Progress of Cancer Nanotechnology as Diagnostics, Therapeutics, and Theranostics Nanomedicine: Preclinical Promise and Translational Challenges
Abstract
:1. Introduction
2. Cancer Nanotechnology: Contemporary Research in Diagnosis and Therapy
2.1. Nanomedicines for Cancer Therapy
2.1.1. Targeted Cancer Chemotherapy
2.1.2. Photodynamic Therapy
2.1.3. Photothermal Therapy
2.2. Nanomedicine as Cancer Diagnostics
2.2.1. Gold NPs
2.2.2. Iron Oxide NPs
2.2.3. Quantum Dots
2.2.4. Gadolinium-Incorporated NPs
2.2.5. Carbon Nanotubes and Graphenes
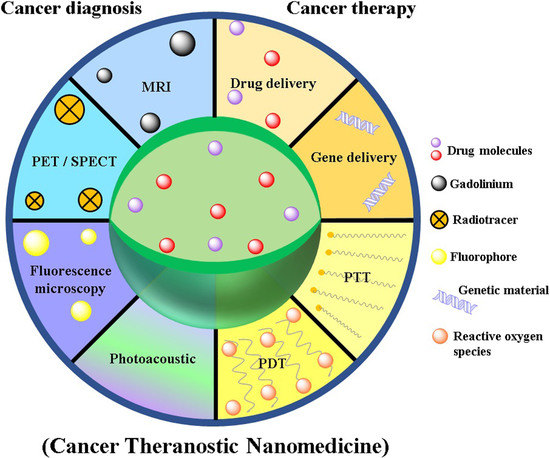
2.3. Nanomedicine as Cancer Theranostics
2.3.1. SPIONs
2.3.2. Gold Nano-Theranostic
2.3.3. Quantum Dots
2.3.4. Carbon Nanotubes, Carbon Dots, and Graphene Theranostic
2.3.5. Revenue and Market Potential of Cancer Nanomedicines
3. Challenges in Clinical Translation and Approval of Cancer Nanomedicine
Toxicity Apprehension
4. Expert Opinion and Future Directions
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Available online: https://www.who.int/news-room/fact-sheets/detail/cancer (accessed on 15 November 2020).
- Stephen, B.J.; Suchanti, S.; Mishra, R.; Singh, A. Cancer Nanotechnology in Medicine: A Promising Approach for Cancer Detection and Diagnosis. Crit. Rev. Ther. Drug Carrier Syst. 2020, 37, 375–405. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, J.; Akhter, S.; Ahmed Khan, M.; Wahajuddin, M.; H Greig, N.; Amjad Kamal, M.; Midoux, P.; Pichon, C. Engineered nanoparticles against MDR in cancer: The state of the art and its prospective. Curr. Pharm. Des. 2016, 22, 4360–4373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmad, J.; Ahmad, M.Z.; Akhter, H. Surface-Engineered Cancer Nanomedicine: Rational Design and Recent Progress. Curr. Pharm. Des. 2020, 26, 1181–1190. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; van der Meel, R.; Chen, X.; Lammers, T. The EPR effect and beyond: Strategies to improve tumor targeting and cancer nanomedicine treatment efficacy. Theranostics 2020, 10, 7921. [Google Scholar] [CrossRef] [PubMed]
- Akhter, S.; Amin, S.; Ahmad, J.; Khan, S.; Anwar, M.; Ahmad, M.Z.; Rahman, Z.; Ahmad, F.J. Nanotechnology to combat multidrug resistance in cancer. In Resistance to Targeted ABC Transporters in Cancer; Springer: Cham, Switzerland, 2015; pp. 245–272. [Google Scholar]
- Indoria, S.; Singh, V.; Hsieh, M.F. Recent advances in theranostic polymeric nanoparticles for cancer treatment: A review. Int. J. Pharm. 2020, 582, 119314. [Google Scholar] [CrossRef] [PubMed]
- Nezhadi, S.; Saadat, E.; Handali, S.; Dorkoosh, F. Nanomedicine and chemotherapeutics drug delivery: Challenges and opportunities. J. Drug Target. 2020, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.; Kim, D. Nanoconfinement-mediated cancer theranostics. Arch. Pharm. Res. 2020, 43, 110–117. [Google Scholar] [CrossRef]
- Haider, N.; Fatima, S.; Taha, M.; Rizwanullah, M.; Firdous, J.; Ahmad, R.; Mazhar, F.; Khan, M.A. Nanomedicines in Diagnosis and Treatment of Cancer: An Update. Curr. Pharm. Des. 2020, 26, 1216–1231. [Google Scholar] [CrossRef]
- Sakr, T.M.; El-Hashash, M.A.; El-Mohty, A.A.; Essa, B.M. 99mTc-gallic-gold nanoparticles as a new imaging platform for tumor targeting. Appl. Radiat. Isot. 2020, 164, 109269. [Google Scholar] [CrossRef]
- El-Ghareb, W.I.; Swidan, M.M.; Ibrahim, I.T.; Abd El-Bary, A.; Tadros, M.I.; Sakr, T.M. 99mTc-Doxorubicin-loaded gallic acid-gold nanoparticles (99mTc-DOX-loaded GA-Au NPs) as a multifunctional theranostic agent. Int. J. Pharm. 2020, 586, 119514. [Google Scholar] [CrossRef]
- Thakor, A.S.; Gambhir, S.S. Nanooncology: The future of cancer diagnosis and therapy. CA Cancer J. Clin. 2013, 63, 395–418. [Google Scholar] [CrossRef] [PubMed]
- Salvioni, L.; Rizzuto, M.A.; Bertolini, J.A.; Pandolfi, L.; Colombo, M.; Prosperi, D. Thirty years of cancer nanomedicine: Success, frustration, and hope. Cancers 2019, 11, 1855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, R.; Billone, P.S.; Mullett, W.M. Nanomedicine in action: An overview of cancer nanomedicine on the market and in clinical trials. J. Nanomater. 2013, 2013, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Chang, E.H. Nanomedicines: Improving current cancer therapies and diagnosis. Nanomedicine 2007, 3, 339. [Google Scholar] [CrossRef]
- Rizwanullah, M.; Alam, M.; Harshita Mir, S.R.; Rizvi, M.M.A.; Amin, S. Polymer-Lipid Hybrid Nanoparticles: A Next-Generation Nanocarrier for Targeted Treatment of Solid Tumors. Curr. Pharm. Des. 2020, 26, 1206–1215. [Google Scholar] [CrossRef] [PubMed]
- Norouzi, M.; Amerian, M.; Amerian, M.; Atyabi, F. Clinical applications of nanomedicine in cancer therapy. Drug Discov. Today 2020, 25, 107–125. [Google Scholar] [CrossRef] [PubMed]
- Akhter, M.H.; Rizwanullah, M.; Ahmad, J.; Ahsan, M.J.; Mujtaba, M.A.; Amin, S. Nanocarriers in advanced drug targeting: Setting novel paradigm in cancer therapeutics. Artif. Cells Nanomed. Biotechnol. 2018, 46, 873–884. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liao, J.; Jia, Y.; Wu, Y.; Shi, K.; Yang, D.; Li, P.; Qian, Z. Physical-, chemical-, and biological-responsive nanomedicine for cancer therapy. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020, 12, e1581. [Google Scholar] [CrossRef]
- Hejmady, S.; Pradhan, R.; Alexander, A.; Agrawal, M.; Singhvi, G.; Gorain, B.; Tiwari, S.; Kesharwani, P.; Dubey, S.K. Recent advances in targeted nanomedicine as promising antitumor therapeutics. Drug Discov. Today 2020. [Google Scholar] [CrossRef]
- Juan, A.; Cimas, F.J.; Bravo, I.; Pandiella, A.; Ocaña, A.; Alonso-Moreno, C. Antibody Conjugation of Nanoparticles as Therapeutics for Breast Cancer Treatment. Int. J. Mol. Sci. 2020, 21, 6018. [Google Scholar] [CrossRef]
- Zhang, H.; Dong, S.; Li, Z.; Feng, X.; Xu, W.; Tulinao, C.M.S.; Jiang, Y.; Ding, J. Biointerface engineering nanoplatforms for cancer-targeted drug delivery. Asian J. Pharm. Sci. 2020, 15, 397–415. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Fan, T.; Xie, Z.; Zeng, Q.; Xue, P.; Zheng, T.; Chen, Y.; Luo, X.; Zhang, H. Advances in nanomaterials for photodynamic therapy applications: Status and challenges. Biomaterials 2020, 237, 119827. [Google Scholar] [CrossRef]
