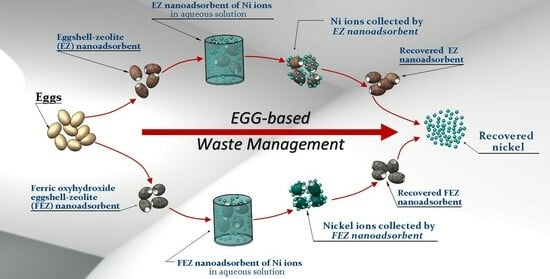
Innovative Low-Cost Composite Nanoadsorbents Based on Eggshell Waste for Nickel Removal from Aqueous Media
Abstract
:1. Introduction
2. Methods
2.1. Materials
2.2. Instrumentation
3. Preparation of Adsorbents
3.1. EZ Nanoadsorbent
3.2. FEZ Nanoadsorbent
3.3. Batch Adsorption Study
3.3.1. Kinetic Study
3.3.2. Adsorbent Performance
3.3.3. Desorption Study
3.3.4. Kinetic Studies
3.3.5. Thermodynamic Study
3.3.6. Statistical Analysis
4. Results
4.1. BET Analysis
4.2. FT-IR Spectroscopy
4.3. SEM Analysis
4.4. XRD Study
4.5. Adsorption Properties
4.5.1. Effect of Adsorbent
4.5.2. Effect of Initial Concentration on Nickel Removal Efficiency
4.5.3. Effect of pH
4.5.4. Effect of Contact Time
- (a)
- In the first stage (0–120 min), adsorption increases rapidly, a phenomenon that could be attributed to the high availability of active sites in the adsorbent;
- (b)
- In the second stage (120–240 min), attenuation of the adsorption rate occurs, as a result of the decrease in available active sites;
- (c)
- In the third stage (240–460 min), metal adsorption exhibits a plateau trend, indicating saturation of the active sites, after reaching equilibrium. The data suggest that the optimal time necessary for the adsorption process to reach equilibrium for each of the adsorbents is 240 min.
4.5.5. Effect of Temperature on the Adsorption Process
4.5.6. Nickel Removal Efficiency—Comparative Analysis between the Adsorbent and Starting Materials
4.5.7. Adsorption Isotherms
4.5.8. Thermodynamic Study
4.5.9. Adsorption Kinetic Study
4.5.10. Insight into Adsorption
4.5.11. Comparison of Nickel Removal Efficiency among Other Adsorbents
4.5.12. Desorption Study and Adsorbent Regeneration
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- EPA—United States Environmental Protection Agency. Technical Documentation: Drought. 2021. Available online: https://www.epa.gov/climate-indicators/climate-change-indicators-drought (accessed on 28 December 2022).
- El-Naggar, A.; Ahmed, N.; Mosa, A.; Niazi, N.K.; Yousaf, B.; Sharma, A.; Sarkar, B.; Cai, Y.; Chang, S.X. Nickel in soil and water: Sources, biogeochemistry, and remediation using biochar. J. Hazard. Mater. 2021, 419, 126421. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, K.; Nansai, K.; Matsubae, K.; Tomita, M.; Takayanagi, W.; Nagasaka, T. Global land-use change hidden behind nickel consumption. Sci. Total Environ. 2017, 586, 730–737. [Google Scholar] [CrossRef] [PubMed]
- Yokoi, R.; Watari, T.; Motoshita, M. Future greenhouse gas emissions from metal production: Gaps and opportunities towards climate goals. Energy Environ. Sci. 2022, 15, 146. [Google Scholar] [CrossRef]
- EFSA Panel on Contaminants in the Food Chain (CONTAM); Schrenk, D.; Bignami, M.; Bodin, L.; Chipman, J.K.; Del Mazo, J.; Grasl-Kraupp, B.; Hogstrand, C.; Hoogenboom, L.R.; Leblanc, J.C.; et al. Update of the risk assessment of nickel in food and drinking water. EFSA J. 2020, 18, e06268. [Google Scholar]
- Genchi, G.; Carocci, A.; Lauria, G.; Sinicropi, M.S.; Catalano, A. Nickel: Human health and environmental toxicology. Int. J. Environ. Res. Public Health 2020, 17, 679. [Google Scholar] [CrossRef]
