Carbon Quantum Dots: Synthesis, Structure, Properties, and Catalytic Applications for Organic Synthesis
Abstract
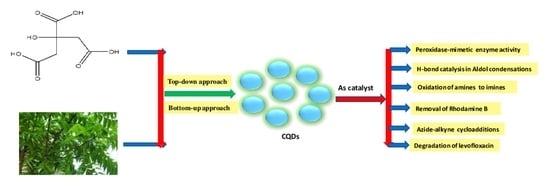
:1. Introduction
2. Synthesis Approach
2.1. Top-Down Approach
2.1.1. Laser Ablation Method
2.1.2. Electrochemical Method
2.1.3. Arch Discharge Method
2.2. Bottom-Up Approach
2.2.1. Hydrothermal Method
2.2.2. Combustion Method
2.2.3. Pyrolysis Method
2.2.4. Microwave Irradiation Method
2.2.5. Template Method
3. Structure of CQDs
4. Optical Properties of Carbon Quantum Dots (CQDs)
4.1. Absorbance
4.2. Photoluminescence
4.2.1. Fluorescence
4.2.2. Phosphorescence
5. Application of Carbon Quantum Dots as a Catalyst
5.1. CQDs as a Catalyst for the Peroxidase-Mimetic Enzyme Activity
5.2. CQDs as a Catalyst for Selective Oxidation of Alcohols to Aldehydes
5.3. CQDs as a Catalyst for Selective Oxidation of Amine to Imine
5.4. CQDs as a Catalyst in the Synthesis of Multisubstituted 4H Pyran with Indole Moieties
5.5. As a Photocatalyst for High-Efficiency Cyclohexane Oxidation
5.6. As a Catalyst for the Removal of Rhodamine B
5.7. As a Catalyst in Azide-Alkyne Cycloadditions
5.8. As H-Bond Catalysis in Aldol Condensations
5.9. As a Catalyst for the Ring Opening of Epoxides
5.10. As a Catalyst for the Degradation of Levofloxacin
5.11. CQDs as Electrocatalyst
6. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability
Acknowledgments
Conflicts of Interest
References
- Clancy, A.; Bayazit, M.K.; Hodge, S.A.; Skipper, N.T.; Howard, C.A.; Shaffer, M.S.P. Charged Carbon Nanomaterials: Redox Chemistries of Fullerenes, Carbon Nanotubes, and Graphenes. Chem. Rev. 2018, 118, 7363–7408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, H.-S.; Jeon, I.; Xiang, R.; Seo, S.; Lee, J.-W.; Li, C.; Pal, A.; Manzhos, S.; Goorsky, M.S.; Yang, Y.; et al. Achieving high efficiency in solution-processed perovskite solar cells using C60/C70 mixed fullerenes. ACS Appl. Mater. Interfaces 2018, 46, 39590–39598. [Google Scholar] [CrossRef] [PubMed]
- Georgakilas, V.; Perman, J.A.; Tucek, J.; Zboril, R. Broad Family of Carbon Nanoallotropes: Classification, Chemistry, and Applications of Fullerenes, Carbon Dots, Nanotubes, Graphene, Nanodiamonds, and Combined Superstructures. Chem. Rev. 2015, 115, 4744–4822. [Google Scholar] [CrossRef] [PubMed]
- Rao, R.; Pint, C.L.; Islam, A.E.; Weatherup, R.S.; Hofmann, S.; Meshot, E.R.; Wu, F.; Zhou, C.; Dee, N.; Amama, P.B.; et al. Carbon nanotubes and related nanomaterials: Critical advances and challenges for synthesis toward mainstream commercial applica-tions. ACS Nano 2018, 12, 11756–11784. [Google Scholar] [CrossRef] [Green Version]
- Patel, K.D.; Singh, R.K.; Kim, H.W. Carbon-based nanomaterials as an emerging platform for theranostics. Materials Horizons 2019, 3, 434–469. [Google Scholar] [CrossRef]
- Wang, X.; Feng, Y.; Dong, P.; Huang, J. A Mini Review on Carbon Quantum Dots: Preparation, Properties, and Electrocatalytic Application. Front. Chem. 2019, 7, 671. [Google Scholar] [CrossRef]
- Zuo, J.; Tao, J.; Zhao, X.; Xiong, X.; Xiao, S.; Zhu, Z. Preparation and application of fluorescent carbon dots. J. Nanomater. 2015, 2015, 787862. [Google Scholar] [CrossRef] [Green Version]
- Xu, X.; Ray, R.; Gu, Y.; Ploehn, H.J.; Gearheart, L.; Raker, K.; Scrivens, W.A. Electrophoretic Analysis and Purification of Fluorescent Single-Walled Carbon Nanotube Fragments. J. Am. Chem. Soc. 2004, 126, 12736–12737. [Google Scholar] [CrossRef]
- Sun, Y.-P.; Zhou, B.; Lin, Y.; Wang, W.; Fernando, K.S.; Pathak, P.; Meziani, M.J.; Harruff, B.A.; Wang, X.; Wang, H. Quantum-Sized Carbon Dots for Bright and Colorful Photoluminescence. J. Am. Chem. Soc. 2006, 128, 7756–7757. [Google Scholar] [CrossRef]
- Ahmad, F.; Khan, A.M. Carbon quantum dots: Nanolights. Int. J. Petrochem. Sci. Eng. 2017, 2, 247–250. [Google Scholar] [CrossRef] [Green Version]
- Yang, S.; Sun, J.; Li, X.; Zhou, W.; Wang, Z.; He, P.; Ding, G.; Xie, X.; Kang, Z.; Jiang, M. Large-scale fabrication of heavy doped carbon quantum dots with tunable-photoluminescence and sensitive fluorescence detection. J. Mater. Chem. A 2014, 2, 8660–8667. [Google Scholar] [CrossRef]
- Guo, H.; Liu, Z.; Shen, X.; Wang, L. One-Pot Synthesis of Orange Emissive Carbon Quantum Dots for All-Type High Color Rendering Index White Light-Emitting Diodes. ACS Sustain. Chem. Eng. 2022, 10, 8289–8296. [Google Scholar] [CrossRef]
- Toma, E.E.; Stoian, G.; Cojocaru, B.; Parvulescu, V.I.; Coman, S.M. ZnO/CQDs Nanocomposites for Visible Light Photodegradation of Organic Pollutants. Catalysts 2022, 12, 952. [Google Scholar] [CrossRef]
- Subedi, S.; Rella, A.K.; Trung, L.G.; Kumar, V.; Kang, S.-W. Electrically Switchable Anisometric Carbon Quantum Dots Exhibiting Linearly Polarized Photoluminescence: Syntheses, Anisotropic Properties, and Facile Control of Uniaxial Orientation. ACS Nano 2022, 16, 6480–6492. [Google Scholar] [CrossRef]
- Gu, L.; Zhang, J.; Yang, G.; Tang, Y.; Zhang, X.; Huang, X.; Zhai, W.; Fodjo, E.K.; Kong, C. Green preparation of carbon quantum dots with wolfberry as on-off-on nanosensors for the detection of Fe3+ and l-ascorbic acid. Food Chem. 2022, 376, 131898. [Google Scholar] [CrossRef]
- Chen, Y.; Xue, B. A review on quantum dots modified g-C3N4-based photocatalysts with improved photocatalytic activity. Catalysts 2020, 1, 142. [Google Scholar] [CrossRef] [Green Version]
