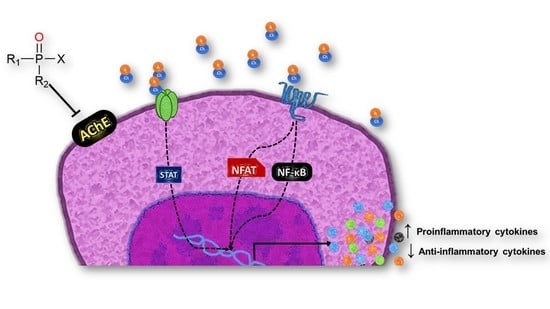
Organophosphorus Pesticides as Modulating Substances of Inflammation through the Cholinergic Pathway
Abstract
:1. Organophosphorus Pesticides
Mechanism of Action of OPs and Toxicity
2. Cholinergic System
3. Immunotoxicity of OPs through the Cholinergic System
4. Cytokine-Mediated Modulation of the Inflammatory Process by OP Exposure
5. Therapeutic Strategies to Mitigate the Long-Term Inflammatory Effects of Acute OP Intoxication
6. Lower Vertebrates as a Biomedical Model
| OPs | Dose | Exposure Time | Effects | Tissue/Cell Line | Organism Model | References |
|---|---|---|---|---|---|---|
| Diazinon | LC50-7.830 ppm, ½ LC50-3.915 ppm | 96 h | ↓ AChE activity ↑ ACh levels | Spleen mononuclear cells | Nile tilapia (O. niloticus) | [22]. |
| Diazinon | 0.97, 1.95 and 3.91 mg/L | 6, 12 and 24 h | ↓ AChE activity ↓mAChR, nAChR concentration and ↑ ACh levels. | Spleen mononuclear cells | Nile tilapia (O. niloticus) | [23]. |
| Diazoxon | 1 nm, 1 µM, and 10 µM | 24 h | ↓ (M3, M4, M5) receptors and nAChR-β2 expression. | Spleen mononuclear cells | Nile tilapia (O. niloticus) | [24]. |
| Diazinon | 1.96 mg/L | 96 h | ↑ Respiratory burst and IgM concentration | Spleen mononuclear cells | Nile tilapia (O. niloticus) | [61]. |
| Diazinon | 0.97, 1.95 and 3.91 mg/L | 6 and 24 h | Alterations in Ca2+ flux and pERK 1/2. ↑ Cellular senescence ↓ mitchondrial membrane. potential ↑ apoptotic cells. | Spleen mononuclear cells | Nile tilapia (O. niloticus) | [107]. |
| Chlorpyrifos | 0.422 and 0.211 mg/L) | 96 h | ↓ Phagocytic Capacity. | Peripheral blood | Nile tilapia (O. niloticus) | [145]. |
| Diazinon | LC50-7.830 ppm | 96 h | ↓ Phagocytic capacity and cellular proliferation. | Spleen mononuclear cells | Nile tilapia (O. niloticus) | [146]. |
| Diazinon | 0.97, 1.95 and 3.91 mg/L | 6 and 24 h | ↑ Reactive oxygen species ↓ Phagocytic activity | Peripheral blood mononuclear cells | Nile tilapia (O. niloticus) | [147]. |
| Chlorpyrifos | 0.051 mg/L | 96 h | ↓ IgM levels and deregulation in lysozyme activity. | Spleen mononuclear cells | Nile tilapia (O. niloticus) | [148]. |
| Diazinon | 0.97, 1.95 and 3.91 mg/L | 12 and 24 h | ↑ Protein oxidative damage. | Liver and gills | Nile tilapia (O. niloticus) | [149]. |
| Diazinon | 0.97, 1.95 and 3.91 mg/L | 6 and 24 h | ↑ Neutrophil extracellular traps (NETs) induction. | Spleen mononuclear cells | Nile tilapia (O. niloticus) | [150]. |
| Diazoxon | M | 1 h and 2 h | ↓ Ca2+ flux against PMA and ionomycin stimulation. ↓ ERK1/2 phosphorylation. ↓ Mitochondrial membrane potential. ↑Apoptotic and cellular senescence. | Spleen mononuclear cells | Nile tilapia (O. niloticus) | [151]. |
| Temephos | 10 mg/L | 7 and 21 days | AChE inhibition ↑ ACh levels | Smooth muscle | Guppy fish (Poecilia reticulata) | [153]. |
| Temephos | 10 mg/L | 7 days | ↓ Phagocytic capacity | Spleen mononuclear cells | Guppy fish (P. reticulata) | [154]. |
| Temephos | 10 mg/L | 7, 14, and 21 days | ↑ Leucocytes death | Spleen mononuclear cells | Guppy fish (P. reticulata) | [155]. |
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Al-Ghanim, K.A. Acute toxicity and effects of sub-lethal malathion exposure on biochemical and haematological parameters of Oreochromis niloticus. Sci. Res. Essays 2012, 7, 1674–1680. [Google Scholar] [CrossRef]
- Sidhu, G.K.; Singh, S.; Kumar, V.; Dhanjal, D.S.; Datta, S.; Singh, J. Toxicity, monitoring and biodegradation of organophosphate pesticides: A review. Crit. Rev. Environ. Sci. Technol. 2019, 49, 1135–1187. [Google Scholar] [CrossRef]
- International Code of Conduct on the Distribution and Use of Pesticides. Available online: http://apps.who.int/iris/bitstream/handle/10665/70602/WHO_HTM_NTD_WHOPES_2010.7_spa.pdf;jsessionid=6BF8CBB80C94FB71C65499FC33ACB281?sequence=1 (accessed on 28 February 2022).
- Cancer Clusters. Available online: https://www.cdc.gov/nceh/clusters/fallon/organophosfaq.htm#:~:text=Organophosphates%20are%20the%20most%20widely,in%20the%20body%20called%20acetylcholinesterase (accessed on 28 February 2022).
