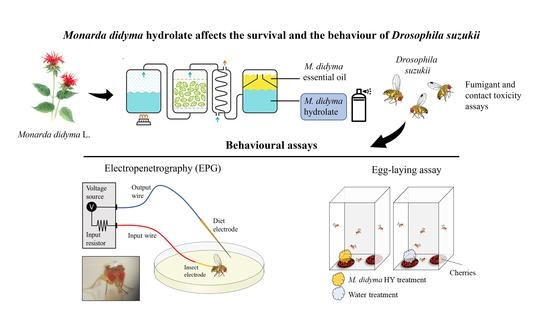
Monarda didyma Hydrolate Affects the Survival and the Behaviour of Drosophila suzukii
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Insect Rearing
2.2. Monarda didyma: Plant Material and HY Extraction
2.3. M. didyma HY Composition
2.4. M. didyma HY Fumigant Toxicity Assay
2.5. M. didyma HY Contact Toxicity Assay
2.6. Survival Assay
2.7. Gene Expression Analysis after M. didyma Treatment
2.8. Dye-Labelling Food Intake Assay
2.9. Feeding Behaviour by Electropenetrography (EPG)
2.10. Laboratory Egg-Laying Assay
2.11. Cherry Oviposition Assay
2.12. Statistical Analyses
3. Results
3.1. M. didyma HY Composition
3.2. M. didyma HY Fumigant and Contact Toxicity Assay
3.3. Effect of M. didyma Contact Exposure on Survival and Detoxification Enzyme Expression
3.4. Dye-Labelling Food Intake Assay
3.5. Feeding Behaviour by Electrical Penetration Graph (EPG)
3.6. Egg-Laying Assays
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Robu, V.; Covaci, G.; Popescu, I.M. The use of essential oils in organic farming. Res. J. Agric. Sci. 2015, 47, 134. [Google Scholar]
- Isman, M.B. Botanical insecticides in the twenty-first century—Fulfilling their promise? Annu. Rev. Entomol. 2020, 65, 233–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, Y.; Harmon, P.F.; Treadwell, D.D.; Carrillo, D.; Sarkhosh, A.; Brecht, J.K. Biocontrol potential of essential oils in organic horticulture systems: From farm to fork. Front. Nutr. 2022, 8, 805138. [Google Scholar] [CrossRef] [PubMed]
- Sarac, A.; Tunc, I. Toxicity of essential oil vapours to stored-product insects. Z. Pflanzenkrankh. Pflanzenschutz 1995, 102, 69–74. [Google Scholar]
- Shaaya, E.; Kostjukovsky, M. Efficacy of phyto-oils as contact insecticides and fumigants for the control of stored-product insects. In Insecticides with Novel Modes of Action. Mechanism and Application; Ishaaya, I., Degheele, D., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; pp. 171–187. [Google Scholar] [CrossRef]
- Isman, M.B. Botanical insecticides, deterrents, and repellents in modern agriculture and an increasingly regulated world. Annu. Rev. Entomol. 2006, 51, 45–66. [Google Scholar] [CrossRef] [Green Version]
- Haddi, K.; Turchen, L.M.; Viteri Jumbo, L.O.; Guedes, R.N.; Pereira, E.J.; Aguiar, R.W.; Oliveira, E.E. Rethinking biorational insecticides for pest management: Unintended effects and consequences. Pest Manag. Sci. 2020, 76, 2286–2293. [Google Scholar] [CrossRef]
- Turchen, L.M.; Cosme-Júnior, L.; Guedes, R.N.C. Plant-derived insecticides under meta-analyses: Status, biases, and knowledge gaps. Insects 2020, 11, 532. [Google Scholar] [CrossRef]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef]
- Werdin González, J.O.; Gutiérrez, M.M.; Murray, A.P.; Ferrero, A.A. Biological activity of essential oils from Aloysia polystachya and Aloysia citriodora (Verbenaceae) against the soybean pest Nezara viridula (Hemiptera: Pentatomidae). Nat. Prod. Commun. 2010, 5, 301–306. [Google Scholar] [CrossRef]
