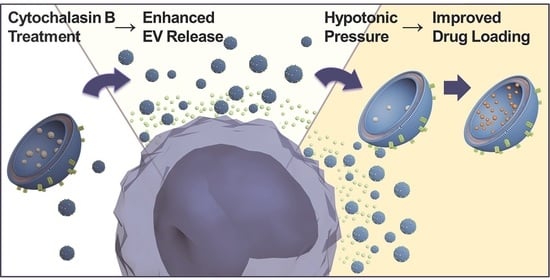
Cytochalasin B Treatment and Osmotic Pressure Enhance the Production of Extracellular Vesicles (EVs) with Improved Drug Loading Capacity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. EV Production and Isolation
2.3. Dendrimer Preparation, Characterization, and Modification
2.4. Nanoparticle Tracking Analysis (NTA)
2.5. Hypotonic Solution Preparation
2.6. Assessment of Cytotoxicity of CB Treatment and Osmotic Pressure
2.7. Protein Concentration Measurement
2.8. Transmission Electron Microscopy (TEM) Imaging
2.9. Western Blotting
2.10. Cell Uptake and Confocal Microscopy Imaging
2.11. Dendrimer/DOX Loading
2.12. Cell Viability Measurement
2.13. Statistical Analysis
3. Results and Discussion
3.1. Cytochalasin B Treatment Increases EV Secretion
3.2. Hypo-Osmotic Pressure Helps Release Intracellular Proteins without Affecting CB Activity
3.3. CB Treatment and Hypo-Omotic Pressure Synergistically Enhances Loading Capacity
3.4. The Combination of CB Treatment and Hypo-Osmotic-Pressure-Improved Drug Delivery
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- de Jong, O.G.; Kooijmans, S.A.A.; Murphy, D.E.; Jiang, L.; Evers, M.J.W.; Sluijter, J.P.G.; Vader, P.; Schiffelers, R.M. Drug delivery with extracellular vesicles: From imagination to innovation. ACC Chem. Res. 2019, 52, 1761–1770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Niel, G.; D’Angelo, G.; Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Kalra, H.; Drummen, G.P.; Mathivanan, S. Focus on extracellular vesicles: Introducing the next small big thing. Int. J. Mol. Sci. 2016, 17, 170. [Google Scholar] [CrossRef] [Green Version]
- O’Brien, K.; Breyne, K.; Ughetto, S.; Laurent, L.C.; Breakefield, X.O. Rna delivery by extracellular vesicles in mammalian cells and its applications. Nat. Rev. Mol. Cell Biol. 2020, 21, 585–606. [Google Scholar] [CrossRef] [PubMed]
- Bu, J.; Lee, T.H.; Poellmann, M.J.; Rawding, P.A.; Jeong, W.-J.; Hong, R.S.; Hyun, S.H.; Eun, H.S.; Hong, S. Tri-modal liquid biopsy: Combinational analysis of circulating tumor cells, exosomes, and cell-free dna using machine learning algorithm. Clin. Transl. Med. 2021, 11, e499. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Vader, P.; Schiffelers, R.M. Extracellular vesicles for nucleic acid delivery: Progress and prospects for safe rna-based gene therapy. Gene Ther. 2017, 24, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Kamerkar, S.; LeBleu, V.S.; Sugimoto, H.; Yang, S.; Ruivo, C.F.; Melo, S.A.; Lee, J.J.; Kalluri, R. Exosomes facilitate therapeutic targeting of oncogenic kras in pancreatic cancer. Nature 2017, 546, 498–503. [Google Scholar] [CrossRef]
- Haney, M.J.; Klyachko, N.L.; Zhao, Y.; Gupta, R.; Plotnikova, E.G.; He, Z.; Patel, T.; Piroyan, A.; Sokolsky, M.; Kabanov, A.V.; et al. Exosomes as drug delivery vehicles for parkinson’s disease therapy. J. Control. Release 2015, 207, 18–30. [Google Scholar] [CrossRef] [Green Version]
- Kim, M.S.; Haney, M.J.; Zhao, Y.; Yuan, D.; Deygen, I.; Klyachko, N.L.; Kabanov, A.V.; Batrakova, E.V. Engineering macrophage-derived exosomes for targeted paclitaxel delivery to pulmonary metastases: In vitro and in vivo evaluations. Nanomedicine 2018, 14, 195–204. [Google Scholar] [CrossRef]
- Tian, Y.; Li, S.; Song, J.; Ji, T.; Zhu, M.; Anderson, G.J.; Wei, J.; Nie, G. A doxorubicin delivery platform using engineered natural membrane vesicle exosomes for targeted tumor therapy. Biomaterials 2014, 35, 2383–2390. [Google Scholar] [CrossRef]
