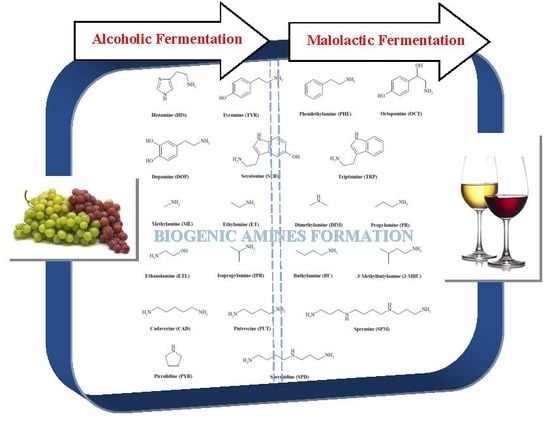
Accumulation of Biogenic Amines in Wine: Role of Alcoholic and Malolactic Fermentation
Abstract
:1. Introduction
2. Analytical Determination of BAs in Wine
3. BAs and Alcoholic Fermentation
4. BAs and Malolactic Fermentation
5. Conclusions
Acknowledgments
Author contributions
Conflicts of Interest
References
- European Food Safety Authority (EFSA). Scientific opinion on risk based control of biogenic amine formation in fermented foods. EFSA J. 2011, 9, 2393. [Google Scholar]
- Spano, G.; Russo, P.; Lonvaud-Funel, A.; Lucas, P.; Alexandre, H.; Grandvalet, C.; Coton, E.; Coton, M.; Barnavon, L.; Bach, B.; et al. Biogenic amines in fermented food. Eur. J. Clin. Nutr. 2010, 64, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Garde-Cerdan, T.; Martinez-Gil, A.M.; Lorenzo, C.; Lara, J.F.; Pardo, F.; Salinas, M.R. Implications of nitrogen compounds during alcoholic fermentation from some grape varieties at different maturation stages and cultivation systems. Food Chem. 2011, 124, 106–116. [Google Scholar] [CrossRef]
- Smit, A.Y.; du Toit, W.J.; du Toit, M. Biogenic amines in wine: Understanding the headache. S. Afr. J. Enol. Vitic. 2008, 29, 109–127. [Google Scholar] [CrossRef]
- Polo, L.; Ferrer, S.; Pea-Gallego, A.; Hernndez-Orte, P.; Pardo, I. Biogenic amine synthesis in high quality Tempranillo wines. Relationship with lactic acid bacteria and vinification conditions. Ann. Microbiol. 2010, 61, 191–198. [Google Scholar] [CrossRef]
- Martuscelli, M.; Arfelli, G.; Manetta, A.C.; Suzzi, G. Biogenic amines content as a measure of the quality of wines of Abruzzo (Italy). Food Chem. 2013, 140, 590–597. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Commission Regulation (EC) No. 2073/2005 of November 2005 on Microbiological Criteria for Foodstuffs. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=CELEX:32005R2073:en:NOT (accessed on 18 July 2017).
- Ten Brink, B.; Damink, C.; Joosten, H.M.L.J.; Huis in’t Veld, J.H.H. Occurrence and formation of biologically active amines in foods. Int. J. Food Microbiol. 1990, 11, 73–84. [Google Scholar] [CrossRef]
- Bremer, P.J.; Fletcher, G.C.; Osborne, C. Scombrotoxin in Seafood; New Zealand Institute for Crop and Food Research Limited: Christchurch, New Zealand; A Crown Research Institute: Wellington, New Zealand, 2003. Available online: www.crop.cri.nz/home/research/marine/pathogens/Scombrotoxin.pdf (accessed on 16 March 2011).
- Anonymous; Australian Food Standards Code 2001. Part D: Fish and fish products. In Standards D1 and D2; Version 18; ANSTAT: South Melbourne, VIC, Australia, 2001. [Google Scholar]
- Anonymous. Chapter 7: Scombrotoxin (histamine) formation (a chemical hazard). In Fish and Fisheries Products Hazards and Controls Guidance, 3rd ed.; Office Food Safety, Center for Food Safety and Applied Nutrition, Food and Drug Administration: Washington, DC, USA, 2001. Available online: http://www.fda.gov/Food/GuidanceComplianceRegulatoryInformation/GuidanceDocuments/Seafood/FishandFisheriesProductsHazardsandControlsGuide/ucm089637.htm (accessed on 16 March 2011).
