Towards Accelerated Autolysis? Dynamics of Phenolics, Proteins, Amino Acids and Lipids in Response to Novel Treatments and during Ageing of Sparkling Wine
Abstract
:1. Introduction
2. Materials and Methods
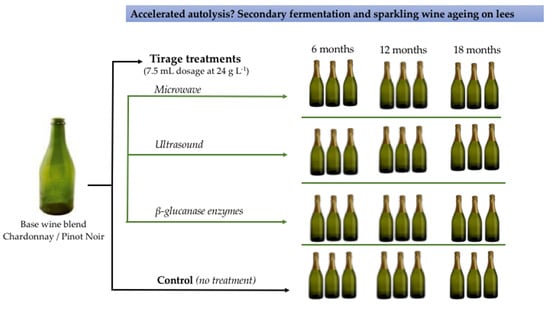
2.1. Yeast Treatments and Preparation of Sparkling Wines
2.2. Analysis of Basic Wine Composition
2.3. Analysis of Proteins
2.4. Analysis of Amino Acids
2.5. Analysis of Lipids
2.6. Statistical Analyses
3. Results
3.1. Basic Composition of Sparkling Wines
3.2. Total Phenolics of Sparkling Wines
3.3. Total Protein of Sparkling Wines
3.4. Free Amino Acid Content of Sparkling Wines
3.5. Lipid Compositon of Sparkling Wines
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Charpentier, C.; Feuillat, M. Yeast autolysis. In The Yeasts; Rose, A.H., Harrison, J.S., Eds.; Academic Press: New York, NY, USA, 1992; Volume 3, pp. 315–386. [Google Scholar]
- Babayan, T.L.; Bezrukov, M.G. Autolysis in Yeasts. Acta Biotechnol. 1985, 5, 129–136. [Google Scholar] [CrossRef]
- Alexandre, H.; Guilloux-Benatier, M. Yeast autolysis in sparkling wine—A review. Aust. J. Grape Wine Res. 2006, 12, 119–127. [Google Scholar] [CrossRef]
- Feuillat, M.; Charpentier, C. Autolysis of Yeasts in Champagne. Am. J. Enol. Vitic. 1982, 33, 6–13. [Google Scholar]
- Charpentier, C.; Aussenac, J.; Charpentier, M.; Prome, J.C.; Duteurtre, B.; Feuillat, M. Release of nucleotides and nucleosides during yeast autolysis: Kinetics and potential impact on flavor. J. Agric. Food Chem. 2005, 53, 3000–3007. [Google Scholar] [CrossRef] [PubMed]
- Buxaderas, S.; Lopez-Tamames, E. Sparkling wines: Features and trends from tradition. Adv. Food Nutr. Res. 2012, 66, 1–45. [Google Scholar]
- Conde, B.C.; Bouchard, E.; Culbert, J.A.; Wilkinson, K.L.; Fuentes, S.; Howell, K.S. Soluble protein and amino acid content affects the foam quality of sparkling wine. J. Agric. Food Chem. 2017, 65, 9110–9119. [Google Scholar] [CrossRef]
- Gallart, M.; Lopez-Tamames, E.; Suberbiola, G.; Buxaderas, S. Influence of fatty acids on wine foaming. J. Agric. Food Chem. 2002, 50, 7042–7045. [Google Scholar] [CrossRef]
- Martínez-Rodriíguez, A.J.; Pueyo, E. Sparkling wines and yeast autolysis. In Wine Chemistry and Biochemistry; Moreno-Arribas, M.V., Polo, M.C., Eds.; Springer: New York, NY, USA, 2009; pp. 61–80. [Google Scholar]
- Nunez, Y.P.; Carrascosa, A.V.; Gonzalez, R.; Polo, M.C.; Martinez-Rodriguez, A.J. Effect of accelerated autolysis of yeast on the composition and foaming properties of sparkling wines elaborated by a champenoise method. J. Agric. Food Chem. 2005, 53, 7232–7237. [Google Scholar] [CrossRef]
- Klis, F.M.; Boorsma, A.; De Groot, P.W. Cell wall construction in Saccharomyces cerevisiae. Yeast 2006, 23, 185–202. [Google Scholar] [CrossRef]
- Martinez-Rodriguez, A.J.; Carrascosa, A.V.; Polo, M.C. Release of nitrogen compounds to the extracellular medium by three strains of Saccharomyces cerevisiae during induced autolysis in a model wine system. Int. J. Food Microbiol. 2001, 68, 155–160. [Google Scholar] [CrossRef]