- Sivasubramanian, M.; Chuang, Y.C.; Lo, L.W. Evolution of nanoparticle-mediated photodynamic therapy: From superficial to deep-seated cancers. Molecules 2019, 24, 520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Unterweger, H.; Subatzus, D.; Tietze, R.; Janko, C.; Poettler, M.; Stiegelschmitt, A.; Schuster, M.; Maake, C.; Boccaccini, A.R.; Alexiou, C. Hypericin-bearing magnetic iron oxide nanoparticles for selective drug delivery in photodynamic therapy. Int. J. Nanomedicine 2015, 10, 6985–6996. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, L.; Nurunnabi, M.; Nafiujjaman, M.; Jeong, Y.Y.; Lee, Y.; Huh, K.M. A photosensitizer-conjugated magnetic iron oxide/gold hybrid nanoparticle as an activatable platform for photodynamic cancer therapy. J. Mater. Chem. B 2014, 2, 2929–2937. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Sun, Y.; Yan, R.; Chen, E.; Wang, H.; Ye, D.; Xu, J.-J.; Chen, H.-Y. Rational engineering of semiconductor QDs enabling remarkable 1O2 production for tumor-targeted photodynamic therapy. Biomaterials 2017, 148, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Murakami, T.; Nakatsuji, H.; Inada, M.; Matoba, Y.; Umeyama, T.; Tsujimoto, M.; Isoda, S.; Hashida, M.; Imahori, H. Photodynamic and photothermal effects of semiconducting and metallic-enriched single-walled carbon nanotubes. J. Am. Chem. Soc. 2012, 134, 17862–17865. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, A.S.C.; Rodrigues, C.F.; Moreira, A.F.; Correia, I.J. Strategies to improve the photothermal capacity of gold-based nanomedicines. Acta Biomater. 2020, 116, 105–137. [Google Scholar] [CrossRef]
- Wang, J.; Wu, X.; Shen, P.; Wang, J.; Shen, Y.; Shen, Y.; Webster, T.J.; Deng, J. Applications of Inorganic Nanomaterials in Photothermal Therapy Based on Combinational Cancer Treatment. Int. J. Nanomed. 2020, 15, 1903–1914. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Jain, P.K.; El-Sayed, I.H.; El-Sayed, M.A. Determination of the minimum temperature required for selective photothermal destruction of cancer cells with the use of immunotargeted gold nanoparticles. Photochem. Photobiol. 2006, 82, 412–417. [Google Scholar] [CrossRef]
- Huang, X.; El-Sayed, I.H.; Qian, W.; El-Sayed, M.A. Cancer cell imaging and photothermal therapy in the nearinfrared region by using gold nanorods. J. Am. Chem. Soc. 2006, 128, 2115–2120. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wang, D.; Xi, J.; Au, L.; Siekkinen, A.; Warsen, A.; Li, Z.Y.; Zhang, H.; Xia, Y.; Li, X. Immuno gold nanocages with tailored optical properties for targeted photothermal destruction of cancer cells. Nano Lett. 2007, 7, 1318–1322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jain, P.K.; Huang, X.; El-Sayed, I.H.; El-Sayed, M.A. Noble metals on the nanoscale: Optical and photothermal properties and some applications in imaging, sensing, biology, and medicine. Acc. Chem. Res. 2008, 41, 1578–1586. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; El-Sayed, I.H.; El-Sayed, M.A. Applications of gold nanorods for cancer imaging and photothermal therapy. Methods Mol. Biol. 2010, 624, 343–357. [Google Scholar] [PubMed]
- Kuo, W.S.; Chang, C.N.; Chang, Y.T.; Yang, M.H.; Chien, Y.H.; Chen, S.J.; Yeh, C.S. Gold nanorods in photodynamic therapy, as hyperthermia agents, and in near-infrared optical imaging. Angew. Chem. Int. Ed. Engl. 2010, 49, 2711–2715. [Google Scholar] [CrossRef] [Green Version]
- Terentyuk, G.S.; Maslyakova, G.N.; Suleymanova, L.V.; Khlebtsov, N.G.; Khlebtsov, B.N.; Akchurin, G.G.; Maksimova, I.L.; Tuchin, V.V. Laser-induced tissue hyperthermia mediated by gold nanoparticles: Toward cancer phototherapy. J. Biomed. Opt. 2009, 14, 021016. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.; Bardhan, R.; Bartels, M.; Perez-Torres, C.; Pautler, R.G.; Halas, N.J.; Joshi, A. A molecularly targeted theranostic probe for ovarian cancer. Mol. Cancer Ther. 2010, 9, 1028–1038. [Google Scholar] [CrossRef] [Green Version]
- Arnfield, M.R.; Mathew, R.P.; Tulip, J.; McPhee, M.S. Analysis of tissue optical coefficients using an approximate equation valid for comparable absorption and scattering. Phys. Med. Biol. 1992, 37, 1219–1230. [Google Scholar] [CrossRef]
- McGrath, A.J.; Chien, Y.H.; Cheong, S.; Herman, D.A.; Watt, J.; Henning, A.M.; Gloag, L.; Yeh, C.S.; Tilley, R.D. Gold over branched palladium nanostructures for photothermal cancer therapy. ACS Nano 2015, 9, 12283–12291. [Google Scholar] [CrossRef]
- Shen, S.; Wang, S.; Zheng, R.; Zhu, X.; Jiang, X.; Fu, D.; Yang, W. Magnetic nanoparticle clusters for photothermal therapy with near-infrared irradiation. Biomaterials 2015, 39, 67–74. [Google Scholar] [CrossRef]
- Siegmund, K.D.; Marjoram, P.; Woo, Y.J.; Tavare, S.; Shibata, D. Inferring clonal expansion and cancer stem cell dynamics from DNA methylation patterns in colorectal cancers. Proc. Natl. Acad. Sci. USA 2009, 106, 4828–4833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, M.; Anderson, R.C.; Lan, X.; Conti, P.S.; Chen, K. Recent advances in the development of nanoparticles for multimodality imaging and therapy of cancer. Med. Res. Rev. 2020, 40, 909–930. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, S.; Sharma, D.; Kalia, K.; Tekade, R.K. Tumor microenvironment targeted nanotherapeutics for cancer therapy and diagnosis: A review. Acta Biomater. 2020, 101, 43–68. [Google Scholar] [CrossRef] [PubMed]
- Farzin, A.; Etesami, S.A.; Quint, J.; Memic, A.; Tamayol, A. Magnetic Nanoparticles in Cancer Therapy and Diagnosis. Adv. Healthc. Mater. 2020, 9, e1901058. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Leroueil, P.R.; Majoros, I.J.; Orr, B.G.; Baker, J.R.; Banaszak Holl, M.M. The binding avidity of a nanoparticle-based multivalent targeted drug delivery platform. Chem. Biol. 2007, 14, 107–115. [Google Scholar] [CrossRef] [Green Version]
- Hirn, S.; Semmler-Behnke, M.; Schleh, C.; Wenk, A.; Lipka, J.; Schäffler, M.; Takenaka, S.; Möller, W.; Schmid, S.; Simon, U.; et al. Particle size-dependent and surface chargedependent biodistribution of gold nanoparticles after intravenous administration. Eur. J. Pharm. Biopharm. 2011, 77, 407–416. [Google Scholar] [CrossRef] [Green Version]
- Decuzzi, P.; Godin, B.; Tanaka, T.; Lee, S.Y.; Chiappini, C.; Liu, X.; Ferrari, M. Size and shape effects in the biodistribution of intravascularly injected particles. J. Control. Release 2010, 141, 320–327. [Google Scholar] [CrossRef]
- Gratton, S.E.; Ropp, P.A.; Pohlhaus, P.D.; Luft, J.C.; Madden, V.J.; Napier, M.E.; DeSimone, J.M. The effect of particle design on cellular internalization pathways. Proc. Natl. Acad. Sci. USA 2008, 105, 11613–11618. [Google Scholar] [CrossRef] [Green Version]
- Carnovale, C.; Bryant, G.; Shukla, R.; Bansal, V. Identifying trends in gold nanoparticle toxicity and uptake: Size, shape, capping ligand, and biological corona. ACS Omega 2019, 4, 242–256. [Google Scholar] [CrossRef] [Green Version]
- Gerosa, C.; Crisponi, G.; Nurchi, V.M.; Saba, L.; Cappai, R.; Cau, F.; Faa, G.; Van Eyken, P.; Scartozzi, M.; Floris, G.; et al. Gold Nanoparticles: A New Golden Era in Oncology? Pharmaceuticals 2020, 13, 192. [Google Scholar] [CrossRef]
- Nie, Z.; Petukhova, A.; Kumacheva, E. Properties and emerging applications of self assembled structures made from inorganic nanoparticles. Nat. Nanotechnol. 2010, 5, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Von Maltzahn, G.; Harris, T.J.; Park, J.H.; Min, D.H.; Schmidt, A.J.; Sailor, M.J.; Bhatia, S.N. Nanoparticle self-assembly gated by logical proteolytic triggers. J. Am. Chem. Soc. 2007, 129, 6064–6065. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liang, G.; Ren, H.; Rao, J. A biocompatible condensation reaction for controlled assembly of nanostructures in living cells. Nat. Chem. 2010, 2, 54–60. [Google Scholar] [CrossRef]