- Parya, E.; Rhim, J.-W. Pectin/carbon quantum dots fluorescent film with ultraviolet blocking property through light conversion. Colloids Surf. B Biointerfaces 2022, 219, 112804. [Google Scholar]
- Ajayan, P.M.; Zhou, O.Z. Applications of carbon nanotubes. Carbon Nanotub. 2001, 80, 391–425. [Google Scholar]
- Baptista, F.R.; Belhout, S.A.; Giordani, S.; Quinn, S.J. Recent developments in carbon nanomaterial sensors. Chem. Soc. Rev. 2015, 44, 4433–4453. [Google Scholar] [CrossRef]
- Chen, D.; Tang, L.; Li, J. Graphene-based materials in electrochemistry. Chem. Soc. Rev. 2010, 39, 3157–3180. [Google Scholar] [CrossRef]
- Cayuela, A.; Benítez-Martínez, S.; Soriano, M.L. Carbon nanotools as sorbents and sensors of nanosized objects: The third way of analytical nanoscience and nanotechnology. TrAC Trends Anal. Chem. 2016, 84, 172–180. [Google Scholar] [CrossRef]
- Pardo, J.; Peng, Z.; Leblanc, R.M. Cancer Targeting and Drug Delivery Using Carbon-Based Quantum Dots and Nanotubes. Molecules 2018, 23, 378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sahar, T.; Abnous, K.; Taghdisi, S.M.; Ramezani, M.; Alibolandi, M. Hybrid car-bon-based materials for gene delivery in cancer therapy. J. Control Release 2020, 318, 158–175. [Google Scholar]
- Mingjun, C.; Cao, Y.; Zhu, Y.; Peng, W.; Li, Y.; Zhang, F.; Xia, Q.; Fan, X. Oxidation-Modulated CQDs Derived from Covalent Organic Frameworks as Enhanced Fluorescence Sensors for the Detection of Chromium (VI) and Ascorbic Acid. Ind. Eng. Chem. Res. 2022, 31, 11484–11493. [Google Scholar]
- Murali, G.; Kwon, B.; Kang, H.; Modigunta, J.K.R.; Park, S.; Lee, S.; Lee, H.; Park, Y.H.; Kim, J.; Park, S.Y.; et al. Hematoporphyrin Photosensitizer-Linked Carbon Quantum Dots for Photodynamic Therapy of Cancer Cells. ACS Appl. Nano Mater. 2022, 5, 4376–4385. [Google Scholar] [CrossRef]
- Li, P.; Yu, M.; Ke, X.; Gong, X.; Li, Z.; Xing, X. Cytocompatible Amphipathic Carbon Quantum Dots as Potent Membrane-Active Antibacterial Agents with Low Drug Resistance and Effective Inhibition of Biofilm Formation. ACS Appl. Bio Mater. 2022, 5, 3290–3299. [Google Scholar] [CrossRef]
- Wu, Y.; Qin, D.; Luo, Z.; Meng, S.; Mo, G.; Jiang, X.; Deng, B. High Quantum Yield Boron and Nitrogen Codoped Carbon Quantum Dots with Red/Purple Emissions for Ratiometric Fluorescent IO4– Sensing and Cell Imaging. ACS Sustain. Chem. Eng. 2022, 10, 5195–5202. [Google Scholar] [CrossRef]
- Kaur, A.; Pandey, K.; Kaur, R.; Vashishat, N.; Kaur, M. Nanocomposites of Carbon Quantum Dots and Graphene Quantum Dots: Environmental Applications as Sensors. Chemosensors 2022, 10, 367. [Google Scholar] [CrossRef]
- Sharma, V.; Vishal, V.; Chandan, G.; Bhatia, A.; Chakrabarti, S.; Bera, M. Green, sustainable, and economical synthesis of fluorescent nitrogen-doped carbon quantum dots for applications in optical displays and light-emitting diodes. Mater. Today Sustain. 2022, 19, 100184. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, D.; Zhang, J.; Balogun, M.T.; Wang, P.; Tong, Y.; Huang, Y. Charge Relays via Dual Carbon-Actions on Nanostructured BiVO4 for High Performance Photoelectrochemical Water Splitting. Adv. Funct. Mater. 2022, 32, 2112738. [Google Scholar] [CrossRef]
- Rasooll, M.M.; Zarei, M.; Zolfigol, M.A.; Sepehrmansourie, H.; Omidi, A.; Hasani, M.; Gu, Y. Novel nano-architectured carbon quantum dots (CQDs) with phosphorous acid tags as an efficient catalyst for the synthesis of multisubstituted 4H-pyran with indole moieties under mild conditions. RSC Adv. 2021, 11, 25995–26007. [Google Scholar] [CrossRef]
- Liu, Z.X.; Bin Chen, B.; Liu, M.L.; Zou, H.Y.; Huang, C.Z. Cu(i)-Doped carbon quantum dots with zigzag edge structures for highly efficient catalysis of azide–alkyne cycloadditions. Green Chem. 2017, 19, 1494–1498. [Google Scholar] [CrossRef]
- Fanqing, M.; Wang, Y.; Chen, Z.; Hu, J.; Lu, G.; Ma, W. Synthesis of CQDs@ FeOOH nanoneedles with abundant active edges for efficient electro-catalytic degradation of levofloxacin: Degradation mechanism and toxicity assessment. Appl. Catal. B Environ. 2021, 282, 119597. [Google Scholar]
- Rezaei, A.; Mohammadi, Y.; Ramazani, A.; Zheng, H. Ultrasound-assisted pseudohomogeneous tungstate catalyst for selective oxidation of alcohols to aldehydes. Sci. Rep. 2022, 12, 3367. [Google Scholar] [CrossRef]
- Preethi, M.; Viswanathan, C.; Ponpandian, N. A metal-free, dual catalyst for the removal of Rhodamine B using novel carbon quantum dots from muskmelon peel under sunlight and ultrasonication: A green way to clean the environment. J. Photochem. Photobiol. A Chem. 2022, 426, 113765. [Google Scholar] [CrossRef]
- Ye, J.; Ni, K.; Liu, J.; Chen, G.; Ikram, M.; Zhu, Y. Oxygen-Rich Carbon Quantum Dots as Catalysts for Selective Oxidation of Amines and Alcohols. Chemcatchem 2017, 10, 259–265. [Google Scholar] [CrossRef]
- Ruihua, L.; Huang, H.; Li, H.; Liu, Y.; Zhong, J.; Li, Y.; Zhang, S.; Kang, Z. Metalnanopar-ticle/carbon quantum dot composite as a photocatalyst for high-efficiency cyclohexane oxidation. ACS Catal. 2014, 1, 328–336. [Google Scholar]
- Han, Y.; Huang, H.; Zhang, H.; Liu, Y.; Han, X.; Liu, R.; Li, H.; Kang, Z. Carbon Quantum Dots with Photoenhanced Hydrogen-Bond Catalytic Activity in Aldol Condensations. ACS Catal. 2014, 4, 781–787. [Google Scholar] [CrossRef]
