stabilizer. Some new generations add Si (0.1–0.5%) to improve their properties at high temperatures [
7,
8,
9]. α + β alloys have more than one stabilizer α-phase (such as interstitial) and β-phase until 6%, and this type of phase is the most common, Ti-6Al-4V is the most used Ti-alloy in the world, with almost 50% of production [
7]. β-Ti alloys have a high percentage of β stabilizers, and these alloys can present a martensitic microstructure. For this reason, the microstructure could be more complex [
8,
9,
10].
Titanium and its alloys present a high corrosion resistance, making them a good option for many applications such as those in the biomedical, aerospace, aeronautic, and chemical industries. The corrosion resistance of Ti alloys is due to the formation of an oxide film on the surface. This is because these kinds of alloys are reactive with oxygen, and in air or aqueous media, the layer’s reaction is spontaneous [
11,
12]. Although a passive layer can protect the titanium surface, the quality of this one will depend on factors such as the temperature and pH of the electrolyte in the redox reaction. A correct combination of these factors could create layers that are stable, continuous, and with good surface adherence [
13,
14]. If the oxide layer is penetrated or presents damage, it can regenerate in the presence of oxygen by repassivation. Titanium’s active condition cannot be maintained, and a process of mass transference in the exposed zone occurs due to the pH difference, oxygen admission, or change potential in the metal/electrolyte, provoking the repassivation [
14,
15]. However, ions of Cl
− or Br
− can penetrate and create an unstable layer, making the repassivation process more difficult [
16,
17,
18]. Further, the difference between titanium phases creates a difference in the homogeneity of oxide layer growth [
19].
Different conventional electrochemical techniques such as linear polarization resistance (LPR), potentiodynamic polarization (PP), and electrochemical impedance spectroscopy (EIS) have been implemented to determine the corrosion and kinetic mechanisms of the reactions. However, these techniques can alter the electrochemical system with external signals in electrochemical measurements [
20,
21,
22,
23,
24,
25]. The use of the electrochemical noise (EN) technique for investigation and monitoring of corrosion has allowed many advances in recent years that are interesting for corrosion science. A special advantage of EN measurements is the possibility to detect and analyze the early stages of localized corrosion.
Electrochemical noise describes spontaneous low-level potential and current fluctuations that occur during an electrochemical process. During the corrosion process, predominantly electrochemical cathodic and anodic reactions can cause small transients in electrical charges on the electrode. These transients manifest in the form of potential and current noise that can be exploited in a corrosion map [
25,
26,
27,
28]. Transients are linked to anodic and cathodic reactions as a result of stochastic processes (rupture and re-passivation of the passive film) and deterministic processes (formation and propagation of pitting) [
29,
30]. Potential and/or current transients in time series are associated with initiation and re-passivation of metastable pitting, which provides useful information on the initial process of localized corrosion. EN data can be analyzed by several methods. Perhaps the most used are those related to the frequency domain (power spectral density), time domain (statistical methods such as skewness, kurtosis, localization index (LI), and the variation of in the signal amplitude with time), and time-frequency domains [
31,
32,
33,
34]. LI, skewness, and kurtosis values have been reported as values related to different corrosion types and values referring to the asymmetry of distribution and shape of EN data [
31,
35,
36].
Research has found that statistical analysis has limitations in determining the corrosion type presented in the system. To reduce the uncertainty of statistical methods, it is necessary to employ different and new analysis methods to determine the corrosion mechanism. Wavelet and Hilbert–Huang Transform (HHT) methods are employed in EN chaotic signals analysis. Wavelets help in removing the DC signal from EN data without affecting the original signal (as a polynomial filter that can remove corrosion data) and can establish the corrosion type that occurs on the material surface. HHT can remove the DC signal without affecting the corrosion data and additionally localize the moment systems demand more energy, and the frequency which occurs can be determined by the corrosion mechanisms based on time–frequency–energy analysis [
23,
37,
38,
39,
40,
41,
42].
For the corrosion behavior of Ti-alloys in an NaCl electrolyte, it has been reported that corrosion resistance increases when the passive layer is present because it is difficult for Cl
− ions to pass through when studied by potentiodynamic polarization [
42]. EIS results indicate that it is the same behavior, namely that Cl
− ions have difficulty penetrating the passive layer, which could create instability on the layer, but it will show repassivation because it can present a diffusion process [
43]. In electrolytes of chlorides of Na, Ca, and Mg, titanium presents a growth-passive layer; these electrolytes present instability of Ti-alloys near α, α + β and β, and phases differences [
44,
45,
46]. In H
2SO
4 electrolytes, Ti-alloys have shown a noble behavior by PP and a passivation system [
47].
