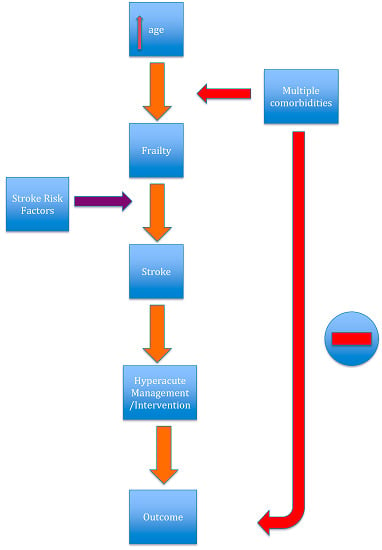
Stroke in Frail Older People
Abstract
:1. Ageing and Stroke
2. Stroke Epidemiology
3. Stroke Recognition
4. Pre-Stroke Function
5. Stroke Aetiology and Risk Factors
6. Hypertension
7. Atrial Fibrillation
8. Antiplatelet Use
9. Cholesterol
10. Access to Stroke Care
Hyper Acute Stroke Management
11. Intravenous Thrombolysis
12. Endovascular Thrombectomy
13. Clot Evacuation
14. Hemicrainectomy
15. Carotid Endarterectomy/Carotid Stenting
16. Outcome
17. Ethical Issues
18. Life Events
19. Death
20. When to Consider End of Life Care
21. Stroke Complications
22. Confusion
23. Mouth Care/Swallowing/Nutrition
24. Conclusions
Conflicts of Interest
References
- Suzman, R.; Beard, J. Global Health and Aging National Institute on Aging National Institutes of Health, U.S. Department of Health and Human Services; World Health Organization (WHO): Geneva, Switzerland, 2011. [Google Scholar]
- Truelsen, T.; Piechowski-Jozwiak, B.; Bonita, R.; Bogousslavsky, J.; Boysen, G. Stroke incidence and prevalence in Europe: A review of available data. Eur. Neurol. 2006, 13, 581–598. [Google Scholar] [CrossRef] [PubMed]
- Breen, L.; Phillips, S.M. Skeletal muscle protein metabolism in the elderly: Interventions to counteract the “anabolic resistance” of ageing. Nutr. Metab. 2011, 8, 68–79. [Google Scholar] [CrossRef] [PubMed]
- Mateen, F.J.; Buchan, A.M.; Hill, M.D. Outcomes of thrombolysis for acute ischemic stroke in octogenarians versus nonagenarians. Stroke 2010, 41, 1833–1835. [Google Scholar] [CrossRef] [PubMed]
- Gilleard, C.; Higgs, P. Third age and baby boomers. Int. J. Ageing Later Life 2007, 2, 13–30. [Google Scholar] [CrossRef]
- Di Carlo, A.; Lamassa, M.; Pracucci, G.; Basile, A.M.; Trefoloni, G.; Vanni, P.; Wolfe, C.D.A.; Tilling, K.; Ebrahim, S.; Inzitari, D. Stroke in the very old. Stroke 1999, 30, 2313–2319. [Google Scholar] [CrossRef] [PubMed]
- Walston, J.; McBurnie, M.A.; Newman, A.; Tracy, R.P.; Kop, W.J.; Hirsch, C.H.; Gottdiener, J.; Fried, L.P. Fraiulty and activation of the inflammation and coagulation systems with and without clinical comorbidities. Arch. Intern. Med. 2002, 162, 2333–2341. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.H.; Weiss, D.; Sourtal, N.; Karunananthan, S.; Quail, J.M.; Wolfson, C.; Bergman, H. Frailty and its association with comorbidity in a community dwelling sample of seniors in Montreal: A cross-sectional study. Aging Clin. Exp. Res. 2010, 22, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Rockwood, K.; Hubbard, R. Frailty and the Geriatrician. Age Ageing 2004, 22, 429–430. [Google Scholar] [CrossRef] [PubMed]
- Feigin, V.L.; Forouzanfar, M.H.; Krishnamurthi, R.; Mensah, G.A.; Connor, M.; Bennett, D.A.; Moran, A.E.; Sacco, R.L.; Anderson, L.; Truelsen, T.; et al. Global and regional burden of stroke during 1990–2010: Findings from the Global burden of disease study 2010. Lancet 2014, 383, 245–254. [Google Scholar] [CrossRef]
- Kadojic, D.; Dikanovic, M.; Bitunac, M.; Vuletic, V.; Cengic, L.; Bijelic, B.R. Epidemiology of stroke. Period. Biol. 2012, 114, 253–257. [Google Scholar]
- Bonita, R.; Beaglehole, R. Stroke prevention in poor countries. Stroke 2007, 38, 2871–2872. [Google Scholar] [CrossRef] [PubMed]
