Lipoic Acid Gold Nanoparticles Functionalized with Organic Compounds as Bioactive Materials
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Apparatus
2.2. Synthesis of Gold Nanoparticles
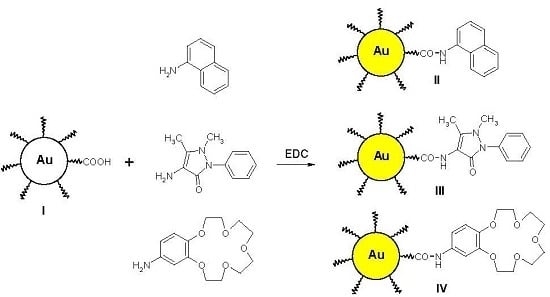
2.3. Functionalization of Gold Nanoparticles
2.4. Synthesis of the Compounds V–VII
2.5. Biological Activity
3. Results and Discussion
3.1. Synthesis of Gold Nanoparticles
3.2. Functionalization of Gold Nanoparticles
3.3. Synthesis of the Compounds V–VII
3.4. Biological Activity
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rai, A.; Prabhune, A.; Perry, C.C. Antibiotic mediated synthesis of gold nanoparticles with potent antimicrobial activity and their application in antimicrobial coatings. J. Mater. Chem. 2010, 20, 6789–6798. [Google Scholar] [CrossRef]
- Felson, D.T.; Anderson, J.J.; Meenan, R.F. The comparative efficacy and toxicity of second-line drugs in rheumatoid arthritis, Results of two meta analyses. Arthritis Rheumatol. 1990, 33, 1449–1461. [Google Scholar] [CrossRef]
- Arvizo, R.R.; Bhattacharyya, S.; Kudgus, R.; Giri, K.; Bhattacharya, R.; Mukherjee, P. Intrinsic therapeutic applications of noble metal nanoparticles: Past, present and future. Chem. Soc. Rev. 2012, 41, 2943–2970. [Google Scholar] [CrossRef] [PubMed]
- Suganya, K.S.; Govindaraju, K.; Kumar, V.G.; Dhas, T.S.; Karthick, V.; Singaravelu, G.; Elanchezhiyan, M. Blue green alga mediated synthesis of gold nanoparticles and its antibacterial efficacy against Gram positive organisms. Mater. Sci. Eng. C 2015, 47, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Xiaoning, L.; Robinson, S.M.; Gupta, A.; Saha, K.; Jiang, Z.; Moyano, D.F.; Sahar, A.; Riley, M.A.; Vincent, M.R. Functional gold nanoparticles as potent antimicrobial agents against multi-drug-resistant bacteria. ACS Nano 2014, 8, 10682–10686. [Google Scholar]
- Niemirowicz, K.; Swiecicka, I.; Wilczewska, A.Z.; Misztalewska, I.; Kalska-Szostko, B.; Bienias, K.; Bucki, R.; Car, H. Gold-functionalized magnetic nanoparticles restrict growth of Pseudomonas aeruginosa. Int. J. Nanomed. 2014, 8, 2217–2224. [Google Scholar]
- Shiang, Y.C.; Ou, C.M.; Chen, S.J.; Ou, T.Y.; Lin, H.J.; Huang, C.C.; Chang, H.T. Highly efficient inhibition of human immunodeficiency virus type 1 reverse transcriptase by aptamers functionalized gold nanoparticles. Nanoscale 2013, 5, 2756–2764. [Google Scholar] [CrossRef] [PubMed]
- Grace, N.A.; Pandian, K. Antibacterial efficacy of aminoglycosidic antibiotics protected gold nanoparticles-A brief study. Int. J. Pharm. 2007, 297, 63–70. [Google Scholar]
- Rastogi, L.; Kora, A.J. Highly stable, protein capped gold nanoparticles as effective drug delivery vehicles for amino-glycosidic antibiotics. Mater. Sci. Eng. C 2012, 32, 1571–1577. [Google Scholar] [CrossRef] [PubMed]
