1. Introduction
Lutein (β,ε-carotene-3,3′-diol; C
40H
56O
2;
Figure 1A) is a xanthophyll carotenoid that is found in many fruits, vegetables, and flowers. Depending on its tissue concentrations, it provides many yellow, orange, and red colours in plants. This natural pigment is also used as a food colourant (food additive code: E 161b) in a wide array of food products and beverages. As a dietary substance, lutein is important for eye health and skin pigmentation [
1,
2,
3,
4,
5,
6,
7,
8].
Marigold (
Tagetes erecta) flowers contain high amounts of carotenoids, which are predominantly fatty esters and diesters of lutein (0.1–0.2% [
w/w] dry matter as carotenoids, of which 80% are lutein diesters) (
Figure 1B) [
9]. In the food industry, lutein is usually extracted from marigold flowers either as its fatty acid esters and diesters (e.g., palmitate, myristate linoleate, laureate) or as free lutein when the extraction is combined with saponification ([
4,
10,
11,
12,
13]. Furthermore, lutein esters are less sensitive to degradation than lutein, with its diesters being more stable than its monoesters [
4].
As well as its colouring and colour-enhancing characteristics, lutein has important chemical and biochemical properties that are relevant for the maintenance of eye health.
In this respect, lutein has two important functions: As a filter of high energy blue light, and as an antioxidant and free radical scavenger. It can thus protect the human body from different types of free radicals, with protection of the skin and eyes from photodamage [
2], combined with anti-cancer activity and cardiovascular protection [
1,
2,
3,
4,
5]. The Joint FAO/WHO Expert Committee on Food Additives set the recommended daily intake for lutein at 0 mg/kg to 2 mg/kg body weight [
14]. Based on additional scientific data, the acceptable daily intake was subsequently set at 1 mg/kg body weight by the European Food Safety Authority [
15]. Furthermore, there are several studies that have reported that daily supplementation of the diet with ~10 mg lutein per adult might provide various health benefits, such as protection from age-related macular degeneration and cataracts [
12,
16,
17,
18,
19]. However, it should be noted that the strength of the available evidence currently does not substantiate the use of lutein in terms of these nutrition and health claims [
20].
As a fat soluble compound, lutein is not soluble in aqueous medium (i.e., solubility in water at 37 °C, 0.197 mg/L; [
21]) which is a considerable limitation to its incorporation into commercial food and beverage products. Furthermore, it is susceptible to degradation in the presence of oxygen, light, and/or increased temperatures. Therefore, there is the need for the development of techniques to improve its stability and to enable its incorporation into water-based matrices [
11,
12,
21,
22]. To overcome these issues, encapsulation is known to efficiently prevent physicochemical degradation. Delivery systems developed for lutein-based compounds include solid lipid nanoparticles, nanocrystals, nanoliposomes, and various emulsions.
Colloidal delivery systems based on mixtures of oil, water, and surfactants are relevant for compound protection and delivery in food and beverage products, such as microemulsions, nanoemulsions, and conventional emulsions. These can be easily made from food-grade ingredients using relatively simple processes, such as mixing and homogenisation at ambient or elevated temperatures. The oil phase of these colloidal systems can also be used to dissolve lipophilic bioactive compounds, to improve their stability and distribution in aqueous systems [
23]. In emulsions, lutein can dissolve in the oil droplets, and this formulation can then be mixed with water. For example, it has been shown that the loss of lutein was higher when entrapped in a single layer emulsion compared to a layer-by-layer emulsion, which was stabilised using gum Arabic [
24,
25]. Frede et al. [
19] compared six types of emulsifiers made with lecithin (i.e., whey protein, β-lactoglobulin, whey protein hydrolysate Biozate 1, and their combinations). The emulsions with combinations of emulsifiers with lecithin gave the best results, due to the small droplet sizes, resistance to creaming, and physical stability. Khalil et al. [
10] showed that lutein esters can be protected from thermal degradation by addition of medium-chain triglyceride (MCT) oil, while MCT-oil-based emulsions prevented degradation by UV light. Another study investigated the binding of lutein in milk-protein complexes, and showed that sodium caseinate is a better carrier for lutein than whey protein isolate [
26]. Similarly, Davidov-Pardo et al. [
17] demonstrated prolonged storage and improved pH and temperature stability of lutein-enriched caseinate emulsions.
