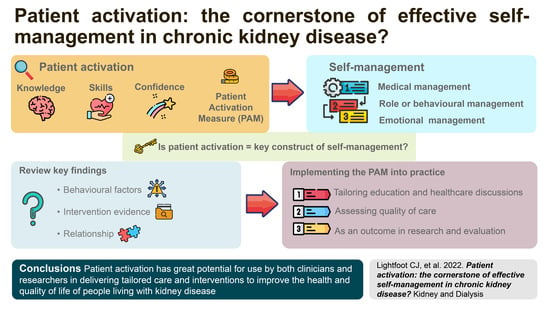
Patient Activation: The Cornerstone of Effective Self-Management in Chronic Kidney Disease?
Abstract
:1. Introduction
2. What Is Self-Management?
Self-Management and CKD
3. What Is Patient Activation?
3.1. Theories of Patient Activation
3.2. Patient Activation and Health Outcomes
4. Patient Activation and Its Role in Effective Self-Management
5. Measuring Patient Activation and Self-Management
6. Strategies to Implement the Patient Activation Measure (PAM)
- (a)
- Tailoring education and healthcare discussions
- (b)
- Assessing quality of care
- (c)
- Using PAM as an outcome in research and evaluation
7. Considerations of Using the PAM
8. Increasing Patient Activation and Self-Management in CKD
9. Patient Perspectives on Improving Patient Activation to Improve CKD Self-Management
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hibbard, J.H.; Greene, J. What the evidence shows about patient activation: Better health outcomes and care experiences; fewer data on costs. Health Aff. 2013, 32, 207–214. [Google Scholar] [CrossRef]
- NHS England. The NHS Long Term Plan. BMJ 2019, 364, 184. [Google Scholar] [CrossRef] [Green Version]
- Hibbard, J.; Gilburt, H. Supporting People to Manage their Health: An Introduction to Patient Activation; The King’s Fund: London, UK, 2014. [Google Scholar]
- Wagner, E.H. Chronic disease management: What will it take to improve care for chronic illness? Eff. Clin. Pract. 1998, 1, 2–4. [Google Scholar]
- Grady, P.A.; Gough, L.L. Self-management: A comprehensive approach to management of chronic conditions. Am. J. Public Health 2014, 104, e25–e31. [Google Scholar] [CrossRef] [PubMed]
- Lorig, K.R.; Holman, H. Self-management education: History, definition, outcomes, and mechanisms. Ann. Behav. Med. 2003, 26, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Corbin, J.M.; Strauss, A. Unending Work and Care: Managing Chronic Illness at Home; Jossey-Bass: San Francisco, CA, USA, 1988. [Google Scholar]
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2012 Clincial Practice Guideline for Evaluation and Management of Chronic Kidney Disease. Kidney Int. 2013, 3, 1–150. [Google Scholar]
- National Institute for Health and Care Excellence (NICE). Chronic Kidney Disease: Assessment and Management; NICE: London, UK, 2021. [Google Scholar]
- Peng, S.; He, J.; Huang, J.; Lun, L.; Zeng, J.; Zeng, S.; Zhang, L.; Liu, X.; Wu, Y. Self-management interventions for chronic kidney disease: A systematic review and meta-analysis. BMC Nephrol. 2019, 20, 142. [Google Scholar] [CrossRef] [PubMed]
- Novak, M.; Costantini, L.; Schneider, S.; Beanlands, H. Approaches to self-management in chronic illness. Semin. Dial. 2013, 26, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Havas, K.; Bonner, A.; Douglas, C. Self-management support for people with chronic kidney disease: Patient perspectives. J. Ren. Care 2016, 42, 7–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tuttle, K.R.; Sunwold, D.; Kramer, H. Can comprehensive lifestyle change alter the course of chronic kidney disease? In Seminars in Nephrology; Elsevier: Amsterdam, The Netherlands, 2009; Volume 5, pp. 512–523. [Google Scholar]
- Teng, H.-L.; Yen, M.; Fetzer, S.; Sung, J.-M.; Hung, S.-Y. Effects of Targeted Interventions on Lifestyle Modifications of Chronic Kidney Disease Patients: Randomized Controlled Trial. West. J. Nurs. Res. 2013, 35, 1107–1127. [Google Scholar] [CrossRef] [PubMed]
- Gibert, S.H.; DeGrazia, D.; Danis, M. Ethics of patient activation: Exploring its relation to personal responsibility, autonomy and health disparities. J. Med. Ethics 2017, 43, 670–675. [Google Scholar] [CrossRef] [PubMed]