- Blasco, L.; Vinas, M.; Villa, T.G. Proteins influencing foam formation in wine and beer: The role of yeast. Int. Microbiol. 2011, 14, 61–71. [Google Scholar]
- Gawel, R.; Day, M.; Van Sluyter, S.C.; Holt, H.; Waters, E.J.; Smith, P.A. White wine taste and mouthfeel as affected by juice extraction and processing. J. Agric. Food Chem. 2014, 62, 10008–10014. [Google Scholar] [CrossRef] [PubMed]
- Waters, E.J.; Pellerin, P.; Brillouet, J.M. A Saccharomyces mannoprotein that protects wine from protein haze. Carbohydr. Polym. 1994, 23, 185–191. [Google Scholar] [CrossRef]
- Moreno-Arribas, V.; Pueyo, E.; Nieto, F.; Martin-Avarez, P.; Polo, M. Influence of the polysaccharides and the nitrogen compounds on foaming properties of sparkling wines. Food Chem. 2000, 70, 309–317. [Google Scholar] [CrossRef]
- Andrés-Lacueva, C.; Lamuela-Raventós, R.M.; Buxaderas, S.; de la Torre-Boronat, M.D.C. Influence of Variety and Aging on Foaming Properties of Cava (Sparkling Wine). J. Agric. Food Chem. 1997, 45, 2520–2525. [Google Scholar] [CrossRef]
- Brissonnet, F.; Maujean, A. Characterization of foaming proteins in a Champagne base wine. Am. J. Enol. Vitic. 1993, 44, 297–301. [Google Scholar]
- Marchal, R.; Jeandet, P.; Robbillard, B. Macromolecules and champagne wine foaming properties: A review. In Macromolecules and Secondary Metabolites of Grapevine and Wines; Jeandet, P., Clément, C., Conreux, A., Eds.; Intercept Lavoisie: Paris, France, 2007; pp. 349–370. [Google Scholar]
- Polo, M.C.; Gonzalez de Llano, M.D.; Ramos, M. Derivatization and liquid chromatographic separation of peptides. In Food Analysis by HPLC; Nollet, M.L., Ed.; Dekker: New York, NY, USA, 1992; pp. 117–140. [Google Scholar]
- Bisson, L.F.; Butzke, C.E. Diagnosis and rectification of stuck and sluggish fermentations. Am. J. Enol. Vitic. 2000, 51, 168–177. [Google Scholar]
- Vincenzi, S.; Marangon, M.; Tolin, S.; Curioni, A. Protein evolution in a white wine during the early stages of winemaking and its relations with wine stability. Aust. J. Grape Wine Res. 2011, 17, 20–27. [Google Scholar] [CrossRef]
- Moreno-Garcia, J.; Mauricio, J.C.; Puig-Pujol, A.; Capdevila, F.; Moreno, J.; Garcia-Martinez, T. Proteomic yeast stress response to pressure in a final stage in the second fermentation during sparkling wine elaboration. In BIO Web of Conferences; EDP Sciences: Les Ulis, France, 2015; Volume 5, p. 020002. [Google Scholar] [CrossRef] [Green Version]
- Ribéreau-Gayon, P.; Dubourdieu, D.; Donèche, B.; Lonvaud, A. Nitrogen Compounds. In Handbook of Enology, the Microbiology of Wine and Vinifications; John Wiley and Sons: Hoboken, NJ, USA, 2006; Volume 1, pp. 109–140. [Google Scholar]
- Moreno-Arribas, V.; Pueyo, E.; Polo, M.; Martınez-Alvarez, P. Changes in the amino acid composition of the different nitrogenous fractions during the aging of wine with yeasts. J. Agric. Food Chem. 1998, 46, 4042–4051. [Google Scholar] [CrossRef]