- Anli, R.E.; Vural, N.; Yilmaz, S.; Vural, Y.H. The determination of biogenic amines in Turkish red wines. J. Food Compos. Anal. 2004, 17, 53–62. [Google Scholar] [CrossRef]
- Onal, A.; Tekkeli, S.E.; Onal, C. A review of the liquid chromatographic methods for the determination of biogenic amines in foods. Food Chem. 2013, 138, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Anli, E.R.; Bayram, M. Biogenic Amines in Wines. Food Rev. Int. 2009, 25, 86–102. [Google Scholar] [CrossRef]
- Ancin-Azpilicueta, C.; Gonzalez-Marco, A.; Jimenez-Moreno, N. Current knowledge about the presence of amines in wine. Crit. Rev. Food Sci. Nutr. 2008, 48, 257–275. [Google Scholar] [CrossRef] [PubMed]
- Mangani, S.; Geurrini, S.; Granchi, L.; Vincenzini, M. Putrescine accumulation in wine: Role of Oenococcus oeni. Curr. Microbiol. 2005, 51, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Beneduce, L.; Romano, A.; Capozzi, V.; Lucas, P.; Barnavon, L.; Bach, B.; Vuchot, P.; Grieco, F.; Spano, G. Biogenic amine in wines. Ann. Microbiol. 2010, 60, 573–578. [Google Scholar] [CrossRef]
- Cecchini, F.; Morassut, M. Effect of grap storage time on biogenic amines content in must. Food Chem. 2010, 123, 263–268. [Google Scholar] [CrossRef]
- Del Prete, V.; Costantini, A.; Cecchini, F.; Morassut, M.; Garcia-Moruno, E. Occurrence of biogenic amines in wine: The role of grapes. Food Chem. 2009, 112, 474–481. [Google Scholar] [CrossRef]
- Guo, Y.-Y.; Yang, Y.-P.; Peng, Q.; Han, Y. Biogenic amines in wine: A review. Int. J. Food Sci. Technol. 2015, 50, 1523–1532. [Google Scholar] [CrossRef]
- Önal, A. Current analytical methods for the determination of biogenic amines in foods. Food Chem. 2007, 103, 1475–1486. [Google Scholar] [CrossRef]
- Coton, M.; Romano, A.; Spano, G.; Ziegler, K.; Vetrano, C.; Desmarias, C.; Lonvaud-Funel, A.; Lucas, P.; Coton, E. Occurence of biogenic amine forming lactic acid bacteria in wine and cider. Food Microbiol. 2010, 27, 1078–1085. [Google Scholar] [CrossRef] [PubMed]
- BediaErim, F. Recent analytical approaches to the analysis of biogenic amines in food samples. Trends Anal. Chem. 2013, 52, 239–247. [Google Scholar] [CrossRef]
- De Borba, B.M.; Rohrer, J.S. Determination of biogenic amines in alcoholic beverages by ion chromatography with suppressed conductivity detection and integrated pulsed amperometric detection. J. Chromatogr. A 2007, 115, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Piasta, A.M.; Jastrzebska, A.; Krzeminski, M.P.; Muzioł, T.M.; Szłyk, E. New procedure of selected biogenic amines determination in wine samples by HPLC. Anal. Chim. Acta 2014, 834, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Herrero, M.; GarcíaCañas, V.; Simo, C.; Cifuentes, A. Recent advances in the application of capillary electromigration methods for food analysis and foodomics. Electrophoresis 2010, 31, 205–228. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, J.O.; Ferreira, M.A. Combined ion-pair extraction and gas chromatography-mass spectrometry for the simultaneous determination of diamines, polyamines and aromatic amines in port wine and grape juice. J. Chromatogr. A 2000, 886, 183–195. [Google Scholar] [CrossRef]
- Lange, J.; Wittmann, C. Enzyme sensor array for the determination of biogenic amines in food samples. Anal. Bioanal. Chem. 2002, 372, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Peña-Gallego, A.; Hernández-Orte, P.; Cacho, J.; Ferreira, V. High-Performance Liquid Chromatography Analysis of Amines in Must and Wine: A Review. Food Rev. Int. 2012, 28, 71–96. [Google Scholar] [CrossRef]