- Hibbard, J.H.; Mahoney, E.R.; Stockard, J.; Tusler, M. Development and testing of a short form of the patient activation measure. Health Serv. Res. 2005, 40, 1918–1930. [Google Scholar] [CrossRef] [Green Version]
- Hibbard, J.H.; Stockard, J.; Mahoney, E.R.; Tusler, M. Development of the Patient Activation Measure (PAM): Conceptualizing and measuring activation in patients and consumers. Health Serv. Res. 2004, 39, 1005–1026. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Greene, J.; Hibbard, J.H. Why Does Patient Activation Matter? An Examination of the Relationships Between Patient Activation and Health-Related Outcomes. J. Gen. Intern. Med. 2012, 27, 520–526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prochaska, J.O.; Velicer, W.F. The transtheoretical model of health behavior change. Am. J. Health Promot. 1997, 12, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Hibbard, J.H. Using systematic measurement to target consumer activation strategies. Med. Care Res. Rev. 2009, 66, 9S–27S. [Google Scholar] [CrossRef]
- Flink, M.; Brandberg, C.; Ekstedt, M. Why patients decline participation in an intervention to reduce re-hospitalization through patient activation: Whom are we missing? Trials 2019, 20, 82. [Google Scholar] [CrossRef] [PubMed]
- Hibbard, J.H.; Greene, J.; Tusler, M. Improving the outcomes of disease management by tailoring care to the patient’s level of activation. Am. J. Manag. Care 2009, 15, 353–360. [Google Scholar] [PubMed]
- Ryan, P. Integrated Theory of Health Behavior Change: Background and intervention development. Clin. Nurse Spec. 2009, 23, 161–170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, S.G.; Curtis, L.M.; Wardle, J.; von Wagner, C.; Wolf, M.S. Skill set or mind set? Associations between health literacy, patient activation and health. PLoS ONE 2013, 8, e74373. [Google Scholar] [CrossRef] [PubMed]
- Greene, J.; Hibbard, J.H.; Tusler, M. How Much do Health Literacy and Patient Activation Contribute to Older Adults’ Ability to Manage Their Health? AARP Public Policy Institute: Washington, DC, USA, 2005. [Google Scholar]
- Serper, M.; Gawron, A.J.; Smith, S.G.; Pandit, A.A.; Dahlke, A.R.; Bojarski, E.A.; Keswani, R.N.; Wolf, M.S. Patient factors that affect quality of colonoscopy preparation. Clin. Gastroenterol. Hepatol. 2014, 12, 451–457. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nijman, J.; Hendriks, M.; Brabers, A.; de Jong, J.; Rademakers, J. Patient activation and health literacy as predictors of health information use in a general sample of Dutch health care consumers. J. Health Commun. 2014, 19, 955–969. [Google Scholar] [CrossRef] [PubMed]
- Rademakers, J.; Nijman, J.; Brabers, A.E.; de Jong, J.D.; Hendriks, M. The relative effect of health literacy and patient activation on provider choice in the Netherlands. Health Policy 2014, 114, 200–206. [Google Scholar] [CrossRef] [Green Version]
- Hibbard, J.H.; Greene, J.; Shi, Y.; Mittler, J.; Scanlon, D. Taking the Long View: How Well Do Patient Activation Scores Predict Outcomes Four Years Later? Med. Care Res. Rev. 2015, 72, 324–337. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, A.; Hibbard, J.H.; Boothroyd, D.B.; Glaseroff, A.; Asch, S.M. Patient activation changes as a potential signal for changes in health care costs: Cohort study of US high-cost patients. J. Gen. Intern. Med. 2018, 33, 2106–2112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Solomon, M.; Wagner, S.L.; Goes, J. Effects of a Web-based intervention for adults with chronic conditions on patient activation: Online randomized controlled trial. J. Med. Internet Res. 2012, 14, e32. [Google Scholar] [CrossRef] [PubMed]