- Gangadaran, P.; Ahn, B.C. Extracellular vesicle- and extracellular vesicle mimetics-based drug delivery systems: New perspectives, challenges, and clinical developments. Pharmaceutics 2020, 12, 442. [Google Scholar] [CrossRef]
- Luan, X.; Sansanaphongpricha, K.; Myers, I.; Chen, H.; Yuan, H.; Sun, D. Engineering exosomes as refined biological nanoplatforms for drug delivery. Acta Pharm. Sin. 2017, 38, 754–763. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ong, S.G.; Chitneni, M.; Lee, K.S.; Ming, L.C.; Yuen, K.H. Evaluation of extrusion technique for nanosizing liposomes. Pharmaceutics 2016, 8, 36. [Google Scholar] [CrossRef] [PubMed]
- Le, Q.V.; Lee, J.; Lee, H.; Shim, G.; Oh, Y.K. Cell membrane-derived vesicles for delivery of therapeutic agents. Acta Pharm. Sin. B 2021, 11, 2096–2113. [Google Scholar] [CrossRef]
- Lu, M.; Zhao, X.; Xing, H.; Xun, Z.; Zhu, S.; Lang, L.; Yang, T.; Cai, C.; Wang, D.; Ding, P. Comparison of exosome-mimicking liposomes with conventional liposomes for intracellular delivery of sirna. Int. J. Pharm. 2018, 550, 100–113. [Google Scholar] [CrossRef] [PubMed]
- Cho, N.J.; Hwang, L.Y.; Solandt, J.J.R.; Frank, C.W. Comparison of extruded and sonicated vesicles for planar bilayer self-assembly. Materials 2013, 6, 3294–3308. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.J.; Wu, J.Y.; Liu, J.; Xu, W.; Qiu, X.; Huang, S.; Hu, X.B.; Xiang, D.X. Artificial exosomes for translational nanomedicine. J. Nanobiotechnol. 2021, 19, 242. [Google Scholar] [CrossRef]
- Antimisiaris, S.G.; Mourtas, S.; Marazioti, A. Exosomes and exosome-inspired vesicles for targeted drug delivery. Pharmaceutics 2018, 10, 218. [Google Scholar] [CrossRef] [Green Version]
- King, H.W.; Michael, M.Z.; Gleadle, J.M. Hypoxic enhancement of exosome release by breast cancer cells. BMC Cancer 2012, 12, 421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, W.; Zhou, X.; Yao, Q.; Liu, Y.; Zhang, H.; Dong, Z. Hif-1-mediated production of exosomes during hypoxia is protective in renal tubular cells. Am. J. Physiol. Ren. Physiol. 2017, 313, F906–F913. [Google Scholar] [CrossRef]
- Gomzikova, M.O.; Zhuravleva, M.N.; Miftakhova, R.R.; Arkhipova, S.S.; Evtugin, V.G.; Khaiboullina, S.F.; Kiyasov, A.P.; Persson, J.L.; Mongan, N.P.; Pestell, R.G.; et al. Cytochalasin b-induced membrane vesicles convey angiogenic activity of parental cells. Oncotarget 2017, 8, 70496–70507. [Google Scholar] [CrossRef]
- Bu, J.; Shim, J.E.; Lee, T.H.; Cho, Y.H. Multi-modal liquid biopsy platform for cancer screening: Screening both cancer-associated rare cells and cancer cell-derived vesicles on the fabric filters for a reliable liquid biopsy analysis. Nano Converg. 2019, 6, 39. [Google Scholar] [CrossRef]
- Park, J.; Lim, J.H.; Jin, H.J.; Namgung, S.; Lee, S.H.; Park, T.H.; Hong, S. A bioelectronic sensor based on canine olfactory nanovesicle-carbon nanotube hybrid structures for the fast assessment of food quality. Analyst 2012, 137, 3249–3254. [Google Scholar] [CrossRef]
- Ahn, S.R.; An, J.H.; Lee, S.H.; Song, H.S.; Jang, J.; Park, T.H. Peptide hormone sensors using human hormone receptor-carrying nanovesicles and graphene fets. Sci. Rep. 2020, 10, 388. [Google Scholar] [CrossRef]
- Sun, C.; Zhou, L.; Gou, M.; Shi, S.; Li, T.; Lang, J. Improved antitumor activity and reduced myocardial toxicity of doxorubicin encapsulated in mpeg-pcl nanoparticles. Oncol. Rep. 2016, 35, 3600–3606. [Google Scholar] [CrossRef] [Green Version]
