1. Introduction
Fibromyalgia syndrome (FMS) is a chronic disorder characterized by widespread and persistent musculoskeletal pain, accompanied by symptoms including fatigue, insomnia, depression, anxiety, and cognitive impairments [
1]. While the etiology of FMS remains unknown, it is widely acknowledged that central pain sensitization and impairments in endogenous pain inhibitory mechanisms play a crucial role in its pathogenesis [
2,
3]. This is expressed in hyperalgesia and allodynia, which together characterize FMS; moreover, patients exhibit reduced thresholds and tolerance to evoked pain, increased responses during protocols measuring pain sensitization, and exaggerated activity in the neuromatrix of pain during painful stimulation [
4,
5,
6,
7,
8,
9].
An important problem in FMS research and its clinical management is the lack of objective markers of the disease and of reliable clinical measures for its diagnosis [
10,
11]. The subjective nature of FMS symptoms, along with the lack of objective indicators, hinders disease comprehension, effective healthcare, and medical and social acceptance. Based on previous research [
12,
13], in 1990 the American College of Rheumatology (ACR) established preliminary criteria for FMS [
1]. However, these criteria have been repeatedly criticized due to the difficulty of using pressure algometry in primary healthcare, the low predictive validity of evoked pain with respect to clinical pain, and the lack of consideration of accompanying symptoms among other aspects [
14,
15].
In 2010, the ACR proposed new criteria, based on the Widespread Pain Index (WPI) and Symptom Severity (SS) scale [
16]. The WPI includes a list of 19 painful body areas. Whereas the SS includes two parts: Part 2a calculates severity scores for fatigue, waking unrefreshed and cognitive symptoms. Part 2b derives a general symptom severity score from a list of 41 symptoms. For FMS diagnosis, one of two conditions must be fulfilled: WPI ≥ 7 and SS ≥ 5, or WPI between 3 and 6 and SS ≥ 9. As in the 1990 criteria, symptom duration of at least the three previous months was required. In 2011 and 2016, these diagnostic criteria were revised again [
17,
18].
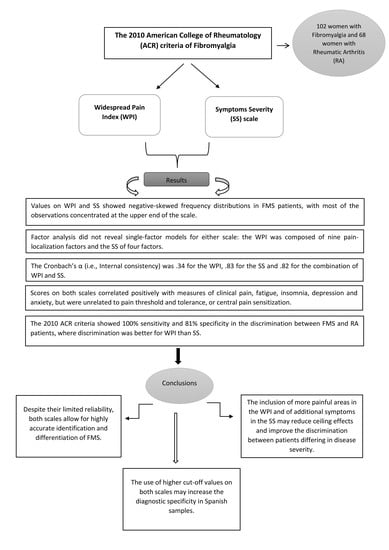
The present study evaluated the reliability and predictive validity of the WPI and SS in a sample of FMS patients recruited with the 1990 ACR criteria. Its objectives were as follows: (1) estimation of the frequency distribution of WPI and SS scores in FMS patients and a sample of patients with rheumatoid arthritis (RA), as a chronic pain syndrome of peripheral origin. (2) Evaluation of the factor structure of WPI and SS. To the best of our knowledge, no previous studies have analyzed the underlying factorial structure of these scales. Based on their aims and composition [
16], we hypothesized that both scales are unidimensional; accordingly, WPI items should load mainly on a painful body-part factor and SS items basically on a pain-severity factor. (3) Assessment of the internal consistency of the WPI and SS. Studies analyzing the internal consistency of both scales together reported Cronbach’s α values of 0.75 in Japanese [
19] and 0.87 in French [
20] samples. A Turkish study reported Cronbach´s α values of 0.78 for the WPI, 0.53 for SS and 0.77 for the two scales together [
21]. (4) Evaluation of the predictive validity of the WPI and SS with respect to the core FMS symptoms; two studies demonstrated inverse associations of WPI and SS with measures of quality of life [
22,
23]. A previous Spanish study reported positive correlations of WPI and SS with fatigue and depression [
23]. In the present study, relationships between WPI and SS scores with self-reported clinical pain, fatigue, insomnia, anxiety, depression, and pain catastrophizing were quantified. (5) Estimation of associations between WPI and SS scores, evoked pain measures (pain threshold and tolerance) and a marker of central pain sensitization. The most well-established hypothesis regarding FMS pathophysiology pertains to central nervous sensitization to pain [
2,
4]. However, to date, no study has analyzed the association of WPI and SS scores with parameters reflecting this phenomenon. (6) Evaluation of the diagnostic accuracy of the 2010 ACR criteria and WPI and SS scores to differentiate between FMS and RA patients. Objectives 2–5 were analyzed only in the FMS sample.