- Werdin González, J.O.; Gutiérrez, M.M.; Murray, A.P.; Ferrero, A.A. Composition and biological activity of essential oils from Labiatae against Nezara viridula (Hemiptera: Pentatomidae) soybean pest. Pest Manag. Sci. 2011, 67, 948–955. [Google Scholar] [CrossRef]
- Brügger, B.P.; Martínez, L.C.; Plata-Rueda, A.; Castro, B.; Soares, M.A.; Wilcken, C.F.; Carvalho, A.G.; Serrão, J.E.; Zanuncio, J.C. Bioactivity of the Cymbopogon citratus (Poaceae) essential oil and its terpenoid constituents on the predatory bug, Podisus nigrispinus (Heteroptera: Pentatomidae). Sci. Rep. 2019, 9, 8358. [Google Scholar] [CrossRef] [PubMed]
- Walsh, D.B.; Bolda, M.P.; Goodhue, R.E.; Dreves, A.J.; Lee, J.C.; Bruck, D.J.; Walton, V.M.; O’neal, S.D.; Zalom, F.G. Drosophila suzukii (Diptera: Drosophilidae): Invasive pest of ripening soft fruit expanding its geographic range and damage potential. J. Integr. Pest Manag. 2011, 1, G1–G7. [Google Scholar] [CrossRef]
- Asplen, M.K.; Anfora, G.; Biondi, A.; Choi, D.S.; Chu, D.; Daane, K.M.; Desneux, N. Invasion biology of spotted wing Drosophila (Drosophila suzukii): A global perspective and future priorities. J. Pest Sci. 2015, 88, 469–494. [Google Scholar] [CrossRef]
- Tait, G.; Mermer, S.; Stockton, D.; Lee, J.; Avosani, S.; Abrieux, A.; Anfora, G.; Beers, E.; Biondi, A.; Burrack, H.; et al. Drosophila suzukii (Diptera: Drosophilidae): A decade of research towards a sustainable integrated pest management program. J. Econ. Entomol. 2021, 114, 1950–1974. [Google Scholar] [CrossRef]
- Cini, A.; Ioriatti, C.; Anfora, G. A review of the invasion of Drosophila suzukii in Europe and a draft research agenda for integrated pest management. Bull. Insectol. 2012, 65, 149–160. [Google Scholar]
- Lee, J.C.; Bruck, D.J.; Curry, H.; Edwards, D.; Haviland, D.R.; Van Steenwyk, R.A.; Yorgey, B.M. The susceptibility of small fruits and cherries to the spotted-wing Drosophila, Drosophila suzukii. Pest Manag. Sci. 2011, 67, 1358–1367. [Google Scholar] [CrossRef]
- Briem, F.; Eben, A.; Gross, J.; Vogt, H. An invader supported by a parasite: Mistletoe berries as a host for food and reproduction of Spotted Wing Drosophila in early spring. J. Pest Sci. 2016, 89, 749–759. [Google Scholar] [CrossRef]
- Kenis, M.; Tonina, L.; Eschen, R.; van der Sluis, B.; Sancassani, M.; Mori, N.; Haye, T.; Helsen, H. Non-crop plants used as host by Drosophila suzukii in Europe. J. Pest Sci. 2016, 89, 735–748. [Google Scholar] [CrossRef] [Green Version]
- Haye, T.; Girod, P.; Cuthbertson, A.G.S.; Wang, X.G.; Daane, K.M.; Hoelmer, K.A.; Baroffio, C.; Zhang, J.P.; Desneux, N. Current SWD IPM tactics and their practical implementation in fruit crops across different regions around the world. J. Pest Sci. 2016, 89, 643–651. [Google Scholar] [CrossRef]
- Civolani, S.; Vaccari, G.; Caruso, S.; Finetti, L.; Bernacchia, G.; Chicca, M.; Cassanelli, S. Evaluation of insecticide adaptive response in Italian population of Drosophila suzukii. Bull. Insectol. 2021, 74, 103–114. [Google Scholar]
