CO2 Plasticization Resistance Membrane for Natural Gas Sweetening Process: Defining Optimum Operating Conditions for Stable Operation
(This article belongs to the Section Polymer Applications)
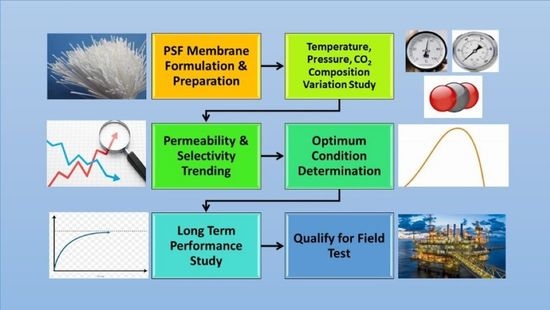
Abstract
:1. Introduction
2. Theory
3. Experimental
3.1. Materials
3.2. Membrane Characterization
3.3. Membrane Performance Testing
- = Permeance of component (cm3/s·cmHg·cm2)
- = Permeate flowrate of component (cm3/s)
- = Differential pressure of component (cm Hg)
- Membrane active area (cm2)
- x, y = Component
- = Differential pressure of component (barg)
- = Mean differential difference of component between feed and retentate (barg)
- = Feed pressure (barg)
- = Component concentration in feed stream (mol%)
- = Retentate pressure (barg)
- = Component concentration in retentate stream (mol%)
- Permeate component pressure (barg)
- = Permeate pressure (barg)
- = Component concentration in permeate stream (mol%).
4. Results and Discussion
4.1. Membrane Characterizations
4.2. Effect of the Feed Pressure on the CO2 Permeation of the PSf Hollow Fibre Membranes at Fixed Operating Temperature
4.3. Effect of the Feed Pressure on the CO2/CH4 Selectivity of the PSf Hollow Fiber Membranes at Fixed Operating Temperature
4.4. Effect of the Feed Temperature on the CO2 Permeation of the PSF Hollow Fiber Membranes at Fixed Operating Pressure
4.5. Effect of the Feed Temperature on the CO2/CH4 Selectivity of the PSf Hollow Fiber Membranes at Fixed Operating Pressure
4.6. Effect of the Feed Composition on the CO2 Permeation and Selectivity of the PSf Hollow Fiber Membranes
4.7. Long-Term Performance Study of the Membrane
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mordor Intelligence. Gas Separation Membrane Market: Growth, Trends, Covid-19 Impact and Forecasts (2022–2027). Available online: https://www.mordorintelligence.com/industry-reports/gas-separation-membrane-market (accessed on 28 May 2022).
- Hwang, H.Y.; Nam, S.Y.; Koh, H.C.; Ha, S.Y.; Barbieri, G.; Drioli, E. The effect of operating conditions on the performance of hollow fiber membrane modules for CO2/M2 separation. J. Ind. Eng. Chem. 2012, 18, 205–211. [Google Scholar] [CrossRef]
- Jia, B.; Chen, Z.; Xian, C. Investigations of CO2 storage capacity and flow behavior in shale formation. J. Pet. Sci. Eng. 2022, 208, 109659. [Google Scholar] [CrossRef]
- Hawthorne, S.B.; Grabanski, C.B.; Jin, L.; Bosshart, N.W.; Miller, D.J. Comparison of CO2 and Produced Gas Hydrocarbons to Recover Crude Oil from Williston Basin Shale and Mudrock Cores at 10.3, 17.2, and 34.5 MPa and 110 °C. Energy Fuels 2021, 35, 6658–6672. [Google Scholar] [CrossRef]
- Kazemi, M.; Borujeni, T. Molecular Dynamic Study of Carbon Dioxide in Carbon Based Organic Nanopores.pdf. In Proceedings of the Society of Petroleum Engineers—Abu Dhabi International Petroleum Exhibition and Conference, Abu Dhabi, UAE, 7 November 2016; p. SPE-181705-MS. [Google Scholar]
- Carta, M.; Bernardo, P. Gas Separation by Membrane Operations. In Encyclopedia of Membranes; Springer: Berlin/Heidelberg, Germany, 2015; pp. 1–3. [Google Scholar] [CrossRef]
- Duthie, X.; Kentish, S.; Powell, C.; Nagai, K.; Qiao, G.; Stevens, G. Operating temperature effects on the plasticization of polyimide gas separation membranes. J. Membr. Sci. 2007, 294, 40–49. [Google Scholar] [CrossRef]