- Poellmann, M.J.; Nair, A.; Bu, J.; Kim, J.K.H.; Kimple, R.J.; Hong, S. Immunoavidity-based capture of tumor exosomes using poly(amidoamine) dendrimer surfaces. Nano Lett. 2020, 20, 5686–5692. [Google Scholar] [CrossRef] [PubMed]
- Bu, J.; Nair, A.; Iida, M.; Jeong, W.J.; Poellmann, M.J.; Mudd, K.; Kubiatowicz, L.J.; Liu, E.W.; Wheeler, D.L.; Hong, S. An avidity-based pd-l1 antagonist using nanoparticle-antibody conjugates for enhanced immunotherapy. Nano Lett. 2020, 20, 4901–4909. [Google Scholar] [CrossRef] [PubMed]
- Bu, J.; Lee, T.H.; Kim, I.S.; Cho, Y.-H. Microfluidic-based mechanical phenotyping of cells for the validation of epithelial-to-mesenchymal-like transition caused by insufficient heat treatment. Sens. Actuators B Chem. 2017, 244, 591–598. [Google Scholar] [CrossRef]
- Bousquet, P.F.; Paulsen, L.A.; Fondy, C.; Lipski, K.M.; Loucy, K.J.; Fondy, T.P. Effects of cytochalasin b in culture and in vivo on murine madison 109 lung carcinoma and on b16 melanoma. Cancer Res. 1990, 50, 1431–1439. [Google Scholar] [PubMed]
- Hwang, J.; Yi, M.; Zhang, X.; Xu, Y.; Jung, J.H.; Kim, D.K. Cytochalasin b induces apoptosis through the mitochondrial apoptotic pathway in hela human cervical carcinoma cells. Oncol. Rep. 2013, 30, 1929–1935. [Google Scholar] [CrossRef]
- Gomzikova, M.; Kletukhina, S.; Kurbangaleeva, S.; Rizvanov, A. Evaluation of cytochalasin b-induced membrane vesicles fusion specificity with target cells. Biomed. Res. Int. 2018, 2018, 7053623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomzikova, M.O.; Zhuravleva, M.N.; Vorobev, V.V.; Salafutdinov, I.I.; Laikov, A.V.; Kletukhina, S.K.; Martynova, E.V.; Tazetdinova, L.G.; Ntekim, A.I.; Khaiboullina, S.F.; et al. Angiogenic activity of cytochalasin b-induced membrane vesicles of human mesenchymal stem cells. Cells 2019, 9, 95. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doyle, L.M.; Wang, M.Z. Overview of extracellular vesicles, their origin, composition, purpose, and methods for exosome isolation and analysis. Cells 2019, 8, 727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pan, X.; Zhao, L.; Quan, J.; Liu, K.; Lai, Y.; Li, Z.; Zhang, Z.; Xu, J.; Xu, W.; Guan, X.; et al. Mir-378a-5p acts as a tumor suppressor in renal cell carcinoma and is associated with the good prognosis of patients. Am. J. Transl. Res. 2019, 11, 2207–2218. [Google Scholar]
- Macknight, A.D. Principles of cell volume regulation. Ren. Physiol. Biochem. 1988, 11, 114–141. [Google Scholar] [CrossRef] [PubMed]
- Calloe, K.; Elmedyb, P.; Olesen, S.P.; Jorgensen, N.K.; Grunnet, M. Hypoosmotic cell swelling as a novel mechanism for modulation of cloned hcn2 channels. Biophys. J. 2005, 89, 2159–2169. [Google Scholar] [CrossRef] [Green Version]
- Hoffmann, E.K.; Lambert, I.H.; Pedersen, S.F. Physiology of cell volume regulation in vertebrates. Physiol. Rev. 2009, 89, 193–277. [Google Scholar] [CrossRef] [PubMed]
- Bu, J.; Cho, Y.-H.; Han, S.-W. Enhancement of isolation sensitivity for the viable heterogeneous circulating tumor cells swelled by hypo-osmotic pressure. RSC Adv. 2017, 7, 49684–49693. [Google Scholar] [CrossRef] [Green Version]
- Fu, W.J.; Kuwahara, M.; Marumo, F. Mechanisms of regulatory volume decrease in collecting duct cells. Jpn. J. Physiol. 1995, 45, 97–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Z.; Irianto, J.; Kazun, S.; Wang, W.; Knight, M.M. The rate of hypo-osmotic challenge influences regulatory volume decrease (rvd) and mechanical properties of articular chondrocytes. Osteoarthr. Cartil. 2015, 23, 289–299. [Google Scholar] [CrossRef] [Green Version]
- Vázquez, E.; Nobles, M.; Valverde, M.A. Defective regulatory volume decrease in human cystic fibrosis tracheal cells because of altered regulation of intermediate conductance ca2+-dependent potassium channels. Proc. Natl. Acad. Sci. USA 2001, 98, 5329–5334. [Google Scholar] [CrossRef] [Green Version]
- Kitadokoro, K.; Bordo, D.; Galli, G.; Petracca, R.; Falugi, F.; Abrignani, S.; Grandi, G.; Bolognesi, M. Cd81 extracellular domain 3d structure: Insight into the tetraspanin superfamily structural motifs. EMBO J. 2001, 20, 12–18. [Google Scholar] [CrossRef] [Green Version]
- Kleemann, R.; Grell, M.; Mischke, R.; Zimmermann, G.; Bernhagen, J. Receptor binding and cellular uptake studies of macrophage migration inhibitory factor (mif): Use of biologically active labeled mif derivatives. J. Interferon Cytokine Res. 2002, 22, 351–363. [Google Scholar] [CrossRef] [PubMed]
- Bu, J.; Nair, A.; Kubiatowicz, L.J.; Poellmann, M.J.; Jeong, W.-J.; Reyes-Martinez, M.; Armstrong, A.J.; George, D.J.; Wang, A.Z.; Zhang, T.; et al. Surface engineering for efficient capture of circulating tumor cells in renal cell carcinoma: From nanoscale analysis to clinical application. Biosens. Bioelectron. 2020, 162, 112250. [Google Scholar] [CrossRef] [PubMed]
- Rawding, P.A.; Bu, J.; Wang, J.; Kim, D.W.; Drelich, A.J.; Kim, Y.; Hong, S. Dendrimers for cancer immunotherapy: Avidity-based drug delivery vehicles for effective anti-tumor immune response. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2021, e1752. [Google Scholar] [CrossRef]
- Nair, A.; Bu, J.; Bugno, J.; Rawding, P.A.; Kubiatowicz, L.J.; Jeong, W.J.; Hong, S. Size-dependent drug loading, gene complexation, cell uptake, and transfection of a novel dendron-lipid nanoparticle for drug/gene co-delivery. Biomacromolecules 2021, 22, 3746–3755. [Google Scholar] [CrossRef]
- Jeong, W.-J.; Bu, J.; Kubiatowicz, L.J.; Chen, S.S.; Kim, Y.; Hong, S. Peptide-nanoparticle conjugates: A next generation of diagnostic and therapeutic platforms? Nano Converg. 2018, 5, 38. [Google Scholar] [CrossRef]
- Jeong, W.J.; Bu, J.; Jafari, R.; Rehak, P.; Kubiatowicz, L.J.; Drelich, A.J.; Owen, R.H.; Nair, A.; Rawding, P.A.; Poellmann, M.J.; et al. Hierarchically multivalent peptide-nanoparticle architectures: A systematic approach to engineer surface adhesion. Adv. Sci. (Weinh) 2021, e2103098. [Google Scholar] [CrossRef] [PubMed]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nair, A.; Bu, J.; Rawding, P.A.; Do, S.C.; Li, H.; Hong, S. Cytochalasin B Treatment and Osmotic Pressure Enhance the Production of Extracellular Vesicles (EVs) with Improved Drug Loading Capacity. Nanomaterials 2022, 12, 3. https://doi.org/10.3390/nano12010003
Nair A, Bu J, Rawding PA, Do SC, Li H, Hong S. Cytochalasin B Treatment and Osmotic Pressure Enhance the Production of Extracellular Vesicles (EVs) with Improved Drug Loading Capacity. Nanomaterials. 2022; 12(1):3. https://doi.org/10.3390/nano12010003
Chicago/Turabian StyleNair, Ashita, Jiyoon Bu, Piper A. Rawding, Steven C. Do, Hangpeng Li, and Seungpyo Hong. 2022. "Cytochalasin B Treatment and Osmotic Pressure Enhance the Production of Extracellular Vesicles (EVs) with Improved Drug Loading Capacity" Nanomaterials 12, no. 1: 3. https://doi.org/10.3390/nano12010003