Membrane Vesicles Derived from Gut Microbiota and Probiotics: Cutting-Edge Therapeutic Approaches for Multidrug-Resistant Superbugs Linked to Neurological Anomalies
Abstract
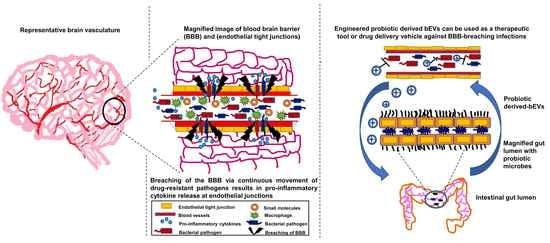
:1. Introduction
2. Blood–Brain Barrier (BBB): A Roadblock to Invading Pathogens
3. Multidrug-Resistant (MDR) Superbugs: A Prominent Case Involving the BBB
4. Bacteria-Derived EVs (bEVs): Nanoscale Vesicles
5. Nanoscale bEVs as Potential Therapeutic Platforms
6. Unresolved Issues with Gut Microbiota-Derived bEVs in Modulating the Gut–Brain Axis: Old Is Gold
6.1. Gut–Brain Axis
6.2. Gut Microbiota-Derived bEVs vs. Eukaryotic-Derived hEVs (Physiological Counterpart)
6.3. Problems Related to Gut Microbiota-Derived bEVs on BBB-Associated Diseases
6.4. Beneficial Roles of Probiotic-Derived bEVs on Gut–Brain-Axis Control
7. Filling Gaps with Probiotic-Derived bEVs against BBB-Breaching Pathogens
8. Remodeling of Probiotic-Derived bEVs against BBB-Invading Pathogens
8.1. Surface-Modified Proteins in Probiotic-Derived bEVs
8.2. Regulation of Neurotransmitters across the BBB
8.3. Quorum-Quenching Proteins
8.4. bEVs as a Drug Delivery Platform to Prevent Degradation and Immune Elimination of Antimicrobials
8.5. CRISPR/Cas-Modified bEVs as Biotherapeutic Agents against BBB-Breach-Related Infections
9. Potential for Application of Probiotic-Derived bEV Platforms against BBB-Associated CNS Infections
10. Future Research and Perspective
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gosselet, F.; Loiola, R.A.; Roig, A.; Rosell, A.; Culot, M. Central nervous system delivery of molecules across the blood-brain barrier. Neurochem. Int. 2021, 144, 104952. [Google Scholar] [CrossRef] [PubMed]
- Muldoon, L.L.; Alvarez, J.I.; Begley, D.J.; Boado, R.J.; Del Zoppo, G.J.; Doolittle, N.D.; Engelhardt, B.; Hallenbeck, J.M.; Lonser, R.R.; Ohlfest, J.R.; et al. Immunologic privilege in the central nervous system and the blood-brain barrier. J. Cereb. Blood Flow Metab. 2013, 33, 13–21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miner, J.J.; Diamond, M.S. Mechanisms of restriction of viral neuroinvasion at the blood-brain barrier. Curr. Opin. Immunol. 2016, 38, 18–23. [Google Scholar] [CrossRef]
- Cain, M.D.; Salimi, H.; Diamond, M.S.; Klein, R.S. Mechanisms of Pathogen Invasion into the Central Nervous System. Neuron 2019, 103, 771–783. [Google Scholar] [CrossRef]
- Li, S.; Nguyen, I.P.; Urbanczyk, K. Common infectious diseases of the central nervous system-clinical features and imaging characteristics. Quant. Imaging Med. Surg. 2020, 10, 2227–2259. [Google Scholar] [CrossRef]
- Sousa, S.A.; Feliciano, J.R.; Pita, T.; Soeiro, C.F.; Mendes, B.L.; Alves, L.G.; Leitao, J.H. Bacterial Nosocomial Infections: Multidrug Resistance as a Trigger for the Development of Novel Antimicrobials. Antibiotics 2021, 10, 942. [Google Scholar] [CrossRef]
- Loscher, W.; Potschka, H. Drug resistance in brain diseases and the role of drug efflux transporters. Nat. Rev. Neurosci. 2005, 6, 591–602. [Google Scholar] [CrossRef]
- Gaddy, J.A.; Actis, L.A. Regulation of Acinetobacter baumannii biofilm formation. Future Microbiol. 2009, 4, 273–278. [Google Scholar] [CrossRef] [Green Version]
- Galea, I. The blood-brain barrier in systemic infection and inflammation. Cell. Mol. Immunol. 2021, 18, 2489–2501. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Hussain, B.; Chang, J. Peripheral inflammation and blood-brain barrier disruption: Effects and mechanisms. CNS Neurosci. Ther. 2021, 27, 36–47. [Google Scholar] [CrossRef]
- Teleanu, R.I.; Preda, M.D.; Niculescu, A.G.; Vladacenco, O.; Radu, C.I.; Grumezescu, A.M.; Teleanu, D.M. Current Strategies to Enhance Delivery of Drugs across the Blood-Brain Barrier. Pharmaceutics 2022, 14, 987. [Google Scholar] [CrossRef] [PubMed]
- Bhowmik, A.; Khan, R.; Ghosh, M.K. Blood brain barrier: A challenge for effectual therapy of brain tumors. BioMed Res. Int. 2015, 2015, 320941. [Google Scholar] [CrossRef] [Green Version]
- Morse, S.V.; Mishra, A.; Chan, T.G.; de Rosales, R.T.M.; Choi, J.J. Liposome delivery to the brain with rapid short-pulses of focused ultrasound and microbubbles. J. Control. Release 2022, 341, 605–615. [Google Scholar] [CrossRef] [PubMed]
- Juhairiyah, F.; de Lange, E.C.M. Understanding Drug Delivery to the Brain Using Liposome-Based Strategies: Studies that Provide Mechanistic Insights Are Essential. AAPS J. 2021, 23, 114. [Google Scholar] [CrossRef] [PubMed]
- Curcio, M.; Cirillo, G.; Rouaen, J.R.C.; Saletta, F.; Nicoletta, F.P.; Vittorio, O.; Iemma, F. Natural Polysaccharide Carriers in Brain Delivery: Challenge and Perspective. Pharmaceutics 2020, 12, 1183. [Google Scholar] [CrossRef]
- Zhang, W.; Mehta, A.; Tong, Z.; Esser, L.; Voelcker, N.H. Development of Polymeric Nanoparticles for Blood-Brain Barrier Transfer-Strategies and Challenges. Adv. Sci. 2021, 8, 2003937. [Google Scholar] [CrossRef]
- Boyes, W.K.; van Thriel, C. Neurotoxicology of Nanomaterials. Chem. Res. Toxicol. 2020, 33, 1121–1144. [Google Scholar] [CrossRef]
- Daraee, H.; Etemadi, A.; Kouhi, M.; Alimirzalu, S.; Akbarzadeh, A. Application of liposomes in medicine and drug delivery. Artif. Cells Nanomed. Biotechnol. 2016, 44, 381–391. [Google Scholar] [CrossRef]
- Fikatas, A.; Dehairs, J.; Noppen, S.; Doijen, J.; Vanderhoydonc, F.; Meyen, E.; Swinnen, J.V.; Pannecouque, C.; Schols, D. Deciphering the Role of Extracellular Vesicles Derived from ZIKV-Infected hcMEC/D3 Cells on the Blood-Brain Barrier System. Viruses 2021, 13, 2363. [Google Scholar] [CrossRef]
- Correa, R.; Caballero, Z.; De Leon, L.F.; Spadafora, C. Extracellular Vesicles Could Carry an Evolutionary Footprint in Interkingdom Communication. Front. Cell. Infect. Microbiol. 2020, 10, 76. [Google Scholar] [CrossRef]
- Hendrix, A.; De Wever, O. Systemically circulating bacterial extracellular vesicles: Origin, fate, and function. Trends Microbiol. 2022, 30, 213–216. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Wang, Z.; Liu, X.; Tyler, B.M. Biogenesis and Biological Functions of Extracellular Vesicles in Cellular and Organismal Communication with Microbes. Front. Microbiol. 2022, 13, 817844. [Google Scholar] [CrossRef] [PubMed]
- Van Beek, L.F.; Surmann, K.; van den Berg van Saparoea, H.B.; Houben, D.; Jong, W.S.P.; Hentschker, C.; Ederveen, T.H.A.; Mitsi, E.; Ferreira, D.M.; van Opzeeland, F.; et al. Exploring metal availability in the natural niche of Streptococcus pneumoniae to discover potential vaccine antigens. Virulence 2020, 11, 1310–1328. [Google Scholar] [CrossRef] [PubMed]
- Malekan, M.; Siadat, S.D.; Aghasadeghi, M.; Shahrokhi, N.; Afrough, P.; Behrouzi, A.; Ahmadi, K.; Mousavi, S.F. Evaluation of protective immunity responses against pneumococcal PhtD and its C-terminal in combination with outer-membrane vesicles as adjuvants. J. Med. Microbiol. 2020, 69, 465–477. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Mandujano, A.; Hernandez-Cortez, C.; Ibarra, J.A.; Castro-Escarpulli, G. The outer membrane vesicles: Secretion system type zero. Traffic 2017, 18, 425–432. [Google Scholar] [CrossRef] [Green Version]