- Sharma, A.; Kumar, V.; Shahzad, B.; Tanveer, M.; Sidhu, G.P.S.; Handa, N.; Kohli, S.K.; Yadav, P.; Bali, A.S.; Parihar, R.D.; et al. Worldwide pesticide usage and its impacts on ecosystem. SN Appl. Sci. 2019, 1, 1446. [Google Scholar] [CrossRef] [Green Version]
- Tudi, M.; Ruan, H.D.; Wang, L.; Lyu, J.; Sadler, R.; Connell, D.; Chu, C.; Phung, D. Agriculture Development, Pesticide Application and Its Impact on the Environment. Int. J. Environ. Res. Public Health 2021, 18, 1112. [Google Scholar] [CrossRef]
- Torres-Palma, R.; Serna-Galvis, E. Sonolysis; Academic Press: Cambridge, MA, USA, 2018; pp. 177–213. [Google Scholar] [CrossRef]
- Fernández, A.; Mancipe, L.; Fernández, A. Intoxicación por organofosforados. Rev. Med. 2010, 18, 84–92. [Google Scholar] [CrossRef] [Green Version]
- Chowdhary, S.; Bhattacharyya, R.; Banerjee, D. Acute organophosphorus poisoning. Clin. Chim. Acta 2014, 431, 66–76. [Google Scholar] [CrossRef]
- Nkinsa, P.N.; Muckle, G.; Ayotte, P.; Lanphear, B.P.; Arbuckle, T.E.; Fraser, W.D.; Bouchard, M.F. Organophosphate pesticides exposure during fetal development and IQ scores in 3 and 4-year old Canadian children. Environ. Res. 2020, 190, 110023. [Google Scholar] [CrossRef]
- Abbas, R.; Leister, C.; El-Gaaloul, M.; Chalon, S.; Sonnichsen, D. Ascending Single-Dose Study of the Safety Profile, Tolerability, and Pharmacokinetics of Bosutinib Coadministered with Ketoconazole to Healthy Adult Subjects. Clin. Therap. 2012, 34, 2011–2019. [Google Scholar] [CrossRef]
- Kaur, K.; Besnier, F.; Glover, K.A.; Nilsen, F.; Aspehaug, V.T.; Fjørtoft, H.B.; Horsberg, T.E. The mechanism (Phe362Tyr mutation) behind resistance in Lepeophtheirus salmonis pre-dates organophosphate use in salmon farming. Sci. Rep. 2017, 7, 12349. [Google Scholar] [CrossRef]
- Oruc, E.; Usta, D. Evaluation of oxidative stress responses and neurotoxicity potential of diazinon in different tissues of Cyprinus carpio. Environ. Toxicol. Pharmacol. 2007, 23, 48–55. [Google Scholar] [CrossRef]
- Fulton, M.H.; Key, P.B. Acetylcholinesterase inhibition in estuarine fish and invertebrates as an indicator of organophosphorus insecticide exposure and effects. Environ. Toxicol. Chem. 2001, 20, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Profile for Diazinon. Available online: https://www.atsdr.cdc.gov/toxprofiles/tp86.pdf (accessed on 28 February 2022).
- Elersek, T.; Filipič, M. Organophosphorous Pestidices Mechanism of Their Toxicity Pesticides—The Impact of Pesticides Expusure; Intech: Rijeka, Croatia, 2011. [Google Scholar] [CrossRef] [Green Version]
- Robb, E.L.; Baker, M.B. Organophosphathe Toxicity. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021; PMID: 29261901. [Google Scholar]
- Vásquez, M.O. Intoxicación por organofosforados. Rev. Med. Sinerg. 2020, 5, e558. [Google Scholar] [CrossRef]
- King, A.M.; Aaron, C.K. Organophosphate and Carbamate Poisoning. Emerg. Med. Clin. N. Am. 2015, 33, 133–151. [Google Scholar] [CrossRef] [PubMed]
- Mileson, B.E.; Chambers, J.E.; Chen, W.L.; Dettbarn, W.; Ehrich, M.; Eldefrawi, A.T.; Gaylor, D.W.; Hamernik, K.; Hodgson, E.; Karczmar, A.G.; et al. Common Mechanism of Toxicity: A Case Study of Organophosphorus Pesticides. Toxicol. Sci. 1998, 41, 8–20. [Google Scholar] [CrossRef] [Green Version]
- Kawashima, K.; Fujii, T.; Moriwaki, Y.; Misawa, H.; Horiguchi, K. Reconciling neuronally and nonneuronally derived acetylcholine in the regulation of immune function. Life Sci. 2012, 1261, 1027–1032. [Google Scholar] [CrossRef]
- Girón-Pérez, M.I.; Zaitseva, G.; Casas-Solis, J.; Santerre, A. Effects of diazinon and diazoxon on the lymphoproliferation rate of splenocytes from Nile tilapia (Oreochromis niloticus): The immunosuppresive effect could involve an increase in acetylcholine levels. Fish Shellfish Immunol. 2008, 25, 517–521. [Google Scholar] [CrossRef] [PubMed]
- Toledo-Ibarra, G.; Díaz-Resendiz, K.; Pavón-Romero, L.; Rojas-García, A.; Medina-Díaz, I.; Girón-Pérez, M. Effects of diazinon on the lymphocytic cholinergic system of Nile tilapia fish (Oreochromis niloticus). Vet. Immunol. Immunopathol. 2016, 176, 58–63. [Google Scholar] [CrossRef]
- Toledo-Ibarra, G.; Girón-Pérez, M.; Covantes-Rosales, C.; Ventura-Ramón, G.; Pérez-Sánchez, G.; López-Torres, A.; Diaz-Resendiz, K.; Becerril-Villanueva, E.; Pavón, L. Alterations in the non-neuronal cholinergic system induced by in-vitro exposure to diazoxon in spleen mononuclear cells of Nile tilapia (O. niloticus). Fish Shellfish Immunol. 2021, 108, 134–141. [Google Scholar] [CrossRef]
- Proskocil, B.; Bruun, D.A.; Thompson, C.M.; Fryer, A.; Lein, P.J. Organophosphorus Pesticides Decrease M2 Muscarinic Receptor Function in Guinea Pig Airway Nerves via Indirect Mechanisms. PLoS ONE 2010, 5, e10562. [Google Scholar] [CrossRef] [Green Version]
- Poojary, A.; Basha, P.M. Cold stress interaction on organophosphate insecticide poisoning: Age-related assessment in rat cerebral cortex. Indian J. Exp. Biol. 2012, 50, 110–116. [Google Scholar]
- Basaure, P.; Guardia-Escote, L.; Cabré, M.; Peris-Sampedro, F.; Sánchez-Santed, F.; Domingo, J.L.; Colomina, M.T. Postnatal chlorpyrifos exposure and apolipoprotein E (APOE) genotype differentially affect cholinergic expression and developmental parameters in transgenic mice. Food Chem. Toxicol. 2018, 118, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, S.; Wang, C.; Tian, H.; Wang, W.; Ru, S. Effects of monocrotophos pesticide on cholinergic and dopaminergic neurotransmitter systems during early development in the sea urchin Hemicentrotus pulcherrimus. Toxicol. Appl. Pharmacol. 2017, 328, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Zhang, X.-G.; Yang, X.; He, Y.-Z. The diagnostic value of butyrylcholinesterase in acute organophosphorus pesticide poisoning. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue= Chin. Crit. Care Med. = Zhongguo Weizhongbing Jijiuyixue 2010, 22, 193–196. [Google Scholar]
- Figueiredo, T.H.; Apland, J.P.; Braga, M.F.M.; Marini, A.M. Acute and long-term consequences of exposure to organophosphate nerve agents in humans. Epilepsia 2018, 59, 92–99. [Google Scholar] [CrossRef] [Green Version]
- Bayrami, M.; Hashemi, T.; Malekirad, A.A.; Ashayeri, H.; Faraji, F.; Abdollahi, M. Electroencephalogram, cognitive state, psychological disorders, clinical symptom, and oxidative stress in horticulture farmers exposed to organophosphate pesticides. Toxicol. Ind. Health 2012, 28, 90–96. [Google Scholar] [CrossRef]
