Breast Cancer: From Pathophysiology to Prevention and Treatment
(Closed)
A topical collection in Cancers (ISSN 2072-6694). This collection belongs to the section "Cancer Therapy".
Viewed by 34778
Share This Topical Collection
Editors
 Prof. Dr. Debra A. Tonetti
Prof. Dr. Debra A. Tonetti
 Prof. Dr. Debra A. Tonetti
Prof. Dr. Debra A. Tonetti
E-Mail
Website
Co-Guest Editor
Department of Biopharmaceutical Sciences, the University of Illinois at Chicago, Chicago, IL 60612, USA
Interests: pharmacology and biology of breast cancer including mechanisms of endocrine-resistance including protein kinase C (PKC) signaling as a mechanism of tamoxifen-resistant breast cancer; breast cancer and lung cancer Patient-Derived Xenografts (PDXs); development of the Selective human Estrogen Receptor Agonists (ShERPAs) TTC-352 in Phase I Clinical Trial for metastatic breast cancer
Topical Collection Information
Dear Colleagues,
Breast cancer continues to be the most commonly diagnosed cancer and the second leading cause of cancer deaths among women. It is subdivided into major subtypes based on the expression of the estrogen receptor (ER) or progesterone receptor (PR) or human epidermal growth factor receptor 2 (HER2). Traditionally, treatment strategies are mainly based on the presence or absence of these markers. Lack of these markers defines Triple Negative Breast Cancer (TNBC). However, the recent successes of novel antibody-drug conjugates (ADCs) in breast cancer are changing the biological landscape of breast cancer subtypes and treatment strategies. Furthermore, about a half of HER2-negative breast cancer show lower levels of HER2 expression (HER2-low) that can be candidates for new antibody-drug conjugate treatments. Trastuzumab deruxtecan demonstrates activity not only in HER2-positive, but also in HER2-low breast cancer patients. This Special Issue will discuss the evolving landscape of breast cancer subtypes and focus on our understanding of the pathophysiology, early detection and practice changing treatments in breast cancer.
We aim to stimulate discussion by bringing together expert opinion and new science from across the field in this Special Issue of Cancers. We welcome submissions that cover any relevant topic, including breast cancer subtypes, chemoprevention, prognostic/predictive markers, novel treatment strategies in breast cancer, CDK4/6 inhibitors, novel antibody-drug conjugates (ADCs) for treatment, and immune checkpoint inhibitors.
Dr. Yesim Gokmen-Polar
Prof. Dr. Debra A. Tonetti
Guest Editors
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 100 words) can be sent to the Editorial Office for announcement on this website.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Cancers is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript.
The Article Processing Charge (APC) for publication in this open access journal is 2900 CHF (Swiss Francs).
Submitted papers should be well formatted and use good English. Authors may use MDPI's
English editing service prior to publication or during author revisions.
Keywords
- breast cancer pathobiology
- breast cancer subtypes
- chemoprevention
- prognostic/predictive markers
- novel treatment strategies in breast cancer
- CDK4/6 inhibitors
- novel antibody-drug conjugates (ADCs)
- immune checkpoint inhibitors
Published Papers (15 papers)
Open AccessReview
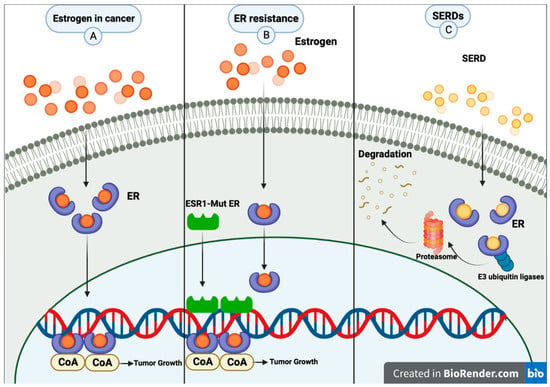
Oral SERD, a Novel Endocrine Therapy for Estrogen Receptor-Positive Breast Cancer
by
Niraj Neupane, Sawyer Bawek, Sayuri Gurusinghe, Elham Moases Ghaffary, Omid Mirmosayyeb, Sangharsha Thapa, Carla Falkson, Ruth O’Regan and Ajay Dhakal
Viewed by 1466
Abstract
Breast cancer is the most common cancer among women worldwide, and estrogen receptor-positive (ER+) breast cancer accounts for a significant proportion of cases. While various treatments are available, endocrine therapies are often the first-line treatment for this type of breast cancer. However, the
[...] Read more.
Breast cancer is the most common cancer among women worldwide, and estrogen receptor-positive (ER+) breast cancer accounts for a significant proportion of cases. While various treatments are available, endocrine therapies are often the first-line treatment for this type of breast cancer. However, the development of drug resistance poses a significant challenge in managing this disease.