- Akhter, S.; Ahmad, M.Z.; Ahmad, F.J.; Storm, G.; Kok, R.J. Gold nanoparticles in theranostic oncology: Current state-of-the-art. Expert Opin. Drug Deliv. 2012, 9, 1225–1243. [Google Scholar] [CrossRef] [PubMed]
- Thaxton, C.S.; Hill, H.D.; Georganopoulou, D.G.; Stoeva, S.I.; Mirkin, C.A. A bio-bar-code assay based upon dithiothreitol-induced oligonucleotide release. Anal. Chem. 2005, 77, 8174–8178. [Google Scholar] [CrossRef]
- Csaki, A.; Moller, R.; Fritzsche, W. Gold nanoparticles as novel label for DNA diagnostics. Expert Rev. Mol. Diagn. 2002, 2, 187–193. [Google Scholar] [CrossRef]
- Oh, J.H.; Lee, J.S. Designed hybridization properties of DNA-gold nanoparticle conjugates for the ultraselective detection of a single-base mutation in the breast cancer gene BRCA1. Anal. Chem. 2011, 83, 7364–7370. [Google Scholar] [CrossRef]
- Radwan, S.H.; Azzazy, H.M. Gold nanoparticles for molecular diagnostics. Expert Rev. Mol. Diagn. 2009, 9, 511–5124. [Google Scholar] [CrossRef]
- Zhou, B.; Yang, J.; Peng, C.; Zhu, J.; Tang, Y.; Zhu, X.; Shen, M.; Zhang, G.; Shi, X. PEGylated polyethylenimine-entrapped gold nanoparticles modified with folic acid for targeted tumor CT imaging. Colloids Surf. B Biointerfaces 2016, 140, 489–496. [Google Scholar] [CrossRef]
- Liu, H.; Wang, H.; Xu, Y.; Guo, R.; Wen, S.; Huang, Y.; Liu, W.; Shen, M.; Zhao, J.; Zhang, G.; et al. Lactobionic acid-modified dendrimer-entrapped gold nanoparticles for targeted computed tomography imaging of human hepatocellular carcinoma. ACS Appl. Mater. Interfaces 2014, 6, 6944–6953. [Google Scholar] [CrossRef]
- Chen, Q.; Li, K.; Wen, S.; Liu, H.; Peng, C.; Cai, H.; Shen, M.; Zhang, G.; Shi, X. Targeted CT/MR dual mode imaging of tumors using multifunctional dendrimer-entrapped gold nanoparticles. Biomaterials 2013, 34, 5200–5209. [Google Scholar] [CrossRef] [PubMed]
- Frey, N.A.; Peng, S.; Cheng, K.; Sun, S. Magnetic nanoparticles: Synthesis, functionalization, and applications in bioimaging and magnetic energy storage. Chem. Soc. Rev. 2009, 38, 2532–2542. [Google Scholar] [CrossRef] [PubMed]
- Ananta, J.S.; Godin, B.; Sethi, R.; Moriggi, L.; Liu, X.; Serda, R.E.; Krishnamurthy, R.; Muthupillai, R.; Bolskar, R.D.; Helm, L.; et al. Geometrical confinement of gadolinium-based contrast agents in nanoporous particles enhances T1 contrast. Nat. Nanotechnol. 2010, 5, 815–821. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stark, D.D.; Weissleder, R.; Elizondo, G.; Hahn, P.F.; Saini, S.; Todd, L.E.; Wittenberg, J.; Ferrucci, J.T. Superparamagnetic iron oxide: Clinical application as a contrast agent for MR imaging of the liver. Radiology 1988, 168, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Weissleder, R.; Elizondo, G.; Wittenberg, J.; Rabito, C.A.; Bengele, H.H.; Josephson, L. Ultrasmall superparamagnetic iron oxide: Characterization of a new class of contrast agents for MR imaging. Radiology 1990, 175, 489–493. [Google Scholar] [CrossRef] [PubMed]
- Michel, S.C.; Keller, T.M.; Fröhlich, J.M.; Fink, D.; Caduff, R.; Seifert, B.; Marincek, B.; Kubik-Huch, R.A. Preoperative breast cancer staging: MR imaging of the axilla with ultrasmall superparamagnetic iron oxide enhancement. Radiology 2002, 225, 527–536. [Google Scholar] [CrossRef]
- Nguyen, B.C.; Stanford, W.; Thompson, B.H.; Rossi, N.P.; Kernstine, K.H.; Kern, J.A.; Robinson, R.A.; Amorosa, J.K.; Mammone, J.F.; Outwater, E.K. Multicenter clinical trial of ultrasmall superparamagnetic iron oxide in the evaluation of mediastinal lymph nodes in patients with primary lung carcinoma. J. Magn. Reson. Imaging 1999, 10, 468–473. [Google Scholar] [CrossRef]
- Harisinghani, M.G.; Barentsz, J.; Hahn, P.F.; Deserno, W.M.; Tabatabaei, S.; van de Kaa, C.H.; de la Rosette, J.; Weissleder, R. Noninvasive detection of clinically occult lymph-node metastases in prostate cancer. N. Engl. J. Med. 2003, 348, 2491–2499. [Google Scholar] [CrossRef] [Green Version]
- Rockall, A.G.; Sohaib, S.A.; Harisinghani, M.G.; Babar, S.A.; Singh, N.; Jeyarajah, A.R.; Oram, D.H.; Jacobs, I.J.; Shepherd, J.H.; Reznek, R.H. Diagnostic performance of nanoparticle-enhanced magnetic resonance imaging in the diagnosis of lymph node metastases in patients with endometrial and cervical cancer. J. Clin. Oncol. 2005, 23, 2813–2821. [Google Scholar] [CrossRef]
- Ross, R.W.; Zietman, A.L.; Xie, W.; Coen, J.J.; Dahl, D.M.; Shipley, W.U.; Kaufman, D.S.; Islam, T.; Guimaraes, A.R.; Weissleder, R.; et al. Lymphotropic nanoparticle-enhanced magnetic resonance imaging (LNMRI) identifies occult lymph node metastases in prostate cancer patients prior to salvage radiation therapy. Clin. Imaging 2009, 33, 301–305. [Google Scholar] [CrossRef]
- Hu, Y.; Li, J.; Yang, J.; Wei, P.; Luo, Y.; Ding, L.; Sun, W.; Zhang, G.; Shi, X.; Shen, M. Facile synthesis of RGD peptide-modified iron oxide nanoparticles with ultrahigh relaxivity for targeted MR imaging of tumors. Biomater. Sci. 2015, 3, 721–732. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; He, H.; Sun, W.; Luo, Y.; Cai, H.; Pan, Y.; Shen, M.; Xia, J.; Shi, X. Hyaluronic acid-modified hydrothermally synthesized iron oxide nanoparticles for targeted tumor MR imaging. Biomaterials 2014, 35, 3666–3677. [Google Scholar] [CrossRef] [PubMed]
- Dai, L.; Liu, Y.; Wang, Z.; Guo, F.; Shi, D.; Zhang, B. One-pot facile synthesis of PEGylated superparamagnetic iron oxide nanoparticles for MRI contrast enhancement. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 41, 161–167. [Google Scholar] [CrossRef] [PubMed]
- McHugh, K.J.; Jing, L.; Behrens, A.M.; Jayawardena, S.; Tang, W.; Gao, M.; Langer, R.; Jaklenec, A. Biocompatible Semiconductor Quantum Dots as Cancer Imaging Agents. Adv Mater. 2018, 30, e1706356. [Google Scholar] [CrossRef] [PubMed]
- Fang, M.; Chen, M.; Liu, L.; Li, Y. Applications of Quantum Dots in Cancer Detection and Diagnosis: A Review. J. Biomed. Nanotechnol. 2017, 13, 1–16. [Google Scholar] [CrossRef]
- Tang, R.; Xue, J.; Xu, B.; Shen, D.; Sudlow, G.P.; Achilefu, S. Tunable ultrasmall visible-to-extended near-infrared emitting silver sulfide quantum dots for integrin-targeted cancer imaging. ACS Nano 2015, 9, 220–230. [Google Scholar] [CrossRef] [Green Version]
- Li, C.; Ji, Y.; Wang, C.; Liang, S.; Pan, F.; Zhang, C.; Chen, F.; Fu, H.; Wang, K.; Cui, D. BRCAA1 antibody- and Her2 antibody-conjugated amphiphilic polymer engineered CdSe/ZnS quantum dots for targeted imaging of gastric cancer. Nanoscale Res. Lett. 2014, 9, 244–256. [Google Scholar] [CrossRef] [Green Version]
- Foda, M.F.; Huang, L.; Shao, F.; Han, H.Y. Biocompatible and highly luminescent near-infrared CuInS₂/ZnS quantum dots embedded silica beads for cancer cell imaging. ACS Appl. Mater. Interfaces 2014, 6, 2011–2017. [Google Scholar] [CrossRef]
- Lux, J.; Sherry, A.D. Advances in gadolinium-based MRI contrast agent designs for monitoring biological processes in vivo. Curr. Opin. Chem. Biol. 2018, 45, 121–130. [Google Scholar] [CrossRef]