- Commission of the European Communities. On the Quality of Water Intended for Human Consumption. Off. J. Eur. Communities 1998, 330, 32–54. [Google Scholar]
- Qasem, N.A.A.; Mohammed, R.H.; Lawal, D.U. Removal of heavy metal ions from wastewater: A comprehensive and critical review. NPJ Clean Water 2021, 4, 36. [Google Scholar] [CrossRef]
- Öden, M.K.; Karasakal, E.N.; Çildir, S. Nickel (II) removal from synthetic wastewater by adsorption using waste pea shell. Int. J. Environ. Trends 2022, 6, 10–20. [Google Scholar]
- Fernández-Reyes, B.; Ortiz-Martínez, K.; Lasalde-Ramírez, J.A.; Hernández-Maldonado, A.J. Engineered adsorbents for the removal of contaminants of emerging concern from water. In Contaminants of Emerging Concern in Water and Wastewater; Butterworth-Heinemann: Oxford, UK, 2020; pp. 3–45. [Google Scholar]
- Krishna Kumar, A.S.; Warchol, J.; Matusik, J.; Tseng, W.L.; Rajesh, N.; Bajda, T. Heavy metal and organic dye removal via a hybrid porous hexagonal boron nitride-based magnetic aerogel. NPJ Clean Water 2022, 5, 24. [Google Scholar] [CrossRef]
- European Commission. Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions, Closing the Loop—An EU Action Plan for the Circular Economy; European Commission: Brussels, Belgium, 2015. [Google Scholar]
- Al Dwairi, R.A.; Al-Rawajfeh, A.E. Removal of cobalt and nickel from wastewater by using Jordan low-cost zeolite and bentonite. J. Univ. Chem. Technol. Metall. 2012, 47, 69–76. [Google Scholar]
- Al-Haj, A.A.; El-Bishtawi, R. Removal of lead and nickel ions using zeolite tuff. J. Chem. Technol. Biotechnol. Int. Res. Process Environ. AND Clean Technol. 1997, 69, 27–34. [Google Scholar]
- Argun, M.E. Use of clinoptilolite for the removal of nickel ions from water: Kinetics and thermodynamics. J. Hazard. Mater. 2008, 150, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Olad, A.; Ahmadi, S.; Rashidzadeh, A. Removal of nickel (II) from aqueous solutions with polypyrrole modified clinoptilolite: Kinetic and isotherm studies. Desalination Water Treat. 2013, 51, 7172–7180. [Google Scholar] [CrossRef]
- Parades-Aguilar, J.; Reyes-Martínez, V.; Bustamante, G.; Almendáriz-Tapia, F.J.; Martínez-Meza, G.; Vílchez-Vargas, R.; Link, A.; Certucha-Barragán, M.T.; Calderón, K. Removal of nickel(II) from wastewater using a zeolite-packed anaerobic bioreactor: Bacterial diversity and community structure shifts. J. Environ. Manag. 2021, 279, 111558. [Google Scholar] [CrossRef] [PubMed]
- Rajic, N.; Stojakovic, D.; Jovanovic, M.; Logar, N.Z.; Mazaj, M.; Kaucic, V. Removal of nickel(II) ions from aqueous solutions using the natural clinoptilolite and preparation of nano-NiO on the exhausted clinoptilolite. Appl. Surf. Sci. 2010, 257, 1524–1532. [Google Scholar] [CrossRef]
- Alquzweeni, S.S.; Alkizwini, R.S. Removal of cadmium from contaminated water using coated chicken bones with double-layer hydroxide (Mg/Fe-LDH). Water 2020, 12, 2303. [Google Scholar] [CrossRef]
- Aman, T.; Kazi, A.A.; Sabri, M.U.; Bano, Q. Potato peels as solid waste for the removal of heavy metal copper(II) from waste water/industrial effluent. Colloids Surf. B Biointerfaces 2008, 63, 116–121. [Google Scholar] [CrossRef]