- Fonarow, G.C.; Reeves, M.J.; Zhao, X.; Olson, D.W.M.; Smith, E.E.; Saver, J.L.; Schwamm, L.H. Age-related differences in characteristics, performance measures, treatment trends, and outcomes in patients with ischaemic stroke. Circulation 2010, 121, 879–891. [Google Scholar] [CrossRef] [PubMed]
- Royal College of Physicians. Clinical Effectiveness and Evaluation Unit on Behalf of the Intercollegiate Stroke Working Party; The Sentinel Stroke National Audit Programme (SSNAP): London, UK, 2016. [Google Scholar]
- Kammersgaard, L.P.; Jorgensen, H.S.; Reith, J.; Nakayama, H.; Pedersen, P.M.; Olsen, T.S. Short- and long-term prognosis for very old stroke patients. The Copenhagen Stroke study. Age Ageing 2004, 33, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Shock, N.W.; Greulich, R.C.; Andres, R.; Lakatta, E.G.; Arenberg, D.; Tobin, J.D. Normal Human Aging: The Baltimore Longitudinal Study of Aging; US Government Printing Office: Washington, DC, USA, 1984.
- Andrews, G.; Clark, M.; Luszcz, M. Successful aging in the Australian longitudinal study of aging: Applying the McArthur Model cross-nationally. J. Soc. Issues 2002, 58, 749–765. [Google Scholar] [CrossRef]
- Pohjasvaara, T.; Erkinjuntti, T.; Vataja, R.; Kaste, M. Comparison of stroke features and disability in daily life in patients with ischaemic stroke aged 55–70 and 71–85 years. Stroke 1997, 28, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Denti, L.; Agosti, M.; Francescheni, M. Outcome predictors of rehabilitation for first stroke in the elderly. Eur. J. Phys. Rehabil. Med. 2008, 44, 3–11. [Google Scholar]
- Jeng, J.S.; Huang, S.J.; Tang, S.C.; Yip, P.K. Predictors of survival and functional outcome in acute stroke patients admitted to the stroke intensive care unit. J. Neurol. Sci. 2008, 270, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Liou, A.K.F.; Clark, R.S.; Henshall, D.C.; Yin, X.-M.; Chen, J. To die or not to die for neurons in ischaemia, traumatic brain injury and epilepsy: A review on the stress-activated signalling and apoptic pathways. Prog. Neurobiol. 2003, 69, 103–142. [Google Scholar] [CrossRef]
- Hebb, A.L.O.; Brandman, D.; Shanker, J.; Hebb, A.O. Visualizing recovery of cognitive function in stroke. J. Behav. Brain Sci. 2013, 3, 641–652. [Google Scholar] [CrossRef]
- Bamford, J.; Sandercock, P.; Dennis, M.; Burn, C.; Warlow, C. Classification and natural history of clinically identifiable subtypes of cerebral infarction. Lancet 1991, 337, 1521–1526. [Google Scholar] [CrossRef]
- Adams, H.P.; Bendixen, B.H.; Kappelle, L.J.; Biller, J.; Love, B.B.; Gordon, D.L.; Marsh, E.E. Classification of subtype of acute ischaemic stroke. Definitions for use in a clinical trial. TOAST Trial of Org 10172 in acute stroke treatment. Stroke 1993, 24, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Pasina, L.; Djade, C.D.; Lucca, U.; Nobili, A.; Tettamanti, M.; Franchi, C.; Salerno, F.; Corrao, S.; Marengoni, A.; Iorio, A.; et al. Association of anticholinergic burden with cognitive and functional status in a cohort of hospitalized elderly: Comparison of the anticholinergic burden scale and the anticholinergic risk scale. Drugs Aging 2013, 30, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.-H.; Wen, Y.-W.; Chen, L.-K.; Hsiao, F.-Y. Effect of polypharmacy, potentially inappropriate medications and anticholinergic burden on clnical outcomes: A retrospective cohort study. CMAJ 2015, 187, E130–E135. [Google Scholar] [CrossRef] [PubMed]