- Sial, A.A.; Roubos, C.R.; Gautam, B.K.; Fanning, P.D.; Van Timmeren, S.; Spies, J.; Petran, A.; Rogers, M.A.; Liburd, Q.E.; Little, B.A.; et al. Evaluation of organic insecticides for management of spotted-wing drosophila (Drosophila suzukii) in berry crops. J. Appl. Entomol. 2019, 143, 593–608. [Google Scholar] [CrossRef]
- Park, C.G.; Jang, M.; Yoon, K.A.; Kim, J. Insecticidal and acetylcholinesterase inhibitory activities of Lamiaceae plant essential oils and their major components against Drosophila suzukii (Diptera: Drosophilidae). Ind. Crops Prod. 2016, 89, 507–513. [Google Scholar] [CrossRef]
- Park, C.; Jang, M.; Shin, E.; Kim, J. Myrtaceae plant essential oils and their β-triketone components as insecticides against Drosophila suzukii. Molecules 2017, 22, 1050. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dam, D.; Molitor, D.; Beyer, M. Natural compounds for controlling Drosophila suzukii. A review. Agron. Sustain. Dev. 2019, 39, 53. [Google Scholar] [CrossRef]
- Kim, J.; Jang, M.; Shin, E.; Kim, J.; Lee, S.H.; Park, C.G. Fumigant and contact toxicity of 22 wooden essential oils and their major components against Drosophila suzukii (Diptera: Drosophilidae). Pestic. Biochem. Physiol. 2016, 133, 35–43. [Google Scholar] [CrossRef] [PubMed]
- de Souza, M.T.; de Souza, M.T.; Bernardi, D.; Krinski, D.; de Melo, D.J.; da Costa Oliveira, D.; Rakes, M.; Zarbin, P.H.G.; de Noronha Sales Maia, B.H.L.; Zawadneak, M.A.C. Chemical composition of essential oils of selected species of Piper and their insecticidal activity against Drosophila suzukii and Trichopria anastrephae. Environ. Sci. Pollut. Res. 2020, 27, 13056–13065. [Google Scholar] [CrossRef] [PubMed]
- Erland, L.A.E.; Rheault, M.R.; Mahmoud, S.S. Insecticidal and oviposition deterrent effects of essential oils and their constituents against the invasive pest Drosophila suzukii (Matsumara) (Diptera:Drosophilidae). Crop Prot. 2015, 78, 20–26. [Google Scholar] [CrossRef] [Green Version]
- Renkema, J.M.; Buitenhuis, R.; Hallett, R.H. Reduced Drosophila suzukii infestation in berries using deterrent compounds and laminate polymer flakes. Insects 2017, 8, 117. [Google Scholar] [CrossRef] [Green Version]
- Wallingford, A.; Helsen, S.P.; Cha, D.H.; Loeb, G.M. Behavioral response of spotted-wing drosophila, Drosophila suzukii Matsumara, to aversive odors and a potential oviposition deterrent in the filed: Drosophila suzukii deterrents. Pest Manag. Sci. 2016, 72, 701–706. [Google Scholar] [CrossRef]
- Finetti, L.; Ferrari, F.; Calò, G.; Cassanelli, S.; De Bastiani, M.; Civolani, S.; Bernacchia, G. Modulation of Drosophila suzukii type 1 tyramine receptor (DsTAR1) by monoterpenes: A potential new target for next generation biopesticides. Pestic. Biochem. Physiol. 2020, 165, 104549. [Google Scholar] [CrossRef]
