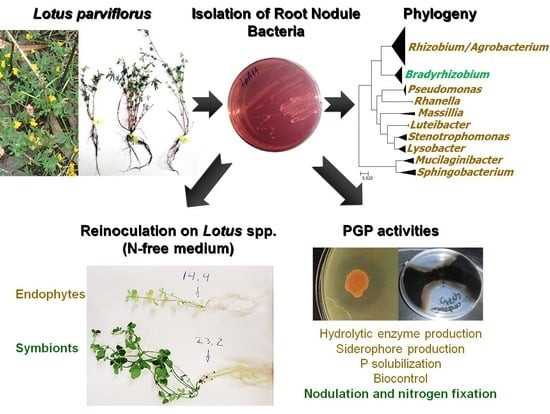
Diversity, Phylogeny and Plant Growth Promotion Traits of Nodule Associated Bacteria Isolated from Lotus parviflorus
Abstract
:1. Introduction
2. Materials and Methods
2.1. Isolation of Bacterial Strains and Growth Conditions
2.2. Repetitive Extragenic Palindromic (REP)-PCR Amplification and Fingerprint Analysis
2.3. PCR Gene Amplification and Sequencing
2.4. NCBI Accession Numbers
- 16S rRNALpA5a: MK611706; LpA5b: MK611741; LpA6: MK611742; LpA7b: MK611744; LpA8: MK611745; LpA9: MK611746; LpA10: MK611747; LpA11: MK611748; LpA12: MK611749; LpA13a: MK611750; LpB5b: MT071928; LpB5d: MT071934; LpB9a: MT071930; LpB10a: MT071937; LpB10b: MT071935; LpB10c: MT071938; LpB10d: MT071936; LpB12a: MT071929; LpB12b: MT071924; LpB13: MT071932; LpB14a: MT071927; LpB14b: MT071931; LpB15b: MT071926; LpB16c: MT071939; LpB16d: MT071925; LpB17b: MT071933; LpB19a: MK611753; LpB23b: MK611754; LpB26: MK611755.
- RecALpA5a: MK671539; LpA5b: MK690337; LpA6: MK671540; LpA7b: MK671541; LpA8: MK690338; LpA9: MK671542; LpA11: MK671543; LpA13a: MK690339; LpB19a: MK671547; LpB23b: MK671548; LpB26: MK671549.
- AtpDLpA5a: MK671550; LpA5b: MK690340; LpA6: MK671551; LpA7b: MK671553; LpA8: MK690341; LpA9: MK671554; LpA10: MK690342; LpA11: MK671555; LpA13a: MK671557; LpB19a: MK671560; LpB23b: MK671561; LpB26: MK671562.
- GlnIILpA8: MK690343; LpA9: MK690344; LpA11: MK671563; LpA13b: MK690346; LpA14: MK690347; LpB19a: MK671564; LpB23b: MK671565; LpB26: MK671566.
- NodCLpA11: MK671567; LpB19a: MK671568; LpB23b: MK671569; LpB26: MK671570.
- NifHLpA11: MK690348; LpB19a: MK671571; LpB23b: MK671572; LpB26: MK671573.