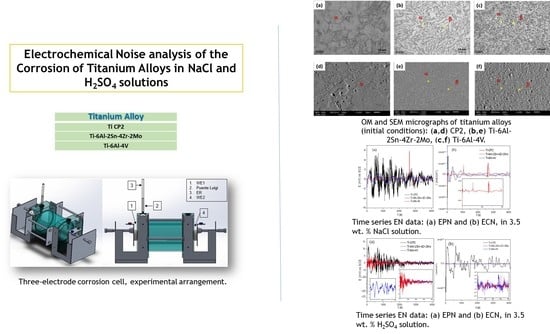
The aim of this research was to study the corrosion behavior of three Ti-alloys, Ti CP2, Ti-6Al-2Sn-4Zr-2Mo, and Ti-6Al-4V, immersed at 3.5 wt. % in H2SO4 and NaCl solutions at room temperature by electrochemical noise technique. Characterization by electrochemical techniques of titanium alloys could find potential applications in the aeronautical industry as in turbine blades and aircraft landing gear.
4. Discussion
Microstructural analysis revealed the porosity in Ti CP2 and Ti-6Al-4V samples, which causes loss of mechanical properties because pores are stress concentrators [
70]. Seah et al. [
71] conclude that although porosity makes material susceptible to localized corrosion, it can also repassivate.
The corrosion resistance of titanium and its alloys depends on the chemical composition of the material. Suitable alloying elements play a major role in the corrosion resistance of these alloys. The porosity of alloys is a compromise between mechanical strength and adequate pore size to obtain certain operating properties [
70].
Nevertheless, an increase in the porosity of metals leads to a lower corrosion potential value, which results in increased susceptibility of porous materials to localized corrosion. It is assumed that the relatively small pores present in the cell walls favor electrolyte placement and oxygen depletion, which is important in the stability and preservation of the oxide layer on titanium. Elements with higher porosity, with open and interconnected pores, allow easier electrolyte flow, which complements the oxygen supply during the passivation process [
72].
Porous titanium showed higher values of
Ecorr when compared to solid counterparts, most probably because of surface oxidation [
72]. Dabrowski et al. [
73] obtained similar results. They found that the influence of porosity of titanium ranged from 45 to 75% on corrosion resistance and showed that material with higher porosity exhibited less susceptibility to corrosion than those of 45% porosity. However, both elements exhibited lower corrosion resistance than the solid Ti [
73]. Chen et al. confirmed that the electrolyte flow can also decrease corrosion rates of solid and porous materials [
74].
In this research, porosity affects homogenous passive layer generation. As pores are high-energy zones, current will accumulate in those areas generating transient. Ti-6Al-2Sn-4Zr-2Mo was the alloy with higher porosity (2.88%), and in NaCl electrolyte, it presents difficulties creating a homogenous passive layer; porosity is one of the factors that interfere with developing a continuous oxide layer. Even though pores affect the passive layer generation, is important to mention that all materials naturally present a porosity percentage that is linked with the manufacturing process. Further, pore diameters are in the order of 1–2 µm, minimizing influence in corrosion behavior. Porosity could be considered as a major impact factor in materials manufacturing by powder metallurgy or select laser melting, where porosity diameter and percentage are higher than in the forging process.
All titanium alloys are susceptible to porosities, and pores’ diameters influence electrochemical behavior. In pores with diameters between 10–100 µm, electrochemical processes of bubbles formation by gas evolution were registered [
75,
76]. For electrochemical noise analysis, Huet [
76] concluded that oxygen evolution generated by pores is represented as fluctuations in PSD. If roughness or pores diameters increase, power spectral order will increase.
Several EN procedures correlating timed dependent fluctuation of current and potential during the corrosion process have been used to indicate the type of corrosion occurring. For instance, it is well recognized that the main source of electrochemical noise is the passive film breakdown process and repassivation process. This is why it is important to study the behavior of titanium alloys with aeronautical applications, since several components are exposed in saline environments and environmental contamination with sulfuric acid.
Recent research in electrochemical characterization of Ti-alloys showed a good corrosion resistance with electrochemical techniques such as PP and EIS. Moreover, Ti alloys do not present damage on the surface [
7,
77]. Researchers have also found that titanium is not affected by pitting corrosion in salt media when the temperature is under 90 °C, that Cl
− ions will not penetrate the surface of alloys, and that the alloy shows passivation [
17,
46,
78,
79,
80,
81,
82,
83,
84,
85].