4. Discussion
The present study evaluated the reliability, factor structure and predictive validity of the WPI and SS scales of the 2010 ACR criteria for FMS. The mean WPI (17.84) and SS (10.05) scores of our FMS patients were higher than those previously reported. In the 2010 Wolfe et al. study [
16], mean scores of 11.4 for WPI and 8 for SS were observed [
16]. In a Spanish FMS sample, reported mean scores were 13 for WPI and 8 for SS. Moreover, in Spanish patients, median scores of 15 for WPI and 8 for SS have been reported [
26]. A possible explanation for this divergence is that our FMS sample was recruited using the 1990 ACR criteria. This supports the notion that the 1990 criteria are stricter than those of 2010 [
16], such that only more severely affected patients are identified.
WPI and SS did not follow a normal distribution. In our FMS patients, the scores were concentrated towards the upper end of the scales (53.9% of them indicated all 19 of the 19 possible painful areas on the WPI, and 64.7% occupied the upper category of 25 or more symptoms on the SS2b); the scores of RA patients were concentrated towards the lower end of the scales (60.3% of them indicated between 0 and 4 painful areas on the WPI, and 81.6% reported between 1 and 10 symptoms in the SS2b). Thus, a strong ceiling effect was observed for the WPI and SS scores in our FMS patients. In accordance with their aim, the scales correctly differentiated between FMS patients and those with AR. The FMS patients indicated more painful areas, increased fatigue, a stronger tendency to wake unrefreshed, more severe cognitive symptoms, and a higher total number of symptoms than those with RA [
40].
One of the reasons for developing the 2010 ACR criteria was to devise an instrument differentiating between individual patients or patient groups according to symptom severity [
16]. However, the observed ceiling effects impede such differentiation between FMS patients and restrict the utility of the scales for clinical evaluations. Furthermore, the deviation from a normal distribution may complicate the statistical analyses. Therefore, it would be useful to include more elements in both scales (painful areas and symptoms) or to introduce dimensional (Likert scale), instead of categorical, response options to improve the discrimination according to clinical severity of FMS patients.
Contrary to our prediction, neither the WPI nor the SS exhibited a single-factor structure. The WPI items were spread across nine factors covering all painful areas represented in the scale. Most of the factors include two body areas except for Factors 1, 2, and 5, which include three body areas, and Factors 8 and 9, which only include one body area. While it is difficult to explain the loads of different body areas on the same factor, the finding challenges the validity of aggregating pain in different body areas to a single symptom scale. The factor structure of the SS was more intuitive; it involved four factors related to (1) the main clinical FMS symptoms (e.g., muscle pain, fatigue, and cognitive problems), (2) emotional-nervousness symptoms (e.g., depression, anxiety, nausea, nervousness, chest pain, blurred vision, and dry mouth), (3) somatic symptoms (e.g., Raynaud’s symptoms, ringing in ears, heartburn, loss/change in taste, hearing difficulties, and loss of appetite), and (4) symptoms associated with the skin and sensory sensitivity (e.g., itching, wheezing, hives/welts, dry eyes, and sun sensitivity).
The internal consistency of the WPI was poor, which accords with the high number of factors revealed by the EFA and reflects the low co-variation of the item on the instrument (where feeling pain in one body area does not necessarily relate to feeling pain in another body area). In addition, the internal consistency of the scale may be limited by the binary response mode and the small number of items [
41]. The SS and the combination of both scales showed satisfactory internal consistency. Most previous studies assessing the internal consistency of the two scales together revealed similar values. While in the present study the Cronbach´s α of the two scales in combination was 0.82, it was 0.87 in the French validation study [
20], 0.77 in the Turkish one [
21], and 0.93 in a previous Spanish study using the revised 2010 criteria with patient´s self-administration procedure [
23]. The WPI and SS scores exhibited a moderate correlation in FMS patients but not in RA patients, suggesting a positive association between the extent of the painful body areas and pain severity.
The WPI and SS demonstrated acceptable predictive validity, as indexed by their associations with clinical symptoms. Despite the skewness of their distributions, making it more difficult to obtain significant correlations, the WPI and the Total Symptoms SS scores correlated positively with the Total Clinical Pain, Current Pain Intensity, Emotional Pain, and Sensorial Pain subscale scores of the MPQ. Similarly, correlations of WPI and SS scores with pain intensity measured by a VAS were demonstrated in previous research [
21]. An important symptom of FMS is fatigue [
16,
42]. Accordingly, SS Fatigue, SS Cognitive Symptoms, the Subtotal SS2a, and SS Total Symptoms scores correlated positively with Fatigue. The SS and WPI scores were also related to emotional variables. In particular, the SS Total Symptoms and Range of Symptoms scores correlated positively with Depression and Catastrophizing. The Total Symptoms and Range of Symptoms scores also correlated positively with Trait Anxiety. This is in line with previous findings of associations between negative emotions and clinical symptoms [
43,
44]. Several studies have shown that depression and anxiety predispose FMS patients to lifestyles that negatively influence clinical course and psychosocial resources [
45,
46]. Furthermore, negative emotions are believed play a role in the exacerbation and maintenance of FMS symptoms [
43,
44]. In a previous study of Spanish patients [
23], WPI and SS scores were also significantly associated with depression and fatigue, with the magnitude of the reported associations being greater than that obtained in our study. One potential explanation for the stronger associations found in that previous study is the use of revised 2011 ACR criteria, with the patients´ self-administered method and the somewhat different SS scale (modified for self-application). We believe that self-administration of the scales might enhance the influence of emotional state on symptom and severity reporting [
44]. Subjective cognitive symptoms, as assessed by the SS2a correlated positively with Current Pain Intensity, Emotional Pain and Fatigue in this study. This accords with neuropsychological studies indicating that cognitive deficits in FMS vary according to clinical pain severity and fatigue [
47,
48].
Analysis of associations of WPI and SS scores with indicators of central nervous sensitization to pain is a relevant question, as this is considered the core pathological mechanism of FMS [
2,
3]. For this purpose, the SREP protocol was used, which was previously demonstrated to be suitable for assessing central pain sensitization in FMS [
5,
6]. Total Symptoms, Range of Symptoms, and Total Symptom Severity (SS) negatively correlated with SREP. These relations are contrary to our predictions and may be explained by the mediating effects of depression and fatigue, which were associated both with SS and SREP scores. When fatigue and depression were statistically controlled no correlations arose between SS and SREP. The 1990 ACR criteria were criticized due to a lack of associations with measures of evoked pain [
49]. In our study, only a significant inverse association with pain threshold was observed for the symptom of waking unrefreshed. This is coherent with previous findings in which FMS patients with poor sleep quality reported greater clinical pain, as well as with the positive association between sleep quality and pain threshold [
50]. Nonetheless, it is evident that, just as with the 1990 ACR criteria, WPI and SS scores do not reliably predict responses to evoked pain.
Patients diagnosed with FMS, compared to RA patients, exhibited higher levels of clinical pain, fatigue, anxiety, depression, insomnia and catastrophizing, a lower pain threshold, lower tolerance of pain, and stronger central pain sensitization, which is coherent with current evidence [
40]. Regarding the diagnostic accuracy of the 2010 ACR criteria for differentiating between the two patients’ groups, all FMS patients diagnosed with the 1990 ACR criteria also fulfilled the 2010 ACR criteria; there was perfect agreement between both criteria, as previously reported [
18]. However, 19.12% of our RA sample fulfilled the 2010 ACR criteria (false positives). In our sample, the 2010 ACR criteria showed 100% sensitivity, as all FMS patients were detected, in addition to good specificity and overall discrimination accuracy. These results contrast with those reported in the previous Spanish study by Segura-Jiménez et al. [
23], in which a sensitivity of 88.3% and specificity of 91.8% were obtained in the discrimination between FMS patients and healthy controls. The greater specificity than sensitivity in that study and the greater accuracy in our study in the discrimination between FMS and RA patients, as opposed to the healthy individuals in that study, is difficult to explain. One reason may be the self-administration method used in that study in comparison to the face-to-face interview conducted in the present study. In a Spanish study using the 2010 ACR criteria to differentiate FMS patients from those with RA and osteoarthritis (OA), a sensitivity of 85.6% and specificity of 73.2% were noted [
26]. Finally, in the original study of Wolfe at al. [
16], an 88.1% of cases were classified correctly. As such, the diagnostic accuracy observed in the present study somewhat exceeded that of previous studies.
The results of our logistic regression analysis showed that WPI and SS also achieved good group discrimination when used separately. However, the WPI was a better predictor than the SS, reflecting the central role of pain in FMS. This is in accordance with previous research [
22], which suggested that the WPI is the better single indicator of disease severity and quality of life in FMS. When the two scales were both included in the logistic regression, a diagnostic accuracy of 100% was obtained. The fact that using the 2010 ACR criteria achieved a specificity of 81%, while using the continuous WPI and SS scores led to perfect group discrimination, underlines the potential to improve the diagnostic criteria. Therefore, we tested the effect of using higher cut-off values in order to increase specificity. When cut-off values were set at WPI ≥14 and SS ≥7, both specificity and overall diagnostic accuracy reached 100%. Whereas previous studies proposed different cut-off values [
51], cross-cultural differences in the expression and rating of symptoms may explain the different results in terms of sensitivity and specificity among different populations [
25,
52]. Taken together, the frequency distribution of the WPI and SS scores, the mean WPI and SS scores obtained in our sample, and the better specificity observed when using higher cut-off values as criteria suggest that the cut-off values of both scores should be increased, at least for Spanish populations.
Arnold et al. [
53] have recently proposed an alternative strategy for the FMS diagnosis in order to overcome the ACR diagnostic criteria limitations. This alternative is part of the ACTTION-APS Pain Taxonomy (AAPT), developed by The Analgesic, Anesthetic, and Addiction Clinical Trial Translations Innovations Opportunities and Networks (ACTTION) public-private partnership with the U.S. Food and Drug Administration (FDA) and the American Pain Society (APS). The ACTTION-APS Pain Taxonomy is a classification system designed for chronic pain [
54], assuming evidence-based diagnostic criteria in a multidimensional framework [
55]. In the case of FMS, the AAPT diagnostic proposal comprised five dimensions: (1) Core Diagnostic Criteria (the presence of pain in 6 or more body sites from a total of 9 possible localizations, sleep disturbance, and fatigue), (2) Common Features (e.g., tenderness, cognitive impairments), (3) Common Medical and Psychiatric Comorbidities, (4) Neurobiological, Psychosocial, and Functional Consequences, and (5) Putative Neurobiological and Psychosocial Mechanisms, Risk Factors, and Protective Factors. A study comparing the 2011 and 2016 ACR criteria with the AAPT criteria found considerable agreement between both sets of criteria, although a lower percentage of correct classifications was observed for AAPT criteria than for ACR criteria [
56]. Furthermore, a higher prevalence of FMS in the German general population was obtained with AAPT criteria than with ACR 2016 criteria [
57]. Thus, AAPT criteria seem to diagnose patients with less symptom severity and fewer pain sites [
57]. Further research is required to assess the reliability and validity of the AAPT criteria [
58].
As limitation of the study, although we have specially care in excluding patients with RA who showed FMS features, it is difficult to completely discard the possibility of the inclusion in the study sample of some cases with dual RA-FMS pathology [
59,
60]. Besides, from the comparison of levels of clinical symptoms of our FMS patients with values of previous studies (see above), it is possible that our sample was biased toward more severely affected patients and higher scores on the WPI, SS, and other clinical scales [
59,
60].