- Finetti, L.; Tiedeman, L.; Zhang, X.; Civolani, S.; Bernacchia, G.; Roeder, T. Monoterpenes alter TAR1-driven physiology in Drosophila species. J. Exp. Biol. 2021, 224, jeb232116. [Google Scholar] [CrossRef] [PubMed]
- Acimovic, M.G.; Teševic, V.V.; Smiljanic, K.T.; Cvetkovic, M.T.; Stankovic, J.M.; Kiprovski, B.M.; Sikora, V.S. Hydrolates—Byproducts of essential oil distillation: Chemical composition, biological activity and potential uses. Adv. Mater. Technol. 2020, 9, 54–70. [Google Scholar] [CrossRef]
- Di Vito, M.; Smolka, A.; Proto, M.R.; Barbanti, L.; Gelmini, F.; Napoli, E.; Bellardi, M.G.; Mattarelli, P.; Beretta, G.; Sanguinetti, M.; et al. Is the antimicrobial activity of hydrolates lower than that of essential oils? Antibiotics 2021, 10, 88. [Google Scholar] [CrossRef] [PubMed]
- Sagdic, O.; Ozcan, M. Antibacterial activity of Turkish spice hydrosols. Food Control 2003, 14, 141–143. [Google Scholar] [CrossRef]
- Rabha, B.; Gopalakrishnam, R.; Baruah, I.; Singh, L. Larvicidal activity of some essential oil hydrolates against dengue and filariasis vectors. J. Med. Res. 2012, 1, 14–16. [Google Scholar]
- Wang, T.H.; Hsia, S.M.; Wu, C.H.; Ko, S.Y.; Chen, M.Y.; Shis, Y.H.; Shien, T.H.; Chuang, L.C.; Wu, C.Y. Evaluation of the antibacterial potential of liquid and vapor phase phenolic essential oil compounds against oral microorganisms. PLoS ONE 2016, 11, e0163147. [Google Scholar] [CrossRef] [Green Version]
- Garzoli, S.; Petralito, S.; Ovidi, E.; Turchetti, G.; Laghezza Masci, V.; Tiezzi, A.; Trilli, J.; Cesa, S.; Antonietta Casadei, M.; Giacomello, P.; et al. Lavandula x intermedia oil and hydrolate: Evaluation of chemical composition and antibacterial activity before and after formulation in nanoemulsion. Ind. Crops Prod. 2020, 145, 112068. [Google Scholar] [CrossRef]
- Dindo, M.L.; Modesto, M.; Rossi, C.; Di Vito, M.; Burgio, G.; Barbanti, L.; Mattarelli, P. Monarda fistulosa hydrolate as antimicrobial agent in articial media for the in vitro rearing of the tachidin parasitoid Exorist larvarum. Entomol. Exp. Appl. 2021, 169, 79–89. [Google Scholar] [CrossRef]
- Petrakis, E.A.; Kimbaris, A.C.; Lykouressis, D.P.; Polissiou, M.G.; Ch Perdikis, D. Hydrosols evaluation in pest control: Insecticidal and settling inhibition potential against Myzus persicae (Sulzer). J. Appl. Entomol. 2015, 139, 260–267. [Google Scholar] [CrossRef]
- Zekri, N.; Handaq, N.; El Caidi, A.; Zair, T.; El Belghiti, M.A. Insecticidal effect of Mentha pulegium L. and Mentha suaveolens Ehrh. hydrosols against a pest of citrus, Toxoptera aurantia (Aphididae). Res. Chem. Intermed. 2016, 42, 1639–1649. [Google Scholar] [CrossRef]
- Oviedo, A.; Van Nieuwenhove, G.; Van Nieuwenhove, C.; Rull, J. Biopesticide effects on pupae and adult mortality of Anastrepha fraterculus and Ceratitis capitata (Diptera: Tephritidae). Austral Entomol. 2017, 57, 457–464. [Google Scholar] [CrossRef]
- Mattarelli, P.; Epifano, F.; Minardi, P.; Di Vito, M.; Modesto, M.; Barbanti, L.; Bellardi, M.G. Chemical composition and antimicrobial activity of essential oils from aerial parts of Monarda didyma and Monarda fistulosa cultivated in Italy. J. Essent. Oil-Bear. Plants 2017, 20, 76–86. [Google Scholar] [CrossRef]