- Pueyo, E.; Martín-Alvarez, P.J.; Polo, M.C. Relationship Between Foam Characteristics and Chemical Composition in Wines and Cavas (Sparkling Wines). Am. J. Enol. Vitic. 1995, 46, 518–524. [Google Scholar]
- Puig-Deu, M.; Lopez-Tamames, E.; Buxaderas, S.; de la Torre-Boronat, M.C. Quality of base and sparkling wines as influenced by the type of fining agent added pre-fermentation. Food Chem. 1999, 66, 35–42. [Google Scholar] [CrossRef]
- Martinez-Lapuente, L.; Guadalupe, Z.; Ayestaran, B.; Perez-Magarino, S. Role of major wine constituents in the foam properties of white and rose sparkling wines. Food Chem. 2015, 174, 330–338. [Google Scholar] [PubMed]
- Pueyo, E.; Martinez-Rodriguez, A.; Polo, M.C.; Santa-Maria, G.; Bartolome, B. Release of lipids during yeast autolysis in a model wine system. J. Agric. Food Chem. 2000, 48, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Rodríguez, A.J.; Polo, M.C.; Carrascosa, A.V. Structural and ultrastructural changes in yeast cells during autolysis in a model wine and in sparkling wines. Int. J. Food Microbiol. 2001, 71, 45–51. [Google Scholar] [CrossRef]
- Chen, E.C.H.; Jamieson, A.M.; Van Gheluwe, G. The Release of Fatty Acids as a Consequence of Yeast Autolysis. J. Am. Soc. Brew. Chem. 1980, 38, 13–18. [Google Scholar] [CrossRef]
- Piton, F.; Charpentier, M.; Troton, D. Cell wall and lipid changes in saccharomyces cerevisiae during aging of champagne wine. Am. J. Enol. Vitic. 1988, 39, 221–226. [Google Scholar]
- Troton, D.; Charpentier, M.; Robillard, B.; Calvayrac, R.; Duteurtre, B. Evolution of the lipid contents of champagne wine during the second fermentation of saccharomyces cerevisiae. Am. J. Enol. Vitic. 1989, 40, 175–182. [Google Scholar]
- Ancin, C.; Ayestaran, B.; Garcia, A.; Garrido, J. Evolution of fatty acids contents in Garnacha and Viura must during fermentation and aging of wine. Z. Für Lebensm. Und-Forsch. A 1998, 206, 143–147. [Google Scholar]
- Ferrari, G.; Meunier, Y.; Feuillat, M. Dosage des acides gras totaux du vin et des levures de vinification. Sci. Aliment. 1987, 7, 61–76. [Google Scholar]
- Herraiz, T.; Herraiz, M.; Reglero, G.; Martin-Alvarez, P.; Cabezudo, M. Changes in the composition of fatty acids during alcoholic fermentation of grape must. Chem. Mikrobiol. Biochem. 1990, 210, 185–188. [Google Scholar]
- Culbert, J.A.; McRae, J.M.; Conde, B.C.; Schmidtke, L.M.; Nicholson, E.L.; Smith, P.A.; Howell, K.S.; Boss, P.K.; Wilkinson, K.L. Influence of production method on the chemical composition, foaming properties, and quality of Australian carbonated and sparkling white wines. J. Agric. Food Chem. 2017, 65, 1378–1386. [Google Scholar] [CrossRef] [PubMed]
- Gawel, R.; Smith, P.A.; Cicerale, S.; Keast, R. The mouthfeel of white wine. Crit. Rev. Food Sci. Nutr. 2018, 58, 2939–2956. [Google Scholar] [CrossRef] [PubMed]
- Gawel, R.; Day, M.; Schulkin, A.; Smith, P.A.; Hererich, M.; Johnson, D. The science of texture. Wine Vit. J. 2013, 2, 34–38. [Google Scholar]
- Sacchi, K.L.; Bisson, L.F.; Adams, D.O. A review of the effect of winemaking techniques on phenolic extraction in red wines. Am. J. Enol. Vitic. 2005, 56, 197–206. [Google Scholar]