- Schaar, V.; Nordstrom, T.; Morgelin, M.; Riesbeck, K. Moraxella catarrhalis outer membrane vesicles carry beta-lactamase and promote survival of Streptococcus pneumoniae and Haemophilus influenzae by inactivating amoxicillin. Antimicrob. Agents Chemother. 2011, 55, 3845–3853. [Google Scholar] [CrossRef] [Green Version]
- Cuesta, C.M.; Guerri, C.; Urena, J.; Pascual, M. Role of Microbiota-Derived Extracellular Vesicles in Gut-Brain Communication. Int. J. Mol. Sci. 2021, 22, 4235. [Google Scholar] [CrossRef]
- Kato, T.; Fahrmann, J.F.; Hanash, S.M.; Vykoukal, J. Extracellular Vesicles Mediate B Cell Immune Response and Are a Potential Target for Cancer Therapy. Cells 2020, 9, 1518. [Google Scholar] [CrossRef]
- Rubio, A.P.D.; D’Antoni, C.L.; Piuri, M.; Perez, O.E. Probiotics, Their Extracellular Vesicles and Infectious Diseases. Front. Microbiol. 2022, 13, 864720. [Google Scholar] [CrossRef]
- Yaghoubfar, R.; Behrouzi, A.; Ashrafian, F.; Shahryari, A.; Moradi, H.R.; Choopani, S.; Hadifar, S.; Vaziri, F.; Nojoumi, S.A.; Fateh, A.; et al. Modulation of serotonin signaling/metabolism by Akkermansia muciniphila and its extracellular vesicles through the gut-brain axis in mice. Sci. Rep. 2020, 10, 22119. [Google Scholar] [CrossRef]
- Heavey, M.K.; Durmusoglu, D.; Crook, N.; Anselmo, A.C. Discovery and delivery strategies for engineered live biotherapeutic products. Trends Biotechnol. 2022, 40, 354–369. [Google Scholar] [CrossRef]
- Kuhn, T.; Koch, M.; Fuhrmann, G. Probiomimetics-Novel Lactobacillus-Mimicking Microparticles Show Anti-Inflammatory and Barrier-Protecting Effects in Gastrointestinal Models. Small 2020, 16, e2003158. [Google Scholar] [CrossRef]
- Shandilya, S.; Kumar, S.; Jha, N.K.; Kesari, K.K.; Ruokolainen, J. Interplay of gut microbiota and oxidative stress: Perspective on neurodegeneration and neuroprotection. J. Adv. Res. 2022, 38, 223–244. [Google Scholar] [CrossRef] [PubMed]
- Jahan, S.; Mukherjee, S.; Ali, S.; Bhardwaj, U.; Choudhary, R.K.; Balakrishnan, S.; Naseem, A.; Mir, S.A.; Banawas, S.; Alaidarous, M.; et al. Pioneer Role of Extracellular Vesicles as Modulators of Cancer Initiation in Progression, Drug Therapy, and Vaccine Prospects. Cells 2022, 11, 490. [Google Scholar] [CrossRef] [PubMed]
- Elsharkasy, O.M.; Nordin, J.Z.; Hagey, D.W.; de Jong, O.G.; Schiffelers, R.M.; Andaloussi, S.E.; Vader, P. Extracellular vesicles as drug delivery systems: Why and how? Adv. Drug Deliv. Rev. 2020, 159, 332–343. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, J.P.; Holme, M.N.; Stevens, M.M. Re-Engineering Extracellular Vesicles as Smart Nanoscale Therapeutics. ACS Nano 2017, 11, 69–83. [Google Scholar] [CrossRef] [Green Version]
- Loureiro, A.; da Silva, G.J. CRISPR-Cas: Converting A Bacterial Defence Mechanism into A State-of-the-Art Genetic Manipulation Tool. Antibiotics 2019, 8, 18. [Google Scholar] [CrossRef] [Green Version]
- Gao, Y.; Gao, K.; Yang, H. CRISPR/Cas: A potential gene-editing tool in the nervous system. Cell Regen. 2020, 9, 12. [Google Scholar] [CrossRef]
- Rhea, E.M.; Banks, W.A. Role of the Blood-Brain Barrier in Central Nervous System Insulin Resistance. Front. Neurosci. 2019, 13, 521. [Google Scholar] [CrossRef] [Green Version]
- Le Govic, Y.; Demey, B.; Cassereau, J.; Bahn, Y.S.; Papon, N. Pathogens infecting the central nervous system. PLoS Pathog. 2022, 18, e1010234. [Google Scholar] [CrossRef]
- Festoff, B.W.; Sajja, R.K.; Cucullo, L. Proximate Mediators of Microvascular Dysfunction at the Blood-Brain Barrier: Neuroinflammatory Pathways to Neurodegeneration. BioMed Res. Int. 2017, 2017, 1549194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shaheryar, Z.A.; Khan, M.A.; Adnan, C.S.; Zaidi, A.A.; Hanggi, D.; Muhammad, S. Neuroinflammatory Triangle Presenting Novel Pharmacological Targets for Ischemic Brain Injury. Front. Immunol. 2021, 12, 748663. [Google Scholar] [CrossRef] [PubMed]
- Connell, J.J.; Chatain, G.; Cornelissen, B.; Vallis, K.A.; Hamilton, A.; Seymour, L.; Anthony, D.C.; Sibson, N.R. Selective permeabilization of the blood-brain barrier at sites of metastasis. J. Natl. Cancer Inst. 2013, 105, 1634–1643. [Google Scholar] [CrossRef] [Green Version]
- Banks, W.A. Characteristics of compounds that cross the blood-brain barrier. BMC Neurol. 2009, 9 (Suppl. 1), S3. [Google Scholar] [CrossRef] [Green Version]
- Pardridge, W.M. Drug transport across the blood-brain barrier. J. Cereb. Blood Flow Metab. 2012, 32, 1959–1972. [Google Scholar] [CrossRef] [PubMed]
- Dando, S.J.; Mackay-Sim, A.; Norton, R.; Currie, B.J.; John, J.A.S.; Ekberg, J.A.; Batzloff, M.; Ulett, G.C.; Beacham, I.R. Pathogens penetrating the central nervous system: Infection pathways and the cellular and molecular mechanisms of invasion. Clin. Microbiol. Rev. 2014, 27, 691–726. [Google Scholar] [CrossRef] [Green Version]
- Novakova, I.; Subileau, E.A.; Toegel, S.; Gruber, D.; Lachmann, B.; Urban, E.; Chesne, C.; Noe, C.R.; Neuhaus, W. Transport rankings of non-steroidal antiinflammatory drugs across blood-brain barrier in vitro models. PLoS ONE 2014, 9, e86806. [Google Scholar] [CrossRef]
- Marchi, N.; Betto, G.; Fazio, V.; Fan, Q.; Ghosh, C.; Machado, A.; Janigro, D. Blood-brain barrier damage and brain penetration of antiepileptic drugs: Role of serum proteins and brain edema. Epilepsia 2009, 50, 664–677. [Google Scholar] [CrossRef] [Green Version]
- Appelboom, G.; Detappe, A.; LoPresti, M.; Kunjachan, S.; Mitrasinovic, S.; Goldman, S.; Chang, S.D.; Tillement, O. Stereotactic modulation of blood-brain barrier permeability to enhance drug delivery. Neuro-Oncology 2016, 18, 1601–1609. [Google Scholar] [CrossRef]
- Salehi, A.; Paturu, M.R.; Patel, B.; Cain, M.D.; Mahlokozera, T.; Yang, A.B.; Lin, T.H.; Leuthardt, E.C.; Yano, H.; Song, S.K.; et al. Therapeutic enhancement of blood-brain and blood-tumor barriers permeability by laser interstitial thermal therapy. Neurooncol. Adv. 2020, 2, vdaa071. [Google Scholar] [CrossRef]
- Karmur, B.S.; Philteos, J.; Abbasian, A.; Zacharia, B.E.; Lipsman, N.; Levin, V.; Grossman, S.; Mansouri, A. Blood-Brain Barrier Disruption in Neuro-Oncology: Strategies, Failures, and Challenges to Overcome. Front. Oncol. 2020, 10, 563840. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, P.S. Antibacterial Discovery: 21st Century Challenges. Antibiotics 2020, 9, 213. [Google Scholar] [CrossRef]
- Saha, M.; Sarkar, A. Review on Multiple Facets of Drug Resistance: A Rising Challenge in the 21st Century. J. Xenobiotics 2021, 11, 197–214. [Google Scholar] [CrossRef]
- Sun, H.; Zhang, Q.; Wang, R.; Wang, H.; Wong, Y.T.; Wang, M.; Hao, Q.; Yan, A.; Kao, R.Y.; Ho, P.L.; et al. Resensitizing carbapenem- and colistin-resistant bacteria to antibiotics using auranofin. Nat. Commun. 2020, 11, 5263. [Google Scholar] [CrossRef] [PubMed]
- Nation, R.L.; Li, J. Colistin in the 21st century. Curr. Opin. Infect. Dis. 2009, 22, 535–543. [Google Scholar] [CrossRef]
- Kim, B.-N.; Peleg, A.Y.; Lodise, T.P.; Lipman, J.; Li, J.; Nation, R.; Paterson, D.L. Management of meningitis due to antibiotic-resistant Acinetobacter species. Lancet Infect. Dis. 2009, 9, 245–255. [Google Scholar] [CrossRef] [Green Version]