ESR1 mutations have been identified as a common mechanism of endocrine therapy resistance in ER+ breast cancer. The first-generation selective estrogen receptor degrader (SERD) fulvestrant has shown some activity against
ESR1 mutant tumors. However, due to its poor bioavailability and need for intramuscular injection, it may not be the optimal therapy for patients. Second-generation SERDs were developed to overcome these limitations. These newer drugs have improved oral bioavailability and pharmacokinetics, making them more convenient and effective for patients. Several oral SERDs are now in phase III trials for early and advanced ER+ breast cancer. This review summarizes the background of oral SERD development, the current status, and future perspectives.
Full article
►▼
Show Figures
Open AccessReview
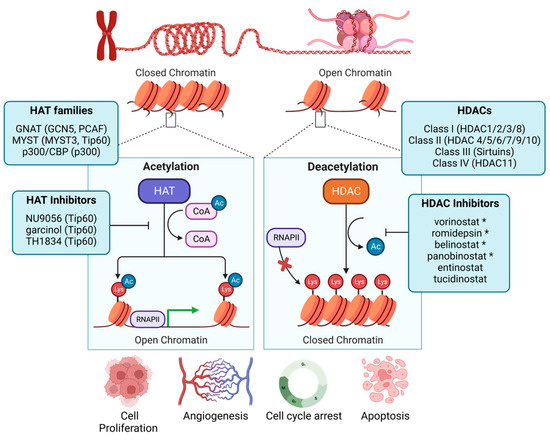
Emerging Role of Epigenetic Modifiers in Breast Cancer Pathogenesis and Therapeutic Response
by
Richard Sean Lee, Kirti Sad, Dorelle V. Fawwal and Jennifer Marie Spangle
Cited by 4 | Viewed by 1696
Abstract
Breast cancer pathogenesis, treatment, and patient outcomes are shaped by tumor-intrinsic genomic alterations that divide breast tumors into molecular subtypes. These molecular subtypes often dictate viable therapeutic interventions and, ultimately, patient outcomes. However, heterogeneity in therapeutic response may be a result of underlying
[...] Read more.
Breast cancer pathogenesis, treatment, and patient outcomes are shaped by tumor-intrinsic genomic alterations that divide breast tumors into molecular subtypes. These molecular subtypes often dictate viable therapeutic interventions and, ultimately, patient outcomes. However, heterogeneity in therapeutic response may be a result of underlying epigenetic features that may further stratify breast cancer patient outcomes. In this review, we examine non-genetic mechanisms that drive functional changes to chromatin in breast cancer to contribute to cell and tumor fitness and highlight how epigenetic activity may inform the therapeutic response. We conclude by providing perspectives on the future of therapeutic targeting of epigenetic enzymes, an approach that holds untapped potential to improve breast cancer patient outcomes.
Full article
►▼
Show Figures
Open AccessArticle
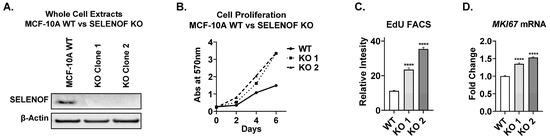
SELENOF Controls Proliferation and Cell Death in Breast-Derived Immortalized and Cancer Cells
by
Roudy C. Ekyalongo, Brenna Flowers, Tanu Sharma, Alexandra Zigrossi, An Zhang, Anaisa Quintanilla-Arteaga, Kanishka Singh and Irida Kastrati
Viewed by 1107
Abstract
SELENOF expression is significantly lower in aggressive breast tumors compared to normal tissue, indicating that its reduction or loss may drive breast tumorigenesis. Deletion of SELENOF in non-tumorigenic immortalized breast epithelial MCF-10A cells resulted in enhanced proliferation, both in adherent culture and matrix-assisted
[...] Read more.
SELENOF expression is significantly lower in aggressive breast tumors compared to normal tissue, indicating that its reduction or loss may drive breast tumorigenesis. Deletion of SELENOF in non-tumorigenic immortalized breast epithelial MCF-10A cells resulted in enhanced proliferation, both in adherent culture and matrix-assisted three-dimmensional (3D) growth. Modulation of SELENOF in vitro through deletion or overexpression corresponded to changes in the cell-cycle regulators p21 and p27, which is consistent with breast tumor expression data from the METABRIC patient database. Together, these findings indicate that SELENOF affects both proliferation and cell death in normal epithelial and breast cancer cells, largely through the regulation of p21 and p27. In glandular cancers like breast cancer, the filling of luminal space is one of the hallmarks of early tumorigenesis. Loss of SELENOF abrogated apoptosis and autophagy, which are required for the formation of hollow acini in MCF-10A cells in matrix-assisted 3D growth, resulting in luminal filling. Conversely, overexpression of SELENOF induced cell death via apoptosis and autophagy. In conclusion, these findings are consistent with the notion that SELENOF is a breast tumor suppressor, and its loss contributes to breast cancer etiology.