Nonionic surfactants, such as poly(oxyethylene) sorbitan esters (i.e., the Tween non-ionic surfactant family) have also been widely used to prepare microemulsions, due to their relatively low cost and efficient emulsifying properties [
23]. Polyoxyethylene sorbitan monooleate (PSM; also known as Tween 80) is a widely established aliphatic non-ionic surfactant that is derived from polyethoxylated sorbitan and oleic acid. It consists of hydrocarbon aliphatic residues (i.e., non-polar tails) and polyoxyethylene groups (i.e., polar regions) attached to sorbitan. PSM is approved by the US Food and Drug Administration for parenteral, oral, and topical applications [
27], and is included on the European Union list of food additives under E 433 (E 432–436). Zhao et al. [
8] prepared lutein-loaded particles with polyvinylpyrrolidone and PSM as the emulsion stabilisers. Their formulation provided good encapsulation efficiency and good water solubility compared to lutein dispersed in water alone, due to the hydrogen bonding between the carrier and the active compound. In a similar approach, zein nanoparticles loaded with 7.5% lutein were stabilised by lecithin and pluronic F127 surfactants. In the presence of these surfactants, the nanoparticles prepared were larger and had a narrower size distribution and stronger zeta potential, and improved encapsulation efficiency [
24].
Similarly, other carriers that undergo self-assembly and/or have compatibility with hydrophobic and hydrophilic compounds can be used here. For example, a suitable candidate is lecithin, which is a phospholipid mixture that contains two non-polar hydrocarbon chains and a zwitterionic polar head group that has both positive and negative charges that derive from its amine and phosphate groups. Lecithin is widely applied as an emulsifying agent in cosmetics, food (food additive code E 322), and pharmaceutical products, due to its low toxicity, biocompatibility, and generally recognized-as-safe regulatory status [
23].
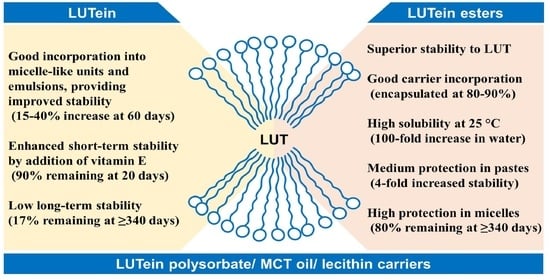
This short overview demonstrates that encapsulation is a promising route for protection of lutein from degradation during food processing and storage. However, the development of efficient and feasible procedures for such encapsulation of lutein-based compounds is still required, in particular for liquid formulations with lutein, which are rarely available on the market. Thus, the aim of the present study was to prepare stable and water-soluble encapsulates of lutein with commercially available 20% lutein and 20% lutein esters powder extracts. We focused on the development of straightforward and feasible approaches with PSM, MCT oil, and sunflower lecithin as the carrier materials. These were prepared as long-lasting water-miscible formulations that are suitable for incorporation into liquid products, and ultimately to develop formulations that are appropriate for industrial production. The materials produced were characterised in terms of particle size, morphology, encapsulation efficiency, and short-term and long-term stability. Different storage conditions were examined, which included stability tests that were performed over up to 300 days at 25 °C in the dark.
3. Results and Discussion
3.1. Encapsulation Efficiency
Encapsulation efficiency was determined for the eight different formulations of the lutein and lutein esters with PSM and MCT oil or lecithin (
Table 2). The composition of the delivery system markedly affected its properties. Overall, all of these encapsulation systems provided high encapsulation efficiencies, in the order of pastes < fine suspensions < emulsions. For the viscous emulsions, nearly complete encapsulation of the lutein was achieved (formulation 3L), with >90% of the lutein esters also entrapped (formulation 3LE).
In the pastes and suspension formulations for the lutein esters (1LE and 2LE, respectively), the encapsulation efficiencies were similar, at just under 80%. For lutein, various finely dispersed suspensions (2L, 2L-1 to 2L-3) were prepared. Decreasing the proportion of PSM (to MCT) from formulations 2L to 2L-1 resulted in 10% reduction in encapsulation efficiency. Conversely, increasing the proportion of MCT oil (to PSM) as formulations 2L vs. 2LE, and 2L-2 vs. 2L-3) had a minor influence on the encapsulation efficiency. This might indicate that low proportions of MCT oil (formulation 2L-3) were already sufficient in the formation of micelle-like units, and therefore even lower amounts should be further investigated. By changing these proportions of PSM and MCT oil, almost 90% entrapment of lutein was achieved (formulation 2L-2). Moreover, by increasing the lutein concentration from 20% to 50% in the extract, the encapsulation efficiency only increased by 6% (comparing sample formulations equivalent to 2L, data not shown). This relatively small increase in lutein entrapment at its considerably higher input concentration suggests that in formulation 2L, the finely dispersed suspension had already approached its loading capacity, which in turn determined the encapsulation efficiencies obtained.