Electrochemical noise cathodic and anodic reactions cause changes in material surfaces: when Ti-alloys are exposed to aqueous solutions, independently of mix, the hydrogen will react by an evolution reaction [
83], given by the next chemical reaction:
This phenomenon occurs in cathodic reactions where hydrogen reacts. On the other hand, the growth of a passive layer in electrochemical conditions is represented according to the following equations [
84,
86]:
where the reaction forms TiO
2 and Ti
2O
3, whose oxides create a stable layer on the metal surface and protect the titanium against corrosion, and oxide development is a slow process. In addition, as was mentioned in the section on wavelets and HHT, in the research of Liu, aggressive ions like chloride, bromide, or sulfate accelerate the anodic process, and ions can migrate in a passive layer according to Liu et al., who showed that, in a test with Cl
−, those ions adhered to material surface together with oxides, as is represented by the chemical reaction [
87]:
Cl
− ions play the role as interstitial element, but as Cl
− is bigger than O, means that the ion will have difficulty sinking into the material, and it generates a diffusion of Cl
− on the surface [
43]. Analysis by wavelets and HHT showed high energy accumulations in zones of low frequencies, because a diffusion process of Cl
− occurred on the Ti-alloys surface. In the case of Ti-6Al-2Sn-4Zr-2Mo, EDP presented energy accumulation at middle frequencies, because Cl
− creates an instability in the TiO
2 layer, and as ions are bigger, it creates changes in electron flows like last the chemical equation shown, but as time advances, the process of instability changes to a controlled process of diffusion. Furthermore, SEM-EDS from
Figure 8c shows how Cl
− is present on the surface, corroborating that diffusion of that element occurs in Ti alloys’ surface.
Factors that permit a non-uniform current distribution on the Ti surface is the inhomogeneity of the alloy according to Yang et al. [
88]; however, a passive film is formed, and this reduces the corrosion kinetic of alloys and increases material life [
89].
Dubent and Mazard [
47] presented that Ti CP2 in H
2SO
4 formed a protective layer that contributed to reduced corrosion rate, but the electrolyte continues to be aggressive. This converges with the results from PSD, where Ti CP2 dissolution presents a value of −104 dBi in H
2SO
4, while in NaCl, the value is −122 dBi. This occurs due to the aggressiveness of redox reaction of H
2SO
4 shown in the next equations, parting from water dissociation [
87,
88,
89,
90]:
According to these reactions, titanium will be dissolved due to the aggressiveness of electrolytes. An H
2 was developed, meaning hydrogen evolved and pH of electrolyte changed. In Ti-6Al-4V, Nabhani et al. [
91] propose that a passive layer is formed on the surface; this layer is formed in a major part of TiO
2 and presents aluminum and vanadium oxides concentrated in phases α and β, respectively; moreover, in the same research, Nabhani concluded that the presence of vanadium oxide increases the corrosion rate because the structure of this oxide could create vacancies and the ions penetration is easier. Wang et al. [
92] report that the passive layer is generated instantaneously, and it also decreases when Cl
− is added, which affects the generation of a passive layer. When Engelkamp [
93] performed experiments with H
2SO
4 added to H
3PO, they demonstrated that when H
2SO
4 is pure, passive layer breakdown is common, as was shown in HHT for that electrolyte in this research. Wang’s and our results confirm that some ions are more severe in Ti alloys, especially Cl
− (and all halides) [
94] because provoking the dissolution of passive-layer H
2SO
4 can generate more material dissolution; however, over a large amount of time, Cl
− can provoke more damage because it prevents the passive layer from being developed, and Cl
− presents a diffusion process. Fattath et al. [
95] also presented a passivation system for Ti alloys under H
2SO
4, which confirms the existence of a passive layer but further confirms the development of a porous layer considering a natural process, this porousness creates instability and changes in measurements.
Beck, Blackburn, and Gao [
96,
97] determined that titanium is susceptible to Cl
− ions, which confirms what was mentioned in the last paragraph; in NaCl electrolyte, the behavior of the passive layer is attributed to Cl
−, which avoids a uniform formation of the layer.
To determine corrosion mechanisms, some authors use statistical methods to related
Rn and
Rp. Al-Mazeedi and Cottis in 2004 [
98] mentioned that it is not possible to realize a measure of
Icorr but that it is possible to estimate a measure from EPN and ECN data, concluding that the use of
Rn to calculate a relation with corrosion rate is correct. However, in 2005, Cottis, Sanchez-Amaya, and Botana [
99] used
Rn to differentiate between corrosion activities in different media and not as an indicator of corrosion rate. In this case,
Rn presents changes in values for Ti alloys in NaCl related to the time series fluctuations and transients; therefore,
Rn’s value is higher in NaCl than in H
2SO
4 because NaCl presents more change in transients due to passive layer instability. This is because
Rn depends on
σE and
σI, and a great variation of
σ is associated to a high amplitude transients or fluctuations. Only Ti-6Al-2Sn-4Zr-2Mo presents an
Rn higher in H
2SO
4 than in NaCl, which can be attributed to the breakdowns and regeneration of oxide layer created on the surface.