- Berbegal, C.; Pardo, I.; Ferrer, S. The use of core-shell high-performance liquid chromatography column technology to improve biogenic amine quantification in wine. J. Sci. Food Agric. 2016, 96, 1556–1561. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Cassou, S.; Saurina, J. Derivatization strategies for the determination of biogenic amines in wines by chromatographic and electrophoretic techniques. J. Chromatogr. B 2011, 879, 1270–1281. [Google Scholar]
- Romero, R.; Jönsson, J.Å.; Gázquez, D.; GraciaBagur, M.; Sánchez-Vinas, M. Multivariate optimization of supported liquid membrane extraction of biogenic amines from wine samples prior to liquid chromatography determination as dabsyl derivatives. J. Sep. Sci. 2002, 25, 584–592. [Google Scholar] [CrossRef]
- Pineda, A.; Carrasco, J.; Peña-Farfal, C.; Henríquez-Aedo, K.; Aranda, M. Preliminary evaluation of biogenic amines content in Chilean young varietal wines by HPLC. Food Control 2012, 23, 251–257. [Google Scholar] [CrossRef]
- Özdestan, Ö.; Üren, A. A method for benzoyl chloride derivatization of biogenic amines for high performance liquid chromatography. Talanta 2009, 78, 1321–1326. [Google Scholar] [CrossRef] [PubMed]
- Herbert, P.; Santos, L.; Alves, A. Simultaneous quantification of primary, secondary amino acids and biogenic amines in musts and wines using OPA/3-MPA/FMOC-Cl fluorescent derivatives. J. Food Sci. 2001, 66, 1319–1325. [Google Scholar] [CrossRef]
- Garcıá-Marino, M.; Trigueros, Á.; Escribano-Bailon, T. Influence of oenological practices on the formation of biogenic amines in quality red wines. J. Food Compos. Anal. 2010, 23, 455–462. [Google Scholar] [CrossRef]
- Nouadje, G.; Simeon, N.; Dedieu, F.; Nertz, M.; Pulg, P.; Couderc, F. Determination of twenty eight biogenic amines and amino acids during wine aging by micellar electrokinetic chromatography and laser-induced fluorescence detection. J. Chromatogr. A 1997, 765, 337–343. [Google Scholar] [CrossRef]
- García-Villar, N.; Hernández-Cassou, S.; Saurina, J. Determination of biogenic amines in wines by pre-column derivatization and high-performance liquid chromatography coupled to mass spectrometry. J. Chromatogr. A 2009, 1216, 6387–6393. [Google Scholar] [CrossRef] [PubMed]
- Bach, B.; Le Quere, S.; Vuchot, P.; Grinbaum, M.; Barnavon, L. Validation of a method for the analysis of biogenic amines: Histamine instability during wine sample storage. Anal. Chim. Acta 2012, 732, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Callejon, R.M.; Troncoso, A.M.; Morales, M.L. Determination of amino acids in grape-derived products: A review. Talanta 2010, 81, 1143–1152. [Google Scholar] [CrossRef] [PubMed]
- Jia, S.; Kang, Y.P.; Park, J.H.; Lee, J.; Kwon, S.W. Determination of biogenic amines in Bokbunja (Rubus coreanus Miq.) wines using a novel ultra-performance liquid chromatography coupled with quadrupole-time of flight mass spectrometry. Food Chem. 2012, 132, 1185–1190. [Google Scholar] [CrossRef] [PubMed]
- Mayr, C.M.; Schieberle, P. Development of stable isotope dilution assays for the simultaneous quantitation of biogenic amines and polyamines in foods by LC-MS/MS. J. Agric. Food Chem. 2012, 60, 3026–3032. [Google Scholar] [CrossRef] [PubMed]
- Romano, A.; Klebanowski, H.; La Guerche, S.; Beneduce, L.; Spano, G.; Murat, M.L.; Lucas, P. Determination of biogenic amines in wine by thin-layer chromatography/densitometry. Food Chem. 2012, 135, 1392–1396. [Google Scholar] [CrossRef] [PubMed]
- Ordonez, J.L.; Troncoso, A.M.; García-Parrilla, M.D.C.; Callejon, R.M. Recent trends in the determination of biogenic amines in fermented beverages—A review. Anal. Chim. Acta 2016, 939, 10–25. [Google Scholar] [CrossRef] [PubMed]
