Membranes Based on Cellulose Nanofibers and Activated Carbon for Removal of Escherichia coli Bacteria from Water
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Palm Fruit Stalks Pulp
2.3. Isolation of Cellulose Nanofibers (CNF)
2.4. Isolation of TEMPO-Oxidized CNF
2.5. Characterization of CNF and TEMPO-Oxidized CNF
2.6. Preparation and Characterization of Activated Carbon
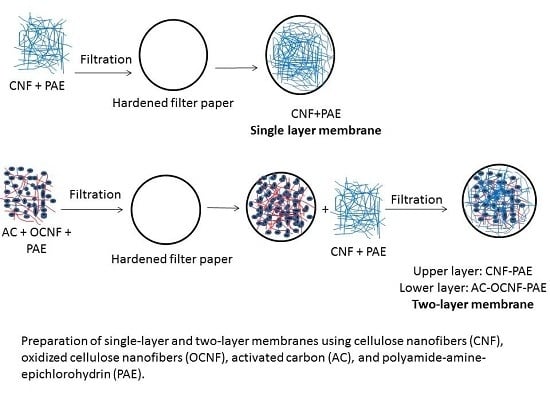
2.7. Nanocellulose Membranes
2.8. Test Microorganism Strain
2.9. Evaluation of Membranes Properties
2.9.1. Pure Water Flux
2.9.2. Rejection Efficiency
2.9.3. Antibacterial Resistance
3. Results and Discussion
3.1. Cellulose Nanofibers
3.2. Activated Carbon Prepared from Olive Stones
3.3. CNF and CNF/AC Membranes
3.4. Water Flux of Membranes
3.5. Removal Efficiency of E. coli Bacteria
3.6. Bacterial Resistance
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhao, Y.; Moser, C.; Lindström, M.E.; Henriksson, G.; Li, J. Cellulose nanofibers from softwood, hardwood, and tunicate: Preparation-structure-membrane performance interrelation. ACS Appl. Mater. Interfaces 2017, 9, 13508–13519. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.L. Bagasse and rice straw nanocellulosic materials and their applications. In Handbook of Polymer Nanocomposites. Processing, Performance and Application. Volume C: Polymer Nanocomposites of Cellulose Nanoparticles; Pandey, J.K., Takagi, H., Nakagaito, A.N., Kim, H.-J., Eds.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 47–64. [Google Scholar]
- Voisin, H.; Bergström, L.; Liu, P.; Mathew, A.P. Nanocellulose-based materials for water purification. Nanomaterials 2017, 7, 57–75. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Burger, C.; Hsiao, B.S.; Chu, B. Ultrafine polysaccharide nanofibrous membranes for water purification. Biomacromolecules 2011, 12, 970–976. [Google Scholar] [CrossRef] [PubMed]
- Rana, D.; Matsuura, T. Surface modification for antifouling membranes. Chem. Rev. 2010, 110, 2448–2471. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Ma, H.; Chu, B.; Hsiao, B.S. Fabrication of cellulose nanofiber-based ultrafiltration membranes by spray coating approach. J. Appl. Polym. Sci. 2017, 134. [Google Scholar] [CrossRef]
- Soyekwo, F.; Zhang, Q.G.; Lin, X.C.; Wu, X.M.; Zhu, A.M.; Liu, Q.L. Facile preparation and separation performances of cellulose nanofibrous membranes. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Wang, Z.; Ma, H.; Hsiao, B.S.; Chu, B. Nanofibrous ultrafiltration membranes containing cross-linked poly(ethylene glycol) and cellulose nanofiber composite barrier layer. Polymer 2014, 55, 366–372. [Google Scholar] [CrossRef]
- Ma, H.; Burger, C.; Hsiao, B.S.; Chu, B. Fabrication and characterization of cellulose nanofiber based thin-membrane nanofibrous composite membranes. J. Membr. Sci. 2014, 454, 272–282. [Google Scholar] [CrossRef]
- Varanasi, S.; Low, Z.-X.; Batchelor, W. Cellulose nanofibre composite membranes—Biodegradable and recyclable UF membranes. J. Chem. Eng. 2015, 265, 138–146. [Google Scholar] [CrossRef]