- Hibbard, J.H.; Greene, J.; Overton, V. Patients with lower activation associated with higher costs; delivery systems should know their patients’ ‘scores’. Health Aff. 2013, 32, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Greene, J.; Hibbard, J.H.; Sacks, R.; Overton, V.; Parrotta, C.D. When patient activation levels change, health outcomes and costs change, too. Health Aff. 2015, 34, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Zimbudzi, E.; Lo, C.; Ranasinha, S.; Kerr, P.G.; Polkinghorne, K.R.; Teede, H.; Usherwood, T.; Walker, R.G.; Johnson, G.; Fulcher, G.; et al. The association between patient activation and self-care practices: A cross-sectional study of an Australian population with comorbid diabetes and chronic kidney disease. Health Expect 2017, 20, 1375–1384. [Google Scholar] [CrossRef] [PubMed]
- Deeny, S.; Thorlby, R.; Steventon, A. Briefing: Reducing Emergency Admissions: Unlocking the Potential of People to Better Manage Their Long-Term Conditions; The Health Foundation: London, UK, 2018. [Google Scholar]
- Gair, R.M.; Stannard, C.; Wong, E.; Hawkins, J.; Van der Veer, S.N.; Farrington, K.; Hope, J.; Fluck, R. Transforming Participation in Chronic Kidney Disease: Programme Report; Renal Association: Bandera, TX, USA, 2019. [Google Scholar]
- Wilkinson, T.J.; Memory, K.E.; Lightfoot, C.J.; Palmer, J.; Smith, A.C. Determinants of patient activation and its association with cardiovascular disease risk in chronic kidney disease: A cross-sectional study. Health Expect. 2021, 24, 843–852. [Google Scholar] [CrossRef]
- Griva, K.; Nandakumar, M.; Ng, J.H.; Lam, K.F.Y.; McBain, H.; Newman, S.P. Hemodialysis Self-management Intervention Randomized Trial (HED-SMART): A Practical Low-Intensity Intervention to Improve Adherence and Clinical Markers in Patients Receiving Hemodialysis. Am. J. Kidney Dis. 2018, 71, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Cukor, D.; Zelnick, L.; Charytan, D.; Shallcross, A.; Mehrotra, R. Patient Activation Measure in Dialysis Dependent Patients in the United States. J. Am. Soc. Nephrol. 2021, 32, 3017–3019. [Google Scholar] [CrossRef] [PubMed]
- Magadi, W.; Lightfoot, C.J.; Memory, K.E.; Santhakumaran, S.; van der Veer, S.N.; Thomas, N.; Gair, R.; Smith, A.C. Patient activation and its association with symptom burden and quality of life across the spectrum of chronic kidney disease stages in England. BMC Nephrol. 2022, 23, 45. [Google Scholar] [CrossRef]
- Schulman-Green, D.; Jaser, S.; Martin, F.; Alonzo, A.; Grey, M.; McCorkle, R.; Redeker, N.S.; Reynolds, N.; Whittemore, R. Processes of self-management in chronic illness. J. Nurs. Scholarsh. Off. Publ. Sigma Tau Int. Honor. Soc. Nurs. 2012, 44, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, H.P.; Poon, B.Y.; Wang, E.; Shortell, S.M. Linking Practice Adoption of Patient Engagement Strategies and Relational Coordination to Patient-Reported Outcomes in Accountable Care Organizations. Milbank Q. 2019, 97, 692–735. [Google Scholar] [CrossRef] [PubMed]
- Newland, P.; Lorenz, R.; Oliver, B.J. Patient activation in adults with chronic conditions: A systematic review. J. Health Psychol. 2020, 26, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Ritter, P.L.; Lorig, K. The English and Spanish Self-Efficacy to Manage Chronic Disease Scale measures were validated using multiple studies. J. Clin. Epidemiol. 2014, 67, 1265–1273. [Google Scholar] [CrossRef] [PubMed]
- Aung, E.; Donald, M.; Williams, G.M.; Coll, J.R.; Doi, S.A. Influence of patient-assessed quality of chronic illness care and patient activation on health-related quality of life. Int. J. Qual. Health Care 2016, 28, 306–310. [Google Scholar] [CrossRef] [Green Version]