- Pradeep Kumar, Y.; Singh, V.K.; Chandra, S.; Bano, D.; Kumar, V.; Talat, M.; Hasan, S.H. Green synthesis of fluorescent carbon quantum dots from azadirachtaindica leaves and their peroxidase-mimetic ac-tivity for the detection of H2O2 and ascorbic acid in common fresh fruits. ACS Biomater. Sci. Eng. 2018, 2, 623–632. [Google Scholar]
- Li, H.; Sun, C.; Ali, M.; Zhou, F.; Zhang, X.; MacFarlane, D.R. Sulfated Carbon Quantum Dots as Efficient Visible-Light Switchable Acid Catalysts for Room-Temperature Ring-Opening Reactions. Angew. Chem. 2015, 127, 8540–8544. [Google Scholar] [CrossRef]
- Wang, Y.; Hu, A. Carbon quantum dots: Synthesis, properties and applications. J. Mater. Chem. C 2014, 34, 6921–6939. [Google Scholar] [CrossRef] [Green Version]
- Sofia, P.; Palomares, E.; Martinez-Ferrero, E. Graphene and carbon quantum dot-based materials in pho-tovoltaic devices: From synthesis to applications. Nanomaterials 2016, 6, 157. [Google Scholar]
- Chae, A.Y.; Choi, S.J.; Paoprasert, N.P.; Park, S.Y.; In, I. Microwave-assisted synthesis of fuorescent carbon quantum dots from an A2/B3 monomer set. RSC Adv. 2017, 7, 12663–12669. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Huang, H.; Cao, W.; Mao, B.; Liu, Y.; Kang, Z. Advances in carbon dots: From the perspective of traditional quantum dots. Mater. Chem. Front. 2020, 4, 1586–1613. [Google Scholar] [CrossRef]
- Li, H.; Xu, Y.; Zhao, L.; Ding, J.; Chen, M.; Chen, G.; Li, Y.; Ding, L. Synthesis of tiny carbon dots with high quantum yield using multi-walled carbon nanotubes as support for selective “turn-off-on” detection of rutin and Al3+. Carbon 2018, 143, 391–401. [Google Scholar] [CrossRef]
- Zhao, M.; Zhang, J.; Xiao, H.; Hu, T.; Jia, J.; Wu, H. Facile in situ synthesis of a carbon quantum dot/graphene heterostructure as an efficient metal-free electrocatalyst for overall water splitting. Chem. Commun. 2019, 55, 1635–1638. [Google Scholar] [CrossRef]
- Yuan, F.; Su, W.; Gao, F. Monolayer 2D polymeric fullerene: A new member of the carbon material family. Chem 2022, 8, 2079–2081. [Google Scholar] [CrossRef]
- Zhu, H.; Wang, X.; Li, Y.; Wang, Z.; Yang, F.; Yang, X. Microwave synthesis of fluorescent carbon nanoparticles with electrochemiluminescence properties. Chem. Commun. 2009, 34, 5118–5120. [Google Scholar] [CrossRef]
- Li, H.; Zhang, Y.; Ding, J.; Wu, T.; Cai, S.; Zhang, W.; Cai, R.; Chen, C.; Yang, R. Synthesis of carbon quantum dots for application of alleviating amyloid-β mediated neurotoxicity. Colloids Surfaces B Biointerfaces 2022, 212, 112373. [Google Scholar] [CrossRef]
- Cui, L.; Ren, X.; Wang, J.; Sun, M. Synthesis of homogeneous carbon quantum dots by ultrafast dual-beam pulsed laser ablation for bioimaging. Mater. Today Nano 2020, 12, 100091. [Google Scholar] [CrossRef]
- Doñate-Buendía, C.; Fernández-Alonso, M.; Lancis, J.; Mínguez-Vega, G. Pulsed laser ablation in liq-uids for the production of gold nanoparticles and carbon quantum dots: From plasmonic to fluorescence and cell labelling. J. Phys. Conf. Ser. 2020, 1537, 012013. [Google Scholar] [CrossRef]
- Jigang, Z.; Booker, C.; Li, R.; Zhou, X.; Sham, T.-K.; Sun, X.; Ding, Z. An electro-chemical avenue to blue luminescent nanocrystals from multiwalled carbon nanotubes (MWCNTs). J. Am. Chem. Soc. 2007, 4, 744–745. [Google Scholar]
- Zhao, Q.-L.; Zhang, Z.-L.; Huang, B.-H.; Peng, J.; Zhang, M.; Pang, D.-W. Facile preparation of low cytotoxicity fluorescent carbon nanocrystals by electrooxidation of graphite. Chem. Commun. 2008, 41, 5116–5118. [Google Scholar] [CrossRef]
- Zheng, L.; Chi, Y.; Dong, Y.; Lin, J.; Wang, B. Electrochemiluminescence of Water-Soluble Carbon Nanocrystals Released Electrochemically from Graphite. J. Am. Chem. Soc. 2009, 131, 4564–4565. [Google Scholar] [CrossRef]
- Deng, J.; Lu, Q.; Mi, N.; Li, H.; Liu, M.; Xu, M.; Tan, L.; Xie, Q.; Zhang, Y.; Yao, S. Electrochemical Synthesis of Carbon Nanodots Directly from Alcohols. Chem. A Eur. J. 2014, 20, 4993–4999. [Google Scholar] [CrossRef]
- Hou, Y.; Lu, Q.; Deng, J.; Li, H.; Zhang, Y. One-pot electrochemical synthesis of functionalized fluorescent carbon dots and their selective sensing for mercury ion. Anal. Chim. Acta 2015, 866, 6974. [Google Scholar] [CrossRef]
- Bottini, M.; Tautz, L.; Huynh, H.; Monosov, E.; Bottini, N.; Dawson, M.I.; Bellucci, S.; Mustelin, T. Covalent decoration of multi-walled carbon nanotubes with silica nanoparticles. Chem. Commun. 2004, 6, 758–760. [Google Scholar] [CrossRef]
- Michelsen, H.A.; Colket, M.B.; Bengtsson, P.-E.; D’anna, A.; Desgroux, P.; Haynes, B.S.; Miller, J.H.; Nathan, G.J.; Pitsch, H.; Wang, H. A review of terminology used to describe soot formation and evo-lution under combustion and pyrolytic conditions. ACS Nano 2020, 10, 12470–12490. [Google Scholar] [CrossRef]
- Su, Y.; Xie, M.; Lu, X.; Wei, H.; Geng, H.; Yang, Z.; Zhang, Y. Facile synthesis and photoelectric properties of carbon dots with upconversion fluorescence using arc-synthesized carbon by-products. RSC Adv. 2013, 4, 4839–4842. [Google Scholar] [CrossRef]
- Biazar, N.; Poursalehi, R.; Delavari, H. Optical and structural properties of carbon dots/TiO2 nanostructures prepared via DC arc discharge in liquid. IP Conf. Proc. 2018, 1920, 020033. [Google Scholar]