- Annane, K.; Lemlikchi, W.; Tingry, S. Efficiency of eggshell as a low-cost adsorbent for removal of cadmium: Kinetic and isotherm studies. Biomass Convers. Biorefinery 2021, 13, 6163–6174. [Google Scholar] [CrossRef]
- Carvalho, J.; Araujo, J.; Castro, F. Alternative low-cost adsorbent for water and wastewater decontamination derived from eggshell waste: An overview. Waste Biomass Valor. 2011, 2, 157–167. [Google Scholar] [CrossRef]
- El-Azazy, M.; El-Shafie, A.S.; Issa, A.A.; Al-Sulaiti, M.; Al-Yafie, J.; Shomar, B.; Al-Saad, K. Potato peels as an adsorbent for heavy metals from aqueous solutions: Eco-structuring of a green adsorbent operating Plackett–Burman design. J. Chem. 2019, 2019, 4926240. [Google Scholar] [CrossRef]
- Zonato, R.D.O.; Estevam, B.R.; Perez, I.D.; Aparecida dos Santos Ribeiro, V.; Boina, R.F. Eggshell as an adsorbent for removing dyes and metallic ions in aqueous solutions. Clean. Chem. Eng. 2022, 2, 100023. [Google Scholar] [CrossRef]
- Waheed, M.; Yousaf, M.; Shehzad, A.; Inam-Ur-Raheem, M.; Khan, M.K.I.; Khan, M.R.; Ahmad, N.; Abdullah; Aadil, R.M. Channeling eggshell waste to valuable and utilizable products: A comprehensive review. Trends Food Sci. Technol. 2020, 106, 78–90. [Google Scholar] [CrossRef]
- Mignardi, S.; Archilletti, L.; Medeghini, L.; De Vito, C. Valorization of eggshell biowaste for sustainable environmental remediation. Sci. Rep. 2020, 10, 2436. [Google Scholar] [CrossRef]
- Chen, D.; Xiao, X.; Yang, K. Removal of phosphate and hexavalent chromium from aqueous solutions by engineered waste eggshell. RSC Adv. 2016, 6, 35332–35339. [Google Scholar] [CrossRef]
- Tripathi, A.; Ranjan, M.R. Heavy metal removal from wastewater using low cost adsorbents. J. Bioremed. Biodeg. 2015, 6, 315. [Google Scholar] [CrossRef]
- Segneanu, A.-E.; Marin, C.N.; Vlase, G.; Cepan, C.; Mihailescu, M.; Muntean, C.; Grozescu, I. Highly efficient engineered waste eggshell-fly ash for cadmium removal from aqueous solution. Sci. Rep. 2022, 12, 9676. [Google Scholar] [CrossRef] [PubMed]
- Tsai, W.T.; Yang, J.M.; Hsu, H.C.; Lin, C.M.; Lin, K.Y.; Chiu, C.H. Development and characterization of mesoporosity in eggshell ground by planetary ball milling. Microporous Mesoporous Mater. 2008, 111, 379–386. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Xin, Y.; Li, C.; Liu, J.; Liu, J.; Liu, Y.; He, W.; Gao, Y. Adsorption of heavy metal with modified eggshell membrane and the in situ synthesis of Cu-Ag/modified eggshell membrane composites. R. Soc. Open Sci. 2018, 5, 180532. [Google Scholar] [CrossRef]
- Chukanov, N.V.; Chervonnyi, A.D. IR Spectra of minerals and related compounds, and reference samples’ data. In Infrared Spectroscopy of Minerals and Related Compounds; Springer Mineralogy; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Zendelska, A.; Golomeova, M.; Jakupi, Š.; Lisičkov, K.; Kuvendžiev, S.; Marinkovski, M. Characterization and application of clinoptilolite for removal of heavy metal ions from water resources. Geol. Maced. 2018, 32, 21–32. [Google Scholar]