- Singleton, V.; Trousdale, E. White Wine Phenolics: Varietal and Processing Differences as Shown by HPLC. Am. J. Enol. Vitic. 1983, 34, 27–34. [Google Scholar]
- Mafata, M.; Buica, A.; du Toit, W.; Panzeri, V.; van Jaarsveld, F.P. The Effect of Grape Temperature on the Sensory Perception of Méthode Cap Classique Wines. S. Afr. J. Enol. 2018, 39, 9. [Google Scholar] [CrossRef] [Green Version]
- Stefenon, C.A.; Bonesi, C.; De, M.; Marzarotto, V.; Barnabe, D.; Spinelli, F.R.; Webber, V.; Vanderlinde, R. Phenolic composition and antioxidant activity in sparkling wines. Modulation by the ageing on lees. Food Chem. 2013, 145, 292–299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salmon, J.M.; Fornairon-Bonnefond, C.; Mazairic, J.P. Interactions between wine lees and polyphenols: Influence on oxygen consumption capacity during simulation of wine aging. Food Sci. 2002, 67, 1604–1609. [Google Scholar] [CrossRef]
- Palomero, F.; Morata, A.; Benito, S.; Gonzalez, M.C.; Suarez-Lepe, J.A. Conventional and enzyme-assisted autolysis during ageing over lees in red wines: Influence on the release of polysaccharides from yeast cell walls and on wine monomeric anthocyanin content. Food Chem. 2007, 105, 838–846. [Google Scholar] [CrossRef]
- Rodriguez-Nogales, J.; Fernandez-Fernandez, E.; Gomez, M.; Vila-Crespo, J. Antioxidant properties of sparkling wines produced with beta-glucanases and commercial yeast preparations. J. Food Sci. 2012, 77, C1005–C1010. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Nogales, J.; Fernández-Fernández, E.; Vila-Crespo, J. Effect of the addition of β-glucanases and commercial yeast preparations on the chemical and sensorial characteristics of traditional sparkling wine. Eur. Food Res. Technol. 2012, 235, 729–744. [Google Scholar] [CrossRef]
- Garcia Martin, J.F.; Guillemet, L.; Feng, C.; Sun, D.W. Cell viability and proteins release during ultrasound-assisted yeast lysis of light lees in model wine. Food Chem. 2013, 141, 934–939. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Loira, I.; Morata, A.; Suárez-Lepe, J.A.; González, M.C.; Rauhut, D. Shortening the ageing on lees process in wines by using ultrasound and microwave treatments both combined with stirring and abrasion techniques. Eur. Food Res. Technol. 2016, 242, 559–569. [Google Scholar] [CrossRef]
- Gnoinski, G.B.; Schmidt, S.A.; Close, D.C.; Goemann, K.; Pinfold, T.L.; Kerslake, F.L. Novel Methods to Manipulate Autolysis in Sparkling Wine: Effects on Yeast. Molecules 2021, 26, 387. [Google Scholar] [CrossRef]
- Laurent, M.; Valade, M. La preparation du levain de tirage: A partir de levures seches actives. Vign. Champen. 2007, 128, 74–95. [Google Scholar]
- Dambergs, R.G.; Gishen, M.; Cozzolino, D. A review of the state of the art, limitations, and perspectives of infrared spectroscopy for the analysis of wine grapes, must, and grapevine tissue. Appl. Spectr. Rev. 2015, 50, 261–278. [Google Scholar] [CrossRef]