- Kim, H.K.; Lee, G.H.; Chang, Y. Gadolinium as an MRI contrast agent. Future Med. Chem. 2018, 10, 639–661. [Google Scholar] [CrossRef]
- Sitharaman, B.; Kissell, K.R.; Harmanth, K.B.; Tran, L.A.; Baikalov, A.; Rusakova, I.; Sun, Y.; Khant, H.A.; Ludtke, S.J.; Chiu, W.; et al. Superparamagnetic gadonanotubes are high-performance MRI contrast agents. Chem. Commun. 2005, 31, 3915–3917. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Regino, C.A.; Koyama, Y.; Hama, Y.; Gunn, A.J.; Bernardo, M.; Kobayashi, H.; Choyke, P.L.; Brechbiel, M.W. Preparation and preliminary evaluation of a biotin-targeted, lectin-targeted dendrimer-based probe for dual-modality magnetic resonance and fluorescence imaging. Bioconjug. Chem. 2007, 18, 1474–1482. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.S.; Park, W.; Na, K. Gadolinium-chelate nanoparticle entrapped human mesenchymal stem cell via photochemical internalization for cancer diagnosis. Biomaterials 2015, 36, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Mi, P.; Kokuryo, D.; Cabral, H.; Kumagai, M.; Nomoto, T.; Aoki, I.; Terada, Y.; Kishimura, A.; Nishiyama, N.; Kataoka, K. Hydrothermally synthesized PEGylated calcium phosphate nanoparticles incorporating Gd-DTPA for contrast enhanced MRI diagnosis of solid tumors. J. Control. Release 2014, 174, 63–71. [Google Scholar] [CrossRef]
- Sheikhpour, M.; Naghinejad, M.; Kasaeian, A.; Lohrasbi, A.; Shahraeini, S.S.; Zomorodbakhsh, S. The Applications of Carbon Nanotubes in the Diagnosis and Treatment of Lung Cancer: A Critical Review. Int. J. Nanomedicine 2020, 15, 7063–7078. [Google Scholar] [CrossRef]
- Gupta, S.; Murthy, C.N.; Prabha, C.R. Recent advances in carbon nanotube based electrochemical biosensors. Int. J. Biol. Macromol. 2018, 108, 687–703. [Google Scholar] [CrossRef]
- Loh, K.P.; Ho, D.; Chiu, G.N.C.; Leong, D.T.; Pastorin, G.; Chow, E.K. Clinical Applications of Carbon Nanomaterials in Diagnostics and Therapy. Adv. Mater. 2018, 30, e1802368. [Google Scholar] [CrossRef]
- Feng, L.; Li, W.; Qu, X. New horizons for diagnostics and therapeutic applications of graphene and graphene oxide. Adv. Mater. 2013, 25, 168–186. [Google Scholar] [CrossRef]
- Yang, K.; Feng, L.; Shi, X.; Liu, Z. Nano-graphene in biomedicine: Theranostic applications. Chem. Soc. Rev. 2013, 42, 530–547. [Google Scholar] [CrossRef]
- Hou, L.; Zhang, H.; Wang, Y.; Wang, L.; Yang, X.; Zhang, Z. Hyaluronic acid-functionalized single-walled carbon nanotubes as tumor-targeting MRI contrast agent. Int. J. Nanomed. 2015, 10, 4507–4520. [Google Scholar]
- Rubio, N.; Hirvonen, L.M.; Chong, E.Z.; Wang, J.T.; Bourgognon, M.; Kafa, H.; Hassan, H.A.; Al-Jamal, W.T.; McCarthy, D.; Hogstrand, C.; et al. Multiphoton luminescence imaging of chemically functionalized multi-walled carbon nanotubes in cells and solid tumors. Chem Commun 2015, 51, 9366–9369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tansi, F.L.; Rüger, R.; Böhm, C.; Steiniger, F.; Raasch, M.; Mosig, A.S.; Kontermann, R.E.; Teichgräber, U.K.; Fahr, A.; Hilger, I. Rapid Target Binding and Cargo Release of Activatable Liposomes Bearing HER2 and FAP Single-Chain Antibody Fragments Reveal Potentials for Image-Guided Delivery to Tumors. Pharmaceutics 2020, 12, 972. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.O.; Pinho, J.O.; Lopes, J.M.; Almeida, A.J.; Gaspar, M.M.; Reis, C. Current Trends in Cancer Nanotheranostics: Metallic, Polymeric, and Lipid-Based Systems. Pharmaceutics 2019, 11, 22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roma-Rodrigues, C.; Pombo, I.; Raposo, L.; Pedrosa, P.; Fernandes, A.R.; Baptista, P.V. Nanotheranostics Targeting the Tumor Microenvironment. Front. Bioeng. Biotechnol. 2019, 7, 197. [Google Scholar] [CrossRef] [Green Version]
- Shim, M.S.; Kim, C.S.; Ahn, Y.C.; Chen, Z.; Kwon, Y.J. Combined multimodal optical imaging and targeted gene silencing using stimuli-transforming nanotheragnostics. J. Am. Chem. Soc. 2010, 132, 8316–8324. [Google Scholar] [CrossRef] [Green Version]
- Santhosh, P.B.; Ulrih, N.P. Multifunctional superparamagnetic iron oxide nanoparticles: Promising tools in cancer theranostics. Cancer Lett. 2013, 336, 8–17. [Google Scholar] [CrossRef]
- Zhao, S.; Yu, X.; Qian, Y.; Chen, W.; Shen, J. Multifunctional magnetic iron oxide nanoparticles: An advanced platform for cancer theranostics. Theranostics 2020, 10, 6278–6309. [Google Scholar] [CrossRef]
- Li, K.; Nejadnik, H.; Daldrup-Link, H.E. Next-generation superparamagnetic iron oxide nanoparticles for cancer theranostics. Drug Discov. Today 2017, 22, 1421–1429. [Google Scholar] [CrossRef]
- Medarova, Z.; Pham, W.; Farrar, C.; Petkova, V.; Moore, A. In vivo imaging of siRNA delivery and silencing in tumours. Nat. Med. 2007, 13, 372–377. [Google Scholar] [CrossRef]
- Maeng, J.H.; Lee, D.H.; Jung, K.H.; Bae, Y.H.; Park, I.S.; Jeong, S.; Jeon, Y.S.; Shim, C.K.; Kim, W.; Kim, J.; et al. Multifunctional doxorubicin loaded superparamagnetic iron oxide nanoparticles for chemotherapy and magnetic resonance imaging in liver cancer. Biomaterials 2010, 31, 4995–5006. [Google Scholar] [CrossRef]
- Jalalian, S.H.; Taghdisi, S.M.; Shahidi Hamedani, N.; Kalat, S.A.; Lavaee, P.; Zandkarimi, M.; Ghows, N.; Jaafari, M.R.; Naghibi, S.; Danesh, N.M.; et al. Epirubicin loaded super paramagnetic iron oxide nanoparticle-aptamer bioconjugate for combined colon cancer therapy and imaging in vivo. Eur. J. Pharm. Sci. 2013, 50, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Nafiujjaman, M.; Revuri, V.; Nurunnabi, M.; Cho, K.J.; Lee, Y.K. Photosensitizer conjugated iron oxide nanoparticles for simultaneous in vitro magneto-fluorescent imaging guided photodynamic therapy. Chem. Commun 2015, 51, 5687–5690. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Luo, L.; Pan, Y.; Luo, S.; Lu, G.; Wu, A. In vivo targeted magnetic resonance imaging and visualized photodynamic therapy in deep-tissue cancers using folic acid-functionalized superparamagnetic-upconversion nanocomposites. Nanoscale 2015, 7, 8946–8954. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Yin, W.; Zheng, X.; Tian, G.; Zhang, X.; Bao, T.; Dong, X.; Wang, Z.; Gu, Z.; Ma, X.; et al. Smart MoS2/Fe3O4 nanotheranostic for magnetically targeted photothermal therapy guided by magnetic resonance/photoacoustic imaging. Theranostics 2015, 5, 931–945. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, Z.; Sun, Y.; Shen, J.; Wei, J.; Yu, C.; Kong, B.; Liu, W.; Yang, H.; Yang, S.; Wang, W. Iron/iron oxide core/shell nanoparticles for magnetic targeting MRI and near-infrared photothermal therapy. Biomaterials 2014, 35, 7470–7478. [Google Scholar] [CrossRef] [PubMed]
- Norouzi, M. Gold Nanoparticles in Glioma Theranostics. Pharmacol. Res. 2020, 156, 104753. [Google Scholar] [CrossRef]
- Navyatha, B.; Nara, S. Gold nanostructures as cancer theranostic probe: Promises and hurdles. Nanomedicine 2019, 14, 766–796. [Google Scholar] [CrossRef]
- Kim, D.; Park, S.; Lee, J.H.; Jeong, Y.Y.; Jon, S. Antibiofouling polymer-coated gold nanoparticles as a contrast agent for in vivo X-ray computed tomography imaging. J. Am. Chem. Soc. 2007, 129, 7661–7665. [Google Scholar] [CrossRef]