Full article
►▼
Show Figures
Open AccessReview
Revisiting Estrogen for the Treatment of Endocrine-Resistant Breast Cancer: Novel Therapeutic Approaches
by
Nivida Shete, Jordan Calabrese and Debra A. Tonetti
Cited by 2 | Viewed by 1679
Abstract
Estrogen receptor (ER)-positive breast cancer is the most common subtype, representing 70–75% of all breast cancers. Several ER-targeted drugs commonly used include the selective estrogen receptor modulator (SERM), tamoxifen (TAM), aromatase inhibitors (AIs) and selective estrogen receptor degraders (SERDs). Through different mechanisms of
[...] Read more.
Estrogen receptor (ER)-positive breast cancer is the most common subtype, representing 70–75% of all breast cancers. Several ER-targeted drugs commonly used include the selective estrogen receptor modulator (SERM), tamoxifen (TAM), aromatase inhibitors (AIs) and selective estrogen receptor degraders (SERDs). Through different mechanisms of action, all three drug classes reduce estrogen receptor signaling. Inevitably, resistance occurs, resulting in disease progression. The counterintuitive action of estrogen to inhibit ER-positive breast cancer was first observed over 80 years ago. High-dose estrogen and diethylstilbestrol (DES) were used to treat metastatic breast cancer accompanied by harsh side effects until the approval of TAM in the 1970s. After the development of TAM, randomized trials comparing TAM to estrogen found similar or slightly inferior efficacy but much better tolerability. After decades of research, it was learned that estrogen induces tumor regression only after a period of long-term estrogen deprivation, and the mechanisms of tumor regression were described. Despite the long history of breast cancer treatment with estrogen, this therapeutic modality is now revitalized due to the development of novel estrogenic compounds with improved side effect profiles, newly discovered predictive biomarkers, the development of non-estrogen small molecules and new combination therapeutic approaches.
Full article
►▼
Show Figures
Open AccessArticle
Taxifolin Inhibits Breast Cancer Growth by Facilitating CD8+ T Cell Infiltration and Inducing a Novel Set of Genes including Potential Tumor Suppressor Genes in 1q21.3
by
Xiaozeng Lin, Ying Dong, Yan Gu, Anil Kapoor, Jingyi Peng, Yingying Su, Fengxiang Wei, Yanjun Wang, Chengzhi Yang, Armaan Gill, Sandra Vega Neira and Damu Tang
Cited by 2 | Viewed by 1414
Abstract
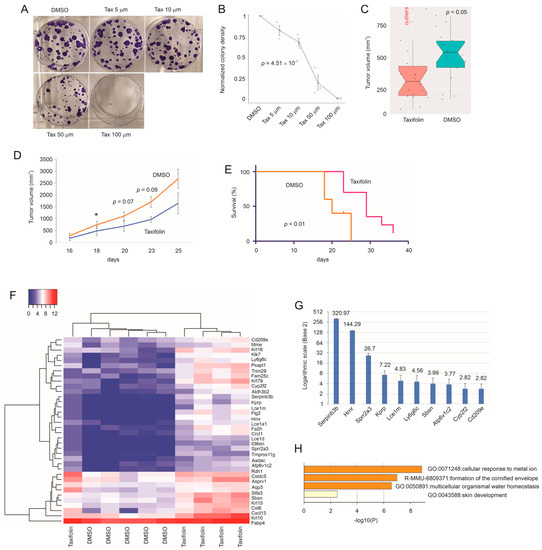
Taxifolin inhibits breast cancer (BC) via novel mechanisms. In a syngeneic mouse BC model, taxifolin suppressed 4T-1 cell-derived allografts. RNA-seq of 4T-1 tumors identified 36 differentially expressed genes (DEGs) upregulated by taxifolin. Among their human homologues, 19, 7, and 2 genes were downregulated
[...] Read more.
Taxifolin inhibits breast cancer (BC) via novel mechanisms. In a syngeneic mouse BC model, taxifolin suppressed 4T-1 cell-derived allografts. RNA-seq of 4T-1 tumors identified 36 differentially expressed genes (DEGs) upregulated by taxifolin. Among their human homologues, 19, 7, and 2 genes were downregulated in BCs, high-proliferative BCs, and BCs with high-fatality risks, respectively. Three genes were established as tumor suppressors and eight were novel to BC, including
HNRN,
KPRP,
CRCT1, and
FLG2. These four genes exhibit tumor suppressive actions and reside in 1q21.3, a locus amplified in 70% recurrent BCs, revealing a unique vulnerability of primary and recurrent BCs with 1q21.3 amplification with respect to taxifolin. Furthermore, the 36 DEGs formed a multiple gene panel (DEG36) that effectively stratified the fatality risk in luminal, HER2+, and triple-negative (TN) equivalent BCs in two large cohorts: the METABRIC and TCGA datasets. 4T-1 cells model human TNBC cells. The DEG36 most robustly predicted the poor prognosis of TNBCs and associated it with the infiltration of CD8+ T, NK, macrophages, and Th2 cells. Of note, taxifolin increased the CD8+ T cell content in 4T-1 tumors. The DEG36 is a novel and effective prognostic biomarker of BCs, particularly TNBCs, and can be used to assess the BC-associated immunosuppressive microenvironment.