- Pop, C.S.; Hussien, M.D.; Popa, M.; Mares, A.; Grumezescu, A.M.; Grigore, R.; Lazar, V.; Chifiriuc, M.C.; Sakizlian, M.; Bezirtzoglou, E.; et al. Metallic-based micro and nanostructures with antimicrobial activity. Curr. Top. Med. Chem. 2015, 15, 1577–1582. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.B.; Norris, D.J.; Bawendi, M.G. Synthesis and characterization of nearly monodisperse CdE (E = sulfur, selenium, tellurium) semiconductor nanocrystallites. J. Am. Chem. Soc. 1993, 115, 8706–8715. [Google Scholar] [CrossRef]
- O’Brien, S.; Brus, L.; Murray, C.B. Synthesis of monodisperse nanoparticles of barium titanate: Toward a generalized strategy of oxide nanoparticle synthesis. J. Am. Chem. Soc. 2001, 123, 12085–12086. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, S.; Mukherjee, T. Photochemical formation of cooper nanoparticles in PVP. Chem. Phys. Lett. 2003, 370, 83–87. [Google Scholar] [CrossRef]
- Gupta, A.K.; Gupta, M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 2010, 26, 3995–4021. [Google Scholar] [CrossRef] [PubMed]
- Haes, A.J.; Hall, W.P.; Chang, L.; Klein, W.L.; van Duyne, R.P. A localized surface plasmon resonance biosensor; first step towards an assay for Alzheimers disease. Nano Lett. 2004, 4, 1029–1034. [Google Scholar] [CrossRef]
- Zu, Y.; Huang, S.; Liao, W.; Lu, Y.; Wang, S. Gold nanoparticles enhanced electroporation for mammalian cell transfection. J. Biomed. Nanotechnol. 2014, 10, 982–992. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Huang, Y.; Jiang, Q.; Sun, Y.; Deng, L.; Liang, Z.; Du, Q.; Zing, J.; Zhao, Y.; Wang, P.C.; et al. Enhanced gene delivery and siRNA silencing by gold nanoparticles coated with charge-reversal polyelectrolyte. ACS Nano 2010, 4, 5505–5511. [Google Scholar] [CrossRef] [PubMed]
- Elbakry, A.; Zaky, A.; Liebl, R.; Rachel, R.; Goepferich, A.; Breunig, M. Layer-by-layer assembled gold nanoparticles for siRNA delivery. Nano Lett. 2009, 9, 2059–2064. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Deshmukh, H.; Rajagopalan, K.; Wang, S. Enhancing electroporation transfection with Au NPs polyplexelectrophoresis. Electrophoresis 2014, 35, 1837–1845. [Google Scholar] [CrossRef] [PubMed]
- Raschke, G.; Kowarik, S.; Franzl, T.; Sonnichesen, C.; Klar, T.A.; Ferdmann, J. Biomolecular recognition based on single gold nanoparticle light scattering. Nano Lett. 2003, 3, 935–938. [Google Scholar] [CrossRef]
- Jacob, S.; Streeper, R.S.; Fogt, D.L.; Hokama, J.Y.; Tritschler, H.J.; Dietze, G.J.; Henriksen, E.J. The antioxidant α-lipoic acid enhances insulin-stimulated glucose metabolism in insulin-resistant rat skeletal muscle. Diabetes 1996, 45, 1024–1029. [Google Scholar] [CrossRef] [PubMed]
- Roux, S.; Garcia, B.; Bridot, J.-L.; Salomé, M.; Marquette, C.; Lemelle, L.; Gillet, P.; Blum, L.; Perriat, P.; Tillement, O. Synthesis, characterization of dihydrolipoic acid capped gold nanoparticles, and functionalization by the electroluminescent luminol. Langmuir 2005, 21, 2526–2536. [Google Scholar] [CrossRef] [PubMed]