- Huang, Q.; Zhang, X.; Jia, A.; Huang, Q.; Jiang, Y.; Xie, L. The Pharmacokinetics/Pharmacodynamics and Neurotoxicity of Tigecycline Intraventricular Injection for the Treatment of Extensively Drug-Resistant Acinetobacter baumannii Intracranial Infection. Infect. Drug Resist. 2022, 15, 4809–4817. [Google Scholar] [CrossRef]
- Yu, X.B.; Huang, Y.Y.; Zhang, X.S.; Wang, Y.Z.; Shi, D.W.; Zhang, C.H.; Chen, J.; Wang, X.R.; Lin, G.Y. Intraventricular colistin sulphate as a last resort therapy in a patient with multidrug-resistant Acinetobacter baumannii induced post-neurosurgical ventriculitis. Br. J. Clin. Pharmacol. 2022, 88, 3490–3494. [Google Scholar] [CrossRef]
- Lin, M.F.; Lan, C.Y. Antimicrobial resistance in Acinetobacter baumannii: From bench to bedside. World J. Clin. Cases 2014, 2, 787–814. [Google Scholar] [CrossRef]
- Wu, X.; Wang, L.; Ye, Y.Z.; Yu, H. Postoperative multidrug-resistant Acinetobacter baumannii meningitis successfully treated with intravenous doxycycline and intraventricular gentamicin: A case report. World J. Clin. Cases 2019, 7, 4342–4348. [Google Scholar] [CrossRef]
- Schneider, F.; Gessner, A.; El-Najjar, N. Efficacy of Vancomycin and Meropenem in Central Nervous System Infections in Children and Adults: Current Update. Antibiotics 2022, 11, 173. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, E.; Sivalingam, K.; Doke, M.; Samikkannu, T. Effects of Drugs of Abuse on the Blood-Brain Barrier: A Brief Overview. Front. Neurosci. 2020, 14, 513. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.-Q.; Zhan, R.-C.; Jia, W.; Zhang, B.-Q.; Wang, J.-J. A case report of intraventricular tigecycline therapy for intracranial infection with extremely drug resistant Acinetobacter baumannii. Medicine 2017, 96, e7703. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; An, Y.; Li, L.; Yi, H. Intrathecal Injection of Tigecycline and Polymyxin B in the Treatment of Extensively Drug-Resistant Intracranial Acinetobacter baumannii Infection: A Case Report and Review of the Literature. Infect. Drug Resist. 2022, 15, 1411–1423. [Google Scholar] [CrossRef]
- Zhong, Y.M.; Zhang, X.H.; Ma, Z.; Liu, W.E. Prevalence of Escherichia coli ST1193 Causing Intracranial Infection in Changsha, China. Trop. Med. Infect. Dis. 2022, 7, 217. [Google Scholar] [CrossRef]
- Coureuil, M.; Join-Lambert, O.; Lécuyer, H.; Bourdoulous, S.; Marullo, S.; Nassif, X. Mechanism of meningeal invasion by Neisseria meningitidis. Virulence 2014, 3, 164–172. [Google Scholar] [CrossRef] [Green Version]
- Cutting, A.S.; Del Rosario, Y.; Mu, R.; Rodriguez, A.; Till, A.; Subramani, S.; Gottlieb, R.A.; Doran, K.S. The Role of Autophagy during Group B Streptococcus Infection of Blood-Brain Barrier Endothelium. J. Biol. Chem. 2014, 289, 35711–35723. [Google Scholar] [CrossRef] [Green Version]
- Parisi, D.N.; Martinez, L.R. Intracellular Haemophilus influenzae invades the brain. Virulence 2014, 5, 645–647. [Google Scholar] [CrossRef] [Green Version]
- Chacko, A.; Delbaz, A.; Walkden, H.; Basu, S.; Armitage, C.W.; Eindorf, T.; Trim, L.K.; Miller, E.; West, N.P.; John, J.A.S.; et al. Chlamydia pneumoniae can infect the central nervous system via the olfactory and trigeminal nerves and contributes to Alzheimer’s disease risk. Sci. Rep. 2022, 12, 2759. [Google Scholar] [CrossRef]
- Guttman, J.A.; Samji, F.N.; Li, Y.; Vogl, A.W.; Finlay, B.B. Evidence that Tight Junctions Are Disrupted Due to Intimate Bacterial Contact and Not Inflammation during Attaching and Effacing Pathogen Infection In Vivo. Infect. Immun. 2006, 74, 6075–6084. [Google Scholar] [CrossRef]
- Doran, K.S.; Engelson, E.J.; Khosravi, A.; Maisey, H.C.; Fedtke, I.; Equils, O.; Michelsen, K.S.; Arditi, M.; Peschel, A.; Nizet, V. Blood-brain barrier invasion by group B Streptococcus depends upon proper cell-surface anchoring of lipoteichoic acid. J. Clin. Investig. 2005, 115, 2499–2507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deatherage, B.L.; Cookson, B.T. Membrane vesicle release in bacteria, eukaryotes, and archaea: A conserved yet underappreciated aspect of microbial life. Infect. Immun. 2012, 80, 1948–1957. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turkina, M.V.; Olofsson, A.; Magnusson, K.E.; Arnqvist, A.; Vikstrom, E. Helicobacter pylori vesicles carrying CagA localize in the vicinity of cell-cell contacts and induce histone H1 binding to ATP in epithelial cells. FEMS Microbiol. Lett. 2015, 362, fnv076. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prados-Rosales, R.; Baena, A.; Martinez, L.R.; Luque-Garcia, J.; Kalscheuer, R.; Veeraraghavan, U.; Camara, C.; Nosanchuk, J.D.; Besra, G.S.; Chen, B.; et al. Mycobacteria release active membrane vesicles that modulate immune responses in a TLR2-dependent manner in mice. J. Clin. Investig. 2011, 121, 1471–1483. [Google Scholar] [CrossRef] [Green Version]
- Volgers, C.; Savelkoul, P.H.M.; Stassen, F.R.M. Gram-negative bacterial membrane vesicle release in response to the host-environment: Different threats, same trick? Crit. Rev. Microbiol. 2018, 44, 258–273. [Google Scholar] [CrossRef] [Green Version]
- de Figueiredo, P.; Ficht, T.A.; Rice-Ficht, A.; Rossetti, C.A.; Adams, L.G. Pathogenesis and immunobiology of brucellosis: Review of Brucella-host interactions. Am. J. Pathol. 2015, 185, 1505–1517. [Google Scholar] [CrossRef]
- Athman, J.J.; Wang, Y.; McDonald, D.J.; Boom, W.H.; Harding, C.V.; Wearsch, P.A. Bacterial Membrane Vesicles Mediate the Release of Mycobacterium tuberculosis Lipoglycans and Lipoproteins from Infected Macrophages. J. Immunol. 2015, 195, 1044–1053. [Google Scholar] [CrossRef] [Green Version]
- Lee, E.Y.; Choi, D.Y.; Kim, D.K.; Kim, J.W.; Park, J.O.; Kim, S.; Kim, S.H.; Desiderio, D.M.; Kim, Y.K.; Kim, K.P.; et al. Gram-positive bacteria produce membrane vesicles: Proteomics-based characterization of Staphylococcus aureus-derived membrane vesicles. Proteomics 2009, 9, 5425–5436. [Google Scholar] [CrossRef]
- Domingues, S.; Nielsen, K.M. Membrane vesicles and horizontal gene transfer in prokaryotes. Curr. Opin. Microbiol. 2017, 38, 16–21. [Google Scholar] [CrossRef]
- Rumbo, C.; Fernandez-Moreira, E.; Merino, M.; Poza, M.; Mendez, J.A.; Soares, N.C.; Mosquera, A.; Chaves, F.; Bou, G. Horizontal transfer of the OXA-24 carbapenemase gene via outer membrane vesicles: A new mechanism of dissemination of carbapenem resistance genes in Acinetobacter baumannii. Antimicrob. Agents Chemother. 2011, 55, 3084–3090. [Google Scholar] [CrossRef]
- Deo, P.; Chow, S.H.; Hay, I.D.; Kleifeld, O.; Costin, A.; Elgass, K.D.; Jiang, J.H.; Ramm, G.; Gabriel, K.; Dougan, G.; et al. Outer membrane vesicles from Neisseria gonorrhoeae target PorB to mitochondria and induce apoptosis. PLoS Pathog. 2018, 14, e1006945. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thay, B.; Wai, S.N.; Oscarsson, J. Staphylococcus aureus alpha-toxin-dependent induction of host cell death by membrane-derived vesicles. PLoS ONE 2013, 8, e54661. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Gao, J.; Wang, Z. Outer membrane vesicles for vaccination and targeted drug delivery. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2019, 11, e1523. [Google Scholar] [CrossRef]