- Wang, L.; Ruan, F.; Lv, T.; Liu, Y.; Deng, D.; Zhao, S.; Wang, H.; Xu, S. One step synthesis of Al/N co-doped carbon nanoparticles with enhanced photoluminescence. J. Lumin. 2015, 158, 1–5. [Google Scholar] [CrossRef]
- Inderbir, S.; Arora, R.; Dhiman, H.; Pahwa, R. Carbon quantum dots: Synthesis, characterization and biomedical applications. Turk. J. Pharm. Sci. 2018, 2, 219–230. [Google Scholar]
- Qu, Y.; Li, X.; Zhang, H.; Huang, R.; Qi, W.; Su, R.; He, Z. Controllable synthesis of a sponge-like Z-scheme N, S-CQDs/Bi2MoO6@ TiO2 film with enhanced photocatalytic and antimicrobial activity under visi-ble/NIR light irradiation. J. Hazard. Mater. 2022, 429, 128310. [Google Scholar] [CrossRef] [PubMed]
- Kaixin, C.; Zhu, Q.; Qi, L.; Guo, M.; Gao, W.; Gao, Q. Synthesis and Properties of Nitro-gen-Doped Carbon Quantum Dots Using Lactic Acid as Carbon Source. Materials 2022, 15, 466. [Google Scholar]
- Henriquez, G.; Ahlawat, J.; Fairman, R.; Narayan, M. Citric Acid-Derived Carbon Quantum Dots Attenuate Paraquat-Induced Neuronal Compromise In Vitro and In Vivo. ACS Chem. Neurosci. 2022, 13, 2399–2409. [Google Scholar] [CrossRef]
- Nammahachak, N.; Aup-Ngoen, K.K.; Asanithi, P.; Horpratum, M.; Chuangchote, S.; Ratanaphan, S.; Surareungchai, W. Hydrothermal synthesis of carbon quantum dots with size tunability via heterogeneous nucleation. RSC Adv. 2022, 12, 31729–31733. [Google Scholar] [CrossRef]
- Jamila, G.S.; Sajjad, S.; Leghari, S.A.K.; Kallio, T.; Flox, C. Glucose derived carbon quantum dots on tungstate-titanate nanocomposite for hydrogen energy evolution and solar light catalysis. J. Nanostruct. Chem. 2021, 12, 611–623. [Google Scholar] [CrossRef]
- Qiu, Y.; Li, D.; Li, Y.; Ma, X.; Li, J. Green carbon quantum dots from sustainable lignocellulosic biomass and its application in the detection of Fe3+. Cellulose 2021, 29, 367–378. [Google Scholar] [CrossRef]
- Aayushi, K.; Maity, B.; Basu, S. Rice Husk-Derived Carbon Quantum Dots-Based Dual-Mode Nano-probe for Selective and Sensitive Detection of Fe3+ and Fluoroquinolones. ACS Biomater. Sci. Eng. 2022, 11, 4764–4776. [Google Scholar]
- El-Brolsy, H.M.E.M.; Hanafy, N.A.N.; El-Kemary, M.A. Fighting Non-Small Lung Cancer Cells Using Optimal Functionalization of Targeted Carbon Quantum Dots Derived from Natural Sources Might Provide Potential Therapeutic and Cancer Bio Image Strategies. Int. J. Mol. Sci. 2022, 23, 13283. [Google Scholar] [CrossRef]
- Kumari, M.; Chaudhary, G.R.; Chaudhary, S.; Umar, A.; Akbar, S.; Baskoutas, S. Bio-Derived Fluorescent Carbon Dots: Synthesis, Properties and Applications. Molecules 2022, 27, 5329. [Google Scholar] [CrossRef]
- Yao, L.; Zhao, M.-M.; Luo, Q.-W.; Zhang, Y.-C.; Liu, T.-T.; Yang, Z.; Liao, M.; Tu, P.; Zeng, K.-W. Carbon Quantum Dots-Based Nanozyme from Coffee Induces Cancer Cell Ferroptosis to Activate Antitumor Immunity. ACS Nano 2022, 16, 9228–9239. [Google Scholar] [CrossRef]
- Zhang, B.; Liu, C.; Liu, Y. A Novel One-Step Approach to Synthesize Fluorescent Carbon Nanoparticles. Eur. J. Inorg. Chem. 2010, 2010, 4411–4414. [Google Scholar] [CrossRef]
- Castañeda-Serna, H.U.; Calderón-Domínguez, G.; García-Bórquez, A.; Salgado-Cruz, M.d.l.P.; Rebollo, R.R.F. Structural and luminescent properties of CQDs produced by microwave and conventional hy-drothermal methods using pelagic Sargassum as carbon source. Opt. Mater. 2022, 126, 112156. [Google Scholar] [CrossRef]
- Hong, Y.; Chen, X.; Zhang, Y.; Zhu, Y.; Sun, J.; Swihart, M.T.; Tan, K.; Dong, L. One-pot hydrothermal synthesis of high quantum yield orange-emitting carbon quantum dots for sensitive detection of per-fluorinated compounds. New J. Chem. 2022, 41, 19658–19666. [Google Scholar] [CrossRef]
- Ye, H.; Liu, B.; Wang, J.; Zhou, C.; Xiong, Z.; Zhao, L. A Hydrothermal Method to Generate Carbon Quantum Dots from Waste Bones and Their Detection of Laundry Powder. Molecules 2022, 27, 6479. [Google Scholar] [CrossRef]
- Huo, X.; Liu, L.; Bai, Y.; Qin, J.; Yuan, L.; Feng, F. Facile synthesis of yellowish-green emitting carbon quantum dots and their applications for phoxim sensing and cellular imaging. Anal. Chim. Acta 2021, 1206, 338685. [Google Scholar] [CrossRef]
- Liu, H.; Ye, T.; Mao, C. Fluorescent Carbon Nanoparticles Derived from Candle Soot. Angew. Chem. 2007, 119, 6593–6595. [Google Scholar] [CrossRef]
- Liu, R.; Wu, D.; Liu, S.; Koynov, K.; Knoll, W.; Li, Q. An Aqueous Route to Multicolor Photoluminescent Carbon Dots Using Silica Spheres as Carriers. Angew. Chem. Int. Ed. 2009, 48, 4598–4601. [Google Scholar] [CrossRef]
- Pan, D.; Zhang, J.; Li, Z.; Wu, C.; Yan, X.; Wu, M. Observation of pH-, solvent-, spin-, and excitation-dependent blue photoluminescence from carbon nanoparticles. Chem. Commun. 2010, 46, 3681–3683. [Google Scholar] [CrossRef]
- Martindale, B.C.M.; Hutton, G.A.M.; Caputo, C.A.; Reisner, E. Solar Hydrogen Production Using Carbon Quantum Dots and a Molecular Nickel Catalyst. J. Am. Chem. Soc. 2015, 137, 6018–6025. [Google Scholar] [CrossRef] [PubMed]
- Rong, M.; Feng, Y.; Wang, Y.; Chen, X. One-pot solid phase pyrolysis synthesis of nitrogen-doped carbon dots for Fe3+ sensing and bioimaging. Sens. Actuators B Chem. 2017, 245, 868–874. [Google Scholar] [CrossRef]