- Warwick, J.; Falaschetti, E.; Rockwood, K.; Mitiniski, A.; Thijs, L.; Beckett, N.; Bulpitt, C.; Peters, R. No evidence that frailty modifies the positive impact of antihypertensives in very elderly people: An investigation of the impact of frailty upon treatment effect in the hypertension in the very elderly trial (HYVET) study. A double blind, placebo controlled study of antihypertensives in people with hypertension aged 80 and over. BMC Med. 2015, 13, 78. [Google Scholar] [CrossRef]
- O’Mahony, D.; O’Sullivan, D.; Byrne, S.; O’Connor, M.N.; Ryan, C.; Gallagher, P. STOPP/START criteria for potentially inappropriate prescribing in oler people version 2. Age Ageing 2015, 44, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Won, C.W.; Kim, S. Use of frailty in deciding clinical treatment goals for chronic disease in elderly patients in the community. JAMDA 2016, 17, 967–969. [Google Scholar] [CrossRef] [PubMed]
- Lane, D.A.; Lip, G.Y.H. Use of CHA2DS2-VASC and HAS-BLED scores to aid decision making for thromboprophylaxis in non-valvular atrial fibrillation. Circulation 2012, 126, 860–865. [Google Scholar] [CrossRef] [PubMed]
- Perera, V.; Bajorek, B.V.; Matthews, S.; Hilmer, S.N. The impact of frailty on the utilization of antithrombotic therapy in older patients. Age Ageing 2009, 38, 156–162. [Google Scholar] [CrossRef] [PubMed]
- La Brooy, B.; Ho, P. New oral anticoagulants: An approach in older people. J. Pharm. Pract. Res. 2015, 45, 217–225. [Google Scholar] [CrossRef]
- Summerbell, J.; Yelland, C.; Woodhouse, K. The kinetics of plasma esterase in relation to old age and frailty. Age Ageing 1990, 12, 128–130. [Google Scholar] [CrossRef]
- Lovelock, C.E.; Cordonnier, C.; Naka, H.; Salman, R.A.; Sudlow, C.L.M.; The Edinburgh Study Group; Sorimachi, T.; Werring, D.J.; Gregoire, S.M.; Imaizumi, T.; et al. Antithrombotic drug use, cerebral microbleeds and intracerebral haemorrhage. Stroke 2010, 41, 1222–1228. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Care Excellence (NICE). Cardiovascular Disease: Risk Management and Reduction, Including Lipid Modification; National Institute for Health and Care Excellence (NICE): London, UK, 2014. [Google Scholar]
- Rudd, A.G.; Hoffman, A.; Down, C.; Pearson, M.; Lowe, D. Access to stroke care in England, Wales and Northern Ireland: The effect of age, gender and weekend admission. Age Ageing 2007, 36, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Stroke Foundation New Zealand. New Zealand Clinical Guidelines for Stroke Management; Stroke Foundation New Zealand: Wellington, New Zealand, 2010. [Google Scholar]
- National Stroke Foundation. Clinical Guidelines for Stroke Management; National Stroke Foundation: Melbourne, Australia, 2010. [Google Scholar]