- Cunha, S.C.; Faria, M.A.; Fernandes, J.O. Gas chromatography-mass spectrometry assessment of amines in port wine and grape juice after fast chloroformate extraction/derivatization. J. Agric. Food Chem. 2011, 59, 8742–8753. [Google Scholar] [CrossRef] [PubMed]
- Daniel, D.; Dos Santos, V.B.; Vidal, D.T.R.; do Lago, C.L. Determination of biogenic amines in beer and wine by capillary electrophoresis-tandem mass spectrometry. J. Chromatogr. A 2015, 1416, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Martín-Álvarez, P.J.; Marcobal, Á.; Polo, C.; Moreno-Arribas, M.V. Influence of technological practices on biogenic amine contents in red wines. Eur. Food Res. Technol. 2006, 222, 420–424. [Google Scholar] [CrossRef]
- Martìnez-Pinilla, O.; Guadalupe, Z.; Hernàndez, Z.; Ayestaràn, B. Amino acids and biogenic amines in red varietal wines: The role of grape variety, malolactic fermentation and vintage. Eur. Food Res. Technol. 2013, 237, 887–895. [Google Scholar] [CrossRef]
- Torrea, D.; Ancin, C. Influence of yeast strain on biogenic amines content in wines: Relationship with the utilization of amino acids during fermentation. Am. J. Enol. Vitic. 2001, 52, 185–190. [Google Scholar]
- Torrea, D.; Ancín, C. Content of biogenic amines in a Chardonnay wine obtained through spontaneous and inoculated fermentation. J. Agric. Food Chem. 2002, 50, 4895–4899. [Google Scholar] [CrossRef] [PubMed]
- Landete, J.M.; Ferrer, S.; Pardo, I. Biogenic amine production by lactic acid bacteria, acetic bacteria and yeast isolated from wine. Food Control 2007, 18, 1569–1574. [Google Scholar] [CrossRef]
- Caruso, M.; Fiorel, C.; Contursi, M.; Salzano, G.; Paparella, A.; Romano, P. Formation of biogenic amines as criteria for the selection of wine yeasts. World J. Microbiol. Biotechnol. 2002, 18, 159–163. [Google Scholar] [CrossRef]
- Wang, Y.-Q.; Ye, D.-Q.; Zhu, B.-Q.; Wu, G.-F.; Duan, C.-Q. Rapid HPLC analysis of amino acids and biogenic amines in wines during fermentation and evaluation of matrix effect. Food Chem. 2014, 163, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Marcobal, Á.; de las Rivas, B.; Moreno-Arribas, M.V.; Muñoz, R. Multiplex PCR method for the simultaneous detection of histamine, tyramine and putrescine producing lactic acid bacteria in foods. J. Food Prot. 2005, 68, 874–878. [Google Scholar] [CrossRef] [PubMed]
- Vidal-Carou, M.C.; Codony-Salcedo, R.; Mariné-Font, A. Histamine and tyramine in Spanish wines: Relationship with total sulfur dioxide level, volatile acidity and malolactic fermentation intensity. Food Chem. 1990, 35, 217–227. [Google Scholar] [CrossRef]
- López-Rituerto, E.; Avenoza, A.; Busto, J.H.; Peregrina, J.M. NMR Study of histidine metabolism during alcoholic and malolactic fermentations of wine and their influence on histamine production. J. Agric. Food Chem. 2013, 61, 9464–9469. [Google Scholar] [CrossRef] [PubMed]
- Granchi, L.; Romano, P.; Mangani, S.; Guerrini, S.; Vincenzini, M. Production of biogenicamines by winemicroorganisms. Bull. OIV 2005, 78, 595–609. [Google Scholar]
- Vigentini, I.; Romano, A.; Compagno, C.; Merico, A.; Molinari, F.; Tirelli, A.; Foschino, R.; Volonterio, G. Physiological and oenological traits of differentDekkera/Brettanomycesbruxellensisstrains under wine-model conditions. FEMS Yeast Res. 2008, 8, 1087–1096. [Google Scholar] [CrossRef] [PubMed]
- Smit, A.Y.; du Toit, W.J.; Stander, M.; du Toit, M. Evaluating the influence of maceration practices on biogenic amine formation in wine. LWT Food Sci. Technol. 2013, 53, 297–307. [Google Scholar] [CrossRef]