- Ma, C.; Zhou, Y.; Yan, W.; He, W.; Liu, Q.; Li, Z.; Wang, H.; Li, X. Predominant catalytic performance of nickel nanoparticles embedded into nitrogen-doped carbon quantum dot-based nanosheets for the nitrore-duction of halogenated nitrobenzene. ACS Sustain. Chem. Eng. 2022, 25, 8162–8171. [Google Scholar] [CrossRef]
- Otten, M.; Hildebrandt, M.; Kühnemuth, R.; Karg, M. Pyrolysis and Solvothermal Synthesis for Carbon Dots: Role of Purification and Molecular Fluorophores. Langmuir 2022, 38, 6148–6157. [Google Scholar] [CrossRef] [PubMed]
- Krishnamoorthy, K.; Veerapandian, M.; Mohan, R.; Kim, S.-J. Investigation of Raman and photoluminescence studies of reduced graphene oxide sheets. Appl. Phys. A 2011, 106, 501–506. [Google Scholar] [CrossRef]
- Zong, J.; Zhu, Y.; Yang, X.; Shen, J.; Li, C. Synthesis of photoluminescent carbogenic dots using mesoporous silica spheres as nanoreactors. Chem. Commun. 2010, 47, 764–766. [Google Scholar] [CrossRef]
- Ahlawat, A.; Rana, P.S.; Solanki, P.R. Studies of photocatalytic and optoelectronic properties of microwave synthesized and polyethyleneimine stabilized carbon quantum dots. Mater. Lett. 2021, 305, 130830. [Google Scholar] [CrossRef]
- Architha, N.; Ragupathi, M.; Shobana, C.; Selvankumar, T.; Kumar, P.; Lee, Y.S.; Selvan, R.K. Microwave-assisted green synthesis of fluorescent carbon quantum dots from Mexican Mint extract for Fe3+ detection and bio-imaging applications. Environ. Res. 2021, 199, 111263. [Google Scholar] [CrossRef]
- Harshita, L.; Yadav, P.; Jain, Y.; Sharma, M.; Reza, M.; Agarwal, M.; Gupta, R. One-pot microwave-assisted synthesis of blue emissive multifunctional NSP co-doped carbon dots as a nanoprobe for sequential detection of Cr (VI) and ascorbic acid in real samples, fluorescent ink and logic gate operation. J. Mol. Liq. 2022, 346, 117088. [Google Scholar]
- Larsson, M.A.; Ramachandran, P.; Jarujamrus, P.; Lee, H.L. Microwave Synthesis of Blue Emissive N-Doped Carbon Quantum Dots as a Fluorescent Probe for Free Chlorine Detection. Sains Malays. 2022, 51, 1197–1212. [Google Scholar] [CrossRef]
- Jeong, G.; Lee, J.M.; Lee, J.a.; Praneerad, J.; Choi, C.A.; Supchocksoonthorn, P.; Roy, A.K.; In, I. Microwave-assisted synthesis of multifunctional fluorescent carbon quantum dots from A4/B2 polyamidation monomer sets. Appl. Surf. Sci. 2021, 542, 148471. [Google Scholar] [CrossRef]
- Bourlinos, A.B.; Stassinopoulos, A.; Anglos, D.; Zboril, R.; Georgakilas, V.; Giannelis, E.P. Photoluminescent Carbogenic Dots. Chem. Mater. 2008, 20, 4539–4541. [Google Scholar] [CrossRef]
- Tang, L.; Ji, R.; Cao, X.; Lin, J.; Jiang, H.; Li, X.; Teng, K.S.; Luk, C.M.; Zeng, S.; Hao, J.; et al. Deep Ultraviolet Photoluminescence of Water-Soluble Self-Passivated Graphene Quantum Dots. ACS Nano 2012, 6, 5102–5110. [Google Scholar] [CrossRef]
- Hola, K.; Bourlinos, A.B.; Kozak, O.; Berka, K.; Siskova, K.M.; Havrdova, M.; Tucek, J.; Safarova, K.; Otyepka, M.; Giannelis, E.P.; et al. Photoluminescence effects of graphitic core size and surface functional groups in carbon dots: COO− induced red-shift emission. Carbon 2014, 70, 279–286. [Google Scholar] [CrossRef]
- Sciortino, A.; Marino, E.; van Dam, B.; Schall, P.; Cannas, M.; Messina, F. Solvatochromism un-ravels the emission mechanism of carbon nanodots. J. Phys. Chem. Lett. 2016, 17, 3419–3423. [Google Scholar] [CrossRef]
- Dager, A.; Uchida, T.; Maekawa, T.; Tachibana, M. Synthesis and characterization of Mono-disperse Carbon Quantum Dots from Fennel Seeds: Photoluminescence analysis using Machine Learning. Sci. Rep. 2019, 9, 14004. [Google Scholar] [CrossRef] [Green Version]
- Zhang, W.; Yu, S.F.; Fei, L.; Jin, L.; Pan, S.; Lin, P. Large-area color controllable remote carbon white-light light-emitting diodes. Carbon 2015, 85, 344–350. [Google Scholar] [CrossRef]
- Martindale, B.C.M.; Hutton, G.A.M.; Caputo, C.A.; Prantl, S.; Godin, R.; Durrant, J.R.; Reisner, E. Enhancing Light Absorption and Charge Transfer Efficiency in Carbon Dots through Graphitization and Core Nitrogen Doping. Angew. Chem. 2017, 129, 6559–6563. [Google Scholar] [CrossRef] [Green Version]
- Tingting, Y.; Wang, H.; Guo, C.; Zhai, Y.; Yang, J.; Yuan, J. A rapid microwave synthesis of green-emissive carbon dots with solid-state fluorescence and pH-sensitive properties. R. Soc. Open Sci. 2018, 7, 180245. [Google Scholar]
- Haitao, L.; Kang, Z.; Liu, Y.; Lee, S.-T. Carbon nanodots: Synthesis, properties and applications. J. Mater. Chem. 2012, 46, 24230–24253. [Google Scholar]
- Zheng, Y.; Yang, D.; Wu, X.; Yan, H.; Zhao, Y.; Feng, B.; Duan, K.; Weng, J.; Wang, J. A facile approach for the synthesis of highly luminescent carbon dots using vitamin-based small organic molecules with benzene ring structure as precursors. RSC Adv. 2015, 5, 90245–90254. [Google Scholar] [CrossRef]
- Hou, H.; Banks, C.E.; Jing, M.; Zhang, Y.; Ji, X. Carbon Quantum Dots and Their Derivative 3D Porous Carbon Frameworks for Sodium-Ion Batteries with Ultralong Cycle Life. Adv. Mater. 2015, 27, 7861–7866. [Google Scholar] [CrossRef]
- Semeniuk, M.; Yi, Z.; Poursorkhabi, V.; Tjong, J.; Jaffer, S.; Lu, Z.-H.; Sain, M. Future per-spectives and review on organic carbon dots in electronic applications. ACS Nano 2019, 6, 6224–6255. [Google Scholar] [CrossRef] [PubMed]