- Zhao, L.; Kim, T.H.; Kim, H.W.; Ahn, J.C.; Kim, S.Y. Surface-enhanced Raman scattering (SERS)-active gold nanochains for multiplex detection and photodynamic therapy of cancer. Acta Biomater. 2015, 20, 155–164. [Google Scholar] [CrossRef]
- Nair, L.V.; Nazeer, S.S.; Jayasree, R.S.; Ajayaghosh, A. Fluorescence imaging assisted photodynamic therapy using photosensitizer-linked gold quantum clusters. ACS Nano 2015, 9, 5825–5832. [Google Scholar] [CrossRef]
- Srivatsan, A.; Jenkins, S.V.; Jeon, M.; Wu, Z.; Kim, C.; Chen, J.; Pandey, R.K. Gold nanocage-photosensitizer conjugates for dual-modal image-guided enhanced photodynamic therapy. Theranostics 2014, 4, 163–174. [Google Scholar] [CrossRef]
- Xia, H.X.; Yang, X.Q.; Song, J.T.; Chen, J.; Zhang, M.Z.; Yan, D.M.; Zhang, L.; Qin, M.Y.; Bai, L.Y.; Zhao, Y.D.; et al. Folic acid-conjugated silica-coated gold nanorods and quantum dots for dual-modality CT and fluorescence imaging and photothermal therapy. J. Mater. Chem. B 2014, 2, 1945–1953. [Google Scholar] [CrossRef] [PubMed]
- Yong, Y.; Cheng, X.; Bao, T.; Zu, M.; Yan, L.; Yin, W.; Ge, C.; Wang, D.; Gu, Z.; Zhao, Y. Tungsten sulfide quantum dots as multifunctional nanotheranostics for in vivo dual-modal image-guided photothermal/radiotherapy synergistic therapy. ACS Nano 2015, 9, 12451–12463. [Google Scholar] [CrossRef] [PubMed]
- Augustine, S.; Singh, J.; Srivastava, M.; Sharma, M.; Das, A.; Malhotra, B.D. Recent advances in carbon based nanosystems for cancer theranostics. Biomater. Sci. 2017, 5, 901–952. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Dougherty, C.A.; Zhu, K.; Hong, H. Theranostic applications of carbon nanomaterials in cancer: Focus on imaging and cargo delivery. J. Control. Release 2015, 210, 230–245. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.T.; Luo, J.; Zhou, Q.; Wang, H. Pharmacokinetics, metabolism and toxicity of carbon nanotubes for biomedical purposes. Theranostics 2012, 2, 271–282. [Google Scholar] [CrossRef] [Green Version]
- Villa, C.H.; Dao, T.; Ahearn, I.; Fehrenbacher, N.; Casey, E.; Rey, D.A.; Korontsvit, T.; Zakhaleva, V.; Batt, C.A.; Philips, M.R.; et al. Single walled carbon nanotubes deliver peptide antigen into dendritic cells and enhance IgG responses to tumor-associated antigens. ACS Nano 2011, 5, 5300–5311. [Google Scholar] [CrossRef]
- Zhang, Z.; Yang, X.; Zhang, Y.; Zeng, B.; Wang, S.; Zhu, T.; Roden, R.B.; Chen, Y.; Yang, R. Delivery of telomerase reverse transcriptase small interfering RNA in complex with positively charged single-walled carbon nanotubes suppresses tumor growth. Clin. Cancer Res. 2006, 12, 4933–4939. [Google Scholar] [CrossRef] [Green Version]
- Cornelissen, B.; Able, S.; Kersemans, V.; Waghorn, P.A.; Myhra, S.; Jurkshat, K.; Crossley, A.; Vallis, K.A. Nanographene oxide-based radioimmunoconstructs for in vivo targeting and SPECT imaging of HER2-positive tumors. Biomaterials 2013, 34, 1146–1154. [Google Scholar] [CrossRef]
- Hong, H.; Zhang, Y.; Engle, J.W.; Nayak, T.R.; Theuer, C.P.; Nickles, R.J.; Barnhart, T.E.; Cai, W. In vivo targeting and positron emission tomography imaging of tumor vasculature with 66Ga-labeled nano-graphene. Biomaterials 2012, 33, 4147–4156. [Google Scholar] [CrossRef] [Green Version]
- Cho, Y.; Kim, H.; Choi, Y. A graphene oxide-photosensitizer complex as an enzyme-activatable theranostic agent. Chem. Commun. 2013, 49, 1202–1204. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhang, Q.; Yang, P.; Yu, X.; Huang, L.Y.; Shen, S.; Cai, S. Manganese oxide-coated carbon nanotubes as dual-modality lymph mapping agents for photothermal therapy of tumor metastasis. ACS Appl. Mater. Interfaces 2016, 8, 3736–3743. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wu, H.; Wang, J.; Yang, Y.; Wu, D.; Zhang, Y.; Zhang, Y.; Zhou, Z.; Yang, S. Graphene oxide-BaGdF5 nanocomposites for multi-modal imaging and photothermal therapy. Biomaterials 2015, 42, 66–77. [Google Scholar] [CrossRef] [PubMed]
- Nurunnabi, M.; Khatun, Z.; Reeck, G.R.; Lee, D.Y.; Lee, Y.K. Photoluminescent graphene nanoparticles for cancer phototherapy and imaging. ACS Appl. Mater. Interfaces 2014, 6, 12413–12421. [Google Scholar] [CrossRef] [PubMed]
- Radnia, F.; Mohajeri, N.; Zarghami, N. New insight into the engineering of green carbon dots: Possible applications in emerging cancer theranostics. Talanta 2020, 209, 120547. [Google Scholar] [CrossRef]
- Zhang, F.; Zhang, M.; Zheng, X.; Tao, S.; Zhang, Z.; Sun, M.; Song, Y.; Zhang, J.; Shao, D.; He, K.; et al. Berberine-based carbon dots for selective and safe cancer theranostics. RSC Adv. 2018, 8, 1168–1173. [Google Scholar] [CrossRef] [Green Version]
- Rosch, J.G.; DuRoss, A.N.; Landry, M.R.; Sun, C. Formulation of Folate-Modified Raltitrexed-Loaded Nanoparticles for Colorectal Cancer Theranostics. Pharmaceutics 2020, 12, 133. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.Y.; Shieh, M.J. Platinum (II) drug-loaded gold nanoshells for chemo-photothermal therapy in colorectal cancer. ACS Appl. Mater. Interfaces 2020, 12, 4254–4264. [Google Scholar] [CrossRef]
- Liu, Z.; Shi, J.; Zhu, B.; Xu, Q. Development of a multifunctional gold nanoplatform for combined chemo-photothermal therapy against oral cancer. Nanomedicine 2020, 15, 661–676. [Google Scholar] [CrossRef]
- Lu, Y.J.; Lin, P.Y.; Huang, P.H.; Kuo, C.Y.; Shalumon, K.T.; Chen, M.Y.; Chen, J.P. Magnetic graphene oxide for dual targeted delivery of doxorubicin and photothermal therapy. Nanomaterials 2018, 8, 193. [Google Scholar] [CrossRef] [Green Version]
- Iancu, S.D.; Albu, C.; Chiriac, L.; Moldovan, R.; Stefancu, A.; Moisoiu, V.; Coman, V.; Szabo, L.; Leopold, N.; Bálint, Z. Assessment of gold-coated iron oxide nanoparticles as negative T2 contrast agent in small animal MRI studies. Int. J. Nanomedicine 2020, 15, 4811. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Wang, Y.; Zhang, C.; Fan, Y.; Li, D.; Cao, X.; Xia, J.; Shi, X.; Guo, R. LDH-stabilized ultrasmall iron oxide nanoparticles as a platform for hyaluronidase-promoted MR imaging and chemotherapy of tumors. Theranostics 2020, 10, 2791. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Liu, L.; Morin, E.E.; Liu, M.; Schwendeman, A. Survey of clinical translation of cancer nanomedicines—Lessons learned from successes and failures. Acc. Chem. Res. 2019, 52, 2445–2461. [Google Scholar] [CrossRef] [PubMed]
- Alexander, A.A.; Jotterand, F. Market considerations for nanomedicines and theranostic nanomedicines. In Cancer Theranostics; Academic Press: Cambridge, MA, USA, 2014; pp. 471–491. [Google Scholar]
- Ferrari, M. Cancer nanotechnology: Opportunities and challenges. Nat. Rev. Cancer 2005, 5, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H. Toward a full understanding of the EPR effect in primary and metastatic tumors as well as issues related to its heterogeneity. Adv. Drug Deliv. Rev. 2015, 91, 3–6. [Google Scholar] [CrossRef]