It should be noted that other studies have determined encapsulation efficiencies for different systems of 87.5% to 93.8% based on concentrated (90%) lutein (e.g., spray-dried water–oil–water emulsions, lutein-loaded polyvinylpyrrolidone particles) [
7,
8]. Furthermore, Li et al. [
29] recently reported an emulsion encapsulation approach where 84.0% encapsulation efficiency was obtained using 90% lutein. Thus, we have demonstrated here that similar encapsulation efficiencies can be achieved with more commercially attractive lutein extracts compared to less feasible pure lutein or highly concentrated lutein powders.
3.2. Stability Tests
Based on the observed encapsulation efficiencies, the lutein ester plus PSM paste (formulation 1LE), and the finely dispersed suspensions of lutein (formulations 2L) and lutein esters (formulation 2LE) plus PSM and MCT oil were further tested in stability tests, in parallel to free lutein and lutein esters extracts. These stability tests were performed to gain more information on the potential shelf-life of the products. The non-encapsulated compounds were studied at 4 °C in the dark, as the initial investigation of stability at 25 °C, and these showed rapid degradation over the first 7 days. The goal of encapsulation was to provide formulations that do not require the use of specific storage approaches, such as refrigeration.
Table 3 shows that all of the formulations that contained the lutein and lutein ester extracts were more stable than the non-encapsulated extracts. The best stability was achieved for formulation 3LE. For both the lutein and lutein esters extracts, the addition of lecithin provided more stable systems for the encapsulation. Furthermore, there was up to 40% increase in the loading capacity of the initial active components achieved with lecithin, compared to PSM without or with MCT oil.
3.3. Stability of the Lutein and Lutein Esters Extracts and Their Pastes and Fine Suspensions
The prolonged stability for the non-encapsulated lutein and lutein esters extracts was monitored over 80 days at 25 °C (
Figure 2A). During this time, the lutein content in the non-encapsulated lutein extract was completely degraded, and the more stable non-encapsulated lutein esters extract reached 35% degradation. These data demonstrated the requirement for encapsulation of these compounds.
3.4. Stability of Lutein and Lutein Esters Syrups with Lecithin
Formulations 3 were prepared as viscous emulsions (i.e., in the form of a syrup) with higher concentrations of the active components than in formulations 1 and 2. In the final product developed that was intended for the market, additional ingredients were also added, such as:
(i) Red orange concentrate (8%, w/w), apple concentrate (5%, w/w) and orange aroma (1.5%, w/w): To improve the organoleptic properties, due to the unpleasant taste of the extracts.
(ii) Vitamin E (0.035%, w/w): To prolong the stability of the active compounds.
The stabilities of the encapsulated compounds in formulation 3L were monitored for 30 days under three different storage conditions: Exposure to light at 25 °C; at 25 °C in the dark; and at 4 °C in the dark (
Figure 3A). The stability of formulation 3L with the addition of vitamin E (formulation 3L-VE) was monitored over 42 days, with these data compared with the formulation without vitamin E (
Figure 3B, 3L). In contrast to rapid degradation of the free lutein at 25 °C (data not shown) and at 4 °C (
Table 3,
Figure 2A), formulation 3L maintained more than 60% of the initial concentration of lutein after 1 month of storage. Relatively comparable protective effects of formulation 3L were observed as storage in the presence of light versus dark, as well as at ambient temperature and with refrigerated storage (
Figure 3A).
However, the marked increase in the stability of the active component was still not considered as sufficient for commercial application to storage prolongation. Therefore, to further improve product stability, formulation 3L was reinforced by the addition of vitamin E. Amongst the tested concentrations, the addition of 0.035% (
w/w) vitamin E provided the longest (10%) improvement to the stability (see
Supplementary Materials, Figure S2). However, this formulation contained 50% of initial concentration of lutein after 42 days of storage in the dark at 25 °C (
Figure 3B). It appeared that the added vitamin E improved the stability of the lutein in the first 20 days of storage, but it did not improve the long-term stability. This appears to be due to gradual degradation by radical-mediated oxidation of vitamin E, which can occur over the timeframe of days or weeks, depending on the environment conditions [
30].