- Kaufhold, S.; Ufer, K.; Hein, M.; Götze, N.; Dohrmann, R. A combined IR and XRD study of natural well crystalline goethites (α-FeOOH). Acta Geochim. 2022, 41, 794–810. [Google Scholar] [CrossRef]
- Park, H.J.; Jeong, S.W.; Yang, J.K.; Kim, B.G.; Lee, S.M. Removal of heavy metals using waste eggshell. J. Environ. Sci. 2007, 19, 1436–1441. [Google Scholar] [CrossRef] [PubMed]
- Theis, T.L.; Richter, R.O. Adsorption reactions of nickel species at oxide surfaces. In Particulates in Water; Kavanaugh, M.C., Leckie, J.O., Eds.; Advances in Chemistry Series; American Chemical Society: Washington, DC, USA, 1980; pp. 73–96. [Google Scholar]
- Yang, L.; He, L.; Xue, J.; Wu, L.; Ma, Y.; Li, H.; Peng, P.; Li, M.; Zhang, Z. Highly efficient nickel (II) removal by sewage sludge biochar supported α-Fe2O3 and α- FeOOH: Sorption characteristics and mechanisms. PLoS ONE 2019, 14, e0218114. [Google Scholar] [CrossRef] [PubMed]
- Cornell, R.M.; Giovanoli, R.; Schneider, W. Review of the hydrolysis of iron(III) and the crystallization of amorphous iron(III) hydroxide hydrate. J. Chem. Technol. Biotechnol. 2007, 46, 115–134. [Google Scholar] [CrossRef]
- Al-Abbad, E.A.; Al Dwairi, R.A. Removal of nickel (II) ions from water by Jordan natural zeolite as sorbent material. J. Saudi Chem. Soc. 2021, 25, 101233. [Google Scholar] [CrossRef]
- Lei, T.; Li, S.-J.; Jiang, F.; Ren, Z.-X.; Wang, L.-L.; Yang, X.-J.; Tang, L.-H.; Wang, S.-X. Adsorption of cadmium ions from an aqueous solution on a highly stable dopamine-modified magnetic nano-adsorbent. Nanoscale Res. Lett. 2019, 14, 352. [Google Scholar] [CrossRef]
- Mohan, S.; Gandhimathi, R. Removal of heavy metal ions from municipal solid waste leachate using coal fly ash as an adsorbent. J. Hazard. Mater. 2009, 169, 351–359. [Google Scholar] [CrossRef]
- Fulazzaky, M.A. Determining the resistance of mass transfer for adsorption of the surfactants onto granular activated carbons from hydrodynamic column. Chem. Eng. J. 2011, 166, 832–840. [Google Scholar] [CrossRef]
- Noble, R.D.; Terry, P.A. Adsorption. In Principles of Chemical Separations with Environmental Applications; Cambridge Series in Chemical Engineering; Cambridge University Press: Cambridge, UK, 2004; pp. 182–213. [Google Scholar]
- Hosseini, S.S.S.; Khosravi, A.; Tavakoli, H.; Esmhosseini, M.; Khezri, S. Natural zeolite for nickel ions removal from aqueous solutions: Optimization and modeling using response surface methodology based on central composite design. Desalination Water Treat. 2015, 57, 16898–16906. [Google Scholar] [CrossRef]
- Kara, A.; Demirbel, E. Kinetic, isotherm and thermodynamic analysis on adsorption of Cr(VI) ions from aqueous solutions by synthesis and characterization of magnetic-poly(divinylbenzene-vinylimidazole) microbeads. Water Air Soil Pollut. 2011, 223, 2387–2403. [Google Scholar] [CrossRef]
- Zhai, Q.Z.; Li, X.D. Efficient removal of cadmium (II) with SBA-15 nanoporous silica: Studies on equilibrium, isotherm, kinetics and thermodynamics. Appl. Water. Sci. 2019, 9, 143. [Google Scholar] [CrossRef]
- Ma, L.; Wei, Q.; Chen, Y.; Song, Q.; Sun, C.; Wang, Z.; Wu, G. Removal of cadmium from aqueous solutions using industrial coal fly ash-nZVI. R. Soc. Open Sci. 2018, 5, 171051. [Google Scholar] [CrossRef] [PubMed]