- Perry, J.; Cotton, J.; Rahman, M.; Brumby, S. Organophosphate exposure and the chronic effects on farmers: A narrative review. Rural Remote Health 2020, 20, 4508. [Google Scholar] [CrossRef]
- De Silva, H.J.; Samarawickrema, N.A.; Wickremasinghe, A.R. Toxicity due to organophosphorus compounds: What about chronic exposure? Trans. R. Soc. Trop. Med. Hyg. 2006, 100, 803–806. [Google Scholar] [CrossRef] [Green Version]
- Thrasher, J.D.; Heuser, G.; Broughton, A. Immunological Abnormalities in Humans Chronically Exposed to Chlorpyrifos. Arch. Environ. Health Int. J. 2002, 57, 181–187. [Google Scholar] [CrossRef]
- Zafar, R.; Munawar, K.; Nasrullah, A.; Haq, S.; Ghazanfar, H.; Sheikh, A.B.; Khan, A.Y. Acute Renal Failure due to Organophosphate Poisoning: A Case Report. Cureus 2017, 9, e1523. [Google Scholar] [CrossRef] [Green Version]
- Cavari, Y.; Landau, D.; Sofer, S.; Leibson, T.; Lazar, I. Organophosphate Poisoning-Induced Acute Renal Failure. Pediatr. Emerg. Care 2013, 29, 646–647. [Google Scholar] [CrossRef]
- Ortega-Miller, J.G.; Yezioro-Rubinsky, S.; Benavides-Pinto, B.C.; Báez-Quintero, L.C. Efectos teratogénicos de insecticidas organofosforados en la etiología de labio y paladar hendido: Revisión de literatura. Rev. Nac. De Odontol. 2017, 13, 13. [Google Scholar] [CrossRef] [Green Version]
- Rodriguez-Diaz, R.; Dando, R.; Jacques-Silva, M.C.; Fachado, A.; Molina, J.; Abdulreda, M.H.; Ricordi, C.; Roper, S.D.; Berggren, P.-O.; Caicedo, A. Alpha cells secrete acetylcholine as a non-neuronal paracrine signal priming beta cell function in humans. Nat. Med. 2011, 17, 888–892. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wessler, I.; Kirkpatrick, C.J.; Racké, K. Non-neuronal acetylcholine, a locally acting molecule, widely distributed in biological systems: Expression and function in humans. Pharmacol. Ther. 1998, 77, 59–79. [Google Scholar] [CrossRef]
- Bhuiyan, M.; Murad, F.; Fant, M. The placental cholinergic system: Localization to the cytotrophoblast and modulation of nitric oxide. Cell Commun. Signal. 2006, 4, 4–7. [Google Scholar] [CrossRef] [Green Version]
- Lev-Lehman, E.; Deutsch, V.; Eldor, A.; Soreq, H. Immature Human Megakaryocytes Produce Nuclear-Associated Acetylcholinesterase. Blood 1997, 89, 3644–3653. [Google Scholar] [CrossRef] [Green Version]
- Kawashima, K.; Fujii, T. The lymphocytic cholinergic system and its biological function. Life Sci. 2003, 72, 2101–2109. [Google Scholar] [CrossRef]
- Resendiz, K.J.G.D.; Toledo-Ibarra, G.A.; Girón-Pérez, M.I. Modulation of Immune Response by Organophosphorus Pesticides: Fishes as a Potential Model in Immunotoxicology. J. Immunol. Res. 2015, 2015, 213836. [Google Scholar] [CrossRef]
- Banks, C.N.; Lein, P.J. A review of experimental evidence linking neurotoxic organophosphorus compounds and inflammation. NeuroToxicology 2012, 33, 575–584. [Google Scholar] [CrossRef] [Green Version]
- Koureas, M.; Rachiotis, G.; Tsakalof, A.; Hadjichristodoulou, C. Increased Frequency of Rheumatoid Arthritis and Allergic Rhinitis among Pesticide Sprayers and Associations with Pesticide Use. Int. J. Environ. Res. Public Health 2017, 14, 865. [Google Scholar] [CrossRef] [Green Version]
- Meyer, A.; Sandler, D.P.; Freeman, L.E.B.; Hofmann, J.; Parks, C.G. Pesticide Exposure and Risk of Rheumatoid Arthritis among Licensed Male Pesticide Applicators in the Agricultural Health Study. Environ. Health Perspect. 2017, 125, 077010. [Google Scholar] [CrossRef]
- Andrew, P.M.; Lein, P.J. Neuroinflammation as a Therapeutic Target for Mitigating the Long-Term Consequences of Acute Organophosphate Intoxication. Front. Pharmacol. 2021, 12, 12. [Google Scholar] [CrossRef] [PubMed]
- Hernández, A.F.; Parrón, T.; Alarcón, R. Pesticides and asthma. Curr. Opin. Allergy Clin. Immunol. 2011, 11, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Benka-Coker, W.; Loftus, C.; Karr, C.; Magzamen, S. Association of Organophosphate Pesticide Exposure and a Marker of Asthma Morbidity in an Agricultural Community. J. Agromed. 2020, 25, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.-L.; Hou, Y.-C.; Wang, I.-K.; Lu, K.-C.; Yen, T.-H. Organophosphate pesticides and new-onset diabetes mellitus: From molecular mechanisms to a possible therapeutic perspective. World J. Diabetes 2021, 12, 1818–1831. [Google Scholar] [CrossRef]
- Wessler, I.K.; Kirkpatrick, C.J. The Non-neuronal Cholinergic System: An Emerging Drug Target in the Airways. Pulm. Pharmacol. Ther. 2001, 14, 423–434. [Google Scholar] [CrossRef]
- Abreu-Villaça, Y.; Filgueiras, C.; Manhães, A.C. Developmental aspects of the cholinergic system. Behav. Brain Res. 2011, 221, 367–378. [Google Scholar] [CrossRef]
- Nizri, E.; Brenner, T. Modulation of inflammatory pathways by the immune cholinergic system. Amino Acids 2011, 45, 73–85. [Google Scholar] [CrossRef]
- Wessler, I.; Kirkpatrick, C.J. Acetylcholine beyond neurons: The non-neuronal cholinergic system in humans. J. Cereb. Blood Flow Metab. 2008, 154, 1558–1571. [Google Scholar] [CrossRef] [Green Version]
- Kawashima, K.; Fujii, T. Extraneuronal cholinergic system in lymphocytes. Pharmacol. Ther. 2000, 86, 29–48. [Google Scholar] [CrossRef]
- Mokarizadeh, A.; Faryabi, M.R.; Rezvanfar, M.A.; Abdollahi, M. A comprehensive review of pesticides and the immune dysregulation: Mechanisms, evidence and consequences. Toxicol. Mech. Methods 2015, 25, 258–278. [Google Scholar] [CrossRef]
- Wang, X.; Yang, Z.; Xue, B.; Shi, H. Activation of the Cholinergic Antiinflammatory Pathway Ameliorates Obesity-Induced Inflammation and Insulin Resistance. Endocrinology 2011, 152, 836–846. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Bouhy, Z.; El-Hakim, Y.A.; Mohamed, E. Chronic Effect of Chlorpyrifos on Biochemical, Immunological Changes and DNA Damage in Juvenile Nile Tilapia (Oreochromis niloticus). Zagazig Vet. J. 2018, 46, 51–59. [Google Scholar] [CrossRef] [Green Version]
- Ahmadi, K.; Mirvaghefei, A.R.; Banaee, M.; Vosoghei, A.R. Effects of long-term diazinon exposure on some immunological and haematological parameters in rainbow trout Oncorhynchus mykiss (Walbaum, 1792). Toxicol. Environ. Health Sci. 2014, 6, 1–7. [Google Scholar] [CrossRef]
- Castillo-Sosa, Y.; Sierra-Fonseca, A.; Martínez-Martínez, A.; Plenge-Tellechea, F. Efecto del diazinón sobre el cultivo de linfocitos de sangre periférica de human. Tecnoscience 2009, 3, 97–106. Available online: https://vocero.uach.mx/index.php/tecnociencia/article/view/734 (accessed on 2 March 2022).