- Laquale, S.; Avato, P.; Argentieri, M.P.; Bellardi, M.G.; D’Addabbo, T. Nematotoxic activity of essential oils from Monarda species. J. Pest Sci. 2018, 91, 1115–1125. [Google Scholar] [CrossRef]
- Mariani, V.; Turci, S.; Michelozzi, M.; Pollini, A.; Bellardi, M.G. Idrolati di Monarda didyma per la difesa delle piante dalle infestazioni di Trialeurodes vaporariorum. Natural 1 2019, 9, 60–66. [Google Scholar]
- Mariani, V.; Turci, S.; Pollini, A.; Biffi, S.; Cencetti, G.; Bellardi, M.G. Prove preliminari di efficacia insetticida dell’idrolato di Monarda didyma contro Trialeurodes vaporariorum. In Atti Giornate Fitopatologiche; IRIS: San Francisco, CA, USA, 2020; pp. 97–104. [Google Scholar]
- Dalton, D.T.; Walton, V.M.; Shearer, P.W.; Walsh, D.B.; Caprile, J.; Isaacs, R. Laboratory survival of Drosophila suzukii under simulated winter conditions of the Pacific Northwest and seasonal field trapping in five primary regions of small and stone fruit production in the United States. Pest Manag. Sci. 2011, 67, 1368–1374. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Zhai, Y.; Lin, Q.; Zhou, X.; Zhang, X.; Liu, T.; Yu, Y. Identification and validation of reference genes for quantitative real-time PCR in Drosophila suzukii (Diptera: Drosophilidae). PLoS ONE 2014, 9, e106800. [Google Scholar] [CrossRef]
- Guedes, R.N.C.; Cervantes, F.A.; Backus, E.A.; Walse, S.S. Electropenetrography of spotted wing drosophila (Drosophila suzukii) on pesticide-treated strawberry. J. Pest Sci. 2019, 93, 91–102. [Google Scholar] [CrossRef]
- Gou, B.; Zhu, E.; He, R.; Stern, U.; Yang, C.-H. High throughput assay to examine egg-laying preferences of individual Drosophila melanogaster. J. Vis. Exp. 2016, 109, e53716. [Google Scholar] [CrossRef] [Green Version]
- Bergé, J.B.; Feyereisen, R.; Amichot, M. Cytochrome P450 monooxygenases and insecticide resistance in insects. Philos. Trans. R. Soc. B 1998, 353, 1701–1705. [Google Scholar] [CrossRef] [Green Version]
- Tu, C.P.; Akgül, B. Drosophila glutathione S-transferases. Methods Enzymol. 2005, 401, 204–226. [Google Scholar] [CrossRef] [PubMed]
- Montella, I.R.; Schama, R.; Valle, D. The classification of esterases: An important gene family involved in insecticide resistance-a review. Memórias Inst. Oswaldo Cruz 2012, 107, 437–449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sarkic, A.; Stappen, I. Essential oils and their single compounds in cosmetics—A critical review. Molecules 2018, 5, 11. [Google Scholar] [CrossRef] [Green Version]
- Naghdi Badi, H.; Abdollahi, M.; Mehrafarin, A.; Ghorbanpour, M.; Tolyat, M.; Qaderi, A.; Ghiaci Yekta, M. An overview on two valuable natural and bioactive compounds, thymol and carvacrol, in medicinal plants. J. Med. Plants 2017, 16, 1–32. [Google Scholar]