- Chauvet-Gelinier, J.-C.; Bonin, B. Stress, anxiety and depression in heart disease patients: A major challenge for cardiac rehabilitation. Ann. Phys. Rehabil. Med. 2017, 60, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Cukor, D.; Coplan, J.; Brown, C.; Friedman, S.; Cromwell-Smith, A.; Peterson, R.A.; Kimmel, P.L. Depression and Anxiety in Urban Hemodialysis Patients. Clin. J. Am. Soc. Nephrol. 2007, 2, 484–490. [Google Scholar] [CrossRef] [Green Version]
- Hibbard, J.H.; Mahoney, E.R.; Stock, R.; Tusler, M. Do increases in patient activation result in improved self-management behaviors? Health Serv. Res. 2007, 42, 1443–1463. [Google Scholar] [CrossRef] [PubMed]
- Hibbard, J.H.; Tusler, M. Assessing activation stage and employing a “next steps” approach to supporting patient self-management. J. Ambul. Care Manag. 2007, 30, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Lightfoot, C.J.; Wilkinson, T.J.; Memory, K.E.; Palmer, J.; Smith, A.C. Reliability and Validity of the Patient Activation Measure in Kidney Disease: Results of Rasch Analysis. Clin. J. Am. Soc. Nephrol. 2021, 16, 880–888. [Google Scholar] [CrossRef] [PubMed]
- Devraj, R.; Wallace, L.S. Application of the content expert process to develop a clinically useful low-literacy Chronic Kidney Disease Self-Management Knowledge Tool (CKD-SMKT). Res. Soc. Adm. Pharm. 2013, 9, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Glasgow, R.E.; Wagner, E.H.; Schaefer, J.; Mahoney, L.D.; Reid, R.J.; Greene, S.M. Development and validation of the Patient Assessment of Chronic Illness Care (PACIC). Med. Care 2005, 43, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Young, L.; Hertzog, M.; Barnason, S. Effects of a home-based activation intervention on self-management adherence and readmission in rural heart failure patients: The PATCH randomized controlled trial. BMC Cardiovasc. Disord. 2016, 16, 176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roberts, N.J.; Kidd, L.; Dougall, N.; Patel, I.S.; McNarry, S.; Nixon, C. Measuring patient activation: The utility of the Patient Activation Measure within a UK context-Results from four exemplar studies and potential future applications. Patient Educ. Couns. 2016, 99, 1739–1746. [Google Scholar] [CrossRef] [PubMed]
- Kearns, R.; Harris-Roxas, B.; McDonald, J.; Song, H.J.; Dennis, S.; Harris, M. Implementing the Patient Activation Measure (PAM) in clinical settings for patients with chronic conditions: A scoping review. Integr. Healthc. J. 2020, 2, e000032. [Google Scholar] [CrossRef]
- Linden, A.; Butterworth, S.W.; Prochaska, J.O. Motivational interviewing-based health coaching as a chronic care intervention. J. Eval. Clin. Pract. 2010, 16, 166–174. [Google Scholar] [CrossRef]
- Chen, S.M.; Creedy, D.; Lin, H.S.; Wollin, J. Effects of motivational interviewing intervention on self-management, psychological and glycemic outcomes in type 2 diabetes: A randomized controlled trial. Int. J. Nurs. Stud. 2012, 49, 637–644. [Google Scholar] [CrossRef] [PubMed]
- UK Kidney Association. PAM and PROM Data. Available online: https://ukkidney.org/audit-research/data-permissions/data/ukrr-ckd-patient-measures-dataset/pam-and-prom-data(accessed on 15 November 2021).
- NHS England. Measurement and Evaluation in Supported Self-Management; NHS England: London, UK, 2021.
- Nair, D.; Cavanaugh, K.L. Measuring Patient Activation as Part of Kidney Disease Policy: Are We There Yet? J. Am. Soc. Nephrol. 2020, 31, 1435–1443. [Google Scholar] [CrossRef]
- Armstrong, N.; Tarrant, C.; Martin, G.; Manktelow, B.; Liz Brewster, L. Independent Evaluation of the Feasibility of Using the Patient Activation Measure in the NHS in England: Early Findings; NHS England: London, UK, 2015.