- Girón-Pérez, M.; Velázquez-Fernández, J.B.; Díaz-Resendiz, K.J.G.; Díaz-Salas, F.; Canto-Montero, C.; Medina-Díaz, I.; Robledo-Marenco, M.; Rojas-García, A.; Zaitseva, G. Immunologic parameters evaluations in Nile tilapia (Oreochromis niloticus) exposed to sublethal concentrations of diazinon. Fish Shellfish Immunol. 2009, 27, 383–385. [Google Scholar] [CrossRef]
- Banaee, M.; Sureda, A.; Mirvaghefi, A.; Ahmadi, K. Effects of diazinon on biochemical parameters of blood in rainbow trout (Oncorhynchus mykiss). Pestic. Biochem. Physiol. 2011, 99, 1–6. [Google Scholar] [CrossRef]
- Alluwaimi, A.M.; Hussein, Y. Diazinon immunotoxicity in mice: Modulation of cytokines level and their gene expression. Toxicology 2007, 236, 123–131. [Google Scholar] [CrossRef]
- Khoshbavar-Rostami, H.; Soltani, M.; Hassan, H. Immune response of great sturgeon (Huso huso) subjected to long-term exposure to sublethal concentration of the organophosphate, diazinon. Aquaculture 2006, 256, 88–94. [Google Scholar] [CrossRef]
- Toledo-Ibarra, G.A.; Rojas-Mayorquín, A.E.; Girón-Pérez, M.I. Influence of the Cholinergic System on the Immune Response of Teleost Fishes: Potential Model in Biomedical Research. Clin. Dev. Immunol. 2013, 2013, 536534. [Google Scholar] [CrossRef]
- Naughton, S.X.; Terry, A.V. Neurotoxicity in acute and repeated organophosphate exposure. Toxicology 2018, 408, 101–112. [Google Scholar] [CrossRef]
- Pavlov, V.A.; Wang, H.; Czura, C.J.; Friedman, S.G.; Tracey, K.J. The Cholinergic Anti-inflammatory Pathway: A Missing Link in Neuroimmunomodulation. Mol. Med. 2003, 9, 125–134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fujii, T.; Mashimo, M.; Moriwaki, Y.; Misawa, H.; Ono, S.; Horiguchi, K.; Kawashima, K. Expression and Function of the Cholinergic System in Immune Cells. Front. Immunol. 2017, 8, 1085. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torrealba, D.; Balasch, J.C.; Criado, M.; Tort, L.; Mackenzie, S.; Roher, N. Functional evidence for the inflammatory reflex in teleosts: A novel α7 nicotinic acetylcholine receptor modulates the macrophage response to dsRNA. Dev. Comp. Immunol. 2018, 84, 279–291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de la Torre, E.; Davel, L.; Jasnis, M.A.; Gotoh, T.; de Lustig, E.S.; Sales, M.E. Muscarinic receptors participation in angiogenic response induced by macrophages from mammary adenocarcinoma-bearing mice. Breast Cancer Res. 2005, 7, R345–R352. [Google Scholar] [CrossRef] [Green Version]
- Kawashima, K. Expression of non-neuronal acetylcholine in lymphocytes and its contribution to the regulation of immune function. Front. Biosci. 2004, 9, 2063–2085. [Google Scholar] [CrossRef] [Green Version]
- Pavlov, V.A.; Ochani, M.; Yang, L.-H.; Gallowitsch-Puerta, M.; Ochani, K.; Lin, X.; Levi, J.; Parrish, W.R.; Rosas-Ballina, M.; Czura, C.J.; et al. Selective α7-nicotinic acetylcholine receptor agonist GTS-21 improves survival in murine endotoxemia and severe sepsis. Crit. Care Med. 2007, 35, 1139–1144. [Google Scholar] [CrossRef]
- Wang, H.; Yu, M.; Ochani, M.; Amella, C.A.; Tanovic, M.; Susarla, S.; Li, J.H.; Wang, H.; Yang, H.; Ulloa, L.; et al. Nicotinic acetylcholine receptor α7 subunit is an essential regulator of inflammation. Nature 2003, 421, 384–388. [Google Scholar] [CrossRef]
- Kostoff, R.N.; Briggs, M.B.; Porter, A.L.; Hernández, A.F.; Abdollahi, M.; Aschner, M.; Tsatsakis, A. The under-reported role of toxic substance exposures in the COVID-19 pandemic. Food Chem. Toxicol. 2020, 145, 111687. [Google Scholar] [CrossRef]
- Galloway, T.; Handy, R. Immunotoxicity of Organophosphorous Pesticides. Ecotoxicology 2003, 12, 345–363. [Google Scholar] [CrossRef]
- Mitra, A.; Sarkar, M.; Chatterjee, C. Modulation of immune response by organophosphate pesticides: Mammals as potential model. Proc. Zoologic. Soc. 2019, 72, 13–24. [Google Scholar] [CrossRef]
- Bakry, N.M.S.; El-Rashidy, A.H.; Eldefrawi, A.T.; Eldefrawi, M.E. Direct actions of organophosphate anticholinesterases on nicotinic and muscarinic acetylcholine receptors. J. Biochem. Toxicol. 1988, 3, 235–259. [Google Scholar] [CrossRef] [PubMed]
- Trailović, S.M.; Marjanović, D.S.; Uzelac, T.V.; Milovanović, M.; Trailović, J.N. Two opposite dose-dependent effects of diazinon on the motor activity of the rat ileum. Res. Vet. Sci. 2017, 112, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Mehrani, H.; Golmanesh, L. Changes in mRNA and protein levels of nicotinic acetylcholine receptors in Diazoxon exposed pC12 cells. Toxicol. In Vitro 2008, 22, 1257–1263. [Google Scholar] [CrossRef] [PubMed]
- Smulders, C.J.G.M.; Bueters, T.J.H.; Vailati, S.; Van Kleef, R.G.D.M.; Vijverberg, H.P.M. Block of Neuronal Nicotinic Acetylcholine Receptors by Organophosphate Insecticides. Toxicol. Sci. 2004, 82, 545–554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Charoenying, T.; Suriyo, T.; Thiantanawat, A.; Chaiyaroj, S.C.; Parkpian, P.; Satayavivad, J. Effects of paraoxon on neuronal and lymphocytic cholinergic systems. Environ. Toxicol. Pharmacol. 2011, 31, 119–128. [Google Scholar] [CrossRef]
- Tarkowski, M.S.; Lutz, W.; Birindelli, S. The lymphocytic cholinergic system and its modulation by organophosphorus pesticides. Int. J. Occup. Med. Environ. Health 2004, 17, 325–337. [Google Scholar]
- Görlach, A.; Bertram, K.; Hudecova, S.; Krizanova, O. Calcium and ROS: A mutual interplay. Redox Biol. 2015, 6, 260–271. [Google Scholar] [CrossRef] [Green Version]
- Adam-Vizi, V.; Starkov, A.A. Calcium and Mitochondrial Reactive Oxygen Species Generation: How to Read the Facts. J. Alzheimer’s Dis. 2010, 20, S413–S426. [Google Scholar] [CrossRef] [Green Version]