- Mautner, A.; Lee, K.-Y.; Lahtinen, P.; Hakalahti, M.; Tammelin, T.; Li, K.; Bismarck, A. Nanopapers for organic solvent nanofiltration. Chem. Commun. 2014, 50, 5778–5781. [Google Scholar] [CrossRef] [PubMed]
- Karim, Z.; Claudpierre, S.; Grahn, M.; Oksman, K.; Mathew, A.P. Nanocellulose based functional membranes for water cleaning: Tailoring of mechanical properties, porosity and metal ion capture. J. Membr. Sci. 2016, 514, 418–428. [Google Scholar] [CrossRef]
- Visanko, M.; Liimatainen, H.; Sirviö, J.A.; Haapala, A.b.; Sliz, R.; Niinimäki, J.; Hormi, O. Porous thin membrane barrier layers from 2,3-dicarboxylic acid cellulose nanofibrils for membrane structures. Carbohydr. Ploym. 2014, 102, 584–589. [Google Scholar] [CrossRef] [PubMed]
- Visanko, M.; Liimatainen, H.; Sirviö, J.A.; Hormi, O. A cross-linked 2,3-dicarboxylic acid cellulose nanofibril network: A nanoporous thin-membrane layer with tailored pore size for composite membranes. Sep. Purif. Technol. 2015, 154, 44–50. [Google Scholar] [CrossRef]
- Wang, R.; Guan, S.; Sato, A.; Wang, X.; Wang, Z.; Yang, R.; Hsiao, B.S.; Chu, B. Nanofibrous microfiltration membranes capable of removing bacteria, viruses and heavy metal ions. J. Membr. Sci. 2013, 446, 376–382. [Google Scholar] [CrossRef]
- Rivera-Utrilla, J.; Sánchez-Polo, M.; Gómez-Serrano, V.; Álvarez, P.M.; Alvim-Ferraz, M.C.M.; Dias, J.M. Activated carbon modifications to enhance its water treatment applications. An overview. J. Hazard. Mater. 2011, 187, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Busscher, H.J.; Dijkstra, R.J.; Engels, E.; Langworthy, D.E.; Collias, D.I.; Bjorkquist, W.D.; Mitchell, M.D.; Van der Mei, H.C. Removal of Two Waterborne Pathogenic Bacterial Strains by Activated Carbon Particles Prior to and after Charge Modification. Environ. Sci. Technol. 2006, 40, 6799–6804. [Google Scholar] [CrossRef] [PubMed]
- Ujang, Z.; Au, Y.L.; Nagaoka, H. Comparative study on microbial removal in immersed membrane filtration (IMF) with and without powdered activated carbon (PAC). Water Sci. Technol. 2002, 46, 109–115. [Google Scholar] [PubMed]
- Dolić, M.B.; Rajaković-Ognjanović, V.N.; Štrbac, S.B.; Dimitrijević, S.I.; Mitrić, M.N.; Onjia, A.E.; Rajaković, L.V. Natural sorbents modified by divalent Cu2+ and Zn2+ ions and their corresponding antimicrobial activity. New Biotechnol. 2017, in press. [Google Scholar]
- Eltugral, N.; Simsir, H.; Karagoz, S.B. Preparation of nano-silver-supported activated carbon using different ligands. Res. Chem. Intermed. 2016, 42, 1663–1676. [Google Scholar] [CrossRef]
- Prashantha Kumar, T.K.M.; Mandlimath, T.R.; Sangeetha, P.; Sakthivel, P.; Revathi, S.K.; Ashok Kumar, S.K.; Sahoo, S.K. Highly efficient performance of activated carbon impregnated with Ag, ZnO and Ag/ZnO nanoparticles as antimicrobial materials. RSC Adv. 2015, 5, 108034–108043. [Google Scholar] [CrossRef]