2.5. Phylogenetic Analysis
2.6. Nodulation Tests
2.7. Tests for Plant-Growth-Promotion (PGP) Traits
3. Results
3.1. Bacterial Diversity
3.2. Phylogenetic Analyses
3.2.1. 16S rRNA Gene
3.2.2. Protein-Encoding Housekeeping Genes
3.2.3. Symbiotic Genes: nodC and nifH
3.3. Nodulation Tests
3.4. PGP Traits
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Allan, G.J.; Francisco-Ortega, J.; Santos-Guerra, A.; Boerner, E.; Zimmer, E.A. Molecular phylogenetic evidence for the geographic origin and classification of Canary Island Lotus (Fabaceae: Loteae). Mol. Phylogenetics Evol. 2004, 32, 123–138. [Google Scholar] [CrossRef] [PubMed]
- Díaz, P.; Borsani, O.; Monza, J. Lotus-related species and their agronomic importance. In Lotus japonicus Handbook; Springer-Verlag: Berlin/Heidelberg, 2005; pp. 25–37. [Google Scholar]
- Márquez, A. Lotus Japonicus, A General Introduction; Springer: Sevilha, Spain, 2005; pp. 3–15. ISBN 978-1-4020-3735-1. [Google Scholar]
- Sato, S.; Nakamura, Y.; Kaneko, T.; Asamizu, E.; Kato, T.; Nakao, M.; Sasamoto, S.; Watanabe, A.; Ono, A.; Kawashima, K.; et al. Genome structure of the legume, Lotus japonicus. DNA Res. 2008, 15, 227–239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Monza, J.; Fabiano, E.; Arias, A. Characterization of an indigenous population of rhizobia nodulating Lotus corniculatus. Soil Biol. Biochem. 1992, 24, 241–247. [Google Scholar] [CrossRef]
- Pankhurst, C.E.; Craig, A.S.; Jones, W.T. Effectiveness of Lotus Root Nodules: I. morphology and flavolan content of nodules formed on Lotus pedunculatus by fast-growing Lotus rhizobia. J. Exp. Bot. 1979, 30, 1085–1093. [Google Scholar] [CrossRef]
- Lorite, M.J.; Muñoz, S.; Olivares, J.; Soto, M.J.; Sanjuán, J. Characterization of strains unlike Mesorhizobium loti that nodulate lotus spp. in saline soils of Granada, Spain. Appl. Environ. Microbiol. 2010, 76, 4019–4026. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Irisarri, P.; Milnitsky, F.; Monza, J.; Bedmar, E.J. Characterization of rhizobia nodulating Lotus subbiflorus from Uruguayan soils. Plant Soil 1996, 180, 39–47. [Google Scholar] [CrossRef]
- Lorite, M.J.; Videira e Castro, I.; Muñoz, S.; Sanjuán, J. Phylogenetic relationship of Lotus uliginosus symbionts with bradyrhizobia nodulating genistoid legumes. FEMS Microbiol. Ecol. 2012, 79, 454–464. [Google Scholar] [CrossRef] [Green Version]
- Cooper, J.E.; Wood, M.; Bjourson, A.J. Nodulation of Lotus pedunculatus in acid rooting solution by fast- and slow-growing rhizobia. Soil Biol. Biochem. 1985, 17, 487–492. [Google Scholar] [CrossRef]
- Estrella, M.J.; Muñoz, S.; Soto, M.J.; Ruiz, O.; Sanjuán, J. Genetic diversity and host range of rhizobia nodulating Lotus tenuis in typical soils of the Salado River Basin (Argentina). Appl. Environ. Microbiol. 2009, 75, 1088–1098. [Google Scholar] [CrossRef] [Green Version]
- Lorite, M.J.; Donate-Correa, J.; del Arco-Aguilar, M.; Pérez Galdona, R.; Sanjuán, J.; León-Barrios, M. Lotus endemic to the Canary Islands are nodulated by diverse and novel rhizobial species and symbiotypes. Syst. Appl. Microbiol. 2010, 33, 282–290. [Google Scholar] [CrossRef]
- León-Barrios, M.; Lorite, M.J.; Donate-Correa, J.; Sanjuán, J. Ensifer meliloti bv. lancerottense establishes nitrogen-fixing symbiosis with Lotus endemic to the Canary Islands and shows distinctive symbiotic genotypes and host range. Syst. Appl. Microbiol. 2009, 32, 413–420. [Google Scholar]