The application of statistical results to determine mechanism was discussed previously by Cottis, Eden, Turgoose, Sun, Mansfeld, Botana, Gaona-Tiburcio, Sanchez Amaya [
29,
30,
54,
58,
97,
98,
99,
100,
101,
102,
103], and looking for a better method, Eden proposed an analysis by skewness and kurtosis and patented it with Reid [
56]. Authors agree with the higher exactitude of skewness and kurtosis compared to the localization index method (LI) [
65,
98,
102,
103,
104,
105,
106], but this also generated discrepancies due to the limitations of statistical analysis for chaotic systems. For this reason, diverse authors [
31,
39,
57,
61,
102,
103,
104,
105,
106,
107] recommend using this method with discretion. The results of skewness and kurtosis presented divergences in NaCl, while skewness showed uniform corrosion results and kurtosis presented pitting, making a divergence in results. This could be explained by the great number of transients of cathodic and anodic reactions. Even though statistical results did not show concordance, they can explain other phenomena: high kurtosis number indicated the presence of transients of high amplitude and high
σ, and, in the case of metals, then generated a passive film. A high kurtosis means instability, or many processes occurring on the surface. On the other hand, skewness is helpful to determine if a cathodic or anodic phenomenon predominates the corrosion system: when current is analyzed, if skewness is positive, it will indicate that cathodic reactions predominate; if it is negative, it indicates that anodic reactions prevail. This behavior is inversely reciprocal for voltage analysis. Skewness is negative only for TiCP2, meaning a predominance of anodic reactions is associated with electron losses; on the other hand, Ti-6Al-2Sn-4Zr-2Mo presented positive skewness, generating electrons gain.
Results of PSD give great information about material [
27,
28,
29,
30] dissolution that cannot be obtained by statistical methods, Ti-6Al-2Sn-4Zr-2Mo presented lower values Ψ
0 in NaCl and H
2SO
4, while Ti CP2 has the higher dissolution material values, denoting that although passivation occurs, the passive layer does not protect the H
2SO
4 reaction, associated with hydrogen evolution and change in electrolyte pH, which increases the aggressiveness. To analyze PSD slope, it is necessary to be careful, because, for some authors, it cannot be mechanistic [
58]. However, if PSD is analyzed by the change in slopes around all the frequencies, it can provide useful information, as Uruchurtu [
63] proposed that slope change means a change in the process if the change slope is around limit frequencies, signifying a charge to passivity or a decrease in pitting rate. All samples showed the change of slope at limit frequency, which means a change to alloy passivity.
Wavelets are a useful technique to analyses complex systems. Statistical and PSD slopes do not give certain information about corrosion mechanisms, and an algorithm to decompose signals has been offered [
69]. Results showed an energy accumulation in crystals D7 and D8, Crystal S8 is related to DC, and crystals of approximation cannot present DC energy [
108]. According to Li, when D8 crystal has the highest energy accumulations, it is because a passive film was formatted, and when energy is higher in crystals D4, D5, and D6, it is associated with a repassivation process, as shown when Ti-6Al-2Sn-4Zr-2Mo are in an NaCl solution. Smith and Macdonald declared that wavelets analysis was able to identify the pitting systems in 2009 and questioned whether it is possible to determine a generalized or passive system. Li, Moshrefi, Lara-Banda, Chui, and Liu [
35,
108,
109,
110,
111,
112,
113,
114,
115,
116] agree with the method to determine passivation and metastable pitting. Asfia et al. also determined that is a good option to eliminate trend without eliminating corrosion data [
105,
114,
117] and that it is helpful to determine corrosion mechanisms in distinct media [
118,
119,
120]. Various authors indicate that the interpretation of wavelet is more useful to determinate corrosion mechanism that statistical methods [
110,
111].
HHT is a technique that can be used in normal or microbiological corrosion processes, and a low-frequency process indicates a passive system [
114]. For localized processes, Hilbert spectra show energy accumulation at middle and high frequencies (10
0 and 10
−1 Hz). A process of metastable pitting/repassivation and a localized corrosion process can also be identified [
119,
120]. All these asseverations from authors about energy distributed at middle frequencies [
34,
48,
69,
109,
118,
120] correspond to a repassivation process as proven in
Figure 6 and
Figure 7, where Ti-alloys presented repassivation, because, for an unstable passive layer, NaCl and H
2SO
4 are created. However, this method is more complete than statistical analysis in finding the corrosion mechanism and not only the corrosion type. Further, given the information about the moment in that a corrosion process begins, this could be a pitting, repassivation, diffusion, or passivation, and it can be related to time-series of ECN and EPN, depending on which one elaborated the Hilbert spectra. Wavelets and HHT presented concordance in results about corrosion mechanism.