- Kapelev, K.G. Results of introducing Monarda as an essential oil plant. Byulleten’ Gos. Nikitsk. Bot. Sada 1976, 2, 35–39. [Google Scholar]
- Marchioni, I.; Najar, B.; Ruffoni, B.; Copetta, A.; Pistelli, L.; Pistelli, L. Bioactive compounds and aroma profile of some Lamiaceae edible flowers. Plants 2020, 9, 691. [Google Scholar] [CrossRef] [PubMed]
- Baradat, P.; Marpeau, A.; Walter, J. Terpene markers. In Genetic Variation in European Populations of Forest Trees; Müller-Starck, G., Ziehe, M., Eds.; Sauerlandeťs: Frankfurt am Main, Germany, 1991; pp. 40–66. [Google Scholar]
- Hanover, J.W. Applications of terpene analysis in forest genetics. New For. 1992, 6, 159–178. [Google Scholar] [CrossRef]
- Langenheim, J.H. Higher plant terpenoids: A phytocentric overview of their ecological roles. J. Chem. Ecol. 1994, 20, 1223–1280. [Google Scholar] [CrossRef]
- Besser, K.; Harper, A.; Welsby, N.; Schauvinhold, I.; Slocombe, S.; Li, Y.; Dixon, R.A.; Broun, P. Divergent regulation of terpenoid metabolism in the trichomes of wild and cultivated tomato species. Plant Physiol. 2009, 149, 499–514. [Google Scholar] [CrossRef] [Green Version]
- Mukrimin, M.; Kovalchuk, A.; Ghimire, R.P.; Kivimäenpää, M.; Sun, H.; Holopainen, J.K.; Asiegbu, F.O. Evaluation of potential genetic and chemical markers for Scots pine tolerance against Heterobasidion annosum infection. Planta 2019, 250, 1881–1895. [Google Scholar] [CrossRef] [Green Version]
- Keesey, I.W.; Jiang, N.; Weißflog, J.; Winz, R.; Svatoš, A.; Wang, C.-H.; Hansson, B.S.; Knaden, M. Plant-Based natural product chemistry for integrated pest management of Drosophila suzukii. J. Chem. Ecol. 2019, 45, 626–637. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Z.; Yang, T.; Zhang, Y.; Wang, L.; Xie, Y. Fumigant toxicity of monoterpenes against fruit fly, Drosophila melanogaster. Ind. Crops Prod. 2016, 81, 147–151. [Google Scholar] [CrossRef]
- Priestley, C.M.; Williamson, E.M.; Wafford, K.A.; Satelle, D.B. Thymol, a constituent of thyme essential oils, is a positive modulator of human GABA and a homo-oligosteric GABA receptor from Drosophila melanogaster. Br. J. Pharmacol. 2003, 140, 1363–1372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Houghton, P.J.; Ren, Y.; Howes, M.J. Acetylcholinesterase inhibitors from plants and fungi. Nat. Prod. Rep. 2006, 23, 181–199. [Google Scholar] [CrossRef] [PubMed]
- Finetti, L.; Roeder, T.; Calò, G.; Bernacchia, G. The insect type 1 tyramine receptors: From structure to behaviour. Insects 2021, 12, 315. [Google Scholar] [CrossRef]
- Terhzaz, S.; Cabrero, P.; Brinzer, R.A.; Halberg, K.A.; Dow, J.A.; Davies, S.A. A novel role of Drosophila cytochrome P450-4e3 in permethrin insecticide tolerance. Insect Biochem. Mol. Biol. 2015, 67, 38–46. [Google Scholar] [CrossRef] [Green Version]
- Scanlan, J.L.; Gledhill-Smith, R.S.; Battlay, P.; Robin, C. Genomic and transcriptomic analyses in Drosophila suggest that the ecdysteroid kinase-like (EcKL) gene family encodes the ‘detoxification-by-phosphorylation’ enzymes of insects. Insect Biochem. Mol. Biol. 2020, 123, 103429. [Google Scholar] [CrossRef]