- Jones, L.B.; Kumar, S.; Bell, C.R.; Crenshaw, B.J.; Coats, M.T.; Sims, B.; Matthews, Q.L. Lipopolysaccharide Administration Alters Extracellular Vesicles in Cell Lines and Mice. Curr. Microbiol. 2021, 78, 920–931. [Google Scholar] [CrossRef] [PubMed]
- Fabrega, M.J.; Aguilera, L.; Gimenez, R.; Varela, E.; Canas, M.A.; Antolin, M.; Badia, J.; Baldoma, L. Activation of Immune and Defense Responses in the Intestinal Mucosa by Outer Membrane Vesicles of Commensal and Probiotic Escherichia coli Strains. Front. Microbiol. 2016, 7, 705. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dean, S.N.; Thakur, M.; Spangler, J.R. Extracellular vesicle production in Gram-positive bacteria. Microb. Biotechnol. 2022, 15, 1055–1057. [Google Scholar] [CrossRef]
- Wang, X.; Thompson, C.D.; Weidenmaier, C.; Lee, J.C. Release of Staphylococcus aureus extracellular vesicles and their application as a vaccine platform. Nat. Commun. 2018, 9, 1379. [Google Scholar] [CrossRef] [Green Version]
- Toyofuku, M.; Carcamo-Oyarce, G.; Yamamoto, T.; Eisenstein, F.; Hsiao, C.C.; Kurosawa, M.; Gademann, K.; Pilhofer, M.; Nomura, N.; Eberl, L. Prophage-triggered membrane vesicle formation through peptidoglycan damage in Bacillus subtilis. Nat. Commun. 2017, 8, 481. [Google Scholar] [CrossRef] [Green Version]
- Raghav, A.; Jeong, G.B. A systematic review on the modifications of extracellular vesicles: A revolutionized tool of nano-biotechnology. J. Nanobiotechnol. 2021, 19, 459. [Google Scholar] [CrossRef]
- Zou, C.; Zhang, Y.; Liu, H.; Wu, Y.; Zhou, X. Extracellular Vesicles: Recent Insights into the Interaction Between Host and Pathogenic Bacteria. Front. Immunol. 2022, 13, 840550. [Google Scholar] [CrossRef]
- Pant, S.; Hilton, H.; Burczynski, M.E. The multifaceted exosome: Biogenesis, role in normal and aberrant cellular function, and frontiers for pharmacological and biomarker opportunities. Biochem. Pharmacol. 2012, 83, 1484–1494. [Google Scholar] [CrossRef] [PubMed]
- Robbins, P.D.; Morelli, A.E. Regulation of immune responses by extracellular vesicles. Nat. Rev. Immunol. 2014, 14, 195–208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brakhage, A.A.; Zimmermann, A.-K.; Rivieccio, F.; Visser, C.; Blango, M.G. Host-derived extracellular vesicles for antimicrobial defense. microLife 2021, 2, uqab003. [Google Scholar] [CrossRef]
- Seo, M.K.; Park, E.J.; Ko, S.Y.; Choi, E.W.; Kim, S. Therapeutic effects of kefir grain Lactobacillus-derived extracellular vesicles in mice with 2,4,6-trinitrobenzene sulfonic acid-induced inflammatory bowel disease. J. Dairy Sci. 2018, 101, 8662–8671. [Google Scholar] [CrossRef] [Green Version]
- Morishita, M.; Horita, M.; Higuchi, A.; Marui, M.; Katsumi, H.; Yamamoto, A. Characterizing Different Probiotic-Derived Extracellular Vesicles as a Novel Adjuvant for Immunotherapy. Mol. Pharm. 2021, 18, 1080–1092. [Google Scholar] [CrossRef]
- Kim, J.H.; Jeun, E.J.; Hong, C.P.; Kim, S.H.; Jang, M.S.; Lee, E.J.; Moon, S.J.; Yun, C.H.; Im, S.H.; Jeong, S.G.; et al. Extracellular vesicle-derived protein from Bifidobacterium longum alleviates food allergy through mast cell suppression. J. Allergy Clin. Immunol. 2016, 137, 507–516.e8. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Hoffmann, J.P.; Chou, C.-W.; Bentrup, K.H.Z.; Fuselier, J.A.; Bitoun, J.P.; Wimley, W.C.; Morici, L.A. Burkholderia thailandensis outer membrane vesicles exert antimicrobial activity against drug-resistant and competitor microbial species. J. Microbiol. 2020, 58, 550–562. [Google Scholar] [CrossRef]
- Costantini, P.E.; Vanpouille, C.; Firrincieli, A.; Cappelletti, M.; Margolis, L.; Palomino, R.A.N. Extracellular Vesicles Generated by Gram-Positive Bacteria Protect Human Tissues Ex Vivo From HIV-1 Infection. Front. Cell. Infect. Microbiol. 2021, 11, 822882. [Google Scholar] [CrossRef]
- Kim, W.; Lee, E.J.; Bae, I.H.; Myoung, K.; Kim, S.T.; Park, P.J.; Lee, K.H.; Pham, A.V.Q.; Ko, J.; Oh, S.H.; et al. Lactobacillus plantarum-derived extracellular vesicles induce anti-inflammatory M2 macrophage polarization in vitro. J. Extracell. Vesicles 2020, 9, 1793514. [Google Scholar] [CrossRef]
- Tartaglia, N.R.; Breyne, K.; Meyer, E.; Cauty, C.; Jardin, J.; Chretien, D.; Dupont, A.; Demeyere, K.; Berkova, N.; Azevedo, V.; et al. Staphylococcus aureus Extracellular Vesicles Elicit an Immunostimulatory Response in vivo on the Murine Mammary Gland. Front. Cell. Infect. Microbiol. 2018, 8, 277. [Google Scholar] [CrossRef]
- Lee, H.; Yun, S.H.; Hyon, J.Y.; Lee, S.Y.; Yi, Y.S.; Choi, C.W.; Jun, S.; Park, E.C.; Kim, S.I. Streptococcus equi-derived extracellular vesicles as a vaccine candidate against Streptococcus equi infection. Vet. Microbiol. 2021, 259, 109165. [Google Scholar] [CrossRef] [PubMed]
- Islam, S.M.S.; Ryu, H.M.; Sohn, S. Tetragenococcus halophilus Alleviates Intestinal Inflammation in Mice by Altering Gut Microbiota and Regulating Dendritic Cell Activation via CD83. Cells 2022, 11, 1903. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Zhang, Q.; Li, W.; Chen, Y.; Shu, C.; Li, Q.; Zhou, J.; Ye, C.; Bai, H.; Sun, W.; et al. Anti-outer Membrane Vesicle Antibodies Increase Antibiotic Sensitivity of Pan-Drug-Resistant Acinetobacter baumannii. Front. Microbiol. 2019, 10, 1379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wawrzeniak, K.; Gaur, G.; Sapi, E.; Senejani, A.G. Effect of Borrelia burgdorferi Outer Membrane Vesicles on Host Oxidative Stress Response. Antibiotics 2020, 9, 275. [Google Scholar] [CrossRef]
- Gonzalez, M.F.; Diaz, P.; Sandoval-Borquez, A.; Herrera, D.; Quest, A.F.G. Helicobacter pylori Outer Membrane Vesicles and Extracellular Vesicles from Helicobacter pylori-Infected Cells in Gastric Disease Development. Int. J. Mol. Sci. 2021, 22, 4823. [Google Scholar] [CrossRef]
- Lee, W.H.; Choi, H.I.; Hong, S.W.; Kim, K.S.; Gho, Y.S.; Jeon, S.G. Vaccination with Klebsiella pneumoniae-derived extracellular vesicles protects against bacteria-induced lethality via both humoral and cellular immunity. Exp. Mol. Med. 2015, 47, e183. [Google Scholar] [CrossRef] [Green Version]
- Cai, W.; Kesavan, D.K.; Wan, J.; Abdelaziz, M.H.; Su, Z.; Xu, H. Bacterial outer membrane vesicles, a potential vaccine candidate in interactions with host cells based. Diagn. Pathol 2018, 13, 95. [Google Scholar] [CrossRef] [Green Version]
- Bottero, D.; Gaillard, M.E.; Errea, A.; Moreno, G.; Zurita, E.; Pianciola, L.; Rumbo, M.; Hozbor, D. Outer membrane vesicles derived from Bordetella parapertussis as an acellular vaccine against Bordetella parapertussis and Bordetella pertussis infection. Vaccine 2013, 31, 5262–5268. [Google Scholar] [CrossRef]
- Armstrong, D.A.; Lee, M.K.; Hazlett, H.F.; Dessaint, J.A.; Mellinger, D.L.; Aridgides, D.S.; Hendricks, G.M.; Abdalla, M.A.K.; Christensen, B.C.; Ashare, A. Extracellular Vesicles from Pseudomonas aeruginosa Suppress MHC-Related Molecules in Human Lung Macrophages. Immunohorizons 2020, 4, 508–519. [Google Scholar] [CrossRef]