Full article
►▼
Show Figures
Open AccessReview
The Challenges of Treating Patients with Breast Cancer and Obesity
by
Alexis LeVee and Joanne Mortimer
Cited by 4 | Viewed by 1865
Abstract
Obesity is defined as a body mass index (BMI) of 30 kg/m
2 or more and is associated with worse outcomes in patients with breast cancer, resulting in an increased incidence of breast cancer, recurrence, and death. The incidence of obesity is increasing,
[...] Read more.
Obesity is defined as a body mass index (BMI) of 30 kg/m
2 or more and is associated with worse outcomes in patients with breast cancer, resulting in an increased incidence of breast cancer, recurrence, and death. The incidence of obesity is increasing, with almost half of all individuals in the United States classified as obese. Patients with obesity present with unique pharmacokinetics and physiology and are at increased risk of developing diabetes mellitus and cardiovascular disease, which leads to specific challenges when treating these patients. The aim of this review is to summarize the impact of obesity on the efficacy and toxicity of systemic therapies used for breast cancer patients, describe the molecular mechanisms through which obesity can affect systemic therapies, outline the existing American Society of Clinical Oncology (ASCO) guidelines for treating patients with cancer and obesity, and highlight additional clinical considerations for treating patients with obesity and breast cancer. We conclude that further research on the biological mechanisms underlying the obesity–breast cancer link may offer new treatment strategies, and clinicals trials that focus on the treatment and outcomes of patients with obesity and all stages of breast cancer are needed to inform future treatment guidelines.
Full article
Open AccessReview
Updated Clinical Evidence on the Role of Adipokines and Breast Cancer: A Review
by
Georgios-Ioannis Verras, Levan Tchabashvili, David-Dimitris Chlorogiannis, Francesk Mulita and Maria-Ioanna Argentou
Cited by 10 | Viewed by 1855
Abstract
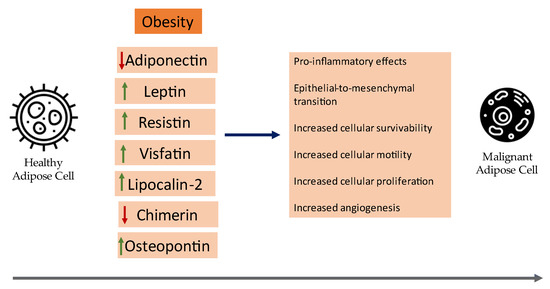
With the recent leaps in medicine, the landscape of our knowledge regarding adipose tissue has changed dramatically: it is now widely regarded as a fully functional endocrine organ. In addition, evidence from observational studies has linked the pathogenesis of diseases like breast cancer
[...] Read more.
With the recent leaps in medicine, the landscape of our knowledge regarding adipose tissue has changed dramatically: it is now widely regarded as a fully functional endocrine organ. In addition, evidence from observational studies has linked the pathogenesis of diseases like breast cancer with adipose tissue and mainly with the adipokines that are secreted in its microenvironment, with the catalog continuously expanding. Examples include leptin, visfatin, resistin, osteopontin, and more. This review aims to encapsulate the current clinical evidence concerning major adipokines and their link with breast cancer oncogenesis. Overall, there have been numerous meta-analyses that contribute to the current clinical evidence, however more targeted larger-scale clinical studies are still expected to solidify their clinical utility in BC prognosis and reliability as follow-up markers.
Full article
►▼
Show Figures
Open AccessCommunication
Cell-of-Origin Targeted Drug Repurposing for Triple-Negative and Inflammatory Breast Carcinoma with HDAC and HSP90 Inhibitors Combined with Niclosamide
by
Udayan Bhattacharya, Mohammad Kamran, Maroua Manai, Massimo Cristofanilli and Tan A. Ince
Cited by 6 | Viewed by 3224
Abstract
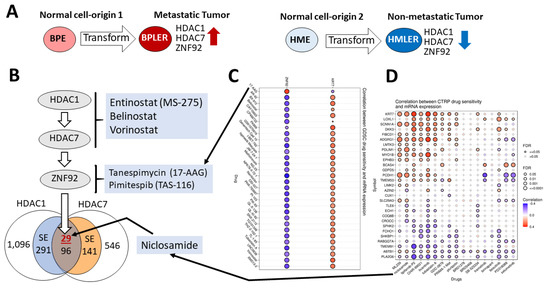
We recently identified a cell-of-origin-specific mRNA signature associated with metastasis and poor outcome in triple-negative carcinoma (TNBC). This TNBC cell-of-origin signature is associated with the over-expression of histone deacetylases and zinc finger protein HDAC1, HDAC7, and ZNF92, respectively. Based on this signature, we
[...] Read more.