- Buccafusco, J.J.; Beach, J.W.; Terry, A. Desensitization of Nicotinic Acetylcholine Receptors as a Strategy for Drug Development. J. Pharmacol. Exp. Ther. 2008, 328, 364–370. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Wu, C.; Tsan, Y.; Hsu, J.; Hung, D.-Z.; Wang, C.-H. Early onset pneumonia in patients with cholinesterase inhibitor poisoning. Respirology 2010, 15, 961–968. [Google Scholar] [CrossRef]
- Faustini, A.; Settimi, L.; Pacifici, R.; Fano, V.; Zuccaro, P.; Forastiere, F. Immunological changes among farmers exposed to phenoxy herbicides: Preliminary observations. Occup. Environ. Med. 1996, 53, 583–585. [Google Scholar] [CrossRef] [PubMed]
- Luebke, B. Pesticide-Induced Immunotoxicity: Are Humans at Risk? Hum. Ecol. Risk Assess. Int. J. 2002, 8, 293–303. [Google Scholar] [CrossRef]
- Dietert, R.R. Developmental immunotoxicology (DIT): Windows of vulnerability, immune dysfunction and safety assessment. J. Immunotoxicol. 2008, 5, 401–412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schäfer, M.; Koppe, F.; Stenger, B.; Brochhausen, C.; Schmidt, A.; Steinritz, D.; Thiermann, H.; Kirkpatrick, C.J.; Pohl, C. Influence of organophosphate poisoning on human dendritic cells. Chem. Interact. 2013, 206, 472–478. [Google Scholar] [CrossRef] [PubMed]
- Xia, C.; Wang, M.; Liang, Q.; Yun, L.; Kang, H.; Fan, L.; Wang, D.; Zhang, G. Changes in monoclonal HLA-DR antigen expression in acute organophosphorus pesticide-poisoned patients. Exp. Ther. Med. 2014, 7, 137–140. [Google Scholar] [CrossRef] [Green Version]
- Costa, C.; Briguglio, G.; Catanoso, R.; Giambò, F.; Polito, I.; Teodoro, M.; Fenga, C. New perspectives on cytokine pathways modulation by pesticide exposure. Curr. Opin. Toxicol. 2020, 19, 99–104. [Google Scholar] [CrossRef]
- Helali, I.; Ferchichi, S.; Maaouia, A.; Aouni, M.; Harizi, H. Modulation of macrophage functionality induced in vitro by chlorpyrifos and carbendazim pesticides. J. Immunotoxicol. 2016, 13, 745–750. [Google Scholar] [CrossRef] [Green Version]
- Li, Q.; Kobayashi, M.; Kawada, T. Organophosphorus pesticides induce apoptosis in human NK cells. Toxicology 2007, 239, 89–95. [Google Scholar] [CrossRef]
- Lasram, M.M.; Lamine, A.J.; Dhouib, I.B.; Bouzid, K.; Annabi, A.; Belhadjhmida, N.; Ben Ahmed, M.; El Fazaa, S.; Abdelmoula, J.; Gharbi, N. Antioxidant and anti-inflammatory effects of N-acetylcysteine against malathion-induced liver damages and immunotoxicity in rats. Life Sci. 2014, 107, 50–58. [Google Scholar] [CrossRef]
- El-Sayed, N.M.; Ahmed, A.A.M.; Selim, M.A.A. Cytotoxic effect of chlorpyrifos is associated with activation of Nrf-2/HO-1 system and inflammatory response in tongue of male Wistar rats. Environ. Sci. Pollut. Res. 2018, 25, 12072–12082. [Google Scholar] [CrossRef] [PubMed]
- Proskocil, B.J.; Grodzki, A.C.G.; Jacoby, D.B.; Lein, P.J.; Fryer, A.D. Organophosphorus Pesticides Induce Cytokine Release from Differentiated Human THP1 Cells. Am. J. Respir. Cell Mol. Biol. 2019, 61, 620–630. [Google Scholar] [CrossRef] [PubMed]
- Kianpour, F.; Mohseni, M.; Beigmohamadi, M.; Yazdinezhad, A.; Ramazani, A.; Hosseini, M.-J.; Sharafi, A. The protective effects of Ziziphora tenuior L. against chlorpyrifos induced toxicity: Involvement of inflammatory and cell death signaling pathways. J. Ethnopharmacol. 2021, 272, 113959. [Google Scholar] [CrossRef]
- Liu, X.; Zhao, X.; Wang, Y.; Hong, J.; Shi, M.; Pfaff, D.; Guo, L.; Tang, H. Triphenyl phosphate permeates the blood brain barrier and induces neurotoxicity in mouse brain. Chemosphere 2020, 252, 126470. [Google Scholar] [CrossRef] [PubMed]
- Ince, S.; Arslan-Acaroz, D.; Demirel, H.H.; Varol, N.; Ozyurek, H.A.; Zemheri, F.; Kucukkurt, I. Taurine alleviates malathion induced lipid peroxidation, oxidative stress, and proinflammatory cytokine gene expressions in rats. Biomed. Pharmacother. 2017, 96, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Weis, G.C.C.; Assmann, C.E.; Mostardeiro, V.B.; Alves, A.D.O.; da Rosa, J.R.; Pillat, M.M.; de Andrade, C.M.; Schetinger, M.R.C.; Morsch, V.M.M.; da Cruz, I.B.M.; et al. Chlorpyrifos pesticide promotes oxidative stress and increases inflammatory states in BV-2 microglial cells: A role in neuroinflammation. Chemosphere 2021, 278, 130417. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, N.; Matsushima, M.; Kawamura, N.; Atsumi, K.; Yamaguchi, T.; Ochi, H.; Kusatsugu, Y.; Oyabu, S.; Hashimoto, N.; Hasegawa, Y.; et al. Modulation of immunological activity on macrophages induced by diazinon. Toxicology 2017, 379, 22–30. [Google Scholar] [CrossRef]
- Tigges, J.; Worek, F.; Thiermann, H.; Wille, T. Organophosphorus pesticides exhibit compound specific effects in rat precision-cut lung slices (PCLS): Mechanisms involved in airway response, cytotoxicity, inflammatory activation and antioxidative defense. Arch. Toxicol. 2021, 96, 321–334. [Google Scholar] [CrossRef]
- Fioranelli, M.; Roccia, M.; Flavin, D.; Cota, L. Regulation of Inflammatory Reaction in Health and Disease. Int. J. Mol. Sci. 2021, 22, 5277. [Google Scholar] [CrossRef]
- Farkhondeh, T.; Mehrpour, O.; Buhrmann, C.; Pourbagher-Shahri, A.M.; Shakibaei, M.; Samarghandian, S. Organophosphorus Compounds and MAPK Signaling Pathways. Int. J. Mol. Sci. 2020, 21, 4258. [Google Scholar] [CrossRef]
- Mostafalou, S.; Eghbal, M.A.; Nili-Ahmadabadi, A.; Baeeri, M.; Abdollahi, M. Biochemical evidence on the potential role of organophosphates in hepatic glucose metabolism toward insulin resistance through inflammatory signaling and free radical pathways. Toxicol. Ind. Health 2012, 28, 840–851. [Google Scholar] [CrossRef]