- Bomben, K.D.; Moulder, J.F.; Stickle, W.F.; Sobol, P.E. Handbook of X-Ray Photoelectron Spectroscopy: A Reference Book of Standard Spectra for Identication and Interpretation of XPS, Physical Electronics, Eden Prairie; Perkin-Elmer Corporation: Waltham, MA, USA, 1995. [Google Scholar]
- Zhou, Y.; Sharma, S.K.; Peng, Z.; Leblanc, R.M. Polymers in Carbon Dots: A Review. Polymers 2017, 9, 67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kolanowska, A.; Dzido, G.; Krzywiecki, M.; Tomczyk, M.M.; Łukowiec, D.; Ruczka, S.; Boncel, S. Carbon Quantum Dots from Amino Acids Revisited: Survey of Renewable Precursors toward High Quan-tum-Yield Blue and Green Fluorescence. ACS Omega 2022, 45, 41165–41176. [Google Scholar] [CrossRef]
- Qiang, S.; Zhang, L.; Li, Z.; Liang, J.; Li, P.; Song, J.; Guo, K.; Wang, Z.; Fan, Q. New Insights into the Cellular Toxicity of Carbon Quantum Dots to Escherichia coli. Antioxidants 2022, 11, 2475. [Google Scholar] [CrossRef]
- Gayen, B.; Palchoudhury, S.; Chowdhury, J. Carbon Dots: A Mystic Star in the World of Nanoscience. J. Nanomater. 2019, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Hu, C.; Yu, C.; Li, M.; Wang, X.; Yang, J.; Zhao, Z.; Eychmüller, A.; Sun, Y.-P.; Qiu, J. Chemically Tailoring Coal to Fluorescent Carbon Dots with Tuned Size and Their Capacity for Cu(II) Detection. Small 2014, 10, 4926–4933. [Google Scholar] [CrossRef]
- Wang, S.; Kirillova, K.; Lehto, X. Travelers’ food experience sharing on social network sites. J. Travel Tour. Mark. 2016, 34, 680–693. [Google Scholar] [CrossRef]
- Jiang, K.; Zhang, L.; Lu, J.; Xu, C.; Cai, C.; Lin, H. Triple-Mode Emission of Carbon Dots: Applications for Advanced Anti-Counterfeiting. Angew. Chem. 2016, 128, 7347–7351. [Google Scholar] [CrossRef]
- Li, F.; Li, Y.; Yang, X.; Han, X.; Jiao, Y.; Wei, T.; Yang, D.; Xu, H.; Nie, G. Highly Fluorescent Chiral N-S-Doped Carbon Dots from Cysteine: Affecting Cellular Energy Metabolism. Angew. Chem. 2018, 130, 2401–2406. [Google Scholar] [CrossRef]
- Anwar, S.; Ding, H.; Xu, M.; Hu, X.; Li, Z.; Wang, J.; Bi, H. Recent advances in synthesis, optical properties, and bio-medical applications of carbon dots. ACS Appl. Bio Mater. 2019, 6, 2317–2338. [Google Scholar] [CrossRef]
- Jhonsi, M.A. Carbon Quantum Dots for Bioimaging. In State of the Art in Nano-Bioimaging; IntechOpen: London, UK, 2018; pp. 35–55. [Google Scholar]
- Konstantinos, D. Carbon quantum dots: Surface passivation and functionalization. Curr. Org. Chem. 2016, 6, 682–695. [Google Scholar]
- Li, L.; Dong, T. Photoluminescence tuning in carbon dots: Surface passivation or/and functionalization, heteroatom doping. J. Mater. Chem. C 2018, 30, 7944–7970. [Google Scholar] [CrossRef]
- Peng, H.; Travas-Sejdic, J. Simple Aqueous Solution Route to Luminescent Carbogenic Dots from Carbohydrates. Chem. Mater. 2009, 21, 5563–5565. [Google Scholar] [CrossRef]
- Wang, L.; Li, W.; Yin, L.; Liu, Y.; Guo, H.; Lai, J.; Han, Y.; Li, G.; Li, M.; Zhang, J.; et al. Full-color fluorescent carbon quantum dots. Sci. Adv. 2020, 6, eabb6772. [Google Scholar] [CrossRef]
- Nisha, P.; Amrita, D. Theoretical study of Dependence of Wavelength on Size of Quantum Dot. Int. J. Sci. Res. Dev. 2016, 4, 126–130. [Google Scholar]
- Kandasamy, G. Recent Advancements in Doped/Co-Doped Carbon Quantum Dots for Multi-Potential Applications. C 2019, 5, 24. [Google Scholar] [CrossRef] [Green Version]
- Darragh, C.; Rocks, C.; Padmanaban, D.B.; Maguire, P.; Svrcek, V.; Mariotti, D. Environmentally friendly nitrogen-doped carbon quantum dots for next generation solar cells. Sustain. Energy Fuels 2017, 7, 1611–1619. [Google Scholar]
- Gao, R.; Wu, Z.; Wang, L.; Liu, J.; Deng, Y.; Xiao, Z.; Fang, J.; Liang, Y. Green Preparation of Fluorescent Nitrogen-Doped Carbon Quantum Dots for Sensitive Detection of Oxytetracycline in Environmental Samples. Nanomaterials 2020, 10, 1561. [Google Scholar] [CrossRef]
- Yu, J.; Liu, C.; Yuan, K.; Lu, Z.; Cheng, Y.; Li, L.; Zhang, X.; Jin, P.; Meng, F.; Liu, H. Luminescence Mechanism of Carbon Dots by Tailoring Functional Groups for Sensing Fe3+ Ions. Nanomaterials 2018, 8, 233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zuo, G.; Xie, A.; Li, J.; Su, T.; Pan, X.; Dong, W. Large Emission Red-Shift of Carbon Dots by Fluorine Doping and Their Applications for Red Cell Imaging and Sensitive Intracellular Ag+ Detection. J. Phys. Chem. C 2017, 121, 26558–26565. [Google Scholar] [CrossRef]
- Baker, S.N.; Baker, G.A. Luminescent carbon nanodots: Emergent nanolights. Angew. Chem. Int. Ed. 2010, 49, 6726–6744. [Google Scholar] [CrossRef] [PubMed]
- Gokus, T.; Nair, R.R.; Bonetti, A.; Böhmler, M.; Lombardo, A.; Novoselov, K.; Geim, A.K.; Ferrari, A.C.; Hartschuh, A. Making Graphene Luminescent by Oxygen Plasma Treatment. ACS Nano 2009, 3, 3963–3968. [Google Scholar] [CrossRef] [Green Version]
- Demchenko, A.P.; Dekaliuk, M.O. Novel fluorescent carbonic nanomaterials for sensing and imaging. Methods Appl. Fluoresc. 2013, 1, 042001. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, R.; Feng, B.; Zhong, X.; Ostrikov, K. Photoluminescence mechanism of carbon dots: Triggering high-color-purity red fluorescence emission through edge amino protonation. Nat. Commun. 2021, 12, 6856. [Google Scholar] [CrossRef]
- Nguyen, H.A.; Srivastava, I.; Pan, D.; Gruebele, M. Unraveling the Fluorescence Mechanism of Carbon Dots with Sub-Single-Particle Resolution. ACS Nano 2020, 14, 6127–6137. [Google Scholar] [CrossRef]