- Wypych, G. Handbook of Plasticizer, 3rd ed.; ChemTec Publishing: Toronto, ON, Canada, 2017; pp. 124–343. ISBN 9781895198973. [Google Scholar]
- Li, P.; Hosseini, S.S.; Zhang, M.; Deng, L.; Xiang, D.; Cao, B. Approaches to Suppress CO2-Induced Plasticization of Polyimide Membranes in Gas. Processes 2019, 7, 51. [Google Scholar]
- Bos, A.; Pünt, I.G.M.; Wessling, M.; Strathmann, H. CO2-induced plasticization phenomena in glassy polymers. J. Membr. Sci. 1999, 155, 67–78. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, Z.; Morisato, A.; Bhuwania, N.; Chinn, D.; Koros, J. Natural gas sweetening using a cellulose triacetate hollow fiber membrane illustrating controlled plasticization benefits. J. Membr. Sci. 2020, 601, 117910. [Google Scholar] [CrossRef]
- Alaslai, N.; Ghanem, B.; Alghunaimi, F.; Litwiller, E.; Pinnau, I. Pure- and mixed-gas permeation properties of highly selective and plasticization resistant hydroxyl-diamine-based 6FDA polyimides for CO2/CH4 separation. J. Membr. Sci. 2016, 505, 100–107. [Google Scholar] [CrossRef] [Green Version]
- Houben, H.J.M.; Borneman, Z.; Nijmeijer, K. Plasticization behavior of crown-ether containing polyimide membranes for the separation of CO2. Sep. Purif. Technol. 2020, 255, 117307. [Google Scholar] [CrossRef]
- Minelli, M.; Oradei, S.; Fiorini, M.; Sarti, G.C. CO2 plasticization effect on glassy polymeric membranes. Polymer 2019, 163, 29–35. [Google Scholar] [CrossRef]
- Yong, W.F.; Kwek, K.H.A.; Liao, K.S.; Chung, T.S. Suppression of aging and plasticization in highly permeable polymers. Polymer 2015, 77, 377–386. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, X.; Paul, D.R. Physical aging of thin glassy polymer films: Free volume interpretation. J. Membr. Sci. 2006, 277, 219–229. [Google Scholar] [CrossRef]
- Genduso, G.; Pinnau, I. Quantification of sorption, diffusion, and plasticization properties of cellulose triacetate films under mixed-gas CO2/CH4 environment. J. Membr. Sci. 2020, 610, 118269. [Google Scholar] [CrossRef]
- Sadrzadeh, M.; Shahidi, K.; Mohammadi, T. Effect of operating parameters on pure and mixed gas permeation properties of a synthesized composite PDMS/PA membrane. J. Membr. Sci. 2009, 342, 327–340. [Google Scholar] [CrossRef]
- Zhang, C.; Yan, J.; Tian, Z.; Liu, X.; Cao, B.; Li, P. Molecular Design of Troger’s Base-Based Polymers Containing Spirobichroman Structure for Gas Separation. Ind. Eng. Chem. Res. 2017, 56, 12783–12788. [Google Scholar] [CrossRef]
- Saimani, S.; Dal-cin, M.M.; Kumar, A.; Kingston, D.M. Separation performance of asymmetric membranes based on PEGDa/PEI semi-interpenetrating polymer network in pure and binary gas mixtures of CO2, N2 and CH4. J. Membr. Sci. 2010, 362, 353–359. [Google Scholar] [CrossRef] [Green Version]
- Achoundong, C.S.K.; Bhuwania, N.; Burgess, S.K.; Karvan, O.; Johnson, J.R.; Koros, W.J. Silane Modification of Cellulose Acetate Dense Films as Materials for Acid Gas Removal. Macromolecules 2013, 46, 5584–5594. [Google Scholar] [CrossRef]
- Adewole, J.K.; Sultan, A.S. Polymeric Membranes for Natural Gas Processing: Polymer Synthesis and Membrane Gas Transport Properties. In Functional Polymers, Polymers and Polymeric Composites; Springer: Cham, Switzerland, 2019. [Google Scholar]