- Cozzolino, D.; Dambergs, R.G. Instrumental analysis of grape, must and wine. In Managing Wine Quality: Volume 1, Viticulture and Wine Quality; Woodhead Publishing Series in Food Science, Technology and Nutrition; Reynolds, A.G., Ed.; Woodhead Publishing Ltd.: Cambridge, UK, 2014; pp. 134–161. [Google Scholar]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ackmann, R. Flame ionization detection applied to thin layer chromatography on coated quartz rods. Methods Enzymol. 1981, 72, 205–252. [Google Scholar]
- Volkman, J.; Nichols, P. Application of thin layer chromatography-flame ionization detection to the analysis of lipids and pollutants in marine and environmental samples. J. Planar Chromatogr. 1991, 4, 19–26. [Google Scholar]
- Boulton, R.B.; Singleton, V.L.; Bisson, L.F.; Kunkee, R.E. Yeast and biochemistry of ethanol fermentation. In Principles and Practices of Winemaking; Springer: Boston, MA, USA, 1996; pp. 139–172. [Google Scholar]
- Martinez Lapuente, L.; Ayestaran, B.; Guadalupe, Z. Influence of wine chemical compounds on the foaming properties of sparkling wines, grapes and wines. In Advances in Production, Processing, Analysis and Valorization; Jordao, A.M., Cosme, F., Eds.; InTechOpen: London, UK, 2018; pp. 195–224. [Google Scholar]
- Pozo-Bayon, M.A.; Hernandez, M.T.; Martin-Alvarez, P.J.; Polo, M.C. Study of low molecular weight phenolic compounds during the aging of sparkling wines manufactured with red and white grape varieties. J. Agric. Food Chem. 2003, 51, 2089–2095. [Google Scholar] [CrossRef]
- Kerslake, F.L.; Jones, J.E.; Close, D.C.; Dambergs, R.G. Bunch exposure effects on the quality of Pinot noir and Chardonnay fruit and base wines for cool climate sparkling wine production. In Proceedings of the 18th International Symposium GIESCO, Porto, Portugal, 7–11 July 2013; Volume 28, pp. 471–477. [Google Scholar]
- Esteruelas, M.; Gonzalez-Royo, E.; Kontoudakis, N.; Orte, A.; Cantos, A.; Canals, J.M.; Zamora, F. Influence of grape maturity on the foaming properties of base wines and sparkling wines (Cava). J. Sci. Food Agric. 2014, 95, 2071–2080. [Google Scholar] [CrossRef]
- Andres-Lacueva, C.; Lopez-Tamames, E.; Lamuela-Raventos, R.; Buxadera, S.; Torre-Boronat, M. Characteristics of sparkling base wines affecting foam behaviour. J. Agric. Food Chem. 1996, 44, 989–995. [Google Scholar] [CrossRef]
- Kollar, R.; Sturdik, E.; Sablaturova, E. Biochemical, morphological and cytochemical studies of enhanced autolysis of Saccharomyces cerevisiae. Folia Microbiol. 1993, 38, 473–478. [Google Scholar] [CrossRef]
| Treatments | ||||||
|---|---|---|---|---|---|---|
| Analyte | Bottle Age (Months) | Control | Microwave | Ultrasound | β-Glucanase Enzyme | p Value (Treatment) |
| Alcohol (% v/v) | 6 | 12.9 ± 0.01 | 13.1 ± 0.5 | 13.0 ± 0.2 | 12.9 ± 0.03 | 0.800 |
| Residual sugar | 6 | 1.18 ± 0.01 | 1.09 ± 0.14 | 1.14 ± 0.05 | 1.09 ± 0.17 | 0.730 |
| (mg L−1) | ||||||
| pH | 6 | 3.09 ± 0.00 | 3.09 ± 0.01 | 3.09 ± 0.00 | 3.09 ± 0.01 | 0.330 |
| 12 | 3.12 ± 0.01 | 3.12 ± 0.01 | 3.12 ± 0.01 | 3.11 ± 0.01 | 0.399 | |