- European Stroke Organisation Writing Committee. Guidelines for management of ischaemic stroke and transient ischaemic attack 2008. Cerebrovasc. Dis. 2008, 25, 457–507. [Google Scholar]
- Sapsonik, G.; Cote, R.; Philips, S.; Gubitz, G.; Bayer, N.; Minuk, J.; Black, S. Stroke ouitcome in those over 80. Stroke 2008, 39, 2310–2317. [Google Scholar] [CrossRef] [PubMed]
- Kernan, W.N.; Ovbiagele, B.; Black, H.R.; Bravata, D.M.; Chimowitz, M.I.; Ezekowitz, M.D.; Fang, M.C.; Fisher, M.; Furie, K.L.; Heck, D.V.; et al. Guidelines for the prevention of stroke in patients with stroke and transient ischaemic attack. Stroke 2014, 45, 2160–2236. [Google Scholar] [CrossRef] [PubMed]
- Intercollegiate Stroke. Working Party. National Clinical Guidelines for Stroke, 5th ed.; Royal College of Physicians: London, UK, 2016. [Google Scholar]
- The ATLANTIS, ECASS, NINDS RT-PA Study Group Investigators. Association of outcome with early stroke treatment: Pooled analysis of ATLANTIS; ECASS and NINDS rt-PA stroke trials. Lancet 2004, 363, 768–774. [Google Scholar]
- Whitely, W.N.; Thompson, D.; Murray, G.; Cohen, G.; Lindley, R.L.; Wardlaw, J.; Sandercock, P. Targetting rtPA in acute ischaemic stroke based on risk of intracranial haemorrhage and poor functional outcome: An analysis of the IST-3 trial. Stroke 2014, 45, 1000–1006. [Google Scholar] [CrossRef] [PubMed]
- Ford, G.A.; Ahmed, N.; Azevedo, E.; Grond, M.; Larrue, V.; Lindsberg, P.J.; Toni, D.; Wahlgren, N. Intravenous Alteplase for stroke in those older than 80 years old. Stroke 2010, 41, 2568–2574. [Google Scholar] [CrossRef] [PubMed]
- Berrouschot, J.; Rother, J.; Glahn, J.; Kucinski, T.; Fiechler, J.; Thomalla, G. Outcome and severe hemorrhagic complications of intravenous thrombolysis with tissue plasminogen activator in very old (≥80 years) stroke patients. Stroke 2005, 36, 2421–2425. [Google Scholar] [CrossRef] [PubMed]
- Wahlgren, N.; Ahmed, N.; Davalos, A.; Hacke, W.; Millan, M.; Muir, K.; Roine, R.O.; Toni, D.; Lees, K.R. Thrombolysis with alteplase 3–4.5 h after acute ischaemic stroke (SITS-ISTR): An observational study. Lancet 2008, 372, 1303–1309. [Google Scholar] [CrossRef]
- Kurr, W.; Aguilar-Pere, M.; Niehaus, L.; Fischer, S.; Schmid, E.; Bazner, H.; Henkes, H. Predictors of outcome after mechanical thrombectomy for anterior circulation large vessel occlusion in patients aged ≥80 years. Cerebrovasc. Dis. 2013, 36, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Gratz, P.P.; Jung, S.; Schroth, G.; Gralla, J.; Mordasini, P.; Hsieh, K.; Heldner, M.R.; Mattle, H.P.; Mono, M.-L.; Fischer, U.; et al. Outcome of standard and high-risk patients with acute circulation stroke after stent retriever thrombectomy. Stroke 2014, 45, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, L.B.; Samsa, G.P. Reliability of the National Institutes of Health Stroke Scale. Stroke 1997, 28, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Finlayson, O.; John, V.; Yeung, R.; Dowlatshahi, D.; Howard, P.; Zhang, L.; Swartz, R.; Aviv, R. Interobserver agreement of ASPECT score distribution for noncontrast CT, CT angiography, and CT perfusion in acute stroke. Stroke 2013, 44, 234–236. [Google Scholar] [CrossRef] [PubMed]