- Favvas, E.P.; Katsaros, F.K.; Papageorgiou, S.K.; Sapalidis, A.A.; Mitropoulos, A.C. A review of the latest development of polyimide based membranes for CO2 separations. React. Funct. Polym. 2017, 120, 104–130. [Google Scholar] [CrossRef]
- Farahdila, K.; Goh, P.S.; Ismail, A.F.; Wan, N.F.W.M.; Mohd, H.M.H.; Soh, W.K.; Yeo, S.Y. Challenges in Membrane Process for Gas Separation from Natural Gas. J. Appl. Membr. Sci. Technol. 2021, 25, 89–105. [Google Scholar] [CrossRef]
- Visser, T.; Masetto, N.; Wessling, M. Materials dependence of mixed gas plasticization behavior in asymmetric membranes. J. Membr. Sci. 2007, 306, 16–28. [Google Scholar] [CrossRef]
- Falbo, F.; Tasselli, F.; Brunetti, A.; Drioli, E.; Barbieri, G. Polyimide hollow fiber membranes for CO2 separation from wet gas mixtures. Braz. J. Chem. Eng. 2014, 31, 1023–1034. [Google Scholar] [CrossRef] [Green Version]
- Ricci, E.; Di, E.; Degli, M.; Liu, L.; Mensitieri, G.; Fabbri, P.; Kentish, S.E.; Grazia, M.; Angelis, D. Towards a systematic determination of multicomponent gas separation with membranes: The case of CO2/CH4 in cellulose acetates. J. Membr. Sci. 2021, 628, 119226. [Google Scholar] [CrossRef]
- Miandoab, E.S.; Kentish, S.E.; Scholes, C.A. Modelling competitive sorption and plasticization of glassy polymeric membranes used in biogas upgrading. J. Membr. Sci. 2021, 617, 118643. [Google Scholar] [CrossRef]
- Genduso, G.; Ghanem, B.S.; Pinnau, I. Experimental mixed-gas permeability, sorption and diffusion of CO2-CH4 mixtures in 6FDA-mPDA polyimide membrane: Unveiling the effect of competitive sorption on permeability selectivity. Membranes 2019, 9, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- White, L.S. Effect of operating environment on membrane performance. Curr. Opin. Chem. Eng. 2020, 28, 105–111. [Google Scholar] [CrossRef]
- Natarajan, P.; Sasikumar, B.; Elakkiya, S.; Arthanareeswaran, G.; Ismail, A.F.; Youravong, W.; Yuliwati, E. Pillared cloisite 15A as an enhancement filler in polysulfone mixed matrix membranes for CO2/N2 and O2/N2 gas separation. J. Nat. Gas. Sci. Eng. 2021, 86, 103720. [Google Scholar] [CrossRef]
- Dehghani Kiadehi, A.; Rahimpour, A.; Jahanshahi, M.; Ghoreyshi, A.A. Novel carbon nano-fibers (CNF)/polysulfone (PSf) mixed matrix membranes for gas separation. J. Ind. Eng. Chem. 2015, 22, 199–207. [Google Scholar] [CrossRef]
- Ilicak, I.; Boroglu, M.S.; Durmus, A.; Boz, I. Journal of Natural Gas Science and Engineering Influence of ZIF-95 on structure and gas separation properties of polyimide-based mixed matrix membranes. J. Nat. Gas. Sci. Eng. 2021, 91, 103941. [Google Scholar] [CrossRef]
- Lokhandwala, K.A.; Baker, R.W. Natural Gas Processing with Membranes. Ind. Eng. Chem. Res. 2008, 1, 2109–2121. [Google Scholar]
- Mushtaq, A.; Mukhtar, H.B.; Shariff, A.M.; Mannan, H.A. A Review: Development of Polymeric Blend Membrane for Removal of CO2 from Natural Gas. Int. J. Eng. Technol. IJET-IJENS 2013, 13, 53–60. [Google Scholar]
- Costello, L.M.; Koros, W.J. Thermally stable polyimide isomers for membrane-based gas separations at elevated temperatures. J. Polym. Sci. Part B Polym. Phys. 1995, 33, 135–146. [Google Scholar] [CrossRef]
- McKeen, L.W. Introduction to Permeation of Plastics and Elastomers. In Permeability Properties of Plastics and Elastomers; Elsevier: Amsterdam, The Netherlands, 2012; pp. 1–20. [Google Scholar] [CrossRef]