As the formulations with lutein extract without and with vitamin E (3L, 3L-VE, respectively) were not sufficiently stable, formulation 3 was prepared with the lutein esters extract and lecithin (formulation 3LE). Moreover, based on our preliminary sensory evaluations, the lutein esters extract had a stronger, bitter taste in comparison to the lutein extract, and might thus be rejected by consumers. Therefore, to improve the taste, the final formulation 3LE also contained commercially available orange aroma, apple juice, and orange juice concentrates, with the equivalent lutein content to formulation 3L. After 250 days, this improved formulation 3LE maintained 80% of the initial concentration of the lutein esters (
Figure 3C). It should also be noted that the same amount of lutein esters in terms of g/L content corresponds to the halved content of lutein after hydrolysis, and furthermore, that esterification of lutein does not impair its bioavailability [
31,
32]. The final concentrations of lutein in products with lutein esters in our formulation was still sufficient for use in the production of food supplements.
3.5. Stability of Lutein and Lutein Esters in Syrups with Lecithin under Accelerated Conditions of Storage
To obtain relevant aging data, and to compare various aging procedures, the stabilities of formulations 3L and 3LE were studied under conditions of accelerated ageing (i.e., at 37 °C), and in real-time (i.e., at 25 °C). Formulations 3L and 3LE were stored under accelerated conditions for at least 56 days (corresponding to ~224 days ageing in real time). After 14 days of accelerated storage (i.e., ~56 days real-time equivalent), the concentration of lutein in formulation 3L started to decrease, and after 56 days of accelerated storage (i.e., ~340 days real-time equivalent), only 17% of the initial concentration remained. The stability of formulation 3L was therefore greatly improved compared to free lutein; however, this was still not sufficient for commercial use. Finally, formulation 3LE with lutein esters provided superior long-term stability, as 95% and 79% of the active components were retained under the conditions of accelerated ageing after 63 days (i.e., ~252 days real-time equivalent) or 91 days (~364 days in real-time), respectively (
Figure 3D).
At this point, it is important to note that various intermediates have been reported to form during the degradation of lutein, such as 1,1,6-trimethyl-1,2-dihydronaphtalene, 2,3-dehydro-α-ionone (two isomers), 3,4-dehydro-β-ionone, 3-oxo-α-ionone, and 3-hydroxy-β-ionone [
33,
34]. However, here, it was not possible to identify any additional compounds by HPLC analysis. The chromatograms of these formulations from the start and during the prolonged storage could only be distinguished by the decreases in areas of the analytes at fixed retention times. Therefore, none of the tested delivery systems saw new peaks on HPLC analysis that would indicate the formation of new compounds (see
Supplementary Materials, Figure S3).
3.6. Colour Stability
Colour stability measurements were performed during the storage. These colour measurements were performed on the day of preparation, and after 1 week, 3 weeks, and 300 days. These data are shown in
Table 4 and
Table 5. Only the pastes and the fine suspensions with the lutein esters were tested for colour stability, as colour changes for the syrups are of less relevance here.
Table 4 and
Table 5 show that both of the formulations examined here (1LE, 2LE) showed stable colour properties over the 300 days of observation. For formulation 1LE (
Table 4), the cumulative ΔE of 2.0 to 2.6 indicates that the colour differences will be perceptible by the human eye through close observation of the sample. For formulation 2LE (
Table 5), the small differences in the cumulative parameter, as ΔE <1, means that the colour differences are not perceptible to the human eye. Visual representation of the samples is provided in the
Supplementary Materials (Figure S4).
3.7. Physical Stability—Particle Size and ζ-Potential
To evaluate the physical stability and suitability of the incorporation of these formulations into food products, the particle sizes and ζ-potentials were determined. The physical stabilities of formulations 1LE, 2LE, and 3LE (all of which contained the lutein esters extract) were monitored by measuring the mean droplet diameter and ζ-potential over 2 months of storage at 25 °C in the dark. These data are shown in
Table 6.
Minor changes in particle size and ζ-potential were seen over 50 days of storage at room temperature (
Table 6), which indicate good physical stability of the samples, especially for formulations 1LE and 3LE. The mean initial particle sizes were from 800 nm to 850 nm for formulations 1LE and 2LE, and ~400 nm for formulation 3LE. After 50 days of storage, there was ~25% loss in particle size for the fine suspension of formulation 2LE, with only ~5% loss for the paste and the emulsion of formulations 1LE and 3LE, respectively.