- Calu, L.; Badea, M.; Chifiriuc, M.C.; Bleotu, C.; David, G.I.; Ioniţă, C.; Măruţescu, L.; Lazăr, V.; Stanică, N.; Soponaru, I.; et al. Synthesis, spectral, thermal, magnetic and biological characterization of Co(II), Ni(II), Cu(II) and Zn(II) complexes with a Schiff base bearing a 1,2,4-triazole pharmacophore. J. Therm. Anal. Calorim. 2015, 120, 375–386. [Google Scholar] [CrossRef]
- Stecoza, C.E.; Cǎproiu, M.T.; Drǎghici, C.; Chifiriuc, M.C.; Drǎcea, N.O. Synthesis, characterization and antimicrobial activity evaluation of some new derivatives of 6,11-dihydrodibenzo[b,e]thiepin 5,5-dioxide. Rev. Chim. 2009, 60, 137–141. [Google Scholar]
- Rosu, T.; Pasculescu, S.; Lazar, V.; Chifiriuc, C.; Cernat, R. Combinations of Cu(II) with mixed ligands derived from 4-amino-2,3-dimethyl-1-phenyl-3-pyrazolin-5-one with biological activity. Molecules 2006, 11, 904–914. [Google Scholar] [CrossRef] [PubMed]
- Limban, C.; Balotescu, M.C.; Missir, A.V.; Chiriţă, I.C.; Bleotu, C. Antimicrobial activity of some new thioureides derived from 2-(4-chlorophenoxymethyl)benzoic acid. Molecules 2008, 13, 567–580. [Google Scholar] [CrossRef] [PubMed]
- Limban, C.; Marutescu, L.; Chifiriuc, M.C. Synthesis, spectroscopic properties and antipathogenic activity of new thiourea derivatives. Molecules 2011, 16, 7593–7607. [Google Scholar] [CrossRef] [PubMed]
- Olar, R.; Badea, M.; Marinescu, D.; Chifiriuc, M.C.; Bleotu, C.; Grecu, M.N.; Iorgulescu, E.; Bucur, M.; Lazar, V.; Finaru, A. Prospects for new antimicrobials based on N,N-dimethylbiguanide complexes as effective agents on both planktonic and adhered microbial strains. Eur. J. Med. Chem. 2010, 45, 2868–2875. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.J.; Ge, Q.F.; Guo, D.W.; Hu, W.X.; Liu, H.Z. Synthesis and anticancer evaluation of alpha-lipoic acid derivatives. Bioorg. Med. Chem. Lett. 2010, 20, 3078–3083. [Google Scholar] [CrossRef] [PubMed]





| Sample | E. coli | P. aeruginosa | E. faecalis | S. aureus | B. subtilis | C. albicans |
|---|---|---|---|---|---|---|
| I | >1 | >1 | >1 | >1 | >1 | >1 |
| II | >1 | >1 | >1 | >1 | 1 | >1 |
| III | >1 | >1 | >1 | >1 | >1 | >1 |
| IV | >1 | >1 | >1 | >1 | >1 | >1 |
| V | >1 | >1 | >1 | >1 | 0.25 | >1 |
| VI | >1 | 0.031 | 1 | >1 | 0.002 | >1 |
| VII | >1 | 0.004 | >1 | >1 | 1 | >1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Turcu, I.; Zarafu, I.; Popa, M.; Chifiriuc, M.C.; Bleotu, C.; Culita, D.; Ghica, C.; Ionita, P. Lipoic Acid Gold Nanoparticles Functionalized with Organic Compounds as Bioactive Materials. Nanomaterials 2017, 7, 43. https://doi.org/10.3390/nano7020043
Turcu I, Zarafu I, Popa M, Chifiriuc MC, Bleotu C, Culita D, Ghica C, Ionita P. Lipoic Acid Gold Nanoparticles Functionalized with Organic Compounds as Bioactive Materials. Nanomaterials. 2017; 7(2):43. https://doi.org/10.3390/nano7020043
Chicago/Turabian StyleTurcu, Ioana, Irina Zarafu, Marcela Popa, Mariana Carmen Chifiriuc, Coralia Bleotu, Daniela Culita, Corneliu Ghica, and Petre Ionita. 2017. "Lipoic Acid Gold Nanoparticles Functionalized with Organic Compounds as Bioactive Materials" Nanomaterials 7, no. 2: 43. https://doi.org/10.3390/nano7020043