- Marques, A.P.; Leităo, M.C.; San Romăo, M.C. Biogenic amines in wines: Influence of oenological factors. Food Chem. 2008, 107, 853–860. [Google Scholar] [CrossRef]
- Tofalo, R.; Patrignani, F.; Lanciotti, R.; Perpetuini, G.; Schirone, M.; Di Gianvito, P.; Pizzoni, D.; Arfelli, G.; Suzzi, G. Aroma profile of Montepulciano d’Abruzzo wine fermented by single and co-culture starters of autochthonous Saccharomyces and non-saccharomyces yeasts. Front. Microbiol. 2016, 610, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Restuccia, D.; Sicari, V.; Pellicanò, T.M.; Spizzirri, U.G.; Loizzo, M.R. The impact of cultivar on polyphenol and biogenic amine profiles in Calabrian red grapes during winemaking. Food Res. Int. 2017, 102, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Benito, Á.; Calderón, F.; Palomero, F.; Benito, S. Combine use of selected Schizosaccharomyces pombe and Lachancea thermotolerans yeast strains as an alternative to the traditional malolactic fermentation in red wine production. Molecules 2015, 20, 9510–9523. [Google Scholar] [CrossRef] [PubMed]
- Benito, Á.; Jeffares, D.; Palomero, F.; Calderón, F.; Bai, F.-Y.; Bähler, J.; Benito, S. Selected Schizosaccharomyces pombe strains have characteristics that are beneficial for winemaking. PLoS ONE 2016, 11, e0151102. [Google Scholar] [CrossRef] [PubMed]
- Lubbers, M.W.; Rodriguez, S.B.; Honey, N.K.; Thornton, R.J. Purification and characterization of urease from Schizosaccharomyces pombe. Can. J. Microbiol. 1996, 42, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Serradilla, J.A.; Luque de Castro, M.D. Role of lees in wine production: A review. Food Chem. 2008, 111, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Lerm, E.; Engelbrecht, L.; du Toit, M. Malolactic fermentation: The ABC’s of MLF. S. Afr. J. Enol. Vitic. 2010, 31, 186–212. [Google Scholar]
- Gardini, F.; Zaccarelli, A.; Belletti, N.; Faustini, F.; Cavazza, A.; Martuscelli, M.; Mastrocola, D.; Suzzi, G. Factors influencing biogenic amine production by a strain of Oenococcus oeni in a model system. Food Control 2005, 16, 609–616. [Google Scholar] [CrossRef]
- Garai, G.; Dueñas, M.T.; Irastorza, A.; Moreno-Arribas, M.V. Biogenic amine production by lactic acid bacteria isolated from cider. Lett. Appl. Microbiol. 2007, 45, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Lucas, P.M.; Wolken, W.A.M.; Claisse, O.; Lolkema, J.S.; Lonvaud-Funel, A. Histamine-producing pathway encoded on an unstable plasmid in Lactobacillus hilgardii 0006. Appl. Environ. Microbiol. 2005, 71, 1417–1424. [Google Scholar] [CrossRef] [PubMed]
- Du Toit, M.; Engelbrecht, L.; Lerm, E.; Krieger-Weber, S. Lactobacillus: The next generation of malolactic fermentation starter cultures—An overview. Food Bioprocess Technol. 2011, 4, 876–906. [Google Scholar] [CrossRef]
- Garcia-Moruno, E.; Muñoz, R. Does Oenococcus oeni produce histamine? Int. J. Food Microbiol. 2012, 157, 121–129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coton, E.; Rollan, G.; Bertrand, A.; Lonvaud-Funel, A. Histamine-producing lactic acid bacteria in wines: Early detection, frequency and distribution. Am. J. Enol. Vitic. 1998, 49, 199–204. [Google Scholar]
- Landete, J.M.; Ferrer, S.; Polo, L.; Pardo, I. Biogenic amines in wines from three Spanish regions. J. Agric. Food Chem. 2005, 53, 1119–1124. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Arribas, V.; Polo, M.C.; Jorganes, F.; Muñoz, R. Screening of biogenic amine production by lactic acid bacteria isolated from grape must and wine. Int. J. Food Microbiol. 2003, 84, 117–123. [Google Scholar] [CrossRef] [Green Version]