- Ramirez, M.I.; Amorim, M.G.; Gadelha, C.; Milic, I.; Welsh, J.A.; Freitas, V.M.; Nawaz, M.; Akbar, N.; Couch, Y.; Makin, L.; et al. Technical challenges of working with extracellular vesicles. Nanoscale 2018, 10, 881–906. [Google Scholar] [CrossRef]
- Turturici, G.; Tinnirello, R.; Sconzo, G.; Geraci, F. Extracellular membrane vesicles as a mechanism of cell-to-cell communication: Advantages and disadvantages. Am. J. Physiol. Cell Physiol. 2014, 306, C621–C633. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Xie, Q.; Zhang, Y.; Ma, W.; Ning, K.; Xiang, J.Y.; Cui, J.; Xiang, H. Combination of probiotics with different functions alleviate DSS-induced colitis by regulating intestinal microbiota, IL-10, and barrier function. Appl. Microbiol. Biotechnol. 2020, 104, 335–349. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.H.; Moon, C.M.; Shin, T.S.; Kim, E.K.; McDowell, A.; Jo, M.K.; Joo, Y.H.; Kim, S.E.; Jung, H.K.; Shim, K.N.; et al. Lactobacillus paracasei-derived extracellular vesicles attenuate the intestinal inflammatory response by augmenting the endoplasmic reticulum stress pathway. Exp. Mol. Med. 2020, 52, 423–437. [Google Scholar] [CrossRef] [PubMed]
- Hurkacz, M.; Dobrek, L.; Wiela-Hojenska, A. Antibiotics and the Nervous System-Which Face of Antibiotic Therapy Is Real, Dr. Jekyll (Neurotoxicity) or Mr. Hyde (Neuroprotection)? Molecules 2021, 26, 7456. [Google Scholar] [CrossRef]
- Ou, J.; Wang, Z.; Liu, X.; Song, B.; Chen, J.; Li, R.; Jia, X.; Huang, R.; Xiang, W.; Zhong, S. Regulatory effects of marine polysaccharides on gut microbiota dysbiosis: A review. Food Chem. X 2022, 15, 100444. [Google Scholar] [CrossRef]
- Shaik, L.; Kashyap, R.; Thotamgari, S.R.; Singh, R.; Khanna, S. Gut-Brain Axis and its Neuro-Psychiatric Effects: A Narrative Review. Cureus 2020, 12, e11131. [Google Scholar] [CrossRef]
- Rutsch, A.; Kantsjo, J.B.; Ronchi, F. The Gut-Brain Axis: How Microbiota and Host Inflammasome Influence Brain Physiology and Pathology. Front. Immunol. 2020, 11, 604179. [Google Scholar] [CrossRef]
- McVey Neufeld, K.A.; Mao, Y.K.; Bienenstock, J.; Foster, J.A.; Kunze, W.A. The microbiome is essential for normal gut intrinsic primary afferent neuron excitability in the mouse. Neurogastroenterol. Motil. 2013, 25, 183-e88. [Google Scholar] [CrossRef]
- Celorrio, M.; Abellanas, M.A.; Rhodes, J.; Goodwin, V.; Moritz, J.; Vadivelu, S.; Wang, L.; Rodgers, R.; Xiao, S.; Anabayan, I.; et al. Gut microbial dysbiosis after traumatic brain injury modulates the immune response and impairs neurogenesis. Acta Neuropathol. Commun. 2021, 9, 40. [Google Scholar] [CrossRef]
- Palomino, R.A.N.; Vanpouille, C.; Costantini, P.E.; Margolis, L. Microbiota-host communications: Bacterial extracellular vesicles as a common language. PLoS Pathog. 2021, 17, e1009508. [Google Scholar] [CrossRef]
- Cheng, H.Y.; Ning, M.X.; Chen, D.K.; Ma, W.T. Interactions Between the Gut Microbiota and the Host Innate Immune Response Against Pathogens. Front. Immunol. 2019, 10, 607. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaparakis-Liaskos, M.; Ferrero, R.L. Immune modulation by bacterial outer membrane vesicles. Nat. Rev. Immunol. 2015, 15, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Shen, Q.; Huang, Z.; Yao, J.; Jin, Y. Extracellular vesicles-mediated interaction within intestinal microenvironment in inflammatory bowel disease. J. Adv. Res. 2022, 37, 221–233. [Google Scholar] [CrossRef] [PubMed]
- Doyle, L.M.; Wang, M.Z. Overview of Extracellular Vesicles, Their Origin, Composition, Purpose, and Methods for Exosome Isolation and Analysis. Cells 2019, 8, 727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Battistelli, M.; Falcieri, E. Apoptotic Bodies: Particular Extracellular Vesicles Involved in Intercellular Communication. Biology 2020, 9, 21. [Google Scholar] [CrossRef] [Green Version]
- Wu, Q.Y.; Liu, B.C.; Ruan, X.Z.; Ma, K.L. Intestinal microbiota-derived membrane vesicles and their role in chronic kidney disease. Biochim. Biophys. Acta Mol. Basis Dis. 2022, 1868, 166478. [Google Scholar] [CrossRef]
- Liu, S.; Gao, J.; Zhu, M.; Liu, K.; Zhang, H.L. Gut Microbiota and Dysbiosis in Alzheimer’s Disease: Implications for Pathogenesis and Treatment. Mol. Neurobiol. 2020, 57, 5026–5043. [Google Scholar] [CrossRef]
- Lee, K.E.; Kim, J.K.; Han, S.K.; Lee, D.Y.; Lee, H.J.; Yim, S.V.; Kim, D.H. The extracellular vesicle of gut microbial Paenalcaligenes hominis is a risk factor for vagus nerve-mediated cognitive impairment. Microbiome 2020, 8, 107. [Google Scholar] [CrossRef]
- Singhrao, S.K.; Olsen, I. Are Porphyromonas gingivalis Outer Membrane Vesicles Microbullets for Sporadic Alzheimer’s Disease Manifestation? J. Alzheimer’s Dis. Rep. 2018, 2, 219–228. [Google Scholar] [CrossRef] [Green Version]
- Pokusaeva, K.; Johnson, C.; Luk, B.; Uribe, G.; Fu, Y.; Oezguen, N.; Matsunami, R.K.; Lugo, M.; Major, A.; Mori-Akiyama, Y.; et al. GABA-producing Bifidobacterium dentium modulates visceral sensitivity in the intestine. Neurogastroenterol. Motil. 2017, 29, e12904. [Google Scholar] [CrossRef]
- Bravo, J.A.; Forsythe, P.; Chew, M.V.; Escaravage, E.; Savignac, H.M.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc. Natl. Acad. Sci. USA 2011, 108, 16050–16055. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neufeld, K.-A.M.; Kang, N.; Bienenstock, J.; Foster, J.A. Effects of intestinal microbiota on anxiety-like behavior. Commun. Integr. Biol. 2014, 4, 492–494. [Google Scholar] [CrossRef] [Green Version]
- Derrien, M.; van Hylckama Vlieg, J.E. Fate, activity, and impact of ingested bacteria within the human gut microbiota. Trends Microbiol. 2015, 23, 354–366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dean, S.N.; Rimmer, M.A.; Turner, K.B.; Phillips, D.A.; Caruana, J.C.; Hervey, W.J.T.; Leary, D.H.; Walper, S.A. Lactobacillus acidophilus Membrane Vesicles as a Vehicle of Bacteriocin Delivery. Front. Microbiol. 2020, 11, 710. [Google Scholar] [CrossRef]
- Choi, J.; Kim, Y.K.; Han, P.L. Extracellular Vesicles Derived from Lactobacillus plantarum Increase BDNF Expression in Cultured Hippocampal Neurons and Produce Antidepressant-like Effects in Mice. Exp. Neurobiol. 2019, 28, 158–171. [Google Scholar] [CrossRef] [PubMed]
- West, C.L.; Stanisz, A.M.; Mao, Y.K.; Champagne-Jorgensen, K.; Bienenstock, J.; Kunze, W.A. Microvesicles from Lactobacillus reuteri (DSM-17938) completely reproduce modulation of gut motility by bacteria in mice. PLoS ONE 2020, 15, e0225481. [Google Scholar] [CrossRef]
- Haas-Neill, S.; Iwashita, E.; Dvorkin-Gheva, A.; Forsythe, P. Effects of Two Distinct Psychoactive Microbes, Lacticaseibacillus rhamnosus JB-1 and Limosilactobacillus reuteri 6475, on Circulating and Hippocampal mRNA in Male Mice. Int. J. Mol. Sci. 2022, 23, 9653. [Google Scholar] [CrossRef]
- Xiao, M.; Huang, X. Unmasking antibiotic-associated neurological disorders: The underminer in Intensive Care Unit. J. Clin. Neurosci. 2021, 91, 131–135. [Google Scholar] [CrossRef]
- Wanleenuwat, P.; Suntharampillai, N.; Iwanowski, P. Antibiotic-induced epileptic seizures: Mechanisms of action and clinical considerations. Seizure 2020, 81, 167–174. [Google Scholar] [CrossRef]