- Merabet, C.; Martens, M.; Mahdhi, M.; Zakhia, F.; Sy, A.; Le Roux, C.; Domergue, O.; Coopman, R.; Bekki, A.; Mars, M.; et al. Multilocus sequence analysis of root nodule isolates from Lotus arabicus (Senegal), Lotus creticus, Argyrolobium uniflorum and Medicago sativa (Tunisia) and description of Ensifer numidicus sp. nov. and Ensifer garamanticus sp. nov. Int. J. Syst. Evol. Microbiol. 2010, 60, 664–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lorite, M.J.; Estrella, M.J.; Escaray, F.J.; Sannazzaro, A.; Videira e Castro, I.M.; Monza, J.; Sanjuán, J.; León-Barrios, M. The Rhizobia-Lotus Symbioses: Deeply Specific and Widely Diverse. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Batista, L.; Tomasco, I.; Lorite, M.J.; Sanjuán, J.; Monza, J. Diversity and phylogeny of rhizobial strains isolated from Lotus uliginosus grown in Uruguayan soils. Appl. Soil Ecol. 2013, 66, 19–28. [Google Scholar] [CrossRef]
- De Meyer, S.E.; Van Hoorde, K.; Vekeman, B.; Braeckman, T.; Willems, A. Genetic diversity of rhizobia associated with indigenous legumes in different regions of Flanders (Belgium). Soil Biol. Biochem. 2011, 43, 2384–2396. [Google Scholar] [CrossRef]
- Avontuur, J.; Palmer, M.; Beukes, C.Y.; Chan, W.; Coetzee, M.; Blom, J.; Stępkowski, T.; Kyrpides, N.C.; Woyke, T.; Shapiro, N.; et al. Genome-informed Bradyrhizobium taxonomy: Where to from here? Syst. Appl. Microbiol. 2019, 42, 427–439. [Google Scholar] [CrossRef]
- Ormeño-Orrillo, E.; Martínez-Romero, E. A Genomotaxonomy View of the Bradyrhizobium Genus. Front. Microbiol. 2019, 10, 1334. [Google Scholar] [CrossRef]
- Bradyrhizobium. Available online: http://www.bacterio.net/bradyrhizobium.html (accessed on 3 September 2019).
- Evans, W.R.; Fleischman, D.E.; Calvert, H.E.; Pyati, P.V.; Alter, G.M.; Rao, N.S. Bacteriochlorophyll and Photosynthetic Reaction Centers in Rhizobium Strain BTAi 1. Appl. Environ. Microbiol. 1990, 56, 3445–3449. [Google Scholar] [CrossRef] [Green Version]
- Giraud, E.; Fleischman, D. Nitrogen-fixing symbiosis between photosynthetic bacteria and legumes. Photosynth. Res. 2004, 82, 115–130. [Google Scholar] [CrossRef]
- Giraud, E.; Xu, L.; Chaintreuil, C.; Gargani, D.; Gully, D.; Sadowsky, M.J. Photosynthetic Bradyrhizobium sp. strain ORS285 is capable of forming nitrogen-fixing root nodules on soybeans (Glycine max). Appl. Environ. Microbiol. 2013, 79, 2459–2462. [Google Scholar] [CrossRef] [Green Version]
- Gevers, D.; Cohan, F.M.; Lawrence, J.G.; Spratt, B.G.; Coenye, T.; Feil, E.J.; Stackebrandt, E.; Van de Peer, Y.; Vandamme, P.; Thompson, F.L.; et al. Opinion: Re-evaluating prokaryotic species. Nat. Rev. Microbiol. 2005, 3, 733–739. [Google Scholar] [CrossRef] [PubMed]
- Kitahara, K.; Miyazaki, K. Revisiting bacterial phylogeny: Natural and experimental evidence for horizontal gene transfer of 16S rRNA. Mob. Genet. Elem. 2013, 3, e24210. [Google Scholar] [CrossRef] [Green Version]
- Glaeser, S.P.; Kämpfer, P. Multilocus sequence analysis (MLSA) in prokaryotic taxonomy. Syst. Appl. Microbiol. 2015, 38, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, O.M.; López, M.V.; Riccillo, P.M. The diversity of rhizobia nodulating beans in Northwest Argentina as a source of more efficient inoculant strains. J. Biotechnol. 2001, 91, 181–188. [Google Scholar] [CrossRef]