- Berbegal, C.; Benavent-Gil, Y.; Navascués, E.; Pardo, I.; Ferrer, S. Lowering histamine formation in a red Ribera del Duero wine (Spain) by using an indigenous O. oeni strain as a malolactic starter. Int. J. Food Microbiol. 2017, 244, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Arribas, V.; Torlois, S.; Joyex, A.; Bertrand, A.; Lonvaud-Funel, A. Isolation, properties and behaviour of tyramineproducing lactic acid bacteria from wine. J. Appl. Microbiol. 2000, 88, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Marcobal, Á.; de las Rivas, B.; Muñoz, R. Methods for the detection of bacteria producing biogenic amines on foods: A survey. J. Consum. Prot. Food Saf. 2006, 1, 187–196. [Google Scholar] [CrossRef] [Green Version]
- Manfroi, L.; Silva, P.H.A.; Rizzon, L.A.; Sabaini, P.S.; Gloria, M.B.A. Influence of alcoholic and malolactic starter cultures on bioactive amines in Merlot wines. Food Chem. 2009, 116, 208–213. [Google Scholar] [CrossRef]
- Arena, M.E.; Manca, de Nadra, M.C. Biogenic amine production by Lactobacillus. J. Appl. Microbiol. 2001, 90, 158–162. [Google Scholar] [CrossRef] [PubMed]
- Henríquez-Aedo, K.; Duràn, D.; Garcia, A.; Hengst, M.B.; Aranda, M. Identification of biogenic amines-producing lactic acid bacteria isolated from spontaneous malolactic fermentation of chilean red wines. LWT Food Sci. Technol. 2016, 68, 183–189. [Google Scholar] [CrossRef]
- Lonvaud-Funel, A. Biogenic amines in wines: Role of lactic acid bacteria. FEMS Microbiol. Lett. 2001, 199, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Martín, F.; Seseña, S.; Izquierdo, P.M.; Palop, M.L. Are Enterococcus populations present during malolactic fermentation of red wine safe? Food Microbiol. 2014, 42, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Capozzi, V.; Ladero, V.; Beneduce, L.; Fernández, M.; Alvarez, M.A.; Benoit, B.; Laurent, B.; Grieco, F.; Spano, G. Isolation and characterization of tyramine-producing Enterococcus faecium strain from red wine. Food Microbiol. 2011, 28, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, P.; Izquierdo, P.M.; Seseña, S.; Palop, M.L. Bacterial biodiversity and dynamics during malolactic fermentation of Tempranillo wines as determined by a culture-independent method (PCR-DGGE). Appl. Microbiol. Biotechnol. 2010, 86, 1555–1562. [Google Scholar] [CrossRef] [PubMed]
- Martín, B.; Garriga, M.; Hugas, M.; Bover-Cid, S.; Veciana-Nogués, M.T.; Aymerich, T. Molecular, technological and safety characterization of gram-positive catalase-positive cocci from slightly fermented sausages. Int. J. Food Microbiol. 2006, 107, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Benavent-Gil, Y.; Berbegal, C.; Lucio, O.; Pardo, I.; Ferrer, S. A new fear in wine: Isolation of Staphylococcus epidermidis histamine producer. Food Control 2016, 62, 142–149. [Google Scholar] [CrossRef]
- Ruiz, P.; Izquierdo, P.M.; Seseña, S.; Palop, M.L. Selection of autochthonous Oenococcus oeni strains according to their oenological properties and vinification results. Int. J. Food Microbiol. 2010, 137, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Orte, P.; Lapeña, A.C.; Peña-Gallego, A.; Astrain, J.; Baron, C.; Pardo, I. Biogenic amine determination in wine fermented in oak barrels. Factors affecting formation. Food Res. Int. 2008, 41, 697–706. [Google Scholar] [CrossRef]