- Hrkach, J.; Von Hoff, D.; Ali, M.M.; Andrianova, E.; Auer, J.; Campbell, T.; De Witt, D.; Figa, M.; Figueiredo, M.; Horhota, A.; et al. Preclinical development and clinical translation of a PSMA-targeted docetaxel nanoparticle with a differentiated pharmacological profile. Sci. Transl. Med. 2012, 4, 128ra39. [Google Scholar] [CrossRef]
- Eliasof, S.; Lazarus, D.; Peters, C.G.; Case, R.I.; Cole, R.O.; Hwang, J.; Schluep, T.; Chao, J.; Lin, J.; Yen, Y.; et al. Correlating preclinical animal studies and human clinical trials of a multifunctional, polymeric nanoparticle. Proc. Natl. Acad. Sci. USA 2013, 110, 15127–15132. [Google Scholar] [CrossRef] [Green Version]
- Zuckerman, J.E.; Gritli, I.; Tolcher, A.; Heidel, J.D.; Lim, D.; Morgan, R.; Chmielowski, B.; Ribas, A.; Davis, M.E.; Yen, Y. Correlating animal and human phase Ia/Ib clinical data with CALAA-01, a targeted, polymer-based nanoparticle containing siRNA. Proc. Natl. Acad. Sci. USA 2014, 111, 11449–11454. [Google Scholar] [CrossRef] [Green Version]
- Meads, M.B.; Gatenby, R.A.; Dalton, W.S. Environment-mediated drug resistance: A major contributor to minimal residual disease. Nat. Rev. Cancer 2009, 9, 665–674. [Google Scholar] [CrossRef]
- Karnik, R.; Gu, F.; Basto, P.; Cannizzaro, C.; Dean, L.; Kyei-Manu, W.; Langer, R.; Farokhzad, O.C. Microfluidic platform for controlled synthesis of polymeric nanoparticles. Nano Lett. 2008, 8, 2906–2912. [Google Scholar] [CrossRef]
- Rolland, J.P.; Maynor, B.W.; Euliss, L.E.; Exner, A.E.; Denison, G.M.; DeSimone, J.M. Direct fabrication and harvesting of monodisperse, shape-specific nanobiomaterials. J. Am. Chem. Soc. 2005, 127, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Mohammadpour, R.; Dobrovolskaia, M.A.; Cheney, D.L.; Greish, K.F.; Ghandehari, H. Subchronic and chronic toxicity evaluation of inorganic nanoparticles for delivery applications. Adv. Drug Deliv. Rev. 2019, 144, 112–132. [Google Scholar] [CrossRef] [PubMed]
- De Matteis, V. Exposure to inorganic nanoparticles: Routes of entry, immune response, biodistribution and in vitro/in vivo toxicity evaluation. Toxics 2017, 5, e29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paris, J.L.; Baeza, A.; Vallet-Regí, M. Overcoming the stability, toxicity, and biodegradation challenges of tumor stimuli-responsive inorganic nanoparticles for delivery of cancer therapeutics. Expert Opin. Drug Deliv. 2019, 16, 1095–1112. [Google Scholar] [CrossRef] [PubMed]
- Damasco, J.A.; Ravi, S.; Perez, J.D.; Hagaman, D.E.; Melancon, M.P. Understanding Nanoparticle Toxicity to Direct a Safe-by-Design Approach in Cancer Nanomedicine. Nanomaterials 2020, 10, E2186. [Google Scholar] [CrossRef]
- Lovrić, J.; Cho, S.J.; Winnik, F.M.; Maysinger, D. Unmodified cadmium telluride quantum dots induce reactive oxygen species formation leading to multiple organelle damage and cell death. Chem. Biol. 2005, 12, 1227–1234. [Google Scholar] [CrossRef] [Green Version]
- De, J.W.H.; Borm, P. Drug delivery and nanoparticles: Applications and hazards. Int. J. Nanomed. 2008, 3, 133–149. [Google Scholar]
- Yen, H.J.; Hsu, S.H.; Tsai, C.L. Cytotoxicity and immunological response of gold and silver nanoparticles of different sizes. Small 2009, 5, 1553–1561. [Google Scholar] [CrossRef]
- Pan, Y.; Neuss, S.; Leifert, A.; Fischler, M.; Wen, F.; Simon, U.; Schmid, G.; Brandau, W.; Jahnen-Dechent, W. Size-dependent cytotoxicity of gold nanoparticles. Small 2007, 3, 1941–1949. [Google Scholar] [CrossRef]
- Adewale, O.B.; Davids, H.; Cairncross, L.; Roux, S. Toxicological behavior of gold nanoparticles on various models: Influence of physicochemical properties and other factors. Int. J. Toxicol. 2019, 38, 357–384. [Google Scholar] [CrossRef]
- Sun, Y.N.; Wang, C.D.; Zhang, X.M.; Ren, L.; Tian, X.H. Shape dependence of gold nanoparticles on in vivo acute toxicological effects and biodistribution. J. Nanosci. Nanotechnol. 2011, 11, 1210–1216. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Liu, Y.; Wang, L.; Xu, L.; Bai, R.; Ji, Y.; Wu, X.; Zhao, Y.; Li, Y.; Chen, C. Surface chemistry and aspect ratiomediated cellular uptake of Au nanorods. Biomaterials 2010, 31, 7606–7619. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Lu, W.; Tovmachenko, O.; Rai, U.S.; Yu, H.; Ray, P.C. Challenge in understanding size and shape dependent toxicity of gold nanomaterials in human skin keratinocytes. Chem. Phys. Lett. 2008, 463, 145–149. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hillyer, J.F.; Albrecht, R.M. Gastrointestinal persorption and tissue distribution of differently sized colloidal gold nanoparticles. J. Pharm. Sci. 2001, 90, 1927–1936. [Google Scholar] [CrossRef]
- Browning, L.M.; Lee, K.J.; Huang, T.; Nallathamby, P.D.; Lowman, J.E.; Xu, X.H. Random walk of single gold nanoparticles in zebrafish embryos leading to stochastic toxic effects on embryonic developments. Nanoscale 2009, 1, 138–152. [Google Scholar] [CrossRef] [Green Version]
- Perreault, F.; Melegari, S.P.; Fuzinatto, C.F.; Bogdan, N.; Morin, M.; Popovic, R.; Matias, W.G. Toxicity of PAMAM-coated gold nanoparticles in different unicellular models. Environ. Toxicol. 2012, 29, 328–336. [Google Scholar] [CrossRef]
- Schmid, G.; Kreyling, W.G.; Simon, U. Toxic effects and biodistribution of ultrasmall gold nanoparticles. Arch Toxicol. 2017, 91, 3011–3037. [Google Scholar] [CrossRef] [Green Version]
- Derfus, A.M.; Chan, W.C.; Bhatia, S.N. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett. 2004, 4, 11–18. [Google Scholar] [CrossRef] [Green Version]
- Jain, K.; Mehra, N.K.; Jain, N.K. Nanotechnology in drug delivery: Safety and toxicity issues. Curr. Pharm. Des. 2015, 21, 4252–4261. [Google Scholar] [CrossRef]
- Kargozar, S.; Hoseini, S.J.; Milan, P.B.; Hooshmand, S.; Kim, H.W.; Mozafari, M. Quantum Dots: A Review from Concept to Clinic. Biotechnol. J. 2020, 15, e2000117. [Google Scholar] [CrossRef]
- Filali, S.; Pirot, F.; Miossec, P. Biological Applications and Toxicity Minimization of Semiconductor Quantum Dots. Trends Biotechnol. 2020, 38, 163–177. [Google Scholar] [CrossRef] [PubMed]
- Weissleder, R.; Stark, D.D.; Engelstad, B.L.; Bacon, B.R.; Compton, C.C.; White, D.L.; Jacobs, P.; Lewis, J. Superparamagnetic iron oxide: Pharmacokinetics and toxicity. AJR Am. J. Roentgenol. 1989, 152, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.W.; Hsu, S.H.; Wang, J.J.; Tsai, J.S.; Lin, K.J.; Wey, S.P.; Chen, F.R.; Lai, C.H.; Yen, T.C.; Sung, H.W. The characteristics, biodistribution, magnetic resonance imaging and biodegradability of superparamagnetic core-shell nanoparticles. Biomaterials 2010, 31, 1316–1324. [Google Scholar] [CrossRef] [PubMed]






| Cancer Nanomedicines | Features | Indications (Approved and or in Clinical Phases) |
|---|---|---|
| DaunoXome® | Daunorubicin citrate encapsulated liposomes of size less than 100 nm/EPR-based passive targeting to tumor | HIV-associated Kaposi’s sarcoma |