- De Meyer, S.E.; De Beuf, K.; Vekeman, B.; Willems, A. A large diversity of non-rhizobial endophytes found in legume root nodules in Flanders (Belgium). Soil Biol. Biochem. 2015, 83, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Gossmann, J.A.; Markmann, K.; Brachmann, A.; Rose, L.E.; Parniske, M. Polymorphic infection and organogenesis patterns induced by a Rhizobium leguminosarum isolate from Lotus root nodules are determined by the host genotype. New Phytol. 2012, 196, 561–573. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez, F.; Angelini, J.; Taurian, T.; Tonelli, M.L.; Fabra, A. Endophytic occupation of peanut root nodules by opportunistic Gammaproteobacteria. Syst. Appl. Microbiol. 2009, 32, 49–55. [Google Scholar] [CrossRef]
- Kan, F.L.; Chen, Z.Y.; Wang, E.T.; Tian, C.F.; Sui, X.H.; Chen, W.X. Characterization of symbiotic and endophytic bacteria isolated from root nodules of herbaceous legumes grown in Qinghai-Tibet plateau and in other zones of China. Arch. Microbiol. 2007, 188, 103–115. [Google Scholar] [CrossRef]
- Muresu, R.; Polone, E.; Sulas, L.; Baldan, B.; Tondello, A.; Delogu, G.; Cappuccinelli, P.; Alberghini, S.; Benhizia, Y.; Benhizia, H.; et al. Coexistence of predominantly nonculturable rhizobia with diverse, endophytic bacterial taxa within nodules of wild legumes. FEMS Microbiol. Ecol. 2008, 63, 383–400. [Google Scholar] [CrossRef] [Green Version]
- Trujillo, M.E.; Alonso-Vega, P.; Rodríguez, R.; Carro, L.; Cerda, E.; Alonso, P.; Martínez-Molina, E. The genus Micromonospora is widespread in legume root nodules: The example of Lupinus angustifolius. Isme J 2010, 4, 1265–1281. [Google Scholar] [CrossRef] [Green Version]
- Zgadzaj, R.; James, E.K.; Kelly, S.; Kawaharada, Y.; de Jonge, N.; Jensen, D.B.; Madsen, L.H.; Radutoiu, S. A Legume Genetic Framework Controls Infection of Nodules by Symbiotic and Endophytic Bacteria. PLoS Genet. 2015, 11, e1005280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mrabet, M.; Mnasri, B.; Romdhane, S.B.; Laguerre, G.; Aouani, M.E.; Mhamdi, R. Agrobacterium strains isolated from root nodules of common bean specifically reduce nodulation by Rhizobium gallicum. Fems Microbiol. Ecol. 2006, 56, 304–309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bai, Y.; Zhou, X.; Smith, D.L. Enhanced Soybean Plant Growth Resulting from Coinoculation of Strains with. Crop Sci. 2003, 43, 1774. [Google Scholar] [CrossRef]
- Tokala, R.K.; Strap, J.L.; Jung, C.M.; Crawford, D.L.; Salove, M.H.; Deobald, L.A.; Bailey, J.F.; Morra, M.J. Novel Plant-Microbe Rhizosphere Interaction Involving Streptomyces lydicus WYEC108 and the Pea Plant (Pisum sativum). Appl. Environ. Microbiol. 2002, 68, 2161–2171. [Google Scholar] [CrossRef] [Green Version]
- Glick, B.R. The enhancement of plant growth by free-living bacteria. Can. J. Microbiol. 1995, 41, 109–117. [Google Scholar] [CrossRef]
- Datta, B.; Chakrabartty, P.K. Siderophore biosynthesis genes of Rhizobium sp. isolated from Cicer arietinum L. 3 Biotech 2014, 4, 391–401. [Google Scholar] [CrossRef] [Green Version]
- Halder, A.K.; Mishra, A.K.; Bhattacharyya, P.; Chakrabartty, P.K. Solubilization of rock phosphate by Rhizobium and Bradyrhizobium. J. Gen. Appl. Microbiol. 1990, 36, 81–92. [Google Scholar] [CrossRef]
- Peix, A.; Rivas-Boyero, A.A.; Mateos, P.F.; Rodriguez-Barrueco, C.; Martínez-Molina, E.; Velazquez, E. Growth promotion of chickpea and barley by a phosphate solubilizing strain of Mesorhizobium mediterraneum under growth chamber conditions. Soil Biol. Biochem. 2001, 33, 103–110. [Google Scholar] [CrossRef]