- De Angelis, G.; Medeghini, L.; Conte, A.M.; Mignardi, S. Recycling of eggshell waste into low-cost adsorbent for Ni removal from wastewater. J. Clean. Prod. 2017, 164, 1497–1506. [Google Scholar] [CrossRef]
- Ayawei, N.; Newton Ebelegi, A.; Wankasi, D. Modelling and interpretation of adsorption isotherms. J. Chem. 2017, 2017, 3039817. [Google Scholar] [CrossRef]
- Mnasri-Ghnimi, S.; Frini-Srasra, N. Removal of heavy metals from aqueous solutions by adsorption using single and mixed pillared clays. Appl. Clay Sci. 2019, 179, 105151. [Google Scholar] [CrossRef]
- Khelifi, O.; Nacef, M.; Affoune, A.M. Biosorption of Nickel(II) ions from aqueous solutions by using Chicken eggshells as low-cost biosorbent. Alger. J. Environ. Sci. Technol. 2016, 2, 107–111. [Google Scholar]
- Darweesh, M.A.; Elgendy, M.Y.; Ayad, M.I.; Ahmed, A.M.M.; Elsayed, N.M.K.; Hammad, W.A. Adsorption isotherm, kinetic, and optimization studies for copper (II) removal from aqueous solutions by banana leaves and derived activated carbon. S. Afr. J. Chem. Eng. 2022, 40, 10–20. [Google Scholar] [CrossRef]
- Ouyang, D.; Zhuo, Y.; Hu, L.; Zeng, Q.; Hu, Y.; He, Z. Research on the adsorption behavior of heavy metal ions by porous material prepared with silicate tailings. Minerals 2019, 9, 291. [Google Scholar] [CrossRef]
- Wang, Z.; Xu, W.; Jie, F.; Zhao, Z.; Zhou, K.; Liu, H. The selective adsorption performance and mechanism of multiwall magnetic carbon nanotubes for heavy metals in wastewater. Sci. Rep. 2021, 11, 16878. [Google Scholar] [CrossRef]
- Nordstrand, J.; Dutta, J. Dynamic Langmuir model: A simpler approach to modeling capacitive deionization. J. Phys. Chem. C 2019, 123, 16479–16485. [Google Scholar] [CrossRef]
- Sahmoune, M.N. Evaluation of thermodynamic parameters for adsorption of heavy metals by green adsorbents. Environ. Chem. Lett. 2019, 17, 697–704. [Google Scholar] [CrossRef]
- Wang, J.; Guo, X. Adsorption kinetic models: Physical meanings, applications, and solving methods. J. Hazard. Mater. 2020, 390, 122156. [Google Scholar] [CrossRef] [PubMed]
- Ebelegi, A.N.; Ayawei, N.; Wankasi, D. Interpretation of adsorption thermodynamics and kinetics. Open J. Phys. Chem. 2020, 10, 166–182. [Google Scholar] [CrossRef]
- Rahmati, A.; Mardani, S. Nanocrystalline NiO structure: Preferred oriented growth nanocrystalline NiO structure: Preferred oriented growth. Turk. J. Phys. 2015, 39, 7. [Google Scholar] [CrossRef]
- Qiao, H.; Wei, Z.; Yang, H.; Zhu, L.; Yan, X. Preparation and characterization of NiO nanoparticles by anodic arc plasma method. J. Nanomater. 2009, 2009, 795928. [Google Scholar] [CrossRef]
- Pérez-Botella, E.; Valencia, S.; Rey, F. Zeolites in adsorption processes: State of the art and future prospects. Chem. Rev. 2022, 122, 17647–17695. [Google Scholar] [CrossRef]
- Sharma, A.K.; Desnavi, S.; Dixit, C.; Varshney, U.; Sharma, A. Extraction of nickel nanoparticles from electroplating waste &their application in production of bio-diesel from biowaste. Int. J. Chem. Eng. Appl. 2014, 6, 156. [Google Scholar]