- Kim, K.S. Investigating Bacterial Penetration of the Blood-Brain Barrier for the Pathogenesis, Prevention, and Therapy of Bacterial Meningitis. ACS Infect. Dis. 2020, 6, 34–42. [Google Scholar] [CrossRef]
- Nau, R.; Sorgel, F.; Eiffert, H. Penetration of drugs through the blood-cerebrospinal fluid/blood-brain barrier for treatment of central nervous system infections. Clin. Microbiol. Rev. 2010, 23, 858–883. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jimenez-Mejias, M.E.; Pichardo-Guerrero, C.; Marquez-Rivas, F.J.; Martin-Lozano, D.; Prados, T.; Pachon, J. Cerebrospinal fluid penetration and pharmacokinetic/pharmacodynamic parameters of intravenously administered colistin in a case of multidrug-resistant Acinetobacter baumannii meningitis. Eur. J. Clin. Microbiol. Infect. Dis. 2002, 21, 212–214. [Google Scholar] [CrossRef] [PubMed]
- Cascio, A.; Conti, A.; Sinardi, L.; Iaria, C.; Angileri, F.F.; Stassi, G.; David, T.; Versaci, A.; Iaria, M.; David, A. Post-neurosurgical multidrug-resistant Acinetobacter baumannii meningitis successfully treated with intrathecal colistin. A new case and a systematic review of the literature. Int. J. Infect. Dis. 2010, 14, e572–e579. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fu, J.; Li, L.; Huo, D.; Yang, R.; Yang, B.; Xu, B.; Yang, X.; Dai, M.; Tan, C.; Chen, H.; et al. Meningitic Escherichia coli alpha-hemolysin aggravates blood-brain barrier disruption via targeting TGFbeta1-triggered hedgehog signaling. Mol. Brain 2021, 14, 116. [Google Scholar] [CrossRef]
- Caporarello, N.; Olivieri, M.; Cristaldi, M.; Scalia, M.; Toscano, M.A.; Genovese, C.; Addamo, A.; Salmeri, M.; Lupo, G.; Anfuso, C.D. Blood-Brain Barrier in a Haemophilus influenzae Type a In Vitro Infection: Role of Adenosine Receptors A2A and A2B. Mol. Neurobiol. 2018, 55, 5321–5336. [Google Scholar] [CrossRef] [PubMed]
- Wegele, C.; Stump-Guthier, C.; Moroniak, S.; Weiss, C.; Rohde, M.; Ishikawa, H.; Schroten, H.; Schwerk, C.; Karremann, M.; Borkowski, J. Non-Typeable Haemophilus influenzae Invade Choroid Plexus Epithelial Cells in a Polar Fashion. Int. J. Mol. Sci. 2020, 21, 5739. [Google Scholar] [CrossRef]
- Ku, Y.H.; Chuang, Y.C.; Chen, C.C.; Lee, M.F.; Yang, Y.C.; Tang, H.J.; Yu, W.L. Klebsiella pneumoniae Isolates from Meningitis: Epidemiology, Virulence and Antibiotic Resistance. Sci. Rep. 2017, 7, 6634. [Google Scholar] [CrossRef] [Green Version]
- Cassidy, B.R.; Zhang, M.; Sonntag, W.E.; Drevets, D.A. Neuroinvasive Listeria monocytogenes infection triggers accumulation of brain CD8+ tissue-resident memory T cells in a miR-155-dependent fashion. J. Neuroinflammation 2020, 17, 259. [Google Scholar] [CrossRef]
- Schubert-Unkmeir, A.; Konrad, C.; Slanina, H.; Czapek, F.; Hebling, S.; Frosch, M. Neisseria meningitidis induces brain microvascular endothelial cell detachment from the matrix and cleavage of occludin: A role for MMP-8. PLoS Pathog. 2010, 6, e1000874. [Google Scholar] [CrossRef] [Green Version]
- Orihuela, C.J.; Mahdavi, J.; Thornton, J.; Mann, B.; Wooldridge, K.G.; Abouseada, N.; Oldfield, N.J.; Self, T.; Ala’Aldeen, D.A.; Tuomanen, E.I. Laminin receptor initiates bacterial contact with the blood brain barrier in experimental meningitis models. J. Clin. Investig. 2009, 119, 1638–1646. [Google Scholar] [CrossRef]
- Chen, H.; Guo, X.; Xie, D.; Dong, X.; Niu, J.; Chen, G. A Clinical Study on the Use of Intraventricular Polymyxin B Supplemented by Continuous External Ventricular Drainage in the Treatment of Drug-Resistant Gram-Negative Bacilli Intracranial Infection. Infect. Drug Resist. 2020, 13, 2963–2970. [Google Scholar] [CrossRef] [PubMed]
- McLoughlin, A.; Rochfort, K.D.; McDonnell, C.J.; Kerrigan, S.W.; Cummins, P.M. Staphylococcus aureus-mediated blood-brain barrier injury: An in vitro human brain microvascular endothelial cell model. Cell. Microbiol. 2017, 19, e12664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anil, A.; Banerjee, A. Pneumococcal Encounter with the Blood-Brain Barrier Endothelium. Front. Cell. Infect. Microbiol. 2020, 10, 590682. [Google Scholar] [CrossRef] [PubMed]
- Iovino, F.; Seinen, J.; Henriques-Normark, B.; van Dijl, J.M. How Does Streptococcus pneumoniae Invade the Brain? Trends Microbiol. 2016, 24, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Zocco, M.A.; dal Verme, L.Z.; Cremonini, F.; Piscaglia, A.C.; Nista, E.C.; Candelli, M.; Novi, M.; Rigante, D.; Cazzato, I.A.; Ojetti, V.; et al. Efficacy of Lactobacillus GG in maintaining remission of ulcerative colitis. Aliment. Pharmacol. Ther. 2006, 23, 1567–1574. [Google Scholar] [CrossRef]
- Tsai, Y.-L.; Tsai, W.-C.; Qing, Z.; Chang, C.-J. Dichotomous effects of microbial membrane vesicles on the regulation of immunity. Med. Microecol. 2020, 3, 100009. [Google Scholar] [CrossRef]
- Ramachandran, G.; Bikard, D. Editing the microbiome the CRISPR way. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2019, 374, 20180103. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Hoffmann, J.P.; Baker, S.M.; Bentrup, K.H.Z.; Wimley, W.C.; Fuselier, J.A.; Bitoun, J.P.; Morici, L.A. Inhibition of Streptococcus mutans biofilms with bacterial-derived outer membrane vesicles. BMC Microbiol. 2021, 21, 234. [Google Scholar] [CrossRef]
- Hu, R.; Lin, H.; Li, J.; Zhao, Y.; Wang, M.; Sun, X.; Min, Y.; Gao, Y.; Yang, M. Probiotic Escherichia coli Nissle 1917-derived outer membrane vesicles enhance immunomodulation and antimicrobial activity in RAW264.7 macrophages. BMC Microbiol. 2020, 20, 268. [Google Scholar] [CrossRef]
- Chee, W.J.Y.; Chew, S.Y.; Than, L.T.L. Vaginal microbiota and the potential of Lactobacillus derivatives in maintaining vaginal health. Microb. Cell Fact. 2020, 19, 203. [Google Scholar] [CrossRef]
- Qin, H.; Zhang, Z.; Hang, X.; Jiang, Y.L. plantarum prevents enteroinvasive Escherichia coli-induced tight junction proteins changes in intestinal epithelial cells. BMC Microbiol. 2009, 9, 63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bajic, S.S.; Canas, M.A.; Tolinacki, M.; Badia, J.; Sanchez, B.; Golic, N.; Margolles, A.; Baldoma, L.; Ruas-Madiedo, P. Proteomic profile of extracellular vesicles released by Lactiplantibacillus plantarum BGAN8 and their internalization by non-polarized HT29 cell line. Sci. Rep. 2020, 10, 21829. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.; Liwinski, T.; Elinav, E. Interaction between microbiota and immunity in health and disease. Cell Res. 2020, 30, 492–506. [Google Scholar] [CrossRef] [PubMed]
- Han, E.C.; Choi, S.Y.; Lee, Y.; Park, J.W.; Hong, S.H.; Lee, H.J. Extracellular RNAs in periodontopathogenic outer membrane vesicles promote TNF-α production in human macrophages and cross the blood–brain barrier in mice. FASEB J. 2019, 33, 13412. [Google Scholar] [CrossRef] [Green Version]
- Al-Nedawi, K.; Mian, M.F.; Hossain, N.; Karimi, K.; Mao, Y.K.; Forsythe, P.; Min, K.K.; Stanisz, A.M.; Kunze, W.A.; Bienenstock, J. Gut commensal microvesicles reproduce parent bacterial signals to host immune and enteric nervous systems. FASEB J. 2015, 29, 684–695. [Google Scholar] [CrossRef]
- Haas-Neill, S.; Forsythe, P. A Budding Relationship: Bacterial Extracellular Vesicles in the Microbiota-Gut-Brain Axis. Int. J. Mol. Sci. 2020, 21, 8899. [Google Scholar] [CrossRef]