- Datta, C.; Basu, P.S. Indole acetic acid production by a Rhizobium species from root nodules of a leguminous shrub, Cajanus cajan. Microbiol. Res. 2000, 155, 123–127. [Google Scholar] [CrossRef]
- Kudoyarova, G.R.; Melentiev, A.I.; Martynenko, E.V.; Timergalina, L.N.; Arkhipova, T.N.; Shendel, G.V.; Kuz’mina, L.Y.; Dodd, I.C.; Veselov, S.Y. Cytokinin producing bacteria stimulate amino acid deposition by wheat roots. Plant Physiol. Biochem. 2014, 83, 285–291. [Google Scholar] [CrossRef]
- Malik, D.K.; Sindhu, S.S. Production of indole acetic acid by Pseudomonas sp.: Effect of coinoculation with Mesorhizobium sp. Cicer on nodulation and plant growth of chickpea (Cicer arietinum). Physiol. Mol. Biol. Plants 2011, 17, 25–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chao, W.-L. Antagonistic activity of Rhizobium spp. against beneficial and plant pathogenic fungi. Lett. Appl. Microbiol. 1990, 10, 213–215. [Google Scholar] [CrossRef]
- Gómez Expósito, R.; Postma, J.; Raaijmakers, J.M.; De Bruijn, I. Diversity and Activity of Lysobacter Species from Disease Suppressive Soils. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ko, H.-S.; Jin, R.-D.; Krishnan, H.B.; Lee, S.-B.; Kim, K.-Y. Biocontrol ability of Lysobacter antibioticus HS124 against Phytophthora blight is mediated by the production of 4-hydroxyphenylacetic acid and several lytic enzymes. Curr. Microbiol. 2009, 59, 608–615. [Google Scholar] [CrossRef] [PubMed]
- Hahn, N.J. The Congo red reaction in bacteria and its usefulness in the identification of rhizobia. Can. J. Microbiol. 1966, 12, 725–733. [Google Scholar] [CrossRef]
- Somasegaran, P.; Hoben, H.J. Handbook for Rhizobia: Methods in legume-rhizobium technology; Springer-Verlag: Berlin, Germany, 1994; ISBN 0-387-94134-7. [Google Scholar]
- Versalovic, J.; Koeuth, T.; Lupski, J.R. Distribution of repetitive DNA sequences in eubacteria and application to fingerprinting of bacterial genomes. Nucleic Acids Res. 1991, 19, 6823–6831. [Google Scholar] [CrossRef]
- Heras, J.; Domínguez, C.; Mata, E.; Pascual, V.; Lozano, C.; Torres, C.; Zarazaga, M. GelJ--a tool for analyzing DNA fingerprint gel images. BMC Bioinform. 2015, 16, 270. [Google Scholar] [CrossRef] [Green Version]
- Sneath, P.; Sokal, R.R. Numerical Taxonomy: The Principles and Practice of Numerical Classification; Freeman & Co: San Francisco, CA, USA, 1973; ISBN 0716706970 9780716706977. [Google Scholar]
- Herrera-Cervera, J.A.; Caballero-Mellado, J.; Laguerre, G.; Tichy, H.-V.; Requena, N.; Amarger, N.; MartÃ-nez-Romero, E.; Olivares, J.; Sanjuan, J. At least five rhizobial species nodulate Phaseolus vulgaris in a Spanish soil. FEMS Microbiol. Ecol. 1999, 30, 87–97. [Google Scholar] [CrossRef]
- Weisburg, W.G.; Barns, S.M.; Pelletier, D.A.; Lane, D.J. 16S ribosomal DNA amplification for phylogenetic study. J. Bacteriol. 1991, 173, 697–703. [Google Scholar] [CrossRef] [Green Version]
- Gaunt, M.W.; Turner, S.L.; Rigottier-Gois, L.; Lloyd-Macgilp, S.A.; Young, J.P. Phylogenies of atpD and recA support the small subunit rRNA-based classification of rhizobia. Int. J. Syst. Evol. Microbiol. 2001, 51, 2037–2048. [Google Scholar] [CrossRef] [Green Version]