- Hassan, A.E.; Chaidhry, S.A.; Grigoryan, M.; Tekle, W.G.; Qureshi, A.I. National trends in utilization and outcomes of endovascular treatment of acute ischaemic stroke patients in the mechanical thrombectomy era. Stroke 2012, 43, 3012–3017. [Google Scholar] [CrossRef] [PubMed]
- Flint, A.C.; Cullen, S.P.; Faigeles, B.S.; Ra, V.A. Predicting long term outcome after endovascular stroke treatment: The totalled health risks in vascular events score. AJNR 2010, 31, 1192–1196. [Google Scholar] [CrossRef] [PubMed]
- Mendelow, A.D.; Gregson, B.A.; Fernandes, H.M.; Murray, G.D.; Teasdale, D.; Hope, T.; Abbas, K.; Shaw, M.D.M.; Barer, D.H. Early surgery vs. initial conservative treatment in patients with spontaneous intracerebral haematomas in the International Surgical Trial in Intracerebral Haemorrhage (STICH): A randomised trial. Lancet 2005, 365, 387–397. [Google Scholar] [CrossRef]
- Morgan, T.; Zuccarello, M.; Narayan, R.; Keyl, P.; Hanley, D. Preliminary findings of the minimally-invasive surgery plus rtPA for intracerebral haemorrhage evacuation (MISTIE) clinical trial. Acta Neurochir. Suppl. 2008, 105, 147–151. [Google Scholar] [PubMed]
- Holtkamp, M.; Buchheim, K.; Unterberg, A.; Hoffman, O.; Schielke Wber, J.R.; Mashur, F. Hemicraniectomy in elderly patients with space occupying media infarction: Improved survival but poor functional outcome. JNNP 2001, 70, 226–228. [Google Scholar] [CrossRef]
- Cheung, A.; Telaghani, C.K.; Wang, J.; Yang, Q.; Mosher, T.J.; Reichwein, R.K.; Cockroft, K.M. Neurological recovery after decompressive crainiectomy for massive ischaemic stroke. Neurocrit. Care 2005, 3, 216–223. [Google Scholar] [CrossRef]
- Juttler, E.; Bosel, J.; Amiri, H.; Schiller, P.; Limprecht, R.; Hacke, W.; Unterberg, A.; DESTINY II Study Group. DESTINY II: Decompressive surgery for the treatment of malignant infarction of the middle cerebral artery II. Int. J. Stroke 2011, 6, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Flores, S.; Berge, E.; Whittle, I.R. Surgical decompression for cerebral oedema in acute ischaemic stroke (Review). Cochrane Database Syst. Rev. 2012. [Google Scholar] [CrossRef]
- Inamasu, J.; Kaito, T.; Watabe, T.; Ganaha, T.; Yamada, Y.; Tanaka, T.; Imizu, S.; Hayashi, T.; Hayakawa, M.; Kato, Y.; et al. Decompressive hemicraniectomy for malignant hemispheric stroke in the elderly: Comparison of outcomes between individuals 61–70 and >70 years of age. J. Stroke Cerebrovasc. Dis. 2013, 22, 1350–1354. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, G.C.; Eliasziw, M.; Barr, H.W.K.; Clagett, G.P.; Barnes, R.W.; Wallace, M.C.; Taylor, D.W.; Haynes, B.; Finan, J.W.; Hachinski, V.C.; et al. The North American Symptomatic Carotid Endarterectomy Trial. Stroke 1999, 30, 1751–1758. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.T.; Comerota, A.J.; Tzilinis, A.; Daoud, Y.; Hammerling, J. Carotid endarterectomy in octogenarians: Does increased age indicate “high risk?”. J. Vasc. Surg. 2005, 41, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Darsalia, V.; Heldmann, U.; Lindvall, O.; Kokaia, Z. Stroke-induced neurogenesis in aged brain. Stroke 2005, 36, 1790–1795. [Google Scholar] [CrossRef] [PubMed]