We recently identified a cell-of-origin-specific mRNA signature associated with metastasis and poor outcome in triple-negative carcinoma (TNBC). This TNBC cell-of-origin signature is associated with the over-expression of histone deacetylases and zinc finger protein HDAC1, HDAC7, and ZNF92, respectively. Based on this signature, we discovered that the combination of three drugs (an HDAC inhibitor, an anti-helminthic Niclosamide, and an antibiotic Tanespimycin that inhibits HSP90) synergistically reduces the proliferation of the twelve tested TNBC cell lines. Additionally, we discovered that four out of five inflammatory breast carcinoma cell lines are sensitive to this combination. Significantly, the concentration of the drugs that are used in these experiments are within or below clinically achievable dose, and the synergistic activity only emerged when all three drugs were combined. Our results suggest that HDAC and HSP90 inhibitors combined with the tapeworm drug Niclosamide can achieve remarkably synergistic inhibition of TNBC and IBC. Since Niclosamide, HDAC, and HSP90 inhibitors were approved for clinical use for other cancer types, it may be possible to repurpose their combination for TNBC and IBC.
Full article
►▼
Show Figures
Open AccessReview
Special Techniques of Adjuvant Breast Carcinoma Radiotherapy
by
Iveta Kolářová, Bohuslav Melichar, Jaroslav Vaňásek, Igor Sirák, Jiří Petera, Kateřina Horáčková, Denisa Pohanková, Zuzana Šinkorová, Oldřich Hošek and Milan Vošmik
Cited by 3 | Viewed by 1695
Abstract
Modern radiotherapy techniques are designed to permit reduced irradiation of healthy tissue, resulting in a diminished risk of adverse effects and shortened recovery times. Several randomized studies have demonstrated the benefits of increased dosage to the tumor bed area in combination with whole
[...] Read more.
Modern radiotherapy techniques are designed to permit reduced irradiation of healthy tissue, resulting in a diminished risk of adverse effects and shortened recovery times. Several randomized studies have demonstrated the benefits of increased dosage to the tumor bed area in combination with whole breast irradiation (WBI). Conventional WBI treatment following breast-conserving procedures, which required 5–7 weeks of daily treatments, has been reduced to 3–4 weeks when using hyperfractionated regimens. The dosage administration improves local control, albeit with poorer cosmesis. The method of accelerated partial breast irradiation (APBI) shortens the treatment period whilst reducing the irradiated volume. APBI can be delivered using intraoperative radiation, brachytherapy, or external beam radiotherapy. Currently available data support the use of external beam partial breast irradiation in selected patients. Modern radiotherapy techniques make it possible to achieve favorable cosmesis in most patients undergoing immediate breast reconstruction surgery, and studies confirm that current methods of external beam radiation allow an acceptable coverage of target volumes both in the reconstructed breast and in the regional lymphatic nodes.
Full article
►▼
Show Figures
Open AccessReview
Trop-2 as a Therapeutic Target in Breast Cancer
by
Elizabeth Sakach, Ruth Sacks and Kevin Kalinsky
Cited by 17 | Viewed by 4231
Abstract
The emergence of Trop-2 as a therapeutic target has given rise to new treatment paradigms for the treatment of patients with advanced and metastatic breast cancer. Trop-2 is most highly expressed in triple negative breast cancer (TNBC), but the receptor is found across
[...] Read more.
The emergence of Trop-2 as a therapeutic target has given rise to new treatment paradigms for the treatment of patients with advanced and metastatic breast cancer. Trop-2 is most highly expressed in triple negative breast cancer (TNBC), but the receptor is found across all breast cancer subtypes. With sacituzumab govitecan, the first FDA-approved, Trop-2 inhibitor, providing a survival benefit in patients with both metastatic TNBC and hormone receptor positive breast cancer, additional Trop-2 directed therapies are under investigation. Ongoing studies of combination regimens with immunotherapy, PARP inhibitors, and other targeted agents aim to further harness the effect of Trop-2 inhibition. Current investigations are also underway in the neoadjuvant and adjuvant setting to evaluate the therapeutic benefit of Trop-2 inhibition in patients with early stage disease. This review highlights the significant impact the discovery Trop-2 has had on our patients with heavily pretreated breast cancer, for whom few treatment options exist, and the future direction of novel Trop-2 targeted therapies.
Full article
Open AccessSystematic Review
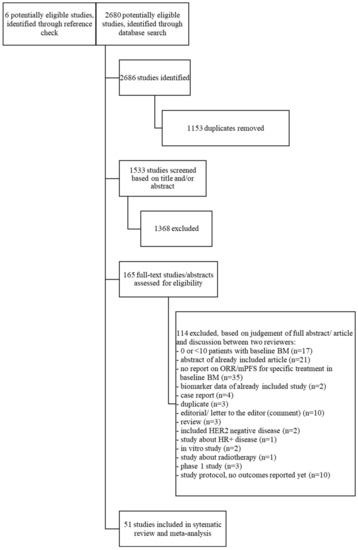
Systemic Therapy for Patients with HER2-Positive Breast Cancer and Brain Metastases: A Systematic Review and Meta-Analysis
by
Inge M. Werter, Sharon Remmelzwaal, George L. Burchell, Tanja D. de Gruijl, Inge R. Konings, Hans J. van der Vliet and C. Willemien Menke-van der Houven van Oordt
Cited by 1 | Viewed by 2735
Abstract
Aim: Patients with HER2-positive (HER2+) metastatic breast cancer (mBC) develop brain metastases (BM) in up to 30% of cases. Treatment of patients with BM can consist of local treatment (surgery and/or radiotherapy) and/or systemic treatment. We undertook a systematic review and meta-analysis to
[...] Read more.