- Stepkowski, T.; Czaplińska, M.; Miedzinska, K.; Moulin, L. The variable part of the dnaK gene as an alternative marker for phylogenetic studies of rhizobia and related alpha Proteobacteria. Syst. Appl. Microbiol. 2003, 26, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Laguerre, G.; Nour, S.M.; Macheret, V.; Sanjuan, J.; Drouin, P.; Amarger, N. Classification of rhizobia based on nodC and nifH gene analysis reveals a close phylogenetic relationship among Phaseolus vulgaris symbionts. Microbiology 2001, 147, 981–993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nguyen, L.-T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Towns, J.; Cockerill, T.; Dahan, M.; Foster, I.; Gaither, K.; Grimshaw, A.; Hazlewood, V.; Lathrop, S.; Lifka, D.; Peterson, G.D.; et al. XSEDE: Accelerating Scientific Discovery. Comput. Sci. Eng. 2014, 16, 62–74. [Google Scholar] [CrossRef]
- Miller, M.A.; Pfeiffer, W.; Schwartz, T. Creating the CIPRES Science Gateway for inference of large phylogenetic trees. In Proceedings of the 2010 Gateway Computing Environments Workshop (GCE); IEEE: New Orleans, LA, USA, 2010; pp. 1–8. [Google Scholar]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.F.; von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef] [Green Version]
- Hoang, D.T.; Chernomor, O.; von Haeseler, A.; Minh, B.Q.; Vinh, L.S. UFBoot2: Improving the Ultrafast Bootstrap Approximation. Mol. Biol. Evol. 2018, 35, 518–522. [Google Scholar] [CrossRef]
- Letunic, I.; Bork, P. Interactive Tree Of Life (iTOL) v4: Recent updates and new developments. Nucleic Acids Res. 2019, 47, W256–W259. [Google Scholar] [CrossRef] [Green Version]
- Jensen, H.L. Nitrogen fixation in leguminous plants. I. General characters of root-nodule bacteria isolated from species of Medicago and Trifolium in Australia. Proc. Linn. Soc. New South Wales 1941, 67, 98–108. [Google Scholar]
- Ferreira, E.M.; Marques, J.F. Selection of Portuguese Rhizobium leguminosarum bv. trifolii strains for production of legume inoculants. Plant Soil 1992, 147, 151–158. [Google Scholar]
- Illmer, P.; Barbato, A.; Schinner, F. Solubilization of hardly-soluble AlPO4 with P-solubilizing microorganisms. Soil Biol. Biochem. 1995, 27, 265–270. [Google Scholar] [CrossRef]
- Beringer, J.E. R factor transfer in Rhizobium leguminosarum. J. Gen. Microbiol. 1974, 84, 188–198. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pérez-Miranda, S.; Cabirol, N.; George-Téllez, R.; Zamudio-Rivera, L.S.; Fernández, F.J. O-CAS, a fast and universal method for siderophore detection. J. Microbiol. Methods 2007, 70, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.C.; Ladha, J.K.; Tripathi, A.K. Evaluation of plant growth promoting and colonization ability of endophytic diazotrophs from deep water rice. J. Biotechnol. 2001, 91, 127–141. [Google Scholar] [CrossRef]
- Brown, J.R.; Masuchi, Y.; Robb, F.T.; Doolittle, W.F. Evolutionary relationships of bacterial and archaeal glutamine synthetase genes. J. Mol. Evol. 1994, 38, 566–576. [Google Scholar] [CrossRef]
- Stępkowski, T.; Zak, M.; Moulin, L.; Króliczak, J.; Golińska, B.; Narożna, D.; Safronova, V.I.; Mądrzak, C.J. Bradyrhizobium canariense and Bradyrhizobium japonicum are the two dominant rhizobium species in root nodules of lupin and serradella plants growing in Europe. Syst. Appl. Microbiol. 2011, 34, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Vinuesa, P.; Silva, C.; Werner, D.; Martínez-Romero, E. Population genetics and phylogenetic inference in bacterial molecular systematics: The roles of migration and recombination in Bradyrhizobium species cohesion and delineation. Mol. Phylogenet. Evol. 2005, 34, 29–54. [Google Scholar] [CrossRef]