- Barritt, A.W.; Smithard, D.G. Role of cerebral cortex plasticity in the recovery of swallowing function following dysphagic stroke. Dysphagia 2009, 24, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Denti, L.; Scoditti, U.; Tonelli, C.; Saccavini, M.; Caminti, C.; Valcavi, R.; Benatti, M.; Ceda, G.P. The poor outcome of ischaemic stroke in very old people: A cohort study and its determinants. JAGS 2010, 58, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Knecht, S.; Hesse, S.; Oster, P. Rehabilitation after stroke. Dtsch. Arztebl. Int. 2011, 108, 600–606. [Google Scholar] [PubMed]
- Ones, K.; Yalcinkaya, E.Y.; Toklu, B.C.; Caglar, N. Effects of age, gender, and cognitive, functional and motor status on functional outcomes of stroke rehabilitation. NeuroRehabilitation 2009, 25, 241–249. [Google Scholar] [PubMed]
- Van Oostenbrugge, R.J.; Hupperts, R.M.M.; Ladder, J. Thrombolysis for acute stroke with special emphasis on the very old: Experience from a single Dutch Centre. J. Neurol. Neurosurg. Psychiatry 2006, 77, 375–377. [Google Scholar] [CrossRef] [PubMed]
- Ergeletzis, D.; Kevorkian, C.G.; Rintala, D. Rehabilitation of the older stroke patient. Am. J. Phys. Med. Rehabil. 2002, 81, 881–889. [Google Scholar] [CrossRef] [PubMed]
- Paolucci, S.; Antonucci, G.; Troisi, E.; Bragoni, M.; Coiro, P.; De Angelis, D.; Pratesi, L.; Venturiero, V.; Grasso, M.G. Aging and stroke rehabilitation Cerebrovasc. Dis. 2003, 15, 98–105. [Google Scholar]
- Kong, K.H.; Chua, K.S.G.; Tow, A.P. Clinical charcteristics and functional outcomes of stroke patients 75 years and older. Arch. Phys. Med. Rehabil. 1998, 79, 1535–1539. [Google Scholar] [CrossRef]
- Faircloth, C.A.; Boylstein, C.; Rittman, M.; Young, M.E.; Gubrium, J. Sudden illness and biographical flow in narratives of stroke recovery. Soc. Health Illn. 2004, 26, 242–261. [Google Scholar] [CrossRef] [PubMed]
- Braijkovic, L.; Godan, A.; Godan, L. Quality of life after stroke in old age: Comparison of persons living in nursing home ad those living in their own home. Croat Med. J. 2009, 50, 182–890. [Google Scholar] [CrossRef]
- Litwin, H. Social network type and morale in old age. Gerontoligist 2001, 41, 516–524. [Google Scholar] [CrossRef]
- Fleming, J.; Farquhar, M.; Cambridge City over-75s Cohort (CC75C) Study Collaboration; Brayne, C.; Barclay, S. Death and the oldest old: Attitudes and preferences for end of life care—Qualitative research within a population-based cohort study. PLoS ONE 2016, 11, e0150686. [Google Scholar] [CrossRef] [PubMed]
- Baijens, L.; Clave, P.; Cras, P.; Ekberg, O.; Forster, A.; Kolb, G.; Leners, J.C.; Maseiro, S.; Mateos, J.; Ortega, O.; et al. ESSD white paper: Oropharyngeal dysphagia as a Geriatric Syndrome. Clin. Interv. Aging 2016, 11, 1403–1428. [Google Scholar] [CrossRef] [PubMed]
- Tyrrell, P.; Smithard, D.G. Fatigue after stroke. Therapy 2005, 2, 865–869. [Google Scholar] [CrossRef]
- Barritt, A.W.; Smithard, D.G. Targetting fatigue in stroke patients. ISRN Neurol. 2011. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, A.-C.; Lindgren, I.; Hallstrom, B.; Norrving, B.; Lindgren, A. Determinates of quality of life in stroke survivors and their informal caregivers. Stroke 2005, 36, 803–808. [Google Scholar] [CrossRef] [PubMed]