- Baldwin, S.R.; Mohapatra, P.; Nagalla, M.; Sindvani, R.; Amaya, D.; Dickson, H.A.; Menuz, K. Identification and characterization of CYPs induced in the Drosophila antenna by exposure to a plant odorant. Sci. Rep. 2021, 11, 20530. [Google Scholar] [CrossRef]
- Wang, Q.; Xu, P.; Sanchez, S.; Duran, P.; Andreazza, F.; Isaacs, R.; Dong, K. Behavioral and physiological responses of Drosophila melanogaster and D. suzukii to volatiles from Plant essential oils. Pest Manag. Sci. 2021, 77, 3698–3705. [Google Scholar] [CrossRef]
- Meng, L.-W.; Yuan, G.-R.; Lu, X.-P.; Jing, T.-X.; Zheng, L.-S.; Yong, H.-X.; Wang, J.-J. Two delta class glutathione S-transferases involved in the detoxification of malathion in Bactrocera dorsalis (Hendel). Pest Manag. Sci. 2019, 75, 1527–1538. [Google Scholar] [CrossRef]
- Tian, F.; Li, C.; Wang, Z.; Liu, J.; Zeng, X. Identification of detoxification genes in imidacloprid-resistant Asian citrus psyllid (Hemiptera: Lividae) and their expression patterns under stress of eight insecticides. Pest Manag. Sci. 2019, 75, 1400–1410. [Google Scholar] [CrossRef] [PubMed]
- De Souza, M.T.; de Souza, M.T.; Bernardi, D.; de Melo, D.J.; Gorgatti Zarbin, P.H.; Cassilha Zawadneak, M.A. Insecticidal and oviposition deterrent effects of essential oils of Baccharis spp. and histological assessment against Drosophila suzukii (Diptera: Drosophilidae). Sci. Rep. 2021, 11, 3944. [Google Scholar] [CrossRef] [PubMed]
- D’Addabbo, T.; Avato, P. Chemical composition and nematicidal properties of sixteen essential oils—A review. Plants 2021, 10, 1368. [Google Scholar] [CrossRef] [PubMed]
- Renkema, J.M.; Wright, D.; Buitenhuis, R.; Hallett, R.H. Plant essential oils and potassium metabisulfite as repellent for D. suzukii (Diptera: Drosophilidae). Sci. Rep. 2017, 6, 21432. [Google Scholar] [CrossRef] [PubMed]
- Ajayi, O.E.; Appel, A.G.; Fadamiro, H.Y. Fumigation toxicity of essential oil monoterpenes to Callobruchus maculatus (Coleoptera: Chrysomelidae: Bruchinae). J. Insects 2014, 2014, 917212. [Google Scholar] [CrossRef] [Green Version]
- Allsopp, E.; Prinsloo, G.; Smart, L.E.; Dewhirst, S.Y. Methyl salicylate, thymol and carvacrol as oviposition deterrents for Frankliniella occidentalis (Pergande) on plum blossoms. Arthropod-Plant Interact. 2014, 8, 421–427. [Google Scholar] [CrossRef]
- Damtie, D.; Mekonnen, Y. Toxicity and oviposition deterrent activities of thyme essential oils against Anopheles arabiensis. Psyche A J. Entomol. 2021, 2021, 6684156. [Google Scholar] [CrossRef]
- Marchetti, E.; Alberghini, S.; Battisti, A.; Squartini, A.; Baronio, P.; Dindo, M.L. Effects of conventional and transgenic Bacillus thuringiensis galleriae toxin on Exorista larvarum (Diptera: Tachinidae), a parasitoid of forest defoliating Lepidoptera. Biocontrol Sci. Technol. 2009, 19, 463–473. [Google Scholar] [CrossRef]