- Díaz-Resendiz, K.J.G.; Ortiz-Lazareno, P.; Rosales, C.E.C.; Trujillo-Lepe, A.; Toledo-Ibarra, G.; Ventura, H.; Girón-Pérez, M. Effect of diazinon, an organophosphate pesticide, on signal transduction and death induction in mononuclear cells of Nile tilapia fish (Oreochromis niloticus). Fish Shellfish Immunol. 2019, 89, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.K.; Raheja, G.; Gill, K.D. Role of muscarinic signal transduction and CREB phosphorylation in dichlorvos-induced memory deficits in rats: An acetylcholine independent mechanism. Toxicology 2009, 256, 175–182. [Google Scholar] [CrossRef]
- Falkenburger, B.H.; Jensen, J.B.; Hille, B. Kinetics of M1 muscarinic receptor and G protein signaling to phospholipase C in living cells. J. Gen. Physiol. 2010, 135, 81–97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lonze, B.; Ginty, D.D. Function and Regulation of CREB Family Transcription Factors in the Nervous System. Neuron 2002, 35, 605–623. [Google Scholar] [CrossRef] [Green Version]
- Eckel-Mahan, K.; Phan, T.; Han, S.; Wang, H.; Chan, G.C.-K.; Scheiner, Z.S.; Storm, D.R. Circadian oscillation of hippocampal MAPK activity and cAMP: Implications for memory persistence. Nat. Neurosci. 2008, 11, 1074–1082. [Google Scholar] [CrossRef] [PubMed]
- Caughlan, A.; Newhouse, K.; Namgung, U.; Xia, Z. Chlorpyrifos Induces Apoptosis in Rat Cortical Neurons that is Regulated by a Balance Between p38 and ERK/JNK MAP Kinases. Toxicol. Sci. 2004, 78, 125–134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wen, A.Y.; Sakamoto, K.M.; Miller, L.S. The Role of the Transcription Factor CREB in Immune Function. J. Immunol. 2010, 185, 6413–6419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghosh, S.; Hayden, M. New regulators of NF-κB in inflammation. Nat. Rev. Immunol. 2008, 8, 837–848. [Google Scholar] [CrossRef]
- Medzhitov, R.; Horng, T. Transcriptional control of the inflammatory response. Nat. Rev. Immunol. 2009, 9, 692–703. [Google Scholar] [CrossRef]
- Medzhitov, R. The spectrum of inflammatory responses. Science 2021, 374, 1070–1075. [Google Scholar] [CrossRef]
- Esquivel-Sentíes, M.; Barrera, I.; Ortega, A.; Vega, L. Organophosphorous pesticide metabolite (DEDTP) induces changes in the activation status of human lymphocytes by modulating the interleukin 2 receptor signal transduction pathway. Toxicol. Appl. Pharmacol. 2010, 248, 122–133. [Google Scholar] [CrossRef] [PubMed]
- Dimitriou, I.D.; Clemenza, L.; Scotter, A.J.; Chen, G.; Guerra, F.M.; Rottapel, R. Putting out the fire: Coordinated suppression of the innate and adaptive immune systems by SOCS1 and SOCS3 proteins. Immunol. Rev. 2008, 224, 265–283. [Google Scholar] [CrossRef] [PubMed]
- Cohney, S.J.; Sanden, D.; Cacalano, N.A.; Yoshimura, A.; Mui, A.; Migone, T.S.; Johnston, J.A. SOCS-3 Is Tyrosine Phosphorylated in Response to Interleukin-2 and Suppresses STAT5 Phosphorylation and Lymphocyte Proliferation. Mol. Cell. Biol. 1999, 19, 4980–4988. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magnarelli, G.; Fonovich, T. Protein phosphorylation pathways disruption by pesticides. Adv. Biol. Chem. 2013, 03, 460–474. [Google Scholar] [CrossRef] [Green Version]
- Lima, A.; Vega, L. Methyl-parathion and organophosphorous pesticide metabolites modify the activation status and interleukin-2 secretion of human peripheral blood mononuclear cells. Toxicol. Lett. 2005, 158, 30–38. [Google Scholar] [CrossRef]
- Malek, T.R. The Biology of Interleukin-2. Annu. Rev. Immunol. 2008, 26, 453–479. [Google Scholar] [CrossRef]
- Costa, L.G. Organophosphorus Compounds at 80: Some Old and New Issues. Toxicol. Sci. 2018, 162, 24–35. [Google Scholar] [CrossRef] [Green Version]
- Eddleston, M.; Buckley, N.; Eyer, P.; Dawson, A. Management of acute organophosphorus pesticide poisoning. Lancet 2008, 371, 597–607. [Google Scholar] [CrossRef] [Green Version]
- Hulse, E.J.; Davies, J.; Simpson, A.J.; Sciuto, A.M.; Eddleston, M. Respiratory Complications of Organophosphorus Nerve Agent and Insecticide Poisoning. Implications for Respiratory and Critical Care. Am. J. Respir. Crit. Care Med. 2014, 190, 1342–1354. [Google Scholar] [CrossRef] [Green Version]
- Kaushal, J.; Khatri, M.; Arya, S.K. A treatise on Organophosphate pesticide pollution: Current strategies and advancements in their environmental degradation and elimination. Ecotoxicol. Environ. Saf. 2021, 207, 111483. [Google Scholar] [CrossRef]
- Bird, S.B.; Gaspari, R.; Dickson, E.W. Early Death Due to Severe Organophosphate Poisoning Is a Centrally Mediated Process. Acad. Emerg. Med. 2003, 10, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Roldan-Tapia, M.; Nieto-Escamez, F.A.; del Águila, E.; Laynez, F.; Parron, T.; Sanchez-Santed, F. Neuropsychological sequelae from acute poisoning and long-term exposure to carbamate and organophosphate pesticides. Neurotoxicol. Teratol. 2006, 28, 694–703. [Google Scholar] [CrossRef] [PubMed]
- Dassanayake, T.; Weerasinghe, V.; Dangahadeniya, U.; Kularatne, K.; Dawson, A.; Karalliedde, L.; Senanayake, N. Cognitive processing of visual stimuli in patients with organophosphate insecticide poisoning. Neurology 2007, 68, 2027–2030. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y. Organophosphate-induced brain damage: Mechanisms, neuropsychiatric and neurological consequences, and potential therapeutic strategies. NeuroToxicology 2012, 33, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Furtado, M.D.A.; Rossetti, F.; Chanda, S.; Yourick, D. Exposure to nerve agents: From status epilepticus to neuroinflammation, brain damage, neurogenesis and epilepsy. NeuroToxicology 2012, 33, 1476–1490. [Google Scholar] [CrossRef]