- Hilari, K.; Needle, J.J.; Harrison, K. What are important factors in health related quality of life for people with aphasia? A systematic review. Arch. Phys. Med. Rehabil. 2012, 93, S86–S95. [Google Scholar] [CrossRef] [PubMed]
- Gusatfason, Y.; Olson, T.; Eriksson, S.; Asplund, K.; Bucht, G. Acute confusion states (Delirium) in stroke patients. Cerebrovasc. Dis. 1991, 1, 257–264. [Google Scholar] [CrossRef]
- Shi, Q.; Presutti, R.; Selchen, D.; Saposnik, G. Delirium in acute stroke. Stroke 2012, 43, 645–649. [Google Scholar] [CrossRef] [PubMed]
- Bray, B.D.; Smith, C.J.; Clod, G.C.; Enderby, P.; James, M.; Paley, L.; Tyrrell, P.; Wolfe, C.D.A.; Rudd, A.G. The association between delays in screening for and assessing dysphagia after acute stroke, and the risk of stroke-associated pneumonia. J. Neurol. Neurosurg. Psychiatry 2016. [Google Scholar] [CrossRef] [PubMed]
- Rofes, L.; Arrelow, V.; Almirall, J.; Cabre, M.; Campins, L.; Garcia-Peris, P.; Speyer, R.; Clave, P. Diagnosis and management of oropharyngeal dysphagia and its nutritional and respiratory complications in the elderly. Gastroeneterol. Res. Pract. 2011. [Google Scholar] [CrossRef] [PubMed]
- Gosney, M.; Martin, M.V.; Wright, A.E. The role of selective decontamination of the digestive tract in acute stroke. Age Ageing 2006, 33, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Kalra, L.; Irshad, S.; Hodsoll, J.; Simpson, M.; Gulliford, M.; Smithard, D.; Patel, A.; Rebello-Mesa, I. Prophylactic antibiotics after acute stroke for reducing pneumonia in patients with dysphagia (STROKE-INF): A prospective, cluster-randomised, open-label, masked endpoint, controlled clinical trial. Lancet 2015, 386, 1835–1844. [Google Scholar] [CrossRef]
- Westendorp, W.F.; Vermeij, J.-D.; Zock, E.; Hooijenga, I.; Kruyt, N.D.; Bosboom, H.J.L.; Kwa, V.I.; Weisfelt, M.; Remmers, M.J.; ten Houten, R.; et al. The preventive antibiotics in stroke study (PASS): A pragmatic randomised open-label masked endpoint clinical trial. Lancet 2015. [Google Scholar] [CrossRef]
- Warunsevitane, A.; Karunatilake, D.; Sim, J.; Lally, F.; Roffe, C. Safety and effect of metoclopramide to prevent pneumonia in patients with stroke fed via nasogastric tube. Stroke 2015, 46, 454–460. [Google Scholar] [CrossRef] [PubMed]
- European Society for Swallowing Disorders (ESSD). ESSD—Position Statement: Screening, Diagnosis and treatment of oropharyngeal dysphagia in stroke patients. Dysphagia 2013, 28, 280–335. [Google Scholar]
- Yoshikawa, M.; Yoshida, M.; Nagasaki, T.; Tanimoto, K.; Tsuga, K.; Akagawa, Y.; Komatsu, T. Aspects of swallowing in healthy dentate elderly persons older than 80 years. J. Gerontol. Med. Sci. 2005, 60A, 506–509. [Google Scholar] [CrossRef]
- Kawashima, K.; Motohashi, Y.; Fujishima, I. Prevalence of dysphagia among community-dwelling elderly individuals as estimated using a questionnaire for dysphagia screening. Dysphagia 2004, 19, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Hudson, H.M.; Daubert, C.R.; Mills, R.H. The interdependency of protein-energy malnutrition, aging and dysphagia. Dysphagia 2000, 15, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Nicosia, M.A.; Hind, J.A.; Roecker, E.B.; Carnes, M.; Doyle, J.; Dengel, G.A.; Robbins, J.A. Age effects on the temporal evolution of isometric and swallowing pressure. J. Gerontol. Med. Sci. 2000, 55A, M634–M640. [Google Scholar] [CrossRef]