- An, Y.; Liu, C.; Li, Y.; Chen, M.; Zheng, Y.; Tian, H.; Shi, R.; He, X.; Lin, X. Preparation of Multicolour Solid Fluorescent Carbon Dots for Light-Emitting Diodes Using Phenylethylamine as a Co-Carbonization Agent. Int. J. Mol. Sci. 2022, 23, 11071. [Google Scholar] [CrossRef]
- Cao, L.; Meziani, M.J.; Sahu, S.; Sun, Y.-P. Photoluminescence Properties of Graphene versus Other Carbon Nanomaterials. Acc. Chem. Res. 2012, 46, 171–180. [Google Scholar] [CrossRef]
- Fang, Y.; Guo, S.; Li, D.; Zhu, C.; Ren, W.; Dong, S.; Wang, E. Easy Synthesis and Imaging Applications of Cross-Linked Green Fluorescent Hollow Carbon Nanoparticles. ACS Nano 2011, 6, 400–409. [Google Scholar] [CrossRef]
- Shen, J.; Zhu, Y.; Chen, C.; Yang, X.; Li, C. Facile preparation and upconversionlumi-nescence of graphene quantum dots. Chem. Commun. 2011, 9, 2580–2582. [Google Scholar] [CrossRef]
- Li, X.; Wang, H.; Shimizu, Y.; Pyatenko, A.; Kawaguchi, K.; Koshizaki, N. Preparation of carbon quantum dots with tunable photoluminescence by rapid laser passivation in ordinary organic solvents. Chem. Commun. 2010, 47, 932–934. [Google Scholar] [CrossRef]
- Nourbakhsh, A.; Cantoro, M.; Vosch, T.; Pourtois, G.; Clemente, F.; van der Veen, M.; Hofkens, J.; Heyns, M.M.; De Gendt, S.; Sels, B.F. Bandgap opening in oxygen plasma-treated graphene. Nanotechnology 2010, 21, 435203. [Google Scholar] [CrossRef]
- Dekaliuk, M.O.; Viagin, O.; Malyukin, Y.V.; Demchenko, A.P. Fluorescent carbon nanomateri-als:“Quantum dots” or nanoclusters? Phys. Chem. Chem. Phys. 2014, 30, 16075–16084. [Google Scholar] [CrossRef]
- Bibekananda, D.; Karak, N. A green and facile approach for the synthesis of water soluble fluorescent carbon dots from banana juice. RSC Adv. 2013, 22, 8286–8290. [Google Scholar]
- Dong, Y.; Shao, J.; Chen, C.; Li, H.; Wang, R.; Chi, Y.; Lin, X.; Chen, G. Blue luminescent graphene quantum dots and graphene oxide prepared by tuning the carbonization degree of citric acid. Carbon 2012, 50, 4738–4743. [Google Scholar] [CrossRef]
- Zhi, B.; Yao, X.; Cui, Y.; Orr, G.; Haynes, C.L. Synthesis, applications and potential photoluminescence mechanism of spectrally tunable carbon dots. Nanoscale 2019, 11, 20411–20428. [Google Scholar] [CrossRef]
- De Caluwé, E.; Halamouá, K.; Van Damme, P.; Adansoniadigitata, L. A review of traditional uses, phytochemistry and pharmacology. Afr. Focus 2010, 1, 11–51. [Google Scholar] [CrossRef]
- Lu, C.; Su, Q.; Yang, X. Ultra-long room-temperature phosphorescent carbon dots: pH sensing and dual-channel detection of tetracyclines. Nanoscale 2019, 11, 16036–16042. [Google Scholar] [CrossRef]
- Gao, W.; Zhang, S.; Wang, G.; Cui, J.; Lu, Y.; Rong, X.; Gao, C. A review on mechanism, applications and influencing factors of carbon quantum dots based photocatalysis. Ceram. Int. 2022, 48, 35986–35999. [Google Scholar] [CrossRef]
- Das, R.; Bandyopadhyay, R.; Pramanik, P. Carbon quantum dots from natural resource: A review. Mater. Today Chem. 2018, 8, 96–109. [Google Scholar] [CrossRef]
- Xu, J.; Tao, J.; Su, L.; Wang, J.; Jiao, T. A Critical Review of Carbon Quantum Dots: From Synthesis toward Applications in Electrochemical Biosensors for the Determination of a Depression-Related Neurotransmitter. Materials 2021, 14, 3987. [Google Scholar] [CrossRef]
- Dong, Y.-L.; Zhang, H.-G.; Rahman, Z.U.; Su, L.; Chen, X.-J.; Hu, J.; Chen, X.-G. Graphene oxide−Fe3O4 magnetic nanocomposites with peroxidase-like activity for colorimetric detection of glucose. Nanoscale 2012, 4, 3969–3976. [Google Scholar] [CrossRef]
- Ryan, B.J.; Carolan, N.; Ó’Fágáin, C. Horseradish and soybean peroxidases: Comparable tools for alternative niches? Trends Biotechnol. 2006, 24, 355–363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, L.; Zhuang, J.; Nie, L.; Zhang, J.; Zhang, Y.; Gu, N.; Wang, T.; Feng, J.; Yang, D.; Perrett, S.; et al. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nat. Nanotechnol. 2007, 2, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Floss, M.A.; Fink, T.; Maurer, F.; Volk, T.; Kreuer, S.; Müller-Wirtz, L.M. Exhaled Aldehydes as Biomarkers for Lung Diseases: A Narrative Review. Molecules 2022, 27, 5258. [Google Scholar] [CrossRef]
- Li, J.; Yao, S.-L.; Liu, S.-J.; Chen, Y.-Q. Fluorescent sensors for aldehydes based on luminescent metal–organic frameworks. Dalton Trans. 2021, 50, 7166–7175. [Google Scholar] [CrossRef]
- Ibáñez, D.; González-García, M.B.; Hernández-Santos, D.; Fanjul-Bolado, P. Spectroelectrochemical Enzyme Sensor System for Acetaldehyde Detection in Wine. Biosensors 2022, 12, 1032. [Google Scholar] [CrossRef]
- Mohammadi, M.; Khazaei, A.; Rezaei, A.; Huajun, Z.; Xuwei, S. Ionic-Liquid-Modified Carbon Quantum Dots as a Support for the Immobilization of Tungstate Ions (WO42−): Heterogeneous Nanocatalysts for the Oxidation of Alcohols in Water. ACS Sustain. Chem. Eng. 2019, 7, 5283–5291. [Google Scholar] [CrossRef]
- Mahamuni, N.N.; Gogate, P.R.; Pandit, A.B. Selective synthesis of sulfoxides from sulfides using ultrasound. Ultrason. Sonochem. 2007, 2, 135–142. [Google Scholar] [CrossRef] [PubMed]