- Capozzi, V.; Russo, P.; Ladero, V.; Fernández, M.; Fiocco, D.; Alvarez, M.A.; Grieco, F.; Spano, G. Biogenic amines degradation by Lactobacillus plantarum: Toward a potential application in wine. Front. Microbiol. 2012, 3, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smit, A.Y.; Engelbrecht, L.; duToit, M. Managing your wine fermentation to reduce the risk of biogenic amine formation. Front. Microbiol. 2012, 3, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buteau, C.; Duitschaever, C.L.; Ashton, G.C. A study of the biogenesis of amines in a Villard Noir Wine. Am. J. Enol. Vitic. 1984, 35, 228–236. [Google Scholar]
- Costantini, A.; Vaudano, E.; delPrete, V.; Danei, M.; García-Moruno, E. Biogenic amine production by contaminating bacteria found in starter preparations used in winemaking. J. Agric. Food Chem. 2009, 57, 10664–10669. [Google Scholar] [CrossRef] [PubMed]
| Sample | BAs | Microorganism/Spontaneous | Analytical Method | Total BAs Content (mg/L) (Harvest Year) | Reference |
|---|---|---|---|---|---|
| Tempranillo | HIS; MET; ETH; TYR; PHE; PUT; CAD | Spontaneous | Derivatization with o-phthaldialdehyde and separation with HPLC coupled to fluorescence detector. | 22.37 (2004) | [47] |
| Merlot | AGM; ETA; ETH; PUT; TYR | S. cerevisiae (106 cells/mL) | Derivatization with o-phthaldialdehyde and separation with HPLC coupled to fluorescence detector. | 6.69 (2004) 25.87 (2005) | [19] |
| Syrah | 9.70 (2004) 23.67 (2005) | ||||
| Cabernet Franc. | 9.65 (2004) 14.02 (2005) | ||||
| Montepulciano | 12.01 (2004) 21.31 (2005) | ||||
| Sangiovese | 10.96 (2004) 18.11 (2005) | ||||
| Carmenere | 8.94 (2004) 20.59 (2005) | ||||
| Cesanese d’Affile | 15.62 (2004) 27.53 (2005) | ||||
| Montepulciano d’Abruzzo | CAD; TRY; PHE; TYR; HIS; ETA; ETH; PUT | S. cerevisiae SRS1 (106 cells/mL) | Derivatization with dansyl chloride and separation by HPLC coupled with PDA detector. | 21–24 (2011) | [61] |
| Arvino | PHE; PUT; HIS; TYR; SPD; SPM | Spontaneous | Derivatization with dansyl chloride and separation with RP-LC-UV with gradient elution (solvents water and acetonitrile). | 23.7 (2016) | [62] |
| Gaglioppo | 41.9 (2016) | ||||
| Greco Nero | 44.0 (2016) | ||||
| Magliocco Canino | 63.1 (2016) | ||||
| Magliocco Dolce | 36.6 (2016) | ||||
| Nocera | 46.8 (2016) | ||||
| Tempranillo | HIS; AGM; SPD; TYR; PUT; TRY; CAD; PHE | Spontaneous | Derivatization with diethyl ethoxy methylene malonate and separation by RP-LC-UV with gradient elution. | 14.6 (2009) 6.9 (2010) | [48] |
| Monastel | 14.3 (2009) 10.2 (2010) | ||||
| Maturana Tinta de Navarrete | 13.9 (2009) 9.6 (2010) | ||||
| Aglianico of Vulture | ETA; MET; AGM; TRY; PHE; PUT; CAD; HIS | Dekkera/B. bruxellensis (5%) | Derivatization with dansyl chloride and separation with RP-LC-UV with gradient elution (solvents water and acetonitrile). | 15.01 (2000) | [52] |
| S. cerevisiae (5%) | 12.4 (2000) | ||||
| Kloeckeraapiculata (5%) | 6.21 (2000) | ||||
| Candida stellata (5%) | 6.73 (2000) | ||||
| Metschnikowiapulcherrima (5%) | 9.60 (2000) | ||||
| Italian red wine | PUT; CAD; SPM | Dekkera/B. bruxellensis (CBS2336 and CBS4601) | Derivatization with dabsyl-chloride and separation with separation with RP-LC-UV with gradient elution (Water and acetonitrile). | 0.40 (2006) | [58] |
| Tempranillo | HIS; TYR; PHE; PUT; CAD | Kluyveromycesthermotolerans/Schizosaccharomyces pombe V2 | UHPLC coupled to fluorescence detector and separation with separation gradient elution (A: methanol/acetonitrile—B: sodium acetate/tetrahydrofuran). | 2.89 (NR) | [63] |
| Selected S. pombe (JB899/Y470) | 1.47 (NR) | [64] | |||
| Selected S. pombe (JB917/CBS1057) | 1.55 (NR) | ||||
| Selected S. pombe (JB873/NCYC3422) | 1.39 (NR) | ||||