- Smithard, D. Dysphagia: A Geriatric giant. Med. Clin. Rev. 2016, 2, 5. [Google Scholar] [CrossRef]
- Holland, G.; Jayasekeran, V.; Pendleton, N.; Horan, M.; Jones, M.; Hamdy, S. Prevalence and symptom profiling of oropharyngeal dysphagia in a community dwelling of an elderly population: A self-reporting questionnaire survey. Dis. Esophagus. 2011, 24, 476–480. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, K.; Aida, J.; Ito, K.; Furuta, M.; Yamahita, Y.; Osaka, K. Nutritional status and dysphagia risk among community dwelling frail older adults. JNHA Nutr. 2014, 18, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Kumlein, S.; Axelsson, K. Stroke patients in nursing homes: Eating, feeding, nutrition and related care. J. Clin. Nurs. 2002, 11, 498–509. [Google Scholar] [CrossRef]
- Smithard, D.G. Feedng and swallowing problems in the institutionalised elderly. Clin. Rehab. 1996, 10, 153–154. [Google Scholar] [CrossRef]
- Smithard, D.G.; O’Neill, P.A.; England, R.E.; Park, C.L.; Wyatt, R.; Martin, D.F.; Morris, J. The natural history of dysphagia following a stroke. Dysphagia 1997, 12, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Kraaijenga, S.A.C.; van der Molen, L.; Stuiver, M.M.; Teertatra, H.J.; Hilgers, F.J.M.; van den Brekel, M.W.M. Effects of strengthening exercises on swallowing musculature and function in senior health subjects: A prospective effectiveness study. Dysphagia 2015, 30, 392–403. [Google Scholar] [CrossRef] [PubMed]
- Karagiannis, M.J.P.; Chivers, J.P.; Karagiannis, T.C. Effects of oral intake of water in patients with oropharyngeal dysphagia. BMC Geriatr. 2011, 11, 9. [Google Scholar] [CrossRef] [PubMed]
- Whelan, K. Inadequate fluid intakes in dysphagic acute stroke. Clin. Nutr. 2001, 20, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Rowat, A.; Graham, C.; Dennis, M. Dehydration in hospital-admitted stroke patients. Stroke 2012, 43, 857–859. [Google Scholar] [CrossRef] [PubMed]
- Cichero, J.A.Y. Thickening agents used for dysphagia management: Effect on bioavailability of medication and feelings of satiety. Nutr. J. 2013, 12, 54. [Google Scholar] [CrossRef] [PubMed]
- Steele, C.M.; Alsanei, W.A.; Ayanikalath, S.; Barbon, C.E.A.; Chen, J.; Cichero, J.A.Y.; Coutts, K.; Dantas, R.O.; Duivestein, J.; Giosa, L.; et al. The influence of food texture and liquid consistency modification on swallowing physiology and function: A systematic review. Dysphagia 2015, 30, 2–26. [Google Scholar] [CrossRef] [PubMed]



© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smithard, D.G. Stroke in Frail Older People. Geriatrics 2017, 2, 24. https://doi.org/10.3390/geriatrics2030024
Smithard DG. Stroke in Frail Older People. Geriatrics. 2017; 2(3):24. https://doi.org/10.3390/geriatrics2030024
Chicago/Turabian StyleSmithard, David G. 2017. "Stroke in Frail Older People" Geriatrics 2, no. 3: 24. https://doi.org/10.3390/geriatrics2030024