- Van Bulck, L.; Claes, K.; Dierickx, K.; Hellemans, A.; Jamar, S.; Smets, S.; Van Pottelbergh, G. Patient and treatment characteristics associated with patient activation in patients undergoing hemodialysis: A cross-sectional study. BMC Nephrol. 2018, 19, 126. [Google Scholar] [CrossRef] [PubMed]
- Bos-Touwen, I.; Jonkman, N.; Westland, H.; Schuurmans, M.; Rutten, F.; de Wit, N.; Trappenburg, J. Tailoring of self-management interventions in patients with heart failure. Curr. Heart Fail. Rep. 2015, 12, 223–235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, M.L.; Zimmerman, L.; Welch, J.L.; Hertzog, M.; Pozehl, B.; Plumb, T. Patient activation with knowledge, self-management and confidence in chronic kidney disease. J. Ren. Care 2016, 42, 15–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vélez-Bermúdez, M.; Christensen, A.J.; Kinner, E.M.; Roche, A.I.; Fraer, M. Exploring the Relationship Between Patient Activation, Treatment Satisfaction, and Decisional Conflict in Patients Approaching End-Stage Renal Disease. Ann. Behav. Med. 2019, 53, 816–826. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, A.J.; Caskey, F.J.; Casula, A.; Inward, C.D.; Ben-Shlomo, Y. Associations with wellbeing and medication adherence in young adults receiving kidney replacement therapy. Clin. J. Am. Soc. Nephrol. 2018, 13, 1669–1679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rivera, E.; Corte, C.; Steffen, A.; DeVon, H.A.; Collins, E.G.; McCabe, P.J. Illness Representation and Self-Care Ability in Older Adults with Chronic Disease. Geriatrics 2018, 3, 45. [Google Scholar] [CrossRef] [Green Version]
- Lo, C.; Zimbudzi, E.; Teede, H.J.; Kerr, P.G.; Ranasinha, S.; Cass, A.; Fulcher, G.; Gallagher, M.; Polkinghorne, K.R.; Russell, G.; et al. Patient-reported barriers and outcomes associated with poor glycaemic and blood pressure control in co-morbid diabetes and chronic kidney disease. J. Diabetes Complicat. 2019, 33, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, N.; Tarrant, C.; Martin, G.; Manktelow, B.; Brewster, L.; Chew, S. Independent Evaluation of the Feasibility of Using the Patient Activation Measure in the NHS in England-Final Report; NHS England: London, UK, 2017.
- Kidd, L.; Lawrence, M.; Booth, J.; Rowat, A.; Russell, S. Development and evaluation of a nurse-led, tailored stroke self-management intervention. BMC Health Serv. Res. 2015, 15, 359. [Google Scholar] [CrossRef] [Green Version]
- Chew, S. i041 Measuring activation: An evaluation of the patient activation measure (PAM). Rheumatology 2018, 57, iii6. [Google Scholar] [CrossRef] [Green Version]
- Welch, J.L.; Johnson, M.; Zimmerman, L.; Russell, C.L.; Perkins, S.M.; Decker, B.S. Self-Management Interventions in Stages 1 to 4 Chronic Kidney Disease: An Integrative Review. West. J. Nurs. Res. 2014, 37, 652–678. [Google Scholar] [CrossRef] [Green Version]
- Hussein, W.F.; Bennett, P.N.; Abra, G.; Watson, E.; Schiller, B. Integrating Patient Activation Into Dialysis Care. Am. J. Kidney Dis. 2022, 79, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Deen, D.; Lu, W.-H.; Rothstein, D.; Santana, L.; Gold, M.R. Asking questions: The effect of a brief intervention in community health centers on patient activation. Patient Educ. Couns. 2011, 84, 257–260. [Google Scholar] [CrossRef]
- Miller, V.M.; Davies, M.J.; Etherton-Beer, C.; McGough, S.; Schofield, D.; Jensen, J.F.; Watson, N. Increasing patient activation through diabetes self-management education: Outcomes of DESMOND in regional Western Australia. Patient Educ. Couns. 2020, 103, 848–853. [Google Scholar] [CrossRef]
- Shah, V.O.; Carroll, C.; Mals, R.; Ghahate, D.; Bobelu, J.; Sandy, P.; Colleran, K.; Schrader, R.; Faber, T.; Burge, M.R. A Home-Based Educational Intervention Improves Patient Activation Measures and Diabetes Health Indicators among Zuni Indians. PLoS ONE 2015, 10, e0125820. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alexander, J.A.; Hearld, L.R.; Mittler, J.N.; Harvey, J. Patient-physician role relationships and patient activation among individuals with chronic illness. Health Serv. Res. 2012, 47, 1201–1223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maeng, D.D.; Martsolf, G.R.; Scanlon, D.P.; Christianson, J.B. Care coordination for the chronically ill: Understanding the patient’s perspective. Health Serv. Res. 2012, 47, 1960–1979. [Google Scholar] [CrossRef] [Green Version]