Aim: Patients with HER2-positive (HER2+) metastatic breast cancer (mBC) develop brain metastases (BM) in up to 30% of cases. Treatment of patients with BM can consist of local treatment (surgery and/or radiotherapy) and/or systemic treatment. We undertook a systematic review and meta-analysis to determine the effect of different systemic therapies in patients with HER2+ mBC and BM. Methods: A systematic search was performed in the databases PubMed, Embase.com, Clarivate Analytics/Web of Science Core Collection and the Wiley/Cochrane Library. Eligible articles included prospective or retrospective studies reporting on the effect of systemic therapy on objective response rate (ORR) and/or median progression free survival (mPFS) in patients with HER2+ mBC and BM. The timeframe within the databases was from inception to 19 January 2022. Fixed-effects meta-analyses were used. Quality appraisal was performed using the ROBINS-I tool. Results: Fifty-one studies were included, involving 3118 patients. Most studies, which contained the largest patient numbers, but also often carried a moderate-serious risk of bias, investigated lapatinib and capecitabine (LC), trastuzumab-emtansine (T-DM1) or pyrotinib. The best quality data and/or highest ORR were described with tucatinib (combined with trastuzumab and capecitabine, TTC) and trastuzumab-deruxtecan (T-DXd). TTC demonstrated an ORR of 47.3% in patients with asymptomatic and/or active BM. T-DXd achieved a pooled ORR of 64% (95% CI 43–85%, I
2 0%) in a heavily pretreated population with asymptomatic BM (3 studies,
n = 96). Conclusions: Though our meta-analysis should be interpreted with caution due to the heterogeneity of included studies and a related serious risk of bias, this review provides a comprehensive overview of all currently available systemic treatment options. T-Dxd and TTC that appear to constitute the most effective systemic therapy in patients with HER2+ mBC and BM, while pyrotinib might be an option in Asian patients.
Full article
►▼
Show Figures
Open AccessReview
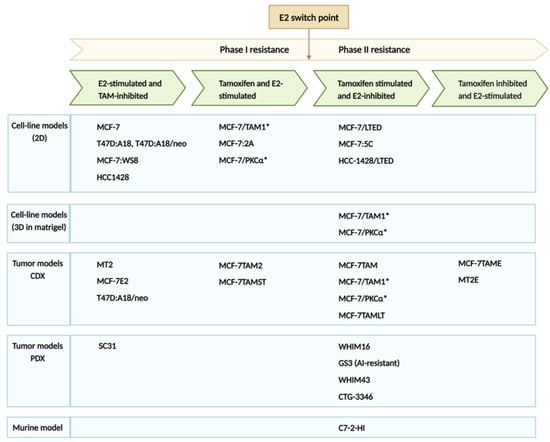
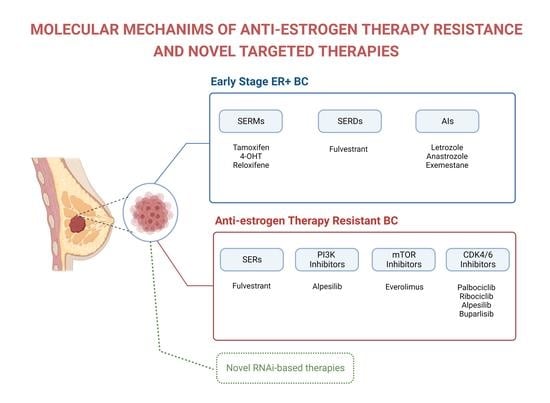
Molecular Mechanisms of Anti-Estrogen Therapy Resistance and Novel Targeted Therapies
by
Rumeysa Ozyurt and Bulent Ozpolat
Cited by 14 | Viewed by 4225
Abstract
Breast cancer (BC) is the most commonly diagnosed cancer in women, constituting one-third of all cancers in women, and it is the second leading cause of cancer-related deaths in the United States. Anti-estrogen therapies, such as selective estrogen receptor modulators, significantly improve survival
[...] Read more.