- Pang, Y.; Zhao, C.; Li, Y.; Li, Q.; Bayongzhong, X.; Peng, D.; Huang, T. Cadmium adsorption performance and mechanism from aqueous solution using red mud modified with amorphous MnO2. Sci. Rep. 2022, 12, 4424. [Google Scholar] [CrossRef]
- Gorzin, F.; Bahri Rasht Abadi, M. Adsorption of Cr(VI) from aqueous solution by adsorbent prepared from paper mill sludge: Kinetics and thermodynamics studies. Adsorpt. Sci. Technol. 2017, 36, 149–169. [Google Scholar] [CrossRef]
- Nasehi, P.; Mahmoudi, B.; Abbaspour, S.F.; Moghaddam, M.S. Cadmium adsorption using novel MnFe2O4-TiO2-UIO-66 magnetic nanoparticles and condition optimization using a response surface methodology. RSC Adv. 2019, 9, 20087–20099. [Google Scholar] [CrossRef]
- Ghose, S.K.; Waychunas, G.A.; Trainor, T.P.; Eng, P.J. Hydrated goethite (α-FeOOH) (100) interface structure: Ordered water and surface functional groups. Geochim. Cosmochim. Acta 2010, 74, 1943–1953. [Google Scholar] [CrossRef]
- Khan, M.N.; Ullah, H.; Naeem, S.; Uddin, J.; Hamid, Y.; Ahmad, W.; Ding, J. Remediation of emerging heavy metals from water using natural adsorbent: Adsorption performance and mechanistic insights. Sustainability 2021, 13, 8817. [Google Scholar] [CrossRef]
- Ho, J.H.; Yeh, Y.N.; Wang, H.W.; Khoo, S.K.; Chen, Y.H.; Chow, C.F. Removal of nickel and silver Ions using eggshells with membrane, eggshell membrane, and eggshells. Food Sci. Technol. Res. 2014, 20, 337–343. [Google Scholar] [CrossRef]
- Mashangwa, T.D. An Investigation into the Efficacy of Eggshells as a Low Cost Adsorbent for the Removal of Potentially Toxic Inorganic Elements from Aqueous Solutions. Master’s Dissertation, Environmental Science. University of South Africa, Cape Town, South Africa, 2016. [Google Scholar]
- Stevens, M.; Batlokwa, B. Removal of nickel (II) and cobalt (II) from wastewater using vinegar-treated eggshell waste biomass. J. Water Resour. Prot. 2017, 9, 931–944. [Google Scholar] [CrossRef]
- Muhamad, N.; Abdullah, N.; Rahman, M.A.; Abas, K.H.; Aziz, A.A.; Othman, M.H.D.; Jaafar, J.; Ismail, A.F. Removal of nickel from aqueous solution using supported zeolite-Y hollow fiber membranes. Environ. Sci. Pollut. Res. 2018, 25, 19054–19064. [Google Scholar] [CrossRef]
- Ismail, M.H.S.; Zhang, X.T.; Adnan, S.N. Application of clinoptilolite in removal of nickel (II) in plating wastewater. World Appl. Sci. J. 2012, 18, 659–664. [Google Scholar]
- Charazińska, S.; Burszta-Adamiak, E.; Lochyński, P. Recent trends in Ni(II) sorption from aqueous solutions using natural materials. Rev. Environ. Sci. Biotechnol. 2022, 21, 105–138. [Google Scholar] [CrossRef]
- Lysenko, V.I. Production and properties of ceramic made from nickel oxide nanopowder. Glass Ceram. 2022, 79, 9–10. [Google Scholar] [CrossRef]
- Sinyoung, S.; Taweekitwanit, E.; Kajitvichyanukul, P. Effects of Nickel on properties of cement mortar derived from the co-burning of industrial waste and its leaching behavior. Adv. Mat. Res. 2015, 1103, 121–127. [Google Scholar] [CrossRef]
- Patra, A.; Dutta, A.; Mohapatra, K.K.; Pradhan, S. Nickel the ultra-micronutrient: Significant for plant growth and metabolism. Food Chem. Toxicol. 2020, 1, 35–37. [Google Scholar]