| 18 | 3.07 ± 0.00 a | 3.06 ± 0.00 b | 3.08 ± 0.02 a | 3.09 ± 0.01 a | 0.022 | |
| p value (time) | <0.0001 | |||||
| TA | 6 | 8.3 ± 0.1 | 8.3 ± 0.1 | 8.2 ± 0.1 | 8.2 ± 0.1 | 0.611 |
| (g L−1) | 12 | 8.0 ± 0.0 | 8.0 ± 0.1 | 8.0 ± 0.0 | 8.0 ± 0.1 | 0.330 |
| 18 | 8.2 ± 0.0 a | 8.1 ± 0.1 b | 8.1 ± 0.1 b | 8.1 ± 0.1 b | 0.028 | |
| p value (time) | <0.0001 | |||||
| Total phenolics | 6 | 0.89 ± 0.03 | 0.87 ± 0.02 | 0.89 ± 0.03 | 0.87 ± 0.01 | 0.373 |
| (au) | 12 | 1.17 ± 0.04 | 1.16 ± 0.02 | 1.15 ± 0.01 | 1.16 ± 0.02 | 0.732 |
| 18 | 1.44 ± 0.02 | 1.38 ± 0.05 | 1.40 ± 0.04 | 1.41 ± 0.01 | 0.222 | |
| p value (time) | <0.0001 | |||||
| Total proteins | 6 | 32.9 ± 1.1 | 32.4 ± 2.4 | 31.3 ± 1.7 | 32.6 ± 1.3 | 0.686 |
| (mg L−1) | 12 | 87.5 ± 4.7 | 80.4 ± 14.9 | 83.6 ± 1.2 | 69.0 ± 10.1 | 0.167 |
| 18 | 75.6 ± 0.5 | 75.5 ± 4.1 | 78.9 ± 1.9 | 77.4 ± 0.5 | 0.096 | |
| p value (time) | <0.0001 | |||||
| Total free amino acids | 6 | 707 ± 40 | 659 ± 65 | 695 ± 17 | 686 ± 13 | 0.780 |
| (mg L−1) | 12 | 634 ± 27 | 633 ± 11 | 619 ± 19 | 622 ± 27 | 0.783 |
| 18 | 616 ± 33 a | 686 ± 20 a | 628 ± 28 a | 694 ± 44 b | 0.044 | |
| p value (time) | 0.005 |
| Treatments | ||||||
|---|---|---|---|---|---|---|
| Analyte | Bottle Age (Months) | Control | Microwave | Ultrasound | β-Glucanase Enzyme | p Value (Treatment) |
| Proline | 6 | 454.0 ± 26.2 | 417.0 ± 42.6 | 447.0 ± 15.9 | 437.0 ± 3.91 | 0.411 |
| 12 | 402 ± 14.0 | 405.0 ± 6.26 | 396.0 ± 20.4 | 394.0 ± 15.1 | 0.758 | |
| 18 | 412.0 ± 22.2 a | 464.0 ± 17.4 b | 421.0 ± 14.6 a | 461.0 ± 25.7 b | 0.028 | |
| p value (time) | 0.002 | |||||
| Arginine | 6 | 51.1 ± 3.16 | 47.8 ± 6.45 | 47.8 ± 0.31 | 47.6 ± 4.83 | 0.721 |
| 12 | 46.1 ± 3.83 | 46.5 ± 2.22 | 46.3 ± 3.16 | 47.8 ± 1.99 | 0.900 | |
| 18 | 39.9 ± 3.42 | 40.7 ± 0.90 | 38.0 ± 2.93 | 44.5 ± 2.47 | 0.079 | |
| p value (time) | 0.0005 | |||||
| Asparagine | 6 | 36.1 ± 3.21 | 35.4 ± 3.29 | 35.7 ± 2.11 | 34.2 ± 0.79 | 0.825 |
| 12 | 22.6 ± 0.38 | 23.7 ± 1.01 | 23.9 ± 0.76 | 23.5 ± 2.36 | 0.692 | |
| 18 | 15.2 ± 0.78 a | 17.6 ± 0.50 b | 16.5 ± 1.06 a | 16.9 ± 0.56 a | 0.012 | |
| p value (time) | <0.0001 | |||||
| Lysine | 6 | 25.4 ± 1.32 | 24.8 ± 1.35 | 25.6 ± 0.50 | 25.4 ± 0.70 | 0.770 |
| 12 | 24.3 ± 0.72 | 24.5 ± 0.32 | 23.0 ± 0.29 | 23.6 ± 1.34 | 0.140 | |
| 18 | 23.1 ± 0.97 | 25.4 ± 1.50 | 22.8 ± 1.63 | 24.7 ± 2.04 | 0.219 | |
| p value (time) | 0.032 | |||||
| Glutamic acid | 6 | 23.5 ± 2.00 | 21.8 ± 1.49 | 23.0 ± 0.36 | 22.8 ± 1.12 | 0.540 |
| 12 | 21.4 ± 1.71 | 21.2 ± 0.35 | 20.1 ± 0.70 | 21.5 ± 1.65 | 0.540 | |
| 18 | 19.1 ± 1.62 | 21.4 ± 0.42 | 19.8 ± 1.44 | 21.9 ± 1.18 | 0.082 | |
| p value (time) | <0.0001 | |||||
| Alanine | 6 | 23.1 ± 2.08 | 21.3 ± 2.02 | 22.3 ± 0.21 | 22.8 ± 1.34 | 0.580 |
| 12 | 22.0 ± 1.81 | 21.3 ± 0.36 | 20.4 ± 0.31 | 21.1 ± 1.57 | 0.500 | |
| 18 | 20.8 ± 0.72 | 22.1 ± 0.14 | 20.9 ± 2.33 | 23.6 ± 1.42 | 0.129 | |
| p value (time) | 0.040 | |||||
| Aspartic acid | 6 | 18.2 ± 1.27 | 17.7 ± 1.50 | 17.6 ± 0.92 | 18.7 ± 1.22 | 0.710 |