| DepoCyt® | Cytarabine encapsulated in multivesicular liposomes Sustained release formulation of cytarabine maintains cytotoxic concentrations of the drug in the cerebrospinal fluid for more than 14 days after a single 50 mg injection | lymphomatous malignant meningitis |
| Doxil® | Doxorubicin hydrochloride encapsulated in stealth liposomes (~100 nm)/passive targeting to tumor via EPR effect | AIDS-associated Kaposi’s sarcoma, multiple myeloma, ovarian cancer |
| Marqibo® | Vincristine sulfate encapsulated in liposomes (~100 nm)/passive targeting to tumor via EPR effect | Acute lymphoid leukemia |
| Mepact™ | Mifamurtide incorporated into large multilamellar liposomes/mononuclear phagocytic system (MSP) targeting | Non-metastasizing osteosarcoma |
| Myocet® | Doxorubicin encapsulated ~180 nm oligolamellar liposomes/MPS targeting; “MPS depot,” slow release of drug into blood circulation resembles | Metastatic breast cancer |
| Oncoprex (Genprex) | Tumor suppressor gene TUSC2/FUS1 encapsulated liposome (~100 nm)/passive targeting to tumor via EPR effect | lung cancer/Clinical phase I/II |
| Oncaspar® | It is a pegaspargase; a PEGylated form of asparaginase for i.m injection | Acute lymphoblastic leukemia |
| Eligard® | PLGA-based leuprolide acetate (synthetic GnRH) microparticles suspension for s.c injection. | Advanced prostate cancer |
| Genexol® | Paclitaxel loaded PEG-PLA block copolymer-based micelles (20–50 nm)/passive targeting to tumor via EPR effect | Metastatic breast cancer, pancreatic cancer |
| Opaxio® | Paclitaxel covalently linked to polyglutamate-based solid NPs/passive targeting to tumor via EPR effect and drug release inside solid tumor via enzymatic hydrolysis of polyglutamate | Glioblastoma |
| Halaven E7389-LF® | Eribulin mesylate liposomal formulation/passive targeting via EPR | Solid tumors/Clinical phase I |
| Abraxane® | Paclitaxel-loaded albumin NPs (<150 nm)/passive targeting via EPR and dissociation of drug-albumin bound helps endothelial transcytosis of paclitaxel via albumin-receptor | Metastatic breast cancer, non-small-cell lung cancer |
| NanoTherm® | Aminosilane-coated superparamagnetic iron oxide NPs (~15 nm)/passive targeting to tumor via EPR, the injected NPs oscillate, generating heat directly within the tumor tissue once exposed to the alternating magnetic field | Clinical phase IV approval (EU) for local heat ablation in glioblastoma, prostate, and pancreatic cancer |
| ThermoDox® | Doxorubicin lysolipid heat-sensitive liposome (~100 nm)/passive targeting to tumor via EPR on i.v injection; when heated to 40 °C–45 °C, liposome releases doxorubicin directly into and around the targeted tumor | Breast cancer, primary liver cancer/Clinical phase III |
| Feridex, Resovist, Combidex (withdrawn), Clariscan (withdrawn) and Feraheme | Iron oxide NPs/passive targeting to tumor via EPR/Feridex® cannot be administered as an i.v bolus; severe back pain, while Resovist® can be administered by fast bolus injection, and therefore imaging of the arterial phase is feasible. Feraheme (Ferumoxytol) iron oxide NPs has a carbohydrate coat. The agent is taken up by macrophages and ultimately the RES | MRI imaging of lymph nodes and certain liver tumor |
| Lipocurc | Liposomal curcumin of size <100 nm/passive targeting by EPR | Solid tumors/Clinical phase I/II |
| MM-302 | HER2-targeted PEGylated liposomal doxorubicin/active and passive targeting to the tumor. | Breast cancer/Clinical phase II/III |
| AuroLase® | PEG-coated silica-gold nanoshells for near IR thermal ablation | Thermal ablation of solid tumors & metastatic lung tumors/Phase I |
| ABI-009 | Rapamycin loaded albumin NPs/passive targeting and endothelial transcytosis via albumin-receptor | Bladder cancer/Clinical phase I/II |
| CRLX301 | Cyclodextrin-based nanoparticle- docetaxel conjugate | Solid tumors/Clinical phase I/II |
| NC-6004 Nanoplatin | Polyamino acid and cisplatin derived PEGylated nano micelles | Solid tumors, lung, bladder, or pancreatic cancers/Clinical phase II/III |
| AZD2811 | Aurora B kinase inhibitor loaded long-circulating NPs | Solid tumors/Clinical phase I |
| Promitil | Mitomycin-C loaded PEGylated liposomal formulation | Solid tumors/Clinical phase I |
| S.No. | Type of Cancer Nanomedicine and Overview | Purpose in Cancer Management | In Vitro/In Vivo Outcome | Ref. |
|---|---|---|---|---|
| 1. | Hypericin-bearing magnetic iron oxide nanoparticles | Therapeutics | Upon irradiation, light-induced cell death showed concentration and time-dependent death of Jurkat T cells because of the generation of reactive oxygen species | [22] |
| 2. | Thiolated heparin–pheophorbide A (PhA) conjugated magnetic iron oxide/gold hybrid nanoparticle | Therapeutics | Phototoxicity and strong fluorescence signals from the NPs result in A549 cells deaths under light irradiation. | [23] |
| 3. | Anti-HER2 (c-erbB-2) conjugated gold nanoshell | Therapeutics | Nanocomplexes when targeted to OVCAR3 cells and irradiated with near infra-red (NIR) laser-caused selective destruction of cancer cells. | [39] |
| 4. | Palladium-gold nanostructures | Therapeutics | -Excellent in vitro and in vivo anticancer activity in HeLa cells and HeLa tumor xenograft of male B9 mice at laser radiations of 808 nm | [41] |
| 5. | Magnetic nanoparticle clusters | Therapeutics | -Excellent in vitro cytotoxicity in A549 cells as well as in A549 tumor-bearing mice model upon NIR irradiation at 808 nm | [42] |
| 6. | Platinum (II) drug-loaded gold nanoshells | Therapeutics | -Upon NIR irradiation, gold-nanoshells promote a significant increment in temperature that was found sufficient to ablate the tumor cells. -Platinum (II) drug-loaded gold nanoshells exhibited a profound inhibition of tumor growth compared to chemotherapy or photothermal therapy given alone. | [130] |
| 7. | PDPN antibody and doxorubicin (DOX) conjugated gold nanoparticles [(PDPNAb)-AuNP-DOX] | Therapeutics | -(PDPNAb)-AuNP-DOXshowed good biocompatibility, drug loading capacity, cellular uptake efficiency, a pH-dependent drug release profile, and a much lower IC50 than free DOX. -The designed (PDPN Ab)-AuNP-DOX system can be applied as a PTT platform because of its high photothermal conversion efficiency. -The administration of (PDPN Ab)-AuNPDOX followed by laser irradiation exhibited an enhanced antitumor effect in in vitro and in vivo model. | [131] |
| 8. | Cetuximab decorated doxorubicin encapsulated magnetic graphene oxide nanoparticles | Therapeutics | -Improved cellular uptake in CT-26 cells via EGFR receptor-mediated endocytosis. -Improved photothermal ablation upon NIR irradiation. -Significant improvement in tumor volume reduction in CT-26 tumor-bearing BALB/c mice. | [132] |
| 9. | Folic acid-modified PEGylated polyethyleneimine (PEI)-entrapped gold nanoparticles (FA-Au PENPs) | Diagnostics | -FA-Au PENPs showed the excellent potential to target FA receptor overexpressed KB-HFAR and KB-LFAR cancer cells -Significantly enhanced in vitro and in vivo CT contrast enhancement compared to non-targeted Au PENPs | [61] |