The particle sizes of the encapsulation formulations depended on the preparation technique, wall materials, and compound–carrier interactions. In the literature, similar approaches that have been applied to lutein encapsulation have included coconut skim milk and whey-protein-stabilised oil–water emulsions [
10,
35] and lipid-core nanocapsules [
11], which reached particle diameters of 0.4 µm to 0.5 µm, 0.7 µm to 1.0 µm, and 0.2 µm, respectively. In the same way, our approaches here produced small particle sizes with good delivery properties, and demonstrated the production of relatively developed morphologies via this straightforward preparation of the paste, fine suspensions, and emulsions.
The ζ-potential with PSM was measured at the beginning and end of the storage periods here, for each the samples taken through the test period. The ζ-potential was similar for the formulations with (formulation 2LE, fine suspension) and without (formulation 1, paste) MCT oil, at −10 mV to −15 mV, and was even weaker for the emulsions (formulation 3LE; around −1 mV). Similarly, the ζ-potential for the lutein-loaded nanocapsules of Brum et al. [
11] were reported to have correspondingly low values (i.e., −5.1 ± 2.22 mV). The particle sizes and ζ-potentials measured here indicated that the particles would remain stable within the suspension, where they would sediment out, which can be prevented by increasing the particle charge, if required. Li et al. [
29] investigated lutein-enriched emulsions that they stabilised with sodium caseinate using a high-pressure homogenisation process, for the influence of the environment conditions on the particle size. Their optimised lutein nanoemulsion droplet mean diameter was 234 ± 3.4 nm, and their ζ-potential was −36.6 ± 1.5 mV, where decreased particle size and increased ζ-potential were achieved by modulation of the homogenisation process, as well as the pH and ionic strength, which demonstrated a similar approach to the present study, where such changes might also be suitable for our delivery systems.
3.8. Microscopy
As a complementary method to dynamic light scattering, dynamic laser 3D microscopy was performed to evaluate the morphology of the formulations and the particle sizes. This showed that the particle sizes for formulations 1LE and 2LE were 2.3 ± 0.9 µm and 1.9 ± 0.9 µm, respectively (see
Supplementary Materials, Figure S6). In comparison, the particle sizes measured by dynamic light scattering were approximately half these values for both of these formulations. This is consistent with our own [
36] and other studies [
37,
38,
39,
40], where these previous observations have indicated that different particle sizes can be measured using different methods. For example, hydration has an important role in particle size determinations. When measured in solution, particles are often smaller, as a semi-fluid layer can form over the outer region of the particle. For this reason, it is important to check the particle size by at least two methods, to gain relevant insight into the particle morphology. Furthermore, 3D microscopy shows that emulsions included bimodal distributions of smaller (~0.1–0.6 µm) and larger (~2–8 µm) particles.
4. Conclusions
In the present study, we have developed an inexpensive, straightforward, and rapid method for encapsulation of extracts of lutein and lutein esters. For the lutein extracts, all of the formulations provided protection of this active compound, and are suitable for products with shelf-lives <1 month. Lutein esters are known for their superior stabilities compared to lutein. Our data here show that the lutein esters are also likely to be better incorporated into these carriers, as indicated by the encapsulation efficiencies (80–90%), the various stability tests, and the evaluations of solubility. For the lutein esters, the paste with PSM (formulation 1LE) provided medium protection from degradation, enabling storage periods approximately 4-fold longer than for the pure compounds. Moreover, both the PSM and MCT oil finely dispersed suspensions (formulation 2LE) and the lecithin emulsion (formulation 3LE) provided the most stable formulations with the lutein esters (300 days storage, up to 80% remaining). The pastes do not appear to greatly prolong the stability of the lutein esters, but did provide a basic means of protection. All of these formulations were physically stable, as the particle sizes and colour parameters were maintained over several months at room temperature in the dark. Due to the bitter taste of the lutein esters, there is the need for the addition of masking agents and flavours in the final products. The paste and finely dispersed suspensions show potential for use in products such as fruit drinks, tea, and other beverages. The emulsions prepared here can be used in a wide variety of food products. Among all of these formulations, the lutein esters in the micellar suspensions and emulsions showed the best overall properties and are the most promising for industrial production and applications, as liquid lutein products remain scarce on the market. In our further ongoing studies, we will focus on the lutein esters in emulsions in terms of prolonged long-term stability studies and planned clinical testing.