- Hassan, M.L.; Bras, J.; Hassan, E.A.; Silard, C.; Mauret, E. Enzyme-assisted isolation of cellulose nanofibers from date palm fruit stalks. Ind. Crops Prod. 2014, 55, 102–108. [Google Scholar] [CrossRef]
- Wise, L.E.; Murphy, M.; D’Addieco, A.A. Chlorite holocellulose, its fractionation and bearing on summative wood analysis and on studies on hemicelluloses. Pap. Trade 1946, 122, 35–43. [Google Scholar]
- Browning, B.L. Methods of Wood Chemistry, Vol. 2; Interscience: New York, NY, USA, 1967. [Google Scholar]
- Hassan, M.L.; Mathew, A.P.; Hassan, E.A.; El-Wakil, N.A.; Oksman, K. Nanofibers from bagasse and rice straw: Process optimization and properties. Wood Sci. Technol. 2012, 46, 193–205. [Google Scholar] [CrossRef]
- Saito, T.; Kimura, S.; Nishiyama, Y.; Isogai, A. Cellulose nanofibers prepared by TEMPO-mediated oxidation of native cellulose. Biomacromolecules 2007, 8, 2458–2491. [Google Scholar] [CrossRef] [PubMed]
- Rwayha, Y.M.N.; Hassan, M.L.; Shehata, M.R. Nanoporous activated carbon from olive stones wastes. J. Sci. Ind. Res. 2017. accepted. [Google Scholar]
- Mautner, A.; Lee, K.-Y.; Tammelin, T.; Mathew, A.P.; Nedoma, A.J.; Li, K.; Bismarck, A. Cellulose nanopapers as tight aqueous ultra-filtration membranes. React. Funct. Ploym. 2015, 86, 209–214. [Google Scholar] [CrossRef]
- Gibbins, E.; Antonio, M.D.; Nair, D.; White, L.S.; Freitas dos Santos, L.M.; Vankelecom, I.F.J.; Livingston, A.G. Observations on solvent flux and solute rejection across solvent resistant nanofiltration membranes. Desalination 2002, 147, 307–313. [Google Scholar] [CrossRef]
- Cheng, W.; He, J.; Chen, M.; Li, D.; Li, H.; Chen, L.; Cao, Y.; Wang, J.; Huang, Y. Preparation, functional characterization and hemostatic mechanism discussion for oxidized microcrystalline cellulose and its composites. Fiber Ploym. 2016, 17, 1277–1286. [Google Scholar] [CrossRef]
- Wu, Y.; He, J.; Cheng, W.; Gu, H.; Guo, Z.; Gao, S.; Huang, Y. Oxidized regenerated cellulose-based hemostat with microscopically gradient structure. Carbohydr. Ploym. 2012, 88, 1023–1032. [Google Scholar] [CrossRef]
- Dineen, P. Antibacterial activity of oxidized regenerated cellulose. Surg. Gynecol. Obstet. 1976, 142, 481–486. [Google Scholar] [PubMed]










© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hassan, M.; Abou-Zeid, R.; Hassan, E.; Berglund, L.; Aitomäki, Y.; Oksman, K. Membranes Based on Cellulose Nanofibers and Activated Carbon for Removal of Escherichia coli Bacteria from Water. Polymers 2017, 9, 335. https://doi.org/10.3390/polym9080335
Hassan M, Abou-Zeid R, Hassan E, Berglund L, Aitomäki Y, Oksman K. Membranes Based on Cellulose Nanofibers and Activated Carbon for Removal of Escherichia coli Bacteria from Water. Polymers. 2017; 9(8):335. https://doi.org/10.3390/polym9080335
Chicago/Turabian StyleHassan, Mohammad, Ragab Abou-Zeid, Enas Hassan, Linn Berglund, Yvonne Aitomäki, and Kristiina Oksman. 2017. "Membranes Based on Cellulose Nanofibers and Activated Carbon for Removal of Escherichia coli Bacteria from Water" Polymers 9, no. 8: 335. https://doi.org/10.3390/polym9080335