- Adewole, J.K.; Ahmad, A.L.; Ismail, S.; Leo, C.P.; Sultan, A.S. Comparative studies on the effects of casting solvent on physico- chemical and gas transport properties of dense polysulfone membrane used for CO2/CH4 separation. J. Appl. Polym. Sci. 2015, 132, 42205. [Google Scholar] [CrossRef]
- Ismail, A.F.; Lorna, W. Penetrant-induced plasticization phenomenon in glassy polymers for gas separation membrane. Sep. Purif. Technol. 2002, 27, 173–194. [Google Scholar] [CrossRef]
- Kadirkhan, F.; Goh, P.S.; Ismail, A.F. Recent Advances of Polymeric Membranes in Tackling Plasticization and Aging. Membranes 2022, 12, 58. [Google Scholar] [CrossRef]
- Halim, M.H.; Kadirkhan, F.; Mustapa, W.N.F.; Soh, W.; Yeo, S. Natural Gas Sweetening Polymeric Membrane: Established Optimum Operating Condition at 70% of CO2 Concentration Fedd Gas Stream. Malaysian J. Fundam. Appl. Sci. 2020, 16, 54–58. [Google Scholar] [CrossRef]
- Sridhar, S.; Smitha, B.; Aminabhavi, T.M. Separation of carbon dioxide from natural gas mixtures through polymeric membranes—A review. Sep. Purif. Rev. 2007, 36, 113–174. [Google Scholar] [CrossRef]
- Ma, C.; Koros, W.J. Physical aging of ester-cross-linked hollow fiber membranes for natural gas separations and mitigation thereof. J. Membr. Sci. 2018, 551, 214–221. [Google Scholar] [CrossRef]
- Siagian, U.W.R.; Raksajati, A.; Himma, N.F.; Khoiruddin, K.; Wenten, I.G. Membrane-based carbon capture technologies: Membrane gas separation vs. membrane contactor. J. Nat. Gas Sci. Eng. 2019, 67, 172–195. [Google Scholar] [CrossRef]
- Al-juaied, M.; Koros, W.J. Performance of natural gas membranes in the presence of heavy hydrocarbons. J. Membr. Sci. 2006, 274, 227–243. [Google Scholar] [CrossRef]
- Liu, Q.; Galizia, M.; Gleason, K.L.; Scholes, C.A.; Paul, D.R.; Benny, D. Influence of toluene on CO2 and CH4 gas transport properties in thermally rearranged (TR) polymers based on 3,3′-dihydroxy-4,4′-diamino-biphenyl (HAB) and 2,2′-bis-(3,4-dicarboxyphenyl) hexafluotopropane dianhydride (6FDA). J. Membr. Sci. 2016, 514, 282–293. [Google Scholar] [CrossRef]












| Gas/Composition (%) | CO2 | C1 | C2 | C3 | N2 |
|---|---|---|---|---|---|
| Set 1 | 15.00 | 76.88 | 3.42 | 2.65 | 2.05 |
| Set 2 | 20.00 | 71.88 | 3.42 | 2.65 | 2.05 |
| Set 3 | 40.00 | 55.90 | 2.00 | 1.60 | 0.50 |
| Set 4 | 70.00 | 28.40 | 1.00 | 0.40 | 0.20 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kadirkhan, F.; Sean, G.P.; Ismail, A.F.; Wan Mustapa, W.N.F.; Halim, M.H.M.; Kian, S.W.; Yean, Y.S. CO2 Plasticization Resistance Membrane for Natural Gas Sweetening Process: Defining Optimum Operating Conditions for Stable Operation. Polymers 2022, 14, 4537. https://doi.org/10.3390/polym14214537
Kadirkhan F, Sean GP, Ismail AF, Wan Mustapa WNF, Halim MHM, Kian SW, Yean YS. CO2 Plasticization Resistance Membrane for Natural Gas Sweetening Process: Defining Optimum Operating Conditions for Stable Operation. Polymers. 2022; 14(21):4537. https://doi.org/10.3390/polym14214537
Chicago/Turabian StyleKadirkhan, Farahdila, Goh Pei Sean, Ahmad Fauzi Ismail, Wan Nurul Ffazida Wan Mustapa, Mohd Hanif Mohamad Halim, Soh Wei Kian, and Yeo Siew Yean. 2022. "CO2 Plasticization Resistance Membrane for Natural Gas Sweetening Process: Defining Optimum Operating Conditions for Stable Operation" Polymers 14, no. 21: 4537. https://doi.org/10.3390/polym14214537