| 10. | Lactobionic acid (LA)-modified dendrimer-entrapped gold nanoparticles (LA-Au DENPs) | Diagnostics | -LA-Au DENPs showed significantly higher cellular internalization in asialoglycoprotein receptors HepG2 and L929 cancer cells -Significantly enhanced in vitro and in vivo CT contrast enhancement compared to non-targeted Au DENPs | [62] |
| 11. | Arginine-glycine-aspartic acid (RGD) peptide-targeted iron oxide nanoparticles | Diagnostics | -Significantly higher in vitro and in vivo r2 relaxivity and selective targeting to the αvβ3 receptor overexpressed U87MG cancer cells | [73] |
| 12. | Polyethyleneimine (PEI)-stabilized hyaluronic acid (HA)-tagged magnetic iron oxide nanoparticles | Diagnostics | -Specifically internalized in CD44 receptor-overexpressed Hela cells -Significantly higher r2 relaxivity and contrast in T2-weighed MR imaging in a tumor model | [74] |
| 13. | Tumor-avid cyclic pentapeptide labeled (Arg-Gly-Asp-DPhe-Lys) NIR emitting silver sulfide quantum dots (QDs) | Diagnostics | -Fluorescence microscopy revealed selective integrin-mediated internalization of targeted quantum dots in 4T1luc cancer cells | [78] |
| 14. | BRCAA1 antibody- and Her2 antibody-conjugated amphiphilic polymer engineered CdSe/ZnS QDs | Diagnostics | -The developed QDs exhibited strong photoluminescence and revealed targeted imaging of in vivo gastric cancer (MGC803) cells | [79] |
| 15. | Pullulan-based gadolinium-chelated nanoparticles | Diagnostics | -Gadolinium nanoparticles showed early detection of the tumor (~3 mm3) within 2 h after the administration of nanoparticles within the small CT26 tumor-bearing mice model by MRI and optical imaging. | [80] |
| 16. | Gold-coated iron oxide nanoparticles (GIONPs) | Diagnostics | -The developed GIONPs showed reduced cytotoxicity, produce a negative T2 signal in the MRI, which makes them a suitable candidate as a contrast agent for MRI applications, and validated in small animals. | [133] |
| 17. | 99 mTc-gallic-gold nanoparticles | Diagnostics | -99 mTc-gallic-gold nanoparticles displayed good stability and cytocompatibility. -It exhibited high uptake in tumor cells after intratumoral and intravenous injection. | [11] |
| 18. | hyaluronic acid (HA)-functionalized GdCl3 entrapped SWNTs to deliver the contrast agent, | Diagnostics | GdCl3-bearing HA-SWNTs showed significantly higher circulation time for MRI. In vivo imaging in S180 cells, tumor-bearing male BALB/c mice revealed that Gd/HA-SWCNTs exhibited the highest tumor-targeting efficiency and T1-relaxivity enhancement. | [92] |
| 19. | Epirubicin-5TR1 aptamer-SPIONs tertiary complex | Theranostics | -Significantly enhanced cellular uptake and cytotoxicity against C26 cells. - MRI demonstrated a high accumulation of the nano-magnets within the tumor site | [103] |
| 20. | pheoA (photosensitizer) conjugated heparin-iron oxide nanoparticles (PheoA–Hep–Fe3O4 NPs) | Theranostics | - Significantly higher T2 signal intensity and cellular uptake of the NPs in KB cells -Excellent dose and time-dependent cytotoxicity | [104] |
| 21. | Folic acid conjugated Protoporphyrin IX (photosensitizer) linked synthesized a near-infrared (NIR) emitting gold quantum cluster capped with lipoic acid | Theranostics | -Photodynamic therapy revealed a higher generation of singlet oxygen generation and the better localization of the NPs on tumor cells. -In vivo study with C6 cell line xenograft of Albino Swiss mice showed that the developed nanocluster is useful for the effective destruction and monitoring of tumor cells. | [112] |
| 22. | Tungsten sulfide (WS2) quantum dots (QDs) | Theranostics | -The developed tungsten sulfide-quantum dots demonstrated a significant increase in signal intensity of X-ray computed tomography/photoacoustic imaging. -The developed system exhibited a synergistic effect of remarkable photothermal therapy/radiotherapy against tumor cell (in HeLa and HepG2 cells). | [115] |
| 23. | Multi-walled carbon nanotubes (MWNTs) coated with manganese oxide (MnO) and polyethylene glycol (PEG) | Theranostics | -Upon NIR laser irradiation multi-walled carbon nanotubes coated with manganese oxide and polyethylene glycol exhibited high photothermal conversion efficiency. -The developed system exhibited powerful dual-modality for lymphatic tracing capability and high efficiency for tumor ablation in in vivo tumor model. | [124] |
| 24. | PEGylated graphene oxide-BaGdF5 nanocomposites (GO/BaGdF5/PEG) | Theranostics | -The developed nanotheranostic system demonstrated a positive magnetic resonance contrast effect and improved X-ray attenuation characteristics than Iohexol. -It enables effective dual-modality for MRI and X-ray computed tomography imaging against in vivo tumor models. | [125] |
| 25. | 99 mTc-doxorubicin-loaded gallic acid-gold nanoparticles (99 mTc-DOX-GA-Au-NPs) | Theranostics | -GA-Au-NPs exhibited increased anti-proliferative activity, with approximately a four-fold lower IC50 value compared to free DOX. The optimized radiolabeling efficiency of 99 mTc-DOX was ≈93%. It showed good physiological stability in mice serum for at least 8 h. -The intratumoral delivery of 99 mTc-DOX-GA-Au-NPs in the tumor-induced mice model showed dramatic improvement in the accumulation of the drug in the tumor. | [12] |
| 26. | LDH-stabilized hyaluronic acid-modified ultrasmall iron oxide nanoparticles loaded with doxorubicin | Theranostics | -The LDH-Fe3O4-HA nanohybrids possess good colloidal stability and cytocompatibility, display an r1 relaxivity ten-fold higher than the pristine ultrasmall Fe3O4. -In vitro experiments demonstrated that LDH-Fe3O4-HA/DOX nanohybrids can specifically target B16 cells overexpressing CD44 receptors and effectively release DOX to the nucleus. -In vivo results show that with the pretreatment of tumor tissue by HAase to degrade the overexpressed HA in the extra-cellular matrix, the designed nanoplatforms have a better tumor penetration for significantly enhanced MR imaging ability of tumors and improved therapeutic outcome with low side effects. | [134] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alshehri, S.; Imam, S.S.; Rizwanullah, M.; Akhter, S.; Mahdi, W.; Kazi, M.; Ahmad, J. Progress of Cancer Nanotechnology as Diagnostics, Therapeutics, and Theranostics Nanomedicine: Preclinical Promise and Translational Challenges. Pharmaceutics 2021, 13, 24. https://doi.org/10.3390/pharmaceutics13010024
Alshehri S, Imam SS, Rizwanullah M, Akhter S, Mahdi W, Kazi M, Ahmad J. Progress of Cancer Nanotechnology as Diagnostics, Therapeutics, and Theranostics Nanomedicine: Preclinical Promise and Translational Challenges. Pharmaceutics. 2021; 13(1):24. https://doi.org/10.3390/pharmaceutics13010024
Chicago/Turabian StyleAlshehri, Sultan, Syed Sarim Imam, Md. Rizwanullah, Sohail Akhter, Wael Mahdi, Mohsin Kazi, and Javed Ahmad. 2021. "Progress of Cancer Nanotechnology as Diagnostics, Therapeutics, and Theranostics Nanomedicine: Preclinical Promise and Translational Challenges" Pharmaceutics 13, no. 1: 24. https://doi.org/10.3390/pharmaceutics13010024