- Bourebaba, Y.; Durán, D.; Boulila, F.; Ahnia, H.; Boulila, A.; Temprano, F.; Palacios, J.M.; Imperial, J.; Ruiz-Argüeso, T.; Rey, L. Diversity of Bradyrhizobium strains nodulating Lupinus micranthus on both sides of the Western Mediterranean: Algeria and Spain. Syst. Appl. Microbiol. 2016, 39, 266–274. [Google Scholar] [CrossRef]
- Mousavi, S.A.; Willems, A.; Nesme, X.; de Lajudie, P.; Lindström, K. Revised phylogeny of Rhizobiaceae: Proposal of the delineation of Pararhizobium gen. nov., and 13 new species combinations. Syst. Appl. Microbiol. 2015, 38, 84–90. [Google Scholar] [CrossRef]
- Park, J.H.; Kim, R.; Aslam, Z.; Jeon, C.O.; Chung, Y.R. Lysobacter capsici sp. nov., with antimicrobial activity, isolated from the rhizosphere of pepper, and emended description of the genus Lysobacter. Int. J. Syst. Evol. Microbiol. 2008, 58, 387–392. [Google Scholar] [CrossRef] [Green Version]
- Puopolo, G.; Sonego, P.; Engelen, K.; Pertot, I. Draft Genome Sequence of Lysobacter capsici AZ78, a Bacterium Antagonistic to Plant-Pathogenic Oomycetes. Genome Announc. 2014, 2. [Google Scholar] [CrossRef] [Green Version]
- Lin, H.; Hu, S.; Liu, R.; Chen, P.; Ge, C.; Zhu, B.; Guo, L. Genome Sequence of Pseudomonas koreensis CRS05-R5, an Antagonistic Bacterium Isolated from Rice Paddy Field. Front. Microbiol 2016, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rafikova, G.F.; Korshunova, T.Y.; Minnebaev, L.F.; Chetverikov, S.P.; Loginov, O.N. A new bacterial strain, Pseudomonas koreensis IB-4, as a promising agent for plant pathogen biological control. Microbiology 2016, 85, 333–341. [Google Scholar] [CrossRef]
- Ganeshan, G.; Kumar, A.M. Pseudomonas fluorescens, a potential bacterial antagonist to control plant diseases. J. Plant Interact. 2005, 1, 123–134. [Google Scholar] [CrossRef]
- Trapet, P.; Avoscan, L.; Klinguer, A.; Pateyron, S.; Citerne, S.; Chervin, C.; Mazurier, S.; Lemanceau, P.; Wendehenne, D.; Besson-Bard, A. The Pseudomonas fluorescens Siderophore Pyoverdine Weakens Arabidopsis thaliana Defense in Favor of Growth in Iron-Deficient Conditions. Plant Physiol. 2016, 171, 675–693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lewis, R.W.; Islam, A.; Opdahl, L.; Davenport, J.R.; Sullivan, T.S. Comparative Genomics, Siderophore Production, and Iron Scavenging Potential of Root Zone Soil Bacteria Isolated from ‘Concord’ Grape Vineyards. Microb. Ecol. 2019, 78, 699–713. [Google Scholar] [CrossRef] [PubMed]
- Gusain, Y.S.; Kamal, R.; Mehta, C.M.; Singh, U.S.; Sharma, A.K. Phosphate solubilizing and indole-3-acetic acid producing bacteria from the soil of Garhwal Himalaya aimed to improve the growth of rice. J. Env. Biol. 2015, 36, 301–307. [Google Scholar]
- Kasotia, A.; Choudhary, D.K. Induced Inorganic Phosphate Solubilization Through N-Methyl-N´-Nitro-N-Nitrosoguanidine Treated Mutants of Pseudomonas koreensis Strain AK-1 (MTCC Number 12058) under Polyethylene Glycol. Proc. Natl. Acad. Sci. USA 2016, 86, 115–123. [Google Scholar] [CrossRef]
- Kwon, S.W.; Kim, J.S.; Park, I.C.; Yoon, S.H.; Park, D.H.; Lim, C.K.; Go, S.J. Pseudomonas koreensis sp. nov., Pseudomonas umsongensis sp. nov. and Pseudomonas jinjuensis sp. nov., novel species from farm soils in Korea. Int. J. Syst. Evol. Microbiol. 2003, 53, 21–27. [Google Scholar] [CrossRef] [Green Version]
- Daimon, H.; Nobuta, K.; Ohe, M.; Harada, J.; Nakayama, Y. Tricