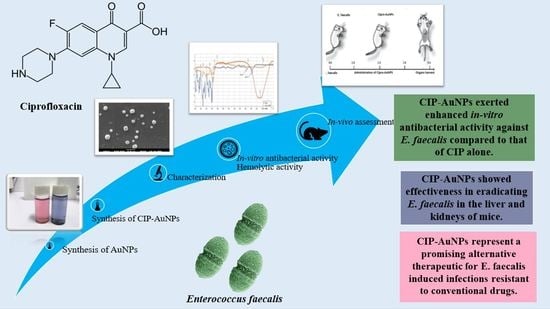
Ciprofloxacin-Loaded Gold Nanoparticles against Antimicrobial Resistance: An In Vivo Assessment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains
2.2. Preparation of AuNPs and CIP-AuNPs
2.3. Characterization of AuNPs and CIP-AuNPs
2.4. Drug Loading Capacity and Encapsulation Efficiency
2.5. Drug Release Efficiency
2.6. Kinetic Analysis of the Drug Release
2.7. In Vitro Stability of CIP-AuNPs
2.8. In Vitro Antibacterial Potential of CIP-AuNPs
2.9. Hemolytic Activity of CIP-AuNPs
2.10. Colonization of E. faecalis in BALB/c Mice
2.11. In Vivo Antibacterial Activity of CIP-AuNPs
2.12. Statistical Analysis
3. Results
3.1. Synthesis of AuNPS and CIP-AuNPs
3.2. CIP Encapsulation Efficiency and CIP Loading Capacity
3.3. Particle Size and Zeta Potential of AuNPs and CIP-AuNPs
3.4. Surface Morphology and Elemental Chemical Composition of AuNPs by SEM–EDS
3.5. Structural Analysis of CIP-AuNPs by FTIR Spectroscopy
3.6. Kinetics of CIP Drug Release from AuNPs
3.7. Stability Tests on CIP-AuNPs
3.8. In Vitro Antibacterial Activity of CIP-AuNPs at the Optimized Dose
3.9. In Vivo Anticolonizing Potential of CIP-AuNPs in an Animal Model
3.10. Hemolytic Activity of CIP-AuNPs
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, D.; Fan, W.; Kishen, A.; Gutmann, J.L.; Fan, B. Evaluation of the Antibacterial Efficacy of Silver Nanoparticles against Enterococcus faecalis Biofilm. J. Endod. 2014, 40, 285–290. [Google Scholar] [CrossRef]
- Sedgley, C.; Buck, G.; Appelbe, O. Prevalence of Enterococcus faecalis at Multiple Oral Sites in Endodontic Patients Using Culture and PCR. J. Endod. 2006, 32, 104–109. [Google Scholar] [CrossRef]
- Saleh, I.M.; Ruyter, I.E.; Haapasalo, M.; Ørstavik, D. Survival of Enterococcus faecalis in infected dentinal tu-bules after root canal filling with different root canal sealers in vitro. Int. Endod. J. 2004, 37, 193–198. [Google Scholar] [CrossRef]
- Liu, H.; Wei, X.; Ling, J.; Wang, W.; Huang, X. Biofilm Formation Capability of Enterococcus faecalis Cells in Starvation Phase and Its Susceptibility to Sodium Hypochlorite. J. Endod. 2010, 36, 630–635. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, M.S.; Clewell, D.B.; Ike, Y.; Shankar, N. Enterococci: From Commensals to Leading Causes of Drug Resistant Infection; Eye and Ear Infirmary: Boston, MA, USA, 2014. [Google Scholar]
- Livermore, D.M.; Blaser, M.; Carrs, O.; Cassell, G.; Fishman, N.; Guidos, R.; Levy, S.; Powers, J.; Norrby, R.; Tillotson, G.; et al. Discovery research: The scientific challenge of finding new antibiotics. J. Antimicrob. Chemother. 2011, 66, 1941–1944. [Google Scholar] [CrossRef] [PubMed]
- Pinto, R.; Lopes, D.; Martins, M.C.L.; Van Dijck, P.; Nunes, C.; Reis, S. Impact of nanosystems in Staphylococcus aureus biofilms treatment. FEMS Microbiol. Rev. 2019, 43, 622–641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vassallo, A.; Silletti, M.F.; Faraone, I.; Milella, L. Nanoparticulate Antibiotic Systems as Antibacterial Agents and Antibiotic Delivery Platforms to Fight Infections. J. Nanomater. 2020, 2020. [Google Scholar] [CrossRef]
- Pugazhendhi, A.; Kumar, S.S.; Manikandan, M.; Saravanan, M. Photocatalytic properties and antimicrobial ef-ficacy of Fe doped CuO nanoparticles against the pathogenic bacteria and fungi. Microb. Pathog. 2018, 122, 84–89. [Google Scholar] [CrossRef]
- Uzair, B.; Liaqat, A.; Iqbal, H.; Menaa, B.; Razzaq, A.; Thiripuranathar, G.; Rana, N.F.; Menaa, F. Green and Cost-Effective Synthesis of Metallic Nanoparticles by Algae: Safe Methods for Translational Medicine. Bioengineering 2020, 7, 129. [Google Scholar] [CrossRef]
- Wang, L.; Hu, C.; Shao, L. The antimicrobial activity of nanoparticles: Present situation and prospects for the future. Int. J. Nanomed. 2017, 12, 1227–1249. [Google Scholar] [CrossRef] [Green Version]
- Dykman, L.; Khlebtsov, N. Gold nanoparticles in biomedical applications: Recent advances and perspectives. Chem. Soc. Rev. 2012, 41, 2256–2282. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Raouf, N.; Al-Enazi, N.M.; Ibraheem, I.B. Green biosynthesis of gold nanoparticles using Galaxaura elongata and characterization of their antibacterial activity. Arab. J. Chem. 2017, 10, S3029–S3039. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Geng, X.; Liu, D.; Li, Z. Near-infrared light-enhanced protease-conjugated gold nanorods as a photo-thermal antimicrobial agent for elimination of exotoxin and biofilms. International journal of nanomedicine. Int. J. Nanomed. 2019, 14, 8047. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rhazouani, A.; Gamrani, H.; El Achaby, M.; Aziz, K.; Gebrati, L.; Uddin, S.; Aziz, F. Synthesis and Toxicity of Graphene Oxide Nanoparticles: A Literature Review of In vitro and In vivo Studies. BioMed Res. Int. 2021, 2021. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, S.N.; Scoffield, J.; Andukuri, A.; Alexander, G.C.; Walker, T.; Kim, S.; Choi, S.C.; Brott, B.C.; Eleazer, P.D.; Lee, J.-Y.; et al. Evaluation of ciprofloxacin and metronidazole encapsulated biomimetic nanomatrix gel on Enterococcus faecalis and Treponema denticola. Biomater. Res. 2015, 19, 9. [Google Scholar] [CrossRef] [Green Version]
- Azad, A.; Rostamifar, S.; Modaresi, F.; Bazrafkan, A.; Rezaie, Z. Assessment of the Antibacterial Effects of Bismuth Nanoparticles against Enterococcus faecalis. BioMed Res. Int. 2020, 2020. [Google Scholar] [CrossRef]
- Parolia, A.; Kumar, H.; Ramamurthy, S.; Davamani, F.; Pau, A. Effectiveness of chitosan-propolis nanoparticle against Enterococcus faecalis biofilms in the root canal. BMC Oral Heal. 2020, 20, 339. [Google Scholar] [CrossRef]
- Arias-Moliz, M.T.; Baca, P.; Solana, C.; Toledano, M.; Medina-Castillo, A.L.; Toledano-Osorio, M.; Osorio, R. Doxycycline-functionalized polymeric nanoparticles inhibit Enterococcus faecalis biofilm formation on dentine. Int. Endod. J. 2021, 54, 413–426. [Google Scholar] [CrossRef]
- Maliszewska, I.; Wróbel, J.; Wanarska, E.; Podhorodecki, A.; Matczyszyn, K. Synergistic effect of methylene blue and biogenic gold nanoparticles against Enterococcus faecalis. Photodiagnosis Photodyn. Ther. 2019, 27, 218–226. [Google Scholar] [CrossRef]
- Balto, H.; Bukhary, S.; Al-Omran, O.; BaHammam, A.; Al-Mutairi, B. Combined Effect of a Mixture of Silver Nanoparticles and Calcium Hydroxide against Enterococcus faecalis Biofilm. J. Endod. 2020, 46, 1689–1694. [Google Scholar] [CrossRef]
- Halkai, K.; Mudda, J.A.; Shivanna, V.; Rathod, V.; Halkai, R. Antibacterial efficacy of biosynthesized silver nanoparticles against Enterococcus faecalis Biofilm: An in vitro study. Contemp. Clin. Dent. 2018, 9, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Liu, Y.; Zhang, H.; Lei, L. Nano-graphene oxide with antisense walR RNA inhibits the pathogenicity of Enterococcus faecalis in periapical periodontitis. J. Dent. Sci. 2019, 15, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Martini, C.; Longo, F.; Castagnola, R.; Marigo, L.; Grande, N.M.; Cordaro, M.; Cacaci, M.; Papi, M.; Palmieri, V.; Bugli, F.; et al. Antimicrobial and Antibiofilm Properties of Graphene Oxide on Enterococcus faecalis. Antibiotics 2020, 9, 692. [Google Scholar] [CrossRef] [PubMed]
- Herizchi, R.; Abbasi, E.; Milani, M.; Akbarzadeh, A. Current methods for synthesis of gold nanoparticles. Artif. Cells Nanomed. Biotechnol. 2016, 44, 596–602. [Google Scholar] [CrossRef]
- Tom, R.T.; Suryanarayanan, V.; Reddy, P.G.; Baskaran, S.; Pradeep, T. Ciprofloxacin-protected gold nanopar-ticles. Langmuir 2004, 20, 1909–1914. [Google Scholar] [CrossRef]
- Topal, G.R.; Devrim, B.; Eryilmaz, M.; Bozkir, A. Design of ciprofloxacin-loaded nano-and micro-composite particles for dry powder inhaler formulations: Preparation, in vitro characterisation, and antimicro-bial efficacy. J. Microencapsul. 2018, 35, 533–547. [Google Scholar] [CrossRef]
- Arafa, M.G.; Mousa, H.A.; Afifi, N.N. Preparation of PLGA-chitosan based nanocarriers for enhancing anti-bacterial effect of ciprofloxacin in root canal infection. Drug Deliv. 2020, 27, 26–39. [Google Scholar] [CrossRef] [Green Version]
- Albuquerque, M.T.; Valera, M.C.; Moreira, C.S.; Bresciani, E.; de Melo, R.M.; Bottino, M.C. Effects of ciprof-loxacin-containing scaffolds on Enterococcus faecalis biofilms. J. Endod. 2015, 41, 710–714. [Google Scholar] [CrossRef]
- Assali, M.; Zaid, A.N.; Abdallah, F.; Almasri, M.; Khayyat, R. Single-walled carbon nanotubes-ciprofloxacin nanoantibiotic: Strategy to improve ciprofloxacin antibacterial activity. Int. J. Nanomed. 2017, 12, 6647–6659. [Google Scholar] [CrossRef] [Green Version]
- Rana, N.F.; Sauvageot, N.; Laplace, J.-M.; Bao, Y.; Nes, I.; Rincé, A.; Posteraro, B.; Sanguinetti, M.; Hartke, A. Redox Balance via Lactate Dehydrogenase Is Important for Multiple Stress Resistance and Virulence in Enterococcus faecalis. Infect. Immun. 2013, 81, 2662–2668. [Google Scholar] [CrossRef] [Green Version]
- Dash, S.; Murthy, P.N.; Nath, L.; Chowdhury, P. Kinetic modeling on drug release from controlled drug delivery systems. Acta Pol. Pharm. Drug Res. 2010, 67, 217–223. [Google Scholar]
- Bettini, R.; Catellani, P.L.; Santi, P.; Massimo, G.; Peppas, N.A.; Colombo, P. Translocation of drug particles in HPMC matrix gel layer: Effect of drug solubility and influence on release rate. J. Control. Release 2001, 70, 383–391. [Google Scholar] [CrossRef]
- Naz, S.S.; Islam, N.U.; Shah, M.R.; Alam, S.S.; Iqbal, Z.; Bertino, M.; Franzel, L.; Ahmed, A. Enhanced biocidal activity of Au nanoparticles synthesized in one pot using 2, 4-dihydroxybenzene carbodithioic acid as a reducing and stabilizing agent. J. Nanobiotechnol. 2013, 11, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muhammad, Z.; Raza, A.; Ghafoor, S.; Naeem, A.; Naz, S.S.; Riaz, S.; Ahmed, W.; Rana, N.F. PEG capped methotrexate silver nanoparticles for efficient anticancer activity and biocompatibility. Eur. J. Pharm. Sci. 2016, 91, 251–255. [Google Scholar] [CrossRef]
- Riaz, S.; Rana, N.F.; Hussain, I.; Tanweer, T.; Nawaz, A.; Menaa, F.; Janjua, H.A.; Alam, T.; Batool, A.; Naeem, A.; et al. Effect of Flavonoid-Coated Gold Nanoparticles on Bacterial Colonization in Mice Organs. Nanomaterials 2020, 10, 1769. [Google Scholar] [CrossRef]
- Bhalerao, S.R.; Rote, A.R. Application of UV spectrophotometric methods for estimation of Ciprofloxacin and tinidazole in combined tablet dosage form. Int. J. Pharmacol. Pharm. Sci. 2012, 4, 4–7. [Google Scholar]
- Bhattacharjee, S. DLS and zeta potential—What they are and what they are not? J Control. Release 2016, 235, 337–351. [Google Scholar] [CrossRef]
- Canepa, E.; Salassi, S.; Simonelli, F.; Ferrando, R.; Rolandi, R.; Lambruschini, C.; Canepa, F.; Dante, S.; Relini, A.; Rossi, G. Non-disruptive uptake of anionic and cationic gold nanoparticles in neutral zwitterionic membranes. Sci. Rep. 2021, 11, 1–11. [Google Scholar] [CrossRef]
- Okoampah, E.; Mao, Y.; Yang, S.; Sun, S.; Zhou, C. Gold nanoparticles-biomembrane interactions: From fundamental to simulation. Colloids Surf. B Biointerfaces 2020, 196, 111312. [Google Scholar] [CrossRef]
- Dreaden, E.C.; Austin, L.; Mackey, M.A.; El-Sayed, M.A. Size matters: Gold nanoparticles in targeted cancer drug delivery. Ther. Deliv. 2012, 3, 457–478. [Google Scholar] [CrossRef] [Green Version]
- Lim, S.; Alam, M.G. Ciprofloxacin-Induced Acute Interstitial Nephritis and Autoimmune Hemolytic Anemia. Ren. Fail. 2003, 25, 647–651. [Google Scholar] [CrossRef] [PubMed]








| CIP Concentration AuNPs | Encapsulation Efficiency (%) | Loading Capacity (%) |
|---|---|---|
| 0.5 mM | 24.43 | 8.85 |
| 1.0 mM | 29.30 | 15.60 |
| 1.5 mM | 30.65 | 28.85 |
| 2.0 mM | 48.92 | 33.81 |
| 2.5 mM | 60.83 | 34.54 |
| CIP-AuNPs | Z-Average (d. nm) | PDI | St Dev (d. nm) | Zeta Potential (mV) |
|---|---|---|---|---|
| 0.5 mM | 24.43 | 0.26 | 6.21 | −32.1 |
| 1.0 mM | 24.09 | 0.301 | 6.044 | −33.3 |
| 1.5 mM | 41 | 0.68 | 10.21 | −19.7 |
| 2.0 mM | 88.2 | 1.000 | 57.4 | −13.4 |
| 2.5 mM | 128.2 | 0.48 | 79.18 | −2.12 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nawaz, A.; Ali, S.M.; Rana, N.F.; Tanweer, T.; Batool, A.; Webster, T.J.; Menaa, F.; Riaz, S.; Rehman, Z.; Batool, F.; et al. Ciprofloxacin-Loaded Gold Nanoparticles against Antimicrobial Resistance: An In Vivo Assessment. Nanomaterials 2021, 11, 3152. https://doi.org/10.3390/nano11113152
Nawaz A, Ali SM, Rana NF, Tanweer T, Batool A, Webster TJ, Menaa F, Riaz S, Rehman Z, Batool F, et al. Ciprofloxacin-Loaded Gold Nanoparticles against Antimicrobial Resistance: An In Vivo Assessment. Nanomaterials. 2021; 11(11):3152. https://doi.org/10.3390/nano11113152
Chicago/Turabian StyleNawaz, Afrah, Syed Mohsin Ali, Nosheen Fatima Rana, Tahreem Tanweer, Amna Batool, Thomas J. Webster, Farid Menaa, Sundus Riaz, Zahra Rehman, Farhat Batool, and et al. 2021. "Ciprofloxacin-Loaded Gold Nanoparticles against Antimicrobial Resistance: An In Vivo Assessment" Nanomaterials 11, no. 11: 3152. https://doi.org/10.3390/nano11113152