Breast cancer (BC) is the most commonly diagnosed cancer in women, constituting one-third of all cancers in women, and it is the second leading cause of cancer-related deaths in the United States. Anti-estrogen therapies, such as selective estrogen receptor modulators, significantly improve survival in estrogen receptor-positive (ER+) BC patients, which represents about 70% of cases. However, about 60% of patients inevitably experience intrinsic or acquired resistance to anti-estrogen therapies, representing a major clinical problem that leads to relapse, metastasis, and patient deaths. The resistance mechanisms involve mutations of the direct targets of anti-estrogen therapies, compensatory survival pathways, as well as alterations in the expression of non-coding RNAs (e.g., microRNA) that regulate the activity of survival and signaling pathways. Although cyclin-dependent kinase 4/6 and phosphatidylinositol 3-kinase (PI3K)/AKT/mammalian target of rapamycin (mTOR) inhibitors have significantly improved survival, the efficacy of these therapies alone and in combination with anti-estrogen therapy for advanced ER+ BC, are not curative in advanced and metastatic disease. Therefore, understanding the molecular mechanisms causing treatment resistance is critical for developing highly effective therapies and improving patient survival. This review focuses on the key mechanisms that contribute to anti-estrogen therapy resistance and potential new treatment strategies alone and in combination with anti-estrogen drugs to improve the survival of BC patients.
Full article
►▼
Show Figures
Open AccessArticle
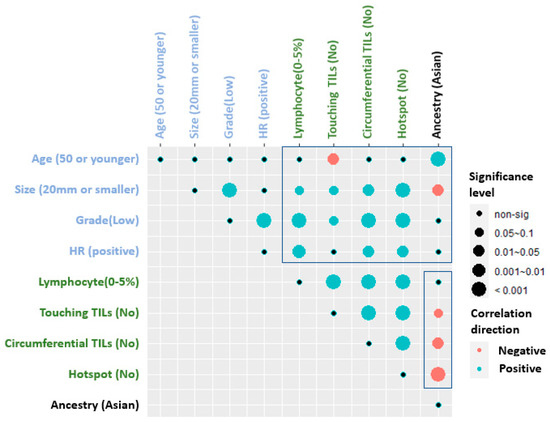
Tumor Infiltrating Lymphocytes in Multi-National Cohorts of Ductal Carcinoma In Situ (DCIS) of Breast
by
Sunil S. Badve, Sanghee Cho, Xiaoyu Lu, Sha Cao, Soumya Ghose, Aye Aye Thike, Puay Hoon Tan, Idris Tolgay Ocal, Daniele Generali, Fabrizio Zanconati, Adrian L. Harris, Fiona Ginty and Yesim Gökmen-Polar
Cited by 1 | Viewed by 1794
Abstract
Tumor-infiltrating lymphocytes (TILs) are prognostic in invasive breast cancer. However, their prognostic significance in ductal carcinoma in situ (DCIS) has been controversial. To investigate the prognostic role of TILs in DCIS outcome, we used different scoring methods for TILs in multi-national cohorts from
[...] Read more.
Tumor-infiltrating lymphocytes (TILs) are prognostic in invasive breast cancer. However, their prognostic significance in ductal carcinoma in situ (DCIS) has been controversial. To investigate the prognostic role of TILs in DCIS outcome, we used different scoring methods for TILs in multi-national cohorts from Asian and European women. Self-described race was genetically confirmed using QC Infinium array combined with radmixture software. Stromal TILs, touching TILs, circumferential TILs, and hotspots were quantified on H&E-stained slides and correlated with the development of second breast cancer events (BCE) and other clinico-pathological variables. In univariate survival analysis, age older than 50 years, hormone receptor positivity and the presence of circumferential TILs were weakly associated with the absence of BCE at the 5-year follow-up in all cohorts (
p < 0.03;
p < 0.02; and
p < 0.02, respectively, adjusted
p = 0.11). In the multivariable analysis, circumferential TILs were an independent predictor of a better outcome (Wald test
p = 0.01), whereas younger age was associated with BCE. Asian patients were younger with larger, higher grade, HR negative DCIS lesions, and higher TIL variables. The spatial arrangement of TILs may serve as a better prognostic indicator in DCIS cases than stromal TILs alone and may be added in guidelines for TILs evaluation in DCIS.
Full article
►▼
Show Figures
Open AccessArticle
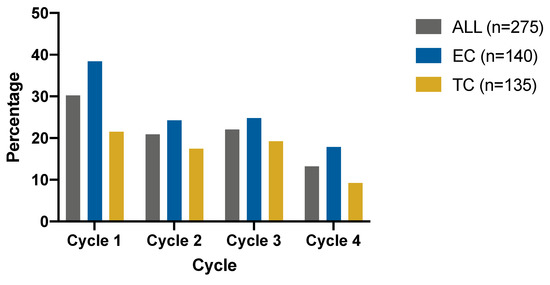
The Comparative Safety of Epirubicin and Cyclophosphamide versus Docetaxel and Cyclophosphamide in Lymph Node-Negative, HR-Positive, HER2-Negative Breast Cancer (ELEGANT): A Randomized Trial
by
Deyue Liu, Jiayi Wu, Caijin Lin, Shuning Ding, Shuangshuang Lu, Yan Fang, Jiahui Huang, Jin Hong, Weiqi Gao, Siji Zhu, Xiaosong Chen, Ou Huang, Jianrong He, Weiguo Chen, Yafen Li, Kunwei Shen and Li Zhu
Cited by 3 | Viewed by 2134
Abstract
Background: In adjuvant settings, epirubicin and cyclophosphamide (EC) and docetaxel and cyclophosphamide (TC) are both optional chemotherapy regimens for lymph node-negative, hormone receptor (HR)-positive, human epidermal receptor 2 (HER2)-negative breast cancer patients. Neutropenia is one of the most common adverse events (AEs) of
[...] Read more.