- Pereira, E.F.R.; Aracava, Y.; DeTolla, L.J.; Beecham, E.J.; Basinger, G.W.; Wakayama, E.J.; Albuquerque, E.X. Animal Models That Best Reproduce the Clinical Manifestations of Human Intoxication with Organophosphorus Compounds. J. Pharmacol. Exp. Ther. 2014, 350, 313–321. [Google Scholar] [CrossRef] [Green Version]
- Xanthos, D.N.; Sandkühler, J. Neurogenic neuroinflammation: Inflammatory CNS reactions in response to neuronal activity. Nat. Rev. Neurosci. 2014, 15, 43–53. [Google Scholar] [CrossRef]
- Olas, B.; Wachowicz, B. Resveratrol and vitamin C as antioxidants in blood platelets. Thromb. Res. 2002, 106, 143–148. [Google Scholar] [CrossRef]
- Zaidi, S.R.; Banu, N. Antioxidant potential of vitamins A, E and C in modulating oxidative stress in rat brain. Clin. Chim. Acta 2004, 340, 229–233. [Google Scholar] [CrossRef]
- Ojha, A.; Srivastava, N. Redox imbalance in rat tissues exposed with organophosphate pesticides and therapeutic potential of antioxidant vitamins. Ecotoxicol. Environ. Saf. 2012, 75, 230–241. [Google Scholar] [CrossRef]
- Deshpande, L.; Blair, R.E.; Huang, B.A.; Phillips, K.F.; DeLorenzo, R.J. Pharmacological blockade of the calcium plateau provides neuroprotection following organophosphate paraoxon induced status epilepticus in rats. Neurotoxicol. Teratol. 2016, 56, 81–86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Ebiary, A.A.; Elsharkawy, R.E.; Soliman, N.A.; Soliman, M.A.; Hashem, A.A. N-acetylcysteine in Acute Organophosphorus Pesticide Poisoning: A Randomized, Clinical Trial. Basic Clin. Pharmacol. Toxicol. 2016, 119, 222–227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fang, J.; She, J.; Lin, F.; Wu, J.-C.; Han, R.; Sheng, R.; Wang, G.; Qin, Z.-H. RRx-001 Exerts Neuroprotection Against LPS-Induced Microglia Activation and Neuroinflammation Through Disturbing the TLR4 Pathway. Front. Pharmacol. 2022, 13, 1219. [Google Scholar] [CrossRef]
- Lin, C.-Y.; Chiang, C.-Y.; Tsai, H.-J. Zebrafish and Medaka: New model organisms for modern biomedical research. J. Biomed. Sci. 2016, 23, 19. [Google Scholar] [CrossRef] [Green Version]
- Wilson, A.B. MHC and adaptive immunity in teleost fishes. Immunogenetics 2017, 69, 521–528. [Google Scholar] [CrossRef]
- Saha, A.; Ghosh, R.K.; Jesna, P.-K.; Choudhury, P.P. Bioindicators of Pesticide Contaminations. Sustain. Agric. Rev. 2021, 48, 185–231. [Google Scholar] [CrossRef]
- Medina-Garza, H. Uso de Biomarcadores en Peces Como Herramienta Para Evaluar la Exposición y Efecto de Contaminantes Ambientales en Cuerpos de Agua. Master’s Thesis, Universidad Autonoma de San Luis Potosí, México, December 2012. [Google Scholar]
- Gonçalves, C.; Marins, A.T.; Amaral, A.M.B.D.; Nunes, M.E.M.; Müller, T.E.; Severo, E.; Feijó, A.; Rodrigues, C.C.; Zanella, R.; Prestes, O.D.; et al. Ecological impacts of pesticides on Astyanax jacuhiensis (Characiformes: Characidae) from the Uruguay river, Brazil. Ecotoxicol. Environ. Saf. 2020, 205, 111314. [Google Scholar] [CrossRef]
- Girón-Pérez, M.I.; Barcelós-García, R.; Vidal-Chavez, Z.G.; Romero-Bañuelos, C.A.; Robledo-Marenco, M.L. Effect of Chlorpyrifos on the Hematology and Phagocytic Activity of Nile Tilapia Cells (Oreochromis niloticus). Toxicol. Mech. Methods 2006, 16, 495–499. [Google Scholar] [CrossRef]
- Girón-Pérez, M.I.; Santerre, A.; Gonzalez-Jaime, F.; Casas-Solis, J.; Hernández-Coronado, M.; Peregrina-Sandoval, J.; Takemura, A.; Zaitseva, G. Immunotoxicity and hepatic function evaluation in Nile tilapia (Oreochromis niloticus) exposed to diazinon. Fish Shellfish Immunol. 2007, 23, 760–769. [Google Scholar] [CrossRef]
- Covantes-Rosales, C.E.; Trujillo-Lepe, A.; Díaz-Resendiz, K.J.G.; Toledo-Ibarra, G.; Ventura, H.; Ortiz-Lazareno, P.; Girón-Pérez, M. Phagocytosis and ROS production as biomarkers in Nile tilapia (Oreochromis niloticus) leukocytes by exposure to organophosphorus pesticides. Fish Shellfish Immunol. 2019, 84, 189–195. [Google Scholar] [CrossRef]
- Díaz-Resendiz, K.G.J.; Girón-Pérez, M.I. Effect of chlorpyrifos on the immune response of Nile tilapia (Oreochromis niloticus). Rev. BioCiencias. 2014, 3, 59–64. [Google Scholar] [CrossRef]
- Toledo-Ibarra, G.A.; Díaz-Resendiz, K.J.G.; Ventura-Ramón, G.H.; González-Jaime, F.; Vega-López, A.; Becerril-Villanueva, E.; Pavón, L.; Girón-Pérez, M.I. Oxidative damage in gills and liver in Nile tilapia (Oreochromis niloticus) exposed to diazinon. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2016, 200, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Covantes-Rosales, C.E.; Toledo-Ibarra, G.A.; González-Navarro, I.; Agraz-Cibrian, J.M.; Girón-Pérez, D.A.; Ventura-Ramon, G.H.; Diaz-Resendiz, K.J.G.; Bueno-Durán, A.Y.; Ponce-Regalado, M.D.; Girón-Pérez, M.I. Diazinon acute exposure induces neutrophil extracellular traps in Nile tilapia (Oreochromis niloticus). Food Agric. Immunol. 2020, 31, 1004–1013. [Google Scholar] [CrossRef]
- Díaz-Resendiz, K.J.G.; Bernal-Ortega, J.; Covantes-Rosales, C.E.; Ortiz-Lazareno, P.C.; Toledo-Ibarra, G.A.; Ventura-Ramon, G.H.; Girón-Pérez, M.I. In-vitro effect of diazoxon, a metabolite of diazinon, on proliferation, signal transduction, and death induction in mononuclear cells of Nile tilapia fish (Oreochromis niloticus). Fish Shellfish Immunol. 2020, 105, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Covantes-Rosales, C.; Toledo-Ibarra, G.; Díaz-Resendíz, K.; Ventura-Ramón, G.; Girón-Pérez, M. Muscarinic acetylcholine receptor expression in brain and immune cells of Oreochromis niloticus. J. Neuroimmunol. 2019, 328, 105–107. [Google Scholar] [CrossRef]
- Toledo-Ibarra, G.A.; Rodríguez-Sánchez, E.J.; Ventura-Ramón, H.G.; Resendiz, K.J.G.D.; Giron, M.I. Cholinergic alterations by exposure to pesticides used in control vector: Guppies fish (Poecilia reticulta) as biological model. Int. J. Environ. Health Res. 2017, 28, 79–89. [Google Scholar] [CrossRef]