| Selected S. pombe V1 | 1.47 (NR) | ||||
| Non-Selected S. pombe (936/CECT12774) | 1.50 (NR) | ||||
| Non-Selected S. pombe (935/CECT12774) | 1.51 (NR) |
| Sample | BAs | Microorganisms | Analytical Method | Total BAs Content (mg/L) (Harvest Year) | Reference |
|---|---|---|---|---|---|
| Periquita | TYR; PUT; HIS | Spontaneous | Derivatization with o-phthaldialdehyde and separation with HPLC coupled to fluorescence detector. | 27.6 (2006) 7.0 (TYR) (2006) | [60] |
| CMS2 (inducer of MLF) | 2.0 (TYR) (2006) | ||||
| Merlot | AGM; ETA; ETH; PUT; TYR | O. oeni (5 × 106 cells/mL) | Derivatization with o-phthaldialdehyde and separation with HPLC coupled to fluorescence detector. | 6.59 (2004) 37.55 (2005) | [19] |
| Syrah | 8.73 (2004) 47.59 (2005) | ||||
| Cabernet Franc. | 6.88 (2004) 31.26 (2005) | ||||
| Montepulciano | 8.43 (2004) 33.85 (2005) | ||||
| Sangiovese | 8.16 (2004) 34.09 (2005) | ||||
| Carmenere | 8.63 (2004) 29.71 (2005) | ||||
| Cesanese d’Affile | 13.64 (2004) 37.80 (2005) | ||||
| Tempranillo | HIS; AGM; SPD; TYR; PUT; TRY; CAD; PHE | Spontaneous | Derivatization with diethyl ethoxy methylene malonate and separation by RP-LC-UV with gradient elution. | 2.227 (2009) 1.313 (2010) | [48] |
| Monastel | 3.772 (2009) 2.236 (2010) | ||||
| Maturana Tinta de Navarrete | 5.019 (2009) 1.646 (2010) | ||||
| Merlot | PUT; SPD; SPM; AGM; CAD; SRT; HIS; TYR; TRY; PHE | Spontaneous * | Derivatization with o-phthaldialdehyde and separation with HPLC coupled to fluorescence detector. | <0.40 (2008) | [79] |
| O. oeni DSM 7008 (6 mg/L) * | 1.93 | ||||
| O. oeni DSM 12923 (6 mg/L) * | 15.5 | ||||
| L. plantarum DSM 4361 (200 mg/L) * | 14.3 | ||||
| Yeast * | 7.94 | ||||
| Spontaneous ** | 12.4 | ||||
| O. oeni DSM 7008 (6 mg/L) ** | 7.4 | ||||
| O. oeni DSM 12923 (6 mg/L) ** | 7.7 | ||||
| L. plantarum DSM 4361 (200 mg/L) ** | 24.1 | ||||
| Yeast ** | 12.9 | ||||
| Spontaneous * | 6.88 | ||||
| O. oeni DSM 7008 (6 mg/L) *** | 9.08 | ||||
| O. oeni DSM 12923 (6 mg/L) *** | 6.23 | ||||
| L. plantarum DSM 4361 (200 mg/L) *** | 14.6 | ||||
| Yeast *** | 9.20 | ||||
| Spontaneous † | 6.43 | ||||
| O. oeni DSM 7008 (6 mg/L) † | 6.13 | ||||
| O. oeni DSM 12923 (6 mg/L) † | 9.81 | ||||
| L. plantarum DSM 4361 (200 mg/L) † | 17.7 | ||||
| Arvino | PHE; PUT; HIS; TYR; SPD; SPM | Spontaneous | Derivatization with dansyl chloride and separation with RP-LC-UV with gradient elution (solvents water and acetonitrile). | 30.0 (2015) | [62] |
| Gaglioppo | 50.3 (2015) | ||||
| Greco Nero | 54.4 (2015) | ||||
| Magliocco Canino | 74.1 (2015) | ||||
| Magliocco Dolce | 43.3 (2015) | ||||
| Nocera | 54.5 (2015) | ||||
| Tempranillo | HIS; MET; ETH; TYR; PHE; PUT; CAD | Spontaneous | Derivatization with o-phthaldialdehyde and separation with HPLC coupled to fluorescence detector. | 22.37 (2004) | [47] |
| Commercial malolacticbacteria | 14.75 (2004) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Restuccia, D.; Loizzo, M.R.; Spizzirri, U.G. Accumulation of Biogenic Amines in Wine: Role of Alcoholic and Malolactic Fermentation. Fermentation 2018, 4, 6. https://doi.org/10.3390/fermentation4010006
Restuccia D, Loizzo MR, Spizzirri UG. Accumulation of Biogenic Amines in Wine: Role of Alcoholic and Malolactic Fermentation. Fermentation. 2018; 4(1):6. https://doi.org/10.3390/fermentation4010006
Chicago/Turabian StyleRestuccia, Donatella, Monica Rosa Loizzo, and Umile Gianfranco Spizzirri. 2018. "Accumulation of Biogenic Amines in Wine: Role of Alcoholic and Malolactic Fermentation" Fermentation 4, no. 1: 6. https://doi.org/10.3390/fermentation4010006