- Mosen, D.M.; Schmittdiel, J.; Hibbard, J.; Sobel, D.; Remmers, C.; Bellows, J. Is patient activation associated with outcomes of care for adults with chronic conditions? J. Ambul. Care Manag. 2007, 30, 21–29. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Level | Description |
|---|---|
 | |
| Measure | Description |
|---|---|
| Chronic Kidney Disease—Self-Management Knowledge Tool (CKD-SMKT) [51] | The CKD-SMKT is a validated 11-item questionnaire, which comprises of several statements of self-management behaviours to which the respondent must indicate if they believe this is ‘true’, ‘false’ or ‘I do not know’, and if they have done this in the last six months (yes or no). Respondents are asked how much they know about their kidney health and to rate this on a five-point Likert scale from “I know everything I need to know” to “I know nothing”. The CKD-SMKT assess CKD disease-specific knowledge of self-management, which is associated with higher patient activation and improved self-management behaviours. |
| Patient Assessment of Care (PACIC) [52] | The PACIC is a validated 20-item tool to assess the extent to which patients with a long-term condition receive care that aligns with the Chronic Care Model (CCM). The items are aggregated to form five subscales: (1) patient activation, (2) delivery system design/decision support, (3) goal setting/tailoring, (4) problem solving/contextual, and (5) follow-up/coordination. Whilst these subscales are congruent to the components of CCM, they do not perfectly map onto the CCM components. Respondents rate how often they experienced the content described in each item during the past six months. Each item is scored on a five-point Likert scale from “almost never to “almost always”. Patient activation (i.e., actions that solicit patient input and involvement in decision-making), goal setting (i.e., acquiring information for and setting of specific, collaborative goal), and problem-solving/contextual (i.e., considering potential barriers and the patient’s social and cultural environment in making treatment plans) counselling all map onto self- management support in the CCM. |
| Patient Activation Measure (PAM) [16] | The PAM-13 is the short form of the 22-item PAM [17] measuring the knowledge, skills, and confidence for self-management. Individuals respond to items such as ‘I know how to prevent further problems with my health condition’ using a 4-point Likert scale ranging from ‘strongly disagree’ (1) to ‘strongly agree’ (4). A ‘not applicable’ (N/A) response is also available. Responses of N/A are scored as 0 and are reported to distinguish those left blank. A continuous activation score is computed from the raw score using an empirically derived calibration table by Insignia Health. The PAM-13 is scored along a Guttman scale (0–100) with higher scores along a unidimensional continuum signifying greater activation. Level 1 (PAM-13 score ≤47; disengagement and disbelief about one’s own role in self-management) encompasses items 1 and 2; Level 2 (47.1–55.1; increasing awareness, confidence, and knowledge in self-management tasks), items 3–8; Level 3 (55.2–67; readiness and taking action), 9–11; and Level 4 (≥67.1; sustainment). |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lightfoot, C.J.; Nair, D.; Bennett, P.N.; Smith, A.C.; Griffin, A.D.; Warren, M.; Wilkinson, T.J. Patient Activation: The Cornerstone of Effective Self-Management in Chronic Kidney Disease? Kidney Dial. 2022, 2, 91-105. https://doi.org/10.3390/kidneydial2010012
Lightfoot CJ, Nair D, Bennett PN, Smith AC, Griffin AD, Warren M, Wilkinson TJ. Patient Activation: The Cornerstone of Effective Self-Management in Chronic Kidney Disease? Kidney and Dialysis. 2022; 2(1):91-105. https://doi.org/10.3390/kidneydial2010012
Chicago/Turabian StyleLightfoot, Courtney J., Devika Nair, Paul N. Bennett, Alice C. Smith, Anthony D. Griffin, Madeleine Warren, and Thomas J. Wilkinson. 2022. "Patient Activation: The Cornerstone of Effective Self-Management in Chronic Kidney Disease?" Kidney and Dialysis 2, no. 1: 91-105. https://doi.org/10.3390/kidneydial2010012