| 12 | 16.2 ± 1.30 | 16.5 ± 0.21 | 15.5 ± 0.30 | 15.3 ± 0.75 | 0.280 | |
| 18 | 14.7 ± 0.95 | 16.4 ± 0.86 | 15.0 ± 0.93 | 17.0 ± 2.11 | 0.175 | |
| p value (time) | 0.0004 | |||||
| Leucine | 6 | 13.2 ± 0.56 | 12.2 ± 1.14 | 13.3 ± 0.10 | 14.2 ± 1.73 | 0.240 |
| 12 | 17.6 ± 2.11 | 15.9 ± 0.40 | 15.6 ± 0.15 | 15.8 ± 0.56 | 0.180 | |
| 18 | 15.5 ± 1.91 | 16.4 ± 0.45 | 15.5 ± 0.85 | 16.8 ± 0.91 | 0.448 | |
| p value (time) | <0.0001 | |||||
| Glycine | 6 | 13.0 ± 0.81 | 12.4 ± 1.31 | 12.7 ± 0.36 | 12.9 ± 1.10 | 0.870 |
| 12 | 11.1 ± 0.79 | 10.2 ± 0.23 | 10.2 ± 0.30 | 10.3 ± 0.61 | 0.190 | |
| 18 | 9.59 ± 1.17 | 10.8 ± 0.43 | 9.96 ± 0.80 | 11.7 ± 2.68 | 0.378 | |
| p value (time) | 0.001 | |||||
| Phenylalanine | 6 | 12.1 ± 0.50 | 11.5 ± 0.69 | 12.0 ± 0.36 | 12.5 ± 0.75 | 0.280 |
| 12 | 13.1 ± 1.27 | 12.0 ± 0.59 | 12.3 ± 0.46 | 12.6 ± 1.21 | 0.520 | |
| 18 | 11.5 ± 0.95 | 12.3 ± 0.44 | 11.7 ± 0.63 | 12.8 ± 0.67 | 0.157 | |
| p value (time) | 0.331 | |||||
| Tyrosine | 6 | 9.0 ± 0.62 | 8.77 ± 0.81 | 9.27 ± 0.06 | 9.63 ± 0.12 | 0.270 |
| 12 | 9.30 ± 0.72 | 8.67 ± 0.58 | 9.00 ± 0.62 | 9.33 ± 1.02 | 0.690 | |
| 18 | 8.03 ± 0.65 | 8.13 ± 0.25 | 8.33 ± 0.40 | 9.57 ± 0.99 | 0.063 | |
| p value (time) | 0.052 | |||||
| Valine | 6 | 6.9 ± 0.20 | 6.6 ± 0.76 | 6.83 ± 0.21 | 6.97 ± 0.06 | 0.710 |
| 12 | 6.67 ± 0.38 | 6.63 ± 0.15 | 6.50 ± 0.10 | 6.57 ± 0.12 | 0.790 | |
| 18 | 6.29 ± 0.54 | 6.92 ± 0.35 | 6.40 ± 0.31 | 7.34 ± 0.53 | 0.065 | |
| p value (time) | 0.037 | |||||
| Serine | 6 | 5.67 ± 0.59 | 5.93 ± 1.14 | 5.67 ± 0.35 | 5.50 ± 0.44 | 0.900 |
| 12 | 5.83 ± 0.45 | 5.50 ± 0.27 | 5.63 ± 0.15 | 5.60 ± 0.44 | 0.700 | |
| 18 | 4.97 ± 0.18 | 5.65 ± 0.22 | 5.16 ± 0.54 | 5.28 ± 0.09 | 0.162 | |
| p value (time) | 0.129 | |||||
| Threonine | 6 | 5.03 ± 0.55 | 5.13 ± 0.55 | 5.23 ± 0.15 | 5.17 ± 0.40 | 0.960 |
| 12 | 4.70 ± 0.27 | 4.70 ± 0.20 | 4.57 ± 0.15 | 4.67 ± 0.25 | 0.860 | |
| 18 | 4.57 ± 0.32 | 5.19 ± 0.09 | 4.83 ± 0.27 | 5.36 ± 1.58 | 0.652 | |
| p value (time) | 0.160 | |||||
| Cysteine | 6 | 3.60 ± 0.27 | 3.27 ± 0.21 | 3.43 ± 0.06 | 3.40 ± 0.17 | 0.280 |
| 12 | 4.17 ± 0.40 | 3.97 ± 0.45 | 4.00 ± 0.53 | 4.17 ± 0.51 | 0.930 | |
| 18 | 4.50 ± 0.36 | 5.14 ± 0.72 | 5.09 ± 0.19 | 5.12 ± 0.49 | 0.352 | |
| p value (time) | <0.0001 | |||||
| Isoleucine | 6 | 3.50 ± 0.10 | 3.47 ± 0.23 | 3.60 ± 0.10 | 3.70 ± 0.20 | 0.380 |
| 12 | 4.30 ± 0.30 | 4.07 ± 0.15 | 3.97 ± 0.06 | 4.00 ± 0.20 | 0.240 | |
| 18 | 3.73 ± 0.32 | 4.04 ± 0.13 | 3.80 ± 0.21 | 4.19 ± 0.46 | 0.284 | |
| p value (time) | 0.001 | |||||
| Methionine | 6 | 2.97 ± 0.25 | 2.97 ± 0.25 | 3.03 ± 0.06 | 2.97 ± 0.12 | 0.960 |
| 12 | 2.80 ± 0.26 | 2.63 ± 0.15 | 2.67 ± 0.06 | 2.67 ± 0.21 | 0.713 | |
| 18 | 2.85 ± 0.13 | 3.11 ± 0.02 | 3.00 ± 0.26 | 3.09 ± 0.18 | 0.277 | |
| p value (time) | 0.002 | |||||
| Tryptophan | 6 | 0.40 ± 0.00 | 0.43 ± 0.06 | 0.43 ± 0.06 | 0.40 ± 0.00 | 0.600 |
| 12, 18 | bd | bd | bd | bd | - | |
| Histidine | 6, 12, 18 | bd | bd | bd | bd | - |
| Glutamine | 6, 12, 18 | bd | bd | bd | bd | - |
| Treatments | ||||||