- Mba, I.E.; Nweze, E.I. Nanoparticles as therapeutic options for treating multidrug-resistant bacteria: Research progress, challenges, and prospects. World J. Microbiol. Biotechnol. 2021, 37, 108. [Google Scholar] [CrossRef]
- Rudramurthy, G.R.; Swamy, M.K.; Sinniah, U.R.; Ghasemzadeh, A. Nanoparticles: Alternatives Against Drug-Resistant Pathogenic Microbes. Molecules 2016, 21, 836. [Google Scholar] [CrossRef]
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar] [CrossRef]
- Van de Waterbeemd, B.; Streefland, M.; van der Ley, P.; Zomer, B.; van Dijken, H.; Martens, D.; Wijffels, R.; van der Pol, L. Improved OMV vaccine against Neisseria meningitidis using genetically engineered strains and a detergent-free purification process. Vaccine 2010, 28, 4810–4816. [Google Scholar] [CrossRef]
- Van der Pol, L.; Stork, M.; van der Ley, P. Outer membrane vesicles as platform vaccine technology. Biotechnol. J. 2015, 10, 1689–1706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meng, W.; He, C.; Hao, Y.; Wang, L.; Li, L.; Zhu, G. Prospects and challenges of extracellular vesicle-based drug delivery system: Considering cell source. Drug Deliv. 2020, 27, 585–598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, Z.; Antenucci, F.; Villumsen, K.R.; Bojesen, A.M. Bacterial Outer Membrane Vesicles as a Versatile Tool in Vaccine Research and the Fight against Antimicrobial Resistance. mBio 2021, 12, e0170721. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Feng, T.; Liu, B.; Qiu, F.; Xu, Y.; Zhao, Y.; Zheng, Y. Engineered exosomes: Desirable target-tracking characteristics for cerebrovascular and neurodegenerative disease therapies. Theranostics 2021, 11, 8926–8944. [Google Scholar] [CrossRef]
- Tiwari, S.; Jamal, S.B.; Hassan, S.S.; Carvalho, P.; Almeida, S.; Barh, D.; Ghosh, P.; Silva, A.; Castro, T.L.P.; Azevedo, V. Two-Component Signal Transduction Systems of Pathogenic Bacteria as Targets for Antimicrobial Therapy: An Overview. Front. Microbiol. 2017, 8, 1878. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dobson, A.; Cotter, P.D.; Ross, R.P.; Hill, C. Bacteriocin production: A probiotic trait? Appl. Environ. Microbiol. 2012, 78, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Jakava-Viljanen, M.; Avall-Jaaskelainen, S.; Messner, P.; Sleytr, U.B.; Palva, A. Isolation of three new surface layer protein genes (slp) from Lactobacillus brevis ATCC 14869 and characterization of the change in their expression under aerated and anaerobic conditions. J. Bacteriol. 2002, 184, 6786–6795. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.Y.; Doody, A.M.; Chen, D.J.; Cremona, G.H.; Shuler, M.L.; Putnam, D.; DeLisa, M.P. Engineered bacterial outer membrane vesicles with enhanced functionality. J. Mol. Biol. 2008, 380, 51–66. [Google Scholar] [CrossRef] [Green Version]
- Uchida, H.; Kinoshita, H.; Kawai, Y.; Kitazawa, H.; Miura, K.; Shiiba, K.; Horii, A.; Kimura, K.; Taketomo, N.; Oda, M.; et al. Lactobacilli binding human A-antigen expressed in intestinal mucosa. Res. Microbiol. 2006, 157, 659–665. [Google Scholar] [CrossRef]
- Klotz, C.; Barrangou, R. Engineering Components of the Lactobacillus S-Layer for Biotherapeutic Applications. Front. Microbiol. 2018, 9, 2264. [Google Scholar] [CrossRef]
- Lee, S.H.; Kim, M.S.; Jung, H.C.; Lee, J.; Lee, J.H.; Lee, H.S.; Kang, S.G. Screening of a novel strong promoter by RNA sequencing and its application to H2 production in a hyperthermophilic archaeon. Appl. Microbiol. Biotechnol. 2015, 99, 4085–4092. [Google Scholar] [CrossRef] [PubMed]
- Parker, A.; Fonseca, S.; Carding, S.R. Gut microbes and metabolites as modulators of blood-brain barrier integrity and brain health. Gut Microbes 2020, 11, 135–157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, L.; Ye, Y.; Gu, L.; Jian, Z.; Stary, C.M.; Xiong, X. Extracellular vesicle-derived miRNA as a novel regulatory system for bi-directional communication in gut-brain-microbiota axis. J. Transl. Med. 2021, 19, 202. [Google Scholar] [CrossRef] [PubMed]
- Layunta, E.; Buey, B.; Mesonero, J.E.; Latorre, E. Crosstalk Between Intestinal Serotonergic System and Pattern Recognition Receptors on the Microbiota-Gut-Brain Axis. Front. Endocrinol. 2021, 12, 748254. [Google Scholar] [CrossRef] [PubMed]
- Wynendaele, E.; Verbeke, F.; Stalmans, S.; Gevaert, B.; Janssens, Y.; Van De Wiele, C.; Peremans, K.; Burvenich, C.; De Spiegeleer, B. Quorum Sensing Peptides Selectively Penetrate the Blood-Brain Barrier. PLoS ONE 2015, 10, e0142071. [Google Scholar] [CrossRef] [Green Version]
- Abib, R.T.; Gaman, A.; Dargel, A.A.; Tamouza, R.; Kapczinski, F.; Gottfried, C.; Leboyer, M. Intracellular Pathogen Infections and Immune Response in Autism. Neuroimmunomodulation 2018, 25, 271–279. [Google Scholar] [CrossRef]
- Garcia-Gutierrez, E.; Narbad, A.; Rodriguez, J.M. Autism Spectrum Disorder Associated with Gut Microbiota at Immune, Metabolomic, and Neuroactive Level. Front. Neurosci. 2020, 14, 578666. [Google Scholar] [CrossRef]
- Jodoin, J.; Demeule, M.; Fenart, L.; Cecchelli, R.; Farmer, S.; Linton, K.J.; Higgins, C.F.; Beliveau, R. P-glycoprotein in blood-brain barrier endothelial cells: Interaction and oligomerization with caveolins. J. Neurochem. 2003, 87, 1010–1023. [Google Scholar] [CrossRef] [Green Version]
- Grill, M.F.; Maganti, R.K. Neurotoxic effects associated with antibiotic use: Management considerations. Br. J. Clin. Pharmacol. 2011, 72, 381–393. [Google Scholar] [CrossRef] [Green Version]
- Fazal, S.; Lee, R. Biomimetic Bacterial Membrane Vesicles for Drug Delivery Applications. Pharmaceutics 2021, 13, 1430. [Google Scholar] [CrossRef]
- Chen, G.; Bai, Y.; Li, Z.; Wang, F.; Fan, X.; Zhou, X. Bacterial extracellular vesicle-coated multi-antigenic nanovaccines protect against drug-resistant Staphylococcus aureus infection by modulating antigen processing and presentation pathways. Theranostics 2020, 10, 7131–7149. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Battalapalli, D.; Hakeem, M.J.; Selamneni, V.; Zhang, P.; Draz, M.S.; Ruan, Z. Engineered CRISPR-Cas systems for the detection and control of antibiotic-resistant infections. J. Nanobiotechnol. 2021, 19, 401. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.H.; van Pijkeren, J.P. CRISPR-Cas9-assisted recombineering in Lactobacillus reuteri. Nucleic Acids Res. 2014, 42, e131. [Google Scholar] [CrossRef] [PubMed]
- Schuster, J.A.; Vogel, R.F.; Ehrmann, M.A. Characterization and distribution of CRISPR-Cas systems in Lactobacillus sakei. Arch. Microbiol. 2019, 201, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Loo, Y.S.; Bose, R.J.; McCarthy, J.R.; Azmi, I.D.M.; Madheswaran, T. Biomimetic bacterial and viral-based nanovesicles for drug delivery, theranostics, and vaccine applications. Drug Discov. Today 2021, 26, 902–915. [Google Scholar] [CrossRef] [PubMed]
- Zambrowicz, B.P.; Sands, A.T. Knockouts model the 100 best-selling drugs—Will they model the next 100? Nat. Rev. Drug Discov. 2003, 2, 38–51. [Google Scholar] [CrossRef]