- Tan, K.H.; Nishida, R. Methyl eugenol: Its occurrence, distribution, and role in nature, especially in relation to insect behavior and pollination. J. Insect Sci. 2012, 12, 56. [Google Scholar] [CrossRef] [Green Version]
- Jankowska, M.; Łozowicka, B.; Kaczyński, P. Comprehensive toxicological study over 160 processing factors of pesticides in selected fruit and vegetables after water, mechanical and thermal processing treatments and their application to human health risk assessment. Sci. Total Environ. 2019, 20, 1156–1167. [Google Scholar] [CrossRef]
- Gaire, S.; Zheng, W.; Scharf, M.E.; Gondhalekar, A.D. Plant essential oil constituents enhance deltamethrin toxicity in a resistant population of bed bugs (Cimex lectularius L.) by inhibiting cytochrome P450 enzymes. Pestic. Biochem. Physiol. 2021, 175, 104829. [Google Scholar] [CrossRef] [PubMed]





| M. didyma HY | Slope ± s.d | LC50 (95% CI) | LC90 (95% CI) | Heterogeneity | χ2 |
|---|---|---|---|---|---|
| 24 h | 0.974 ± 0.099 | / | / | 8.24 | / |
| 48 h | 0.986 ± 0.097 | 5.03 µL mL−1 (2.08–10.11) | 100.04 µL mL−1 (36.28–274.74) | 2.83 | 11.514 |
| Probing | Mean Probing Duration per Insect (min) | Mean Number of Probing Events | Mean Duration of Probing Event (min) |
| 1000 µL mL−1 HY | 8.85 ± 1.75 | 94.46 ± 11.07 | 0.10 ± 0.02 |
| Control | 12.48 ± 2.22 | 157.91 ± 25.02 | 0.10 ± 0.02 |
| Statistical analysis | n.s. | * | n.s. |
| Non-Probing | Mean Non-Probing Duration per Insect (min) | Mean Number of Non-Probing Events | Mean Duration of Non-Probing Event (min) |
|---|---|---|---|
| 1000 µL mL−1 HY | 111.09 ± 1.74 | 122.42 ± 14.09 | 1.26 ± 0.18 |
| Control | 107.29 ± 2.21 | 197.39 ± 30.37 | 0.90 ± 0.17 |
| Statistical analysis | n.s. | * | * |
| Dabbing | Mean Dabbing Duration per Insect (s) | Mean Number of Dabbing Events | Mean Duration of Dabbing Event (s) |
| 1000 µL mL−1 HY | 9.31 ± 3.82 | 27.22 ± 4.68 | 0.50 ± 0.29 |
| Control | 10.17 ± 2.24 | 37.83 ± 8.42 | 0.27 ± 0.02 |
| Statistical analysis | n.s. | n.s. | * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Finetti, L.; Civolani, S.; Mirandola, D.; Benetti, L.; Francati, S.; Albanese, F.; Menicucci, F.; Michelozzi, M.; Bellardi, M.G.; Dindo, M.L.; et al. Monarda didyma Hydrolate Affects the Survival and the Behaviour of Drosophila suzukii. Insects 2022, 13, 280. https://doi.org/10.3390/insects13030280
Finetti L, Civolani S, Mirandola D, Benetti L, Francati S, Albanese F, Menicucci F, Michelozzi M, Bellardi MG, Dindo ML, et al. Monarda didyma Hydrolate Affects the Survival and the Behaviour of Drosophila suzukii. Insects. 2022; 13(3):280. https://doi.org/10.3390/insects13030280
Chicago/Turabian StyleFinetti, Luca, Stefano Civolani, Daniele Mirandola, Lorenzo Benetti, Santolo Francati, Federica Albanese, Felicia Menicucci, Marco Michelozzi, Maria Grazia Bellardi, Maria Luisa Dindo, and et al. 2022. "Monarda didyma Hydrolate Affects the Survival and the Behaviour of Drosophila suzukii" Insects 13, no. 3: 280. https://doi.org/10.3390/insects13030280