Primary Liver Cancer
A topical collection in Cancers (ISSN 2072-6694). This collection belongs to the section "Methods and Technologies Development".
Viewed by 30580Editor
Interests: hepatocellular carcinoma; immune checkpoint inhibitors; molecular-targeted agents; radiofrequency ablation; systemic therapy; trans-arterial chemoembolization; bile duct cancer; pancreatic cancer
Topical Collection Information
Dear Colleagues,
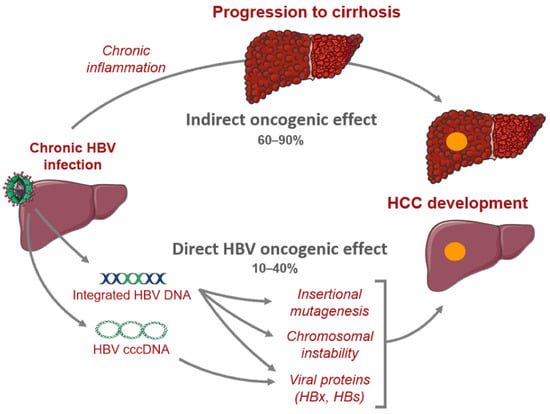
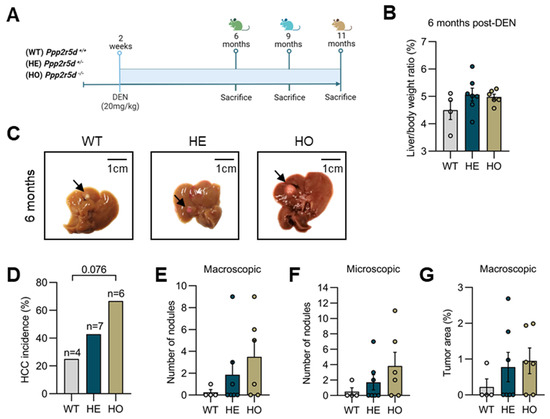
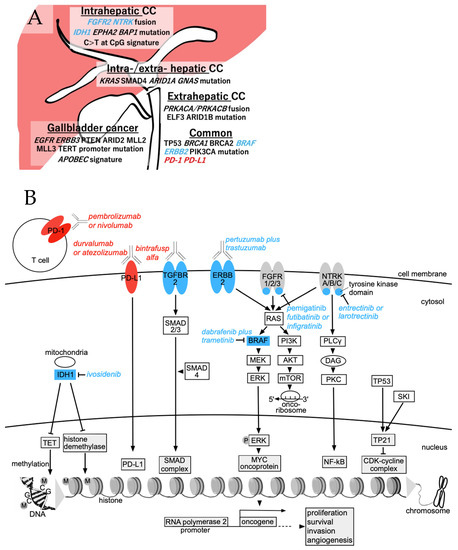
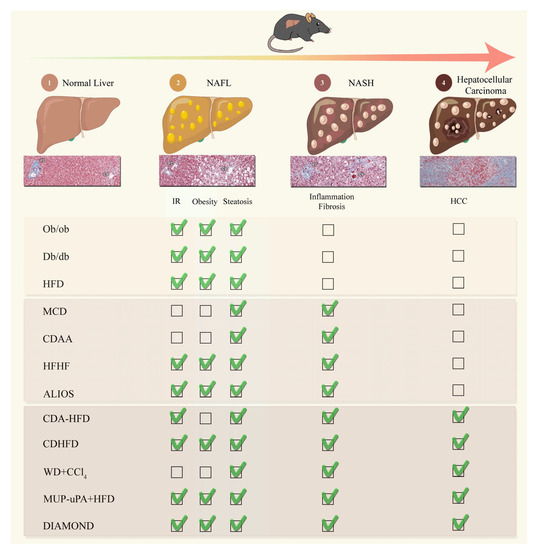
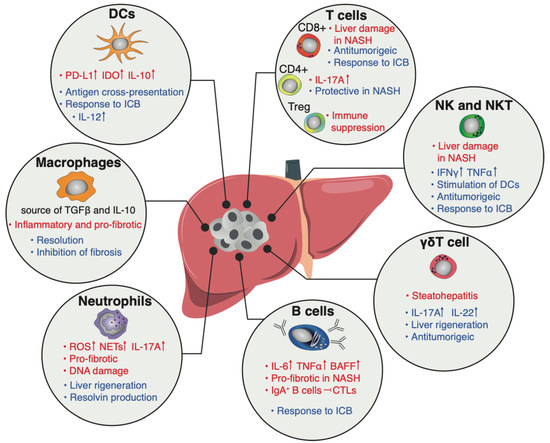
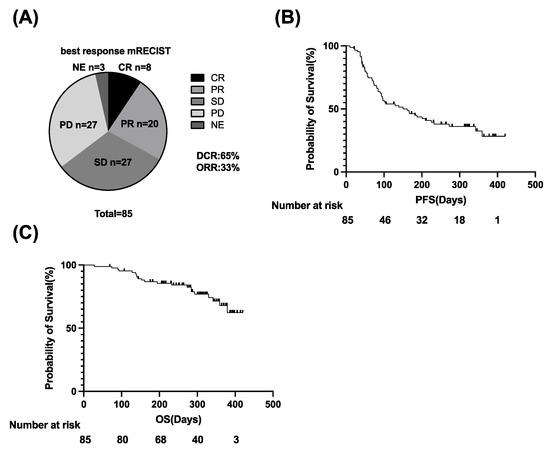
Primary liver cancer is the second most common cause of cancer deaths globally; hepatocellular carcinoma (HCC) and intrahepatic cholangiocarcinoma (iCCA) are the two main histological subtypes. In HCC, potentially curative treatments, such as surgical resection, transplantation or ablation, are basically indicated in patients at the early stage, chemoembolization at the intermediate stage, and systemic therapies at the advanced stage. Molecular-targeted agents, such as the multi-kinase inhibitors sorafenib and lenvatinib, are used as first-line treatments, immunotherapy with atezolizumab plus bevacizumab have been approved more recently, and many more agents as second-line treatments. In iCCA, surgery is the only possible curative treatment. Patients have limited treatment options if worsens after systemic chemotherapy. However, precision medicines, such as the fibroblast growth factor receptor 1–3 inhibitor pemigatinib, have been recently licensed for patients with driver gene mutations.
We are pleased to invite you to contribute with your valuable work to this Special Issue on “Primary Liver Cancer”.
This Special Issue aims to collect papers focused on the recent advance in diagnosis and treatment of primary liver cancer.
In this Special Issue, original research articles and reviews are welcome. Research areas may include (but are not limited to) the following:
- Diagnosis (including biomarkers and imaging techniques)
- Treatment (including surgery, ablation, IVR and systemic treatments)
I look forward to receiving your contributions.
Dr. Masaru Enomoto
Guest Editor
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 100 words) can be sent to the Editorial Office for announcement on this website.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Cancers is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2900 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
Keywords
- primary liver cancer
- hepatobiliary cancer
- hepatocellular carcinoma
- cholangiocarcinoma