Background: In adjuvant settings, epirubicin and cyclophosphamide (EC) and docetaxel and cyclophosphamide (TC) are both optional chemotherapy regimens for lymph node-negative, hormone receptor (HR)-positive, human epidermal receptor 2 (HER2)-negative breast cancer patients. Neutropenia is one of the most common adverse events (AEs) of these regimens. The rate of grade 3–4 neutropenia varies in different studies, and direct comparisons of safety profiles between EC and TC are lacking. Method: ELEGANT (NCT02549677) is a prospective, randomized, open-label, noninferior hematological safety trial. Eligible patients with lymph node-negative HR+/HER2-tumors (1:1) were randomly assigned to received four cycles of EC (90/600 mg/m
2) or TC (75/600 mg/m
2) every three weeks as adjuvant chemotherapy. The primary endpoint was the incidence of grade 3 or 4 neutropenia defined by National Cancer Institute-Common Terminology Criteria for Adverse Events (NCI-CTCAE) version 4.0 on an intention-to-treat basis. Noninferiority was defined as an upper 95% CI less than a noninferiority margin of 15%. Results: In the intention-to-treat population, 140 and 135 patients were randomized into the EC and TC arms, respectively. For the primary endpoint, the rate of grade 3 or 4 neutropenia is 50.71% (95% CI: 42.18%, 59.21%) in the EC arm and 48.15% (95% CI: 39.53%, 56.87%) in the TC arm (95%CI risk difference: −0.100, 0.151), showing the noninferiority of the EC arm. For secondary endpoints, the rate of all-grade anemia is higher in the EC arm (EC 42.86% versus TC 22.96%,
p = 0.0007), and more patients suffer from nausea/vomiting, hair loss, and nail changes (
p < 0.01) in the EC arm. No statistically different disease-free survival was observed between the two arms (
p = 0.13). Conclusion: EC is not inferior to TC in the rate of grade 3 or 4 neutropenia, but more other AEs were observed in the EC group.
Full article
►▼
Show Figures
Open AccessArticle
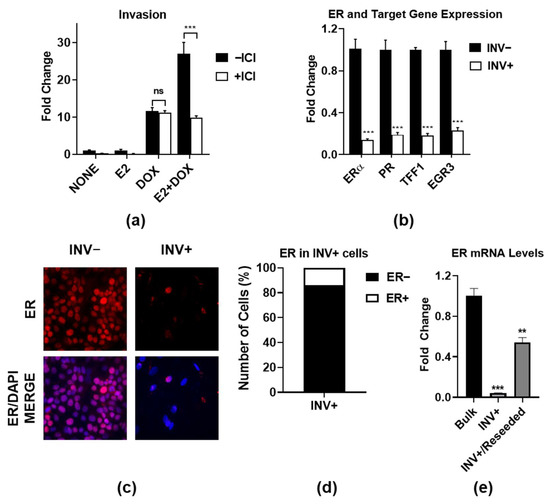
Estrogen Receptor-Regulated Gene Signatures in Invasive Breast Cancer Cells and Aggressive Breast Tumors
by
Emily Smart, Svetlana E. Semina, Luis H. Alejo, Nidhi S. Kansara and Jonna Frasor
Cited by 1 | Viewed by 1828
Abstract
Most metastatic breast cancers arise from estrogen receptor α (ER)-positive disease, and yet the role of ER in promoting metastasis is unclear. Here, we used an ER+ breast cancer cell line that is highly invasive in an ER- and IKKβ-dependent manner. We defined
[...] Read more.
Most metastatic breast cancers arise from estrogen receptor α (ER)-positive disease, and yet the role of ER in promoting metastasis is unclear. Here, we used an ER+ breast cancer cell line that is highly invasive in an ER- and IKKβ-dependent manner. We defined two ER-regulated gene signatures that are specifically regulated in the subpopulations of invasive cells. The first consists of proliferation-associated genes, which is a known function of ER, which actually suppress rather than enhance invasion. The second signature consists of genes involved in essential biological processes, such as organelle assembly and vesicle trafficking. Importantly, the second subpopulation-specific signature is associated with aggressive disease and poor patient outcome, independently of proliferation. These findings indicate a complex interplay between ER-driven proliferation and invasion, and they define new ER-regulated gene signatures that are predictive of aggressive ER+ breast cancer.
Full article
►▼
Show Figures