- An, H.; Luo, H.; Xu, T.; Chang, S.; Chen, Y.; Zhu, Q.; Huang, Y.; Tan, H.; Li, Y.-G. Visible-Light-Driven Oxidation of Amines to Imines in Air Catalyzed by Polyoxometalate–Tris(bipyridine)ruthenium Hybrid Compounds. Inorg. Chem. 2022, 61, 10442–10453. [Google Scholar] [CrossRef]
- Chen, W.; Li, H.; Song, J.; Zhao, Y.; Ma, P.; Niu, J.; Wang, J. Binuclear Ru(III)-Containing Polyoxometalate with Efficient Photocatalytic Activity for Oxidative Coupling of Amines to Imines. Inorg. Chem. 2022, 61, 2076–2085. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, N.; Palasin, K.; Asano, Y. Novel Enzymatic Method for Imine Synthesis via the Oxi-dation of Primary Amines Using D-Amino Acid Oxidase from Porcine Kidney. Catalysts 2022, 5, 511. [Google Scholar] [CrossRef]
- Pyun, J. Graphene oxide as catalyst: Application of carbon materials beyond nanotechnology. Angew. Chem. Int. Ed. 2011, 1, 46–48. [Google Scholar] [CrossRef]
- Su, D.S.; Zhang, J.; Frank, B.; Thomas, A.; Wang, X.; Paraknowitsch, J.; Schlögl, R. Metal-Free Heterogeneous Catalysis for Sustainable Chemistry. Chemsuschem 2010, 3, 169–180. [Google Scholar] [CrossRef]
- Yu, H.; Peng, F.; Tan, J.; Hu, X.; Wang, H.; Yang, J.; Zheng, W. Selective Catalysis of the Aerobic Oxidation of Cyclohexane in the Liquid Phase by Carbon Nanotubes. Angew. Chem. 2011, 123, 4064–4068. [Google Scholar] [CrossRef]
- Shiri, M. Indoles in Multicomponent Processes (MCPs). Chem. Rev. 2012, 112, 3508–3549. [Google Scholar] [CrossRef]
- Vidhya, L.N.; Thirumurugan, P.; Noorulla, K.M.; Perumal, P.T. InCl3 mediated one-pot multicomponent synthesis, anti-microbial, antioxidant and anticancer evaluation of 3-pyranyl indole derivatives. Bioorganic Med. Chem. Lett. 2010, 17, 5054–5061. [Google Scholar]
- Gomha, S.M.; Abdel-Aziz, H.A. Synthesis of new heterocycles derived from 3-(3-methyl-1H-indol-2-yl)-3-oxopropanenitrile as potent antifungal agents. Bull. Korean Chem. Soc. 2012, 9, 2985–2990. [Google Scholar] [CrossRef] [Green Version]
- Fadda, A.A.; El-Mekabaty, A.; Mousa, I.A.; Elattar, K.M. Chemistry of 3-(1H-Indol-3-yl)-3-oxopropanenitrile. Synth. Commun. 2014, 44, 1579–1599. [Google Scholar] [CrossRef]
- Thirumurugan, P.; Nandakumar, A.; Muralidharan, D.; Perumal, P.T. Simple and convenient approach to the Kr€ ohnke pyridine type synthesis of functionalized indol-3-yl pyridine derivatives using 3-cyanoacetyl indole. J. Comb. Chem. 2010, 1, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Irfan, S.M.; Khan, P.; Abid, M.; Khan, M.M. Design, synthesis, and biological evaluation of novel fused spiro-4 H-pyran derivatives as bacterial biofilm disruptor. ACS Omega 2019, 16, 16794–16807. [Google Scholar]
- Kumar, S.P.; Silakari, O. The current status of O-heterocycles: A synthetic and medicinal overview. ChemMedChem 2018, 11, 1071–1087. [Google Scholar]
- Zhao, L.; Sun, Z.; Ma, J. Novel Relationship between Hydroxyl Radical Initiation and Surface Group of Ceramic Honeycomb Supported Metals for the Catalytic Ozonation of Nitrobenzene in Aqueous Solution. Environ. Sci. Technol. 2009, 43, 4157–4163. [Google Scholar] [CrossRef]
- McKeen, J.C.; Yan, Y.S.; Davis, M.E. Proton Conductivity in Sulfonic Acid-Functionalized Zeolite Beta: Effect of Hydroxyl Group. Chem. Mater. 2008, 20, 3791–3793. [Google Scholar] [CrossRef] [Green Version]
- Doyle, A.G.; Jacobsen, E.N. Small-molecule H-bond donors in asymmetric catalysis. Chem. Rev. 2007, 12, 5713–5743. [Google Scholar] [CrossRef]
- Chen, X.; Brauman, J.I. Hydrogen bonding lowers intrinsic nucleophilicity of solvated nucleophiles. J. Am. Chem. Soc. 2008, 45, 15038–15046. [Google Scholar] [CrossRef]
- Kitano, M.; Nakajima, K.; Kondo, J.N.; Hayashi, S.; Hara, M. Protonated Titanate Nanotubes as Solid Acid Catalyst. J. Am. Chem. Soc. 2010, 132, 6622–6623. [Google Scholar] [CrossRef]
- Navalon, S.; Dhakshinamoorthy, A.; Alvaro, M.; Garcia, H. Carbocatalysis by graphene-based materials. Chem. Rev. 2014, 12, 6179–6212. [Google Scholar] [CrossRef]
- Chang, B.; Fu, J.; Tian, Y.; Dong, X. Multifunctionalized Ordered Mesoporous Carbon as an Efficient and Stable Solid Acid Catalyst for Biodiesel Preparation. J. Phys. Chem. C 2013, 117, 6252–6258. [Google Scholar] [CrossRef]
- Fekry, A.M. An Innovative Simple Electrochemical Levofloxacin Sensor Assembled from Carbon Paste Enhanced with Nano-Sized Fumed Silica. Biosensors 2022, 12, 906. [Google Scholar] [CrossRef] [PubMed]
- Sitara, E.; Ehsan, M.F.; Nasir, H.; Iram, M.; Bukhari, A.B. Synthesis, characterization and photocatalytic activity of MoS2/ZnSe heterostructures for the degradation of levofloxacin. Catalysts 2020, 12, 1380. [Google Scholar] [CrossRef]







Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yadav, P.K.; Chandra, S.; Kumar, V.; Kumar, D.; Hasan, S.H. Carbon Quantum Dots: Synthesis, Structure, Properties, and Catalytic Applications for Organic Synthesis. Catalysts 2023, 13, 422. https://doi.org/10.3390/catal13020422
Yadav PK, Chandra S, Kumar V, Kumar D, Hasan SH. Carbon Quantum Dots: Synthesis, Structure, Properties, and Catalytic Applications for Organic Synthesis. Catalysts. 2023; 13(2):422. https://doi.org/10.3390/catal13020422
Chicago/Turabian StyleYadav, Pradeep Kumar, Subhash Chandra, Vivek Kumar, Deepak Kumar, and Syed Hadi Hasan. 2023. "Carbon Quantum Dots: Synthesis, Structure, Properties, and Catalytic Applications for Organic Synthesis" Catalysts 13, no. 2: 422. https://doi.org/10.3390/catal13020422