- Choi, S.Y.; Lim, S.; Cho, G.; Kwon, J.; Mun, W.; Im, H.; Mitchell, R.J. Chromobacterium violaceum delivers violacein, a hydrophobic antibiotic, to other microbes in membrane vesicles. Environ. Microbiol. 2020, 22, 705–713. [Google Scholar] [CrossRef]



| Origin of bEV | Infecting Pathogens | Role of bEV | Reference |
|---|---|---|---|
| Gram-positive bacteria | |||
| Bifidobacterium longum | Food-borne infections | Induction of progenitor cells | [96] |
| Burkholderia spp. | Activity against A. baumannii and S. aureus | N.D. | [97] |
| L. gasseri | Human Immunodeficiency Virus (HIV) | Change in susceptibility pattern of viral infection by regulation of toll-like receptor (TLR)-2 signaling | [98] |
| L. rhamnosus | Superficial infections | M2 Macrophage | [99] |
| S. aureus | Pneumococcal infection | TH1-mediated cell immunity | [100] |
| Streptococcus spp. | Streptococcal infection | Induction of dendritic cells | [101] |
| Tetragenococcus halophilus | Opportunistic pathogens | Anti-inflammatory factor interferon beta (IFN-β) | [102] |
| Gram-negative bacteria | |||
| Acinetobacter spp. | Pan-drug resistant A. baumannii | Activation of IgG and IgM | [103] |
| Borrelia burgdorferi | B. burgdorferi colonization | Stabilizing superoxide | [104] |
| Helicobacter pylori | H. pylori infection | Induction of TH2 immune cells | [105] |
| K. pneumoniae | K. pneumoniae infection | Humoral and cellular immunity | [106] |
| N. meningitis | Meningococcal disease | IgG-mediated response | [107] |
| Pertussis A | Bordetella pertussis infection | Induction of CD4 cells | [108] |
| P. aeruginosa | Lethal dose of P. aeruginosa | Mixed cellular response | [109] |
| Eukaryotic (hEVs) | Bacterial (bEVs) | Common Limitations |
|---|---|---|
| Differentiation between cell surface markers | Lipopolysaccharide (LPS) toxicity | Immunomodulators outburst |
| Inefficient purification of vesicles | High inflammatory responses | Low viability and inefficient growth conditions |
| Lack of heterogeneity | High chance of infection (pathogen-derived bEVs) | High cellular toxicity |
| Category | Eukaryotic-Derived hEVs | Gut-Microbiota-Derived bEVs |
|---|---|---|
| Biogenesis | Generally produced from plasma membrane except exosomes, which originate from endocytic pathway | Gram-negative bacteria: decreased protein linkages between the OM and peptidoglycan, accumulation of unfolded proteins and/or peptidoglycan in the periplasmic space, and explosive cell lysis Gram-positive bacteria: turgor pressure by accumulation of bEVs and the action of cell-wall-degrading enzymes |
| Composition (Cargo) | Multivesicular bodies composed of endosomal proteins; RNA and miRNA are regularly incorporated | Proteins, peptidoglycans, lipids, LPS, lipoteichoic acids (LTA), nucleic acids, and metabolites |
| Major functions | Intercellular communications (cell proliferation, matrix formation, and phagocytosis) | Innate and adaptive immunity, bacterial communications, interaction with host miRNA for movement across intestinal barrier |
| Size | 40–100 nm (exosomes) [124]; 500–2000 nm (apoptotic bodies) and 100–500 nm (microvesicles) [125] | 10–300 nm [126] |
| Origin of bEVs | Roles of bEVs | References |
|---|---|---|
| L. acidophilus | Changes in complex microbial communities | [134] |
| L. plantarum | Enhance the action of brain-derived neurotropic factor (BDNF), lowering the stress level in hippocampus neuron | [135] |
| L. reuteri DSM 17938 | Modulate intestinal and colon motility and enhance gut–brain intercommunication for CNS homeostasis | [136] |
| L. rhamnosus | Reduce the behavioral changes including anxiety and depression | [137] |
| Pathogen | Mode of Pathogenesis | Immunological Factors Contributing BBB Infection | References |
|---|---|---|---|
| A. baumannii | -Meningitis -Catheter-associated infection | Increased inflammatory cell response, toll-like receptor (TLR) altered expression, and proinflammatory cytokine burst within 24 h of infection | [142,143] |
| E. coli | -Endothelial cells -Attenuation of transforming growth factor (TGF)-β 1 signaling | Increased expression of endothelial-derived platelet-derived growth factor receptor (PDFGR) and intercellular adhesion molecule (ICAM), resulting in inflammation | [144] |
| H. influenzae | -Large amount of vascular endothelial growth factor receptor (VEGFR) -Adenosine receptor dysfunction | Endothelial disruption and tight junction altered expression: downregulation of tumor necrosis factor (TNF-α); endothelial proliferation | [145,146] |
| K. pneumoniae | -Cerebrospinal infection -Intracranial infection | Increased production of proinflammatory cytokines and chemokines; induction of hypoxia inducible factor (HIF)-1α | [147] |
| L. monocytogenes | -Vimentin-mediated infection Neuroinflammation | In1F virulent factor-associated downregulation of tight junction and overexpression of PDFGR and ICAM, resulting in inflammation | [148] |
| N. meningitidis | -Secretion of IgA protease -Evasion of immune response | Deformation of adherence junction, triggering IL-6 and IL-8 expression: leukocyte infiltration and infected phagocyte movement | [149,150] |
| P. aeruginosa | -Cerebrospinal infection -Meningitis | Increased production of inflammatory cell response; overproduction of IL-1β and IL-6 | [151] |
| S. aureus | -Brain abscesses and endocarditis -Cytokine burst | Stimulate immune invasion, T cell activation: burst of proinflammatory cytokines; TNF-α, IL-6, and IL-10 overproduction | [152] |
| S. pneumoniae | -Neonatal meningitis -Laminin receptor transcytosis | TNF-α, IL-6, and IL-10 overproduction and increased permeability through anchored tight junction; cleavage of IgA through pneumococci IgA protease | [153,154] |
| Origin of bEV | Physiological Roles | Invading Pathogen(s) | References |
|---|---|---|---|
| Burkholderia thailandensis with quinolone | Synergistic antibiofilm activity | Streptococcus spp. | [158] |
| E. coli Nissle 1917 | Increased anti-inflammatory properties, such as IL-10 and T helper (TH) cell-mediated cytotoxicity | E. coli and S. aureus | [159] |
| L. crispatus and L. jensenii | Antibiofilm and anti-inflammatory effect | Candida albicans | [160] |
| L. paracasei and L. plantarum | Decrease pro-inflammatory cytokine production | Enteroinvasive E. coli | [161] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Srivastava, P.; Kim, K.-s. Membrane Vesicles Derived from Gut Microbiota and Probiotics: Cutting-Edge Therapeutic Approaches for Multidrug-Resistant Superbugs Linked to Neurological Anomalies. Pharmaceutics 2022, 14, 2370. https://doi.org/10.3390/pharmaceutics14112370
Srivastava P, Kim K-s. Membrane Vesicles Derived from Gut Microbiota and Probiotics: Cutting-Edge Therapeutic Approaches for Multidrug-Resistant Superbugs Linked to Neurological Anomalies. Pharmaceutics. 2022; 14(11):2370. https://doi.org/10.3390/pharmaceutics14112370
Chicago/Turabian StyleSrivastava, Prakhar, and Kwang-sun Kim. 2022. "Membrane Vesicles Derived from Gut Microbiota and Probiotics: Cutting-Edge Therapeutic Approaches for Multidrug-Resistant Superbugs Linked to Neurological Anomalies" Pharmaceutics 14, no. 11: 2370. https://doi.org/10.3390/pharmaceutics14112370