- Vischetti, C.; Marini, E.; Casucci, C.; De Bernardi, A. Nickel in the environment: Bioremediation techniques for soils with low or moderate contamination in European Union. Environments 2022, 9, 133. [Google Scholar] [CrossRef]













| Sample | Surface Area (m2/g) | Average Pore Size Diameter (nm) | Total Pore Volume (cm3/g) |
|---|---|---|---|
| Eggshell | 1.311 | 8.347 | 2.03 × 10−3 |
| Zeolite | 12.111 | 15.574 | 38.06 × 10−3 |
| EZ | 19.321 | 7.132 | 11.20 × 10−3 |
| FEZ | 23.901 | 4.023 | 8.10 × 10−3 |
| t-test ** | t Stat: 0.98; t Critical two-tail: 2.44 | ||
| t Stat: −0.72; t Critical two-tail: 2.44 | |||
| Adsorbent Material | Langmuir Model | Freundlich Model | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Qe,exp | Qm | k | RL | R2 | KF | n | R2 | Ea (kJ/mol) | |
| FEZ | 321.1 | 321.0 | 0.283 | 0.861 | 0.9994 | 4.60517 | 1.893 | 0.9999 | 32.4 |
| EZ | 287.9 | 286.8 | 0.223 | 0.783 | 0.9989 | 3.85721 | 1.659 | 0.9998 | 32.2 |
| T (K) | Adsorbents | |||||
|---|---|---|---|---|---|---|
| FEZ | EZ | |||||
| ΔGo (kJ/mol) | ΔHo (kJ/mol) | ΔSo J/(mol K) | ΔGo (kJ/mol) | ΔHo (kJ/mol) | ΔSo J/(mol K) | |
| 295.15 | −10.50 | 28.89 | 154.35 | −7.14 | 24.61 | 138.32 |
| 303.15 | −18.83 | −12.12 | ||||
| 313.15 | −27.15 | −17.03 | ||||
| Adsorbent Material | Qeexp (mg/g) | Pseudo-First Order | Pseudo-Second Order | Intraparticle Diffusion | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Qecalc | K1 | R2 | Qecalc | K2 | R2 | Ki | C | R2 | ||
| FEZ | 321.1 | 322.28 | 0.023 | 0.9994 | 321.58 | 2.781 | 0.9997 | 8.0276 | 33.875 | 0.9764 |
| EZ | 287.9 | 288.35 | 0.011 | 0.9991 | 287.95 | 1.924 | 0.9993 | 6.2782 | 21.466 | 0.9726 |
| Adsorbent Type | Removal Efficiency (%) | Ni Initial Concentration (%) | Reference |
|---|---|---|---|
| eggshell | 90.9 | 100 mg/L | [69] |
| eggshell | 93.5 | 100 ppm | [70] |
| eggshell-derived hydroxyapatite | 91.0 | 100 mg/L | [49] |
| vinegar-treated eggshell waste biomass | 76.5 | 1000 ppm | [71] |
| clinoptilolite | 93.6 | 100 mg/L | [15] |
| zeolite | 58.6 | 1000 mg/L | [40] |
| supported zeolite-Y hollow fiber membranes | 63.0 | 10 mg/L | [72] |
| clinoptilolite | 60.0 | 25 mg/L | [73] |
| EZ | 97.3 | 25.5 mg/L | This study |
| FEZ | 99.9 | 25.5 mg/L | This study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Segneanu, A.-E.; Trusca, R.; Cepan, C.; Mihailescu, M.; Muntean, C.; Herea, D.D.; Grozescu, I.; Salifoglou, A. Innovative Low-Cost Composite Nanoadsorbents Based on Eggshell Waste for Nickel Removal from Aqueous Media. Nanomaterials 2023, 13, 2572. https://doi.org/10.3390/nano13182572
Segneanu A-E, Trusca R, Cepan C, Mihailescu M, Muntean C, Herea DD, Grozescu I, Salifoglou A. Innovative Low-Cost Composite Nanoadsorbents Based on Eggshell Waste for Nickel Removal from Aqueous Media. Nanomaterials. 2023; 13(18):2572. https://doi.org/10.3390/nano13182572
Chicago/Turabian StyleSegneanu, Adina-Elena, Roxana Trusca, Claudiu Cepan, Maria Mihailescu, Cornelia Muntean, Dumitru Daniel Herea, Ioan Grozescu, and Athanasios Salifoglou. 2023. "Innovative Low-Cost Composite Nanoadsorbents Based on Eggshell Waste for Nickel Removal from Aqueous Media" Nanomaterials 13, no. 18: 2572. https://doi.org/10.3390/nano13182572