- Girón-Pérez, D.A.; Hermosillo-Escobedo, A.T.; Macias-Garrigos, K.; Díaz-Resendiz, K.J.G.; Toledo-Ibarra, G.A.; Ventura-Ramón, G.H.; Girón-Pérez, M.I. Altered phagocytic capacity due to acute exposure and long-term post-exposure to pesticides used for vector-borne disease as dengue. Int. J. Environ. Health Res. 2020, 32, 455–462. [Google Scholar] [CrossRef]
- Díaz-Resendiz, K.J.G.; Hermosillo-Escobedo, A.T.; Ventura-Ramón, G.H.; Toledo-Ibarra, G.A.; Girón-Pérez, D.A.; Bueno-Durán, A.Y.; Girón-Pérez, M.I. Death of guppy fish (Poecilia reticulata) leukocytes induced by in vivo exposure to temephos and spinosad. Int. J. Environ. Health Res. 2020, 32, 701–711. [Google Scholar] [CrossRef]



| OPs | Dose | Exposure Time | Cholinergic Effects | Tissue/Cell Line | Organism Model | References |
|---|---|---|---|---|---|---|
| Diazinon | LC50-7.830 ppm, ½ LC50-3.915 ppm | 96 h | ↓ AChE activity ↑ ACh levels | Spleen mononuclear cells | Nile tilapia (Oreochromis niloticus) | [22]. |
| Diazinon | 0.97, 1.95 and 3.91 mg/L | 6, 12, and 24 h | ↓ AChE activity ↓ mAChR, nAChR concentration and ↑ ACh levels. | Spleen mononuclear cells | Nile tilapia (O. niloticus) | [23]. |
| Diazoxon | 1 nm, 1 µM, and 10 µM | 24 h | ↓ (M3, M4, M5) receptors and nAChR β2 expression. | Spleen mononuclear cells | Nile tilapia (O. niloticus) | [24]. |
| Paraoxon | 1 mg/kg | 6 and 24 h | ↓ mAChR M2 function ↑ ACh levels. ↑ mAChR M3 stimulation | Peripheral blood | Guinea Pig | [25]. |
| Chlorpyrifos | LD50 1/3 LD50 | 48 h | ↓ ChAT activity ↓ AChE activity | Cerebral cortex | Male Rat | [26]. |
| Chlorpyrifos | 1 mg/Kg | 1 h and 6 h | ↓ ChAT activity, nAChR α4, and α7 expression ↓ VAChT expression | Forebrain Peripheral blood | Human apoE-TR mice | [27]. |
| Monocrotophos | 0.01, 0.10, or 1.00 mg/L | N/A | ↓ ChAT activity ↓ AChE activity | Embryos | Sea urchin (Hemicentrotus pulcherrimus) | [28]. |
| OPs | Acute exposure | N/A | ↓ BuChE activity | Peripheral blood | Human | [29]. |
| OPs | Dose | Exposure Time | Effects of Cytokines | Inflammation Results | Organism Model | References |
|---|---|---|---|---|---|---|
| Chlorpyrifos, dimethoate | 0–1000 μM | 24 h | IL-10 was significantly downregulated | ↓ DC-specific cell surface markers (i.e., CD83 and CD209). Inhibition of Akt family | DC, differentiated from the monocyte cell line THP-1 | [90]. |
| Chlorpyrifos | 0, 001, 10 μM | 24 h | ↓ Expression of IL-1β and TNF-α | Biphasic responses of lysosomal enzyme activity. inhibition NO release | Macrophages from mouse peritoneum | [93]. |
| Malathion | 200 mg/kg b.w./day | 28 days | ↑ Expression of IL-1β, IL-6 and IFN-γ | ↑ Activities of hepatocellular enzymes in plasma, lipid peroxidation index, CD3+/CD4+ and CD3+/CD4+ percent | Adult male Wistar rats | [95]. |
| chlorpyrifos | 3.375–13.5 mg/kg | 28 days | ↑ Expression of IL-1β and TNF-α | ↑ Activation of NF-kB, cleaved caspase 3 and HO-1 and Nrf-2 pathway Cellular damage in organs | Male Wistar rats | [96]. |
| Parathion, chlorpyrifos, and diazinon | 1–100 μM | 24 h | ↑ Expression of TNF-α, IL-1β PDGF (platelet-derived growth factor) and TGF-β (transforming growth factor-β). the of TNF-α protein. | ↑ NF-κB activation and ↓AChE activity | THP1 cells differentiated into macrophages | [97]. |
| Chlorpyrifos | 6.75 mg/kg | 8 weeks | ↑ Expression of IL-6, TLR-2, IL-1β, TNF-α, and NLPR3 | ↑ Expression of apoptotic genes (Caspase 3, Caspase 9, Caspase 8 and Bax) | Male rats | [98]. |
| Triphenyl phosphate | 0, 50, or 150 mg/kg | 30 days | ↑ Expression of IL-6 and TNF-α | ↑ Inflammation in the thalamus and hippocampus. MAPK signaling pathways were significantly affected. | Male mice (C57/BL6) | [99]. |
| Malathion | 27 mg/kg (1/50 of LD50) | 30 days | ↑ Expression of IF-γ, IL1-β, TNF-α, and NFĸB | ↓AChE levels in serum (30%) and liver (25%) compared to the control group. Lipid peroxidation. | Rats | [100]. |
| Chlorpyrifos | 0.3–300 μM | 24 h | ↑ Expression of IL-1β and NLRP3 | ↑ Oxidative stress production (NO, MDA, and O2∙) | BV-2 microglial cells. | [101]. |
| Diazinon | 10–100 μM | 24 h | Induce expression of TNF-α and IL-6 | ↑ ROS generation. Induced expressions of COX-2, iNOS, and cell-surface molecules CD40, CD86, and MHC class II. ↓phagocytic activity | RAW264.7 cells | [102]. |
| Parathion, Malathion, paraoxon and malaoxon | 100–2000 µmol/L | 24 h | ↑ Expression of IL-6, GM-CSF and MIP-1α | ↓Viability, intracellular GSH and phosphorylation of STAT3. ↑Phosphorylated p38MAPK | Rat precision-cut lung slices | [103]. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Camacho-Pérez, M.R.; Covantes-Rosales, C.E.; Toledo-Ibarra, G.A.; Mercado-Salgado, U.; Ponce-Regalado, M.D.; Díaz-Resendiz, K.J.G.; Girón-Pérez, M.I. Organophosphorus Pesticides as Modulating Substances of Inflammation through the Cholinergic Pathway. Int. J. Mol. Sci. 2022, 23, 4523. https://doi.org/10.3390/ijms23094523
Camacho-Pérez MR, Covantes-Rosales CE, Toledo-Ibarra GA, Mercado-Salgado U, Ponce-Regalado MD, Díaz-Resendiz KJG, Girón-Pérez MI. Organophosphorus Pesticides as Modulating Substances of Inflammation through the Cholinergic Pathway. International Journal of Molecular Sciences. 2022; 23(9):4523. https://doi.org/10.3390/ijms23094523
Chicago/Turabian StyleCamacho-Pérez, Milton Rafael, Carlos Eduardo Covantes-Rosales, Gladys Alejandra Toledo-Ibarra, Ulises Mercado-Salgado, María Dolores Ponce-Regalado, Karina Janice Guadalupe Díaz-Resendiz, and Manuel Iván Girón-Pérez. 2022. "Organophosphorus Pesticides as Modulating Substances of Inflammation through the Cholinergic Pathway" International Journal of Molecular Sciences 23, no. 9: 4523. https://doi.org/10.3390/ijms23094523