|---|---|---|---|---|---|---|
| Analyte | Bottle Age (Months) | Control | Microwave | Ultrasound | β-Glucanase Enzyme | p Value (Treatment) |
| Total lipid | 6 | 6.26 ± 1.32 | 6.94 ± 2.36 | 5.56 ± 0.96 | 4.36 ± 0.62 | 0.127 |
| 12 | 3.95 ± 0.50 a | 4.76 ± 0.29 a | 3.51 ± 0.45 a | 5.58 ± 1.16 b | 0.048 | |
| 18 | 4.13 ± 1.02 a | 4.41 ± 0.33 a | 5.42 ± 0.24 a | 6.61 ± 1.10 b | 0.013 | |
| p value (time) | 0.089 | |||||
| Free fatty acids | 6 | 0.46 ± 0.10 | 0.45 ± 0.11 | 0.41 ± 0.08 | 0.38 ± 0.08 | 0.647 |
| (FFA) | 12 | 0.13 ± 0.03 | 0.13 ± 0.01 | 0.08 ± 0.03 | 0.14 ± 0.05 | 0.173 |
| 18 | 0.10 ± 0.02 a | 0.08 ± 0.06 a | 0.07 ± 0.02 a | 0.19 ± 0.02 b | 0.046 | |
| p value (time) | <0.0001 | |||||
| Phospholipids | 6 | 0.94 ± 0.13 | 2.08 ± 1.78 | 0.98 ± 0.19 | 0.73 ± 0.03 | 0.165 |
| (PL) | 12 | 0.57 ± 0.13 a | 0.62 ± 0.35 a | 0.32 ± 0.09 a | 1.04 ± 0.10 b | 0.014 |
| 18 | 0.47 ± 0.19 a | 0.42 ± 0.08 a | 1.10 ± 0.16 b | 1.14 ± 0.30 b | 0.005 | |
| p value (time) | 0.059 | |||||
| Diacylglycerol | 6 | 1.34 ± 0.78 | 1.06 ± 0.49 | 0.87 ± 0.31 | 0.75 ± 0.26 | 0.421 |
| (DAG) | 12 | 2.30 ± 0.23 a | 2.78 ± 0.15 a | 2.07 ± 0.18 a | 3.13 ± 0.74 b | 0.049 |
| 18 | 2.56 ± 0.71 | 2.61 ± 0.30 | 2.86 ± 0.23 | 3.78 ± 0.58 | 0.055 | |
| p value (time) | <0.0001 | |||||
| Triacylglycerol | 6 | 0.04 ± 0.03 | 0.07 ± 0.07 | 0.04 ± 0.02 | 0.03 ± 0.02 | 0.585 |
| (TAG) | 12 | 0.05 ± 0.03 | 0.06 ± 0.02 | 0.05 ± 0.04 | 0.07 ± 0.06 | 0.760 |
| 18 | 0.02 ± 0.01 a | 0.09 ± 0.01 b | 0.04 ± 0.05 a | 0.07 ± 0.01 a | 0.048 | |
| p value (time) | 0.516 | |||||
| Monoacylglycerol | 6 | 3.08 ± 0.48 | 2.94 ± 0.38 | 2.87 ± 0.57 | 2.22 ± 0.44 | 0.115 |
| (MAG) | 12 | 0.54 ± 0.08 | 0.67 ± 0.08 | 0.65 ± 0.21 | 0.91 ± 0.25 | 0.144 |
| 18 | 0.65 ± 0.11 a | 0.76 ± 0.08 a | 1.04 ± 0.07 b | 1.07 ± 0.16 b | 0.0003 | |
| p value (time) | <0.0001 | |||||
| Sterol esters | 6 | 0.41 ± 0.21 | 0.32 ± 0.26 | 0.40 ± 0.21 | 0.25 ± 0.10 | 0.661 |
| (SE) | 12 | 0.36 ± 0.10 a | 0.51 ± 0.05 b | 0.35 ± 0.04 a | 0.29 ± 0.08 a | 0.034 |
| 18 | 0.32 ± 0.01 | 0.46 ± 0.11 | 0.30 ± 0.04 | 0.37 ± 0.06 | 0.202 | |
| p value (time) | 0.516 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gnoinski, G.B.; Close, D.C.; Schmidt, S.A.; Kerslake, F.L. Towards Accelerated Autolysis? Dynamics of Phenolics, Proteins, Amino Acids and Lipids in Response to Novel Treatments and during Ageing of Sparkling Wine. Beverages 2021, 7, 50. https://doi.org/10.3390/beverages7030050
Gnoinski GB, Close DC, Schmidt SA, Kerslake FL. Towards Accelerated Autolysis? Dynamics of Phenolics, Proteins, Amino Acids and Lipids in Response to Novel Treatments and during Ageing of Sparkling Wine. Beverages. 2021; 7(3):50. https://doi.org/10.3390/beverages7030050
Chicago/Turabian StyleGnoinski, Gail B., Dugald C. Close, Simon A. Schmidt, and Fiona L. Kerslake. 2021. "Towards Accelerated Autolysis? Dynamics of Phenolics, Proteins, Amino Acids and Lipids in Response to Novel Treatments and during Ageing of Sparkling Wine" Beverages 7, no. 3: 50. https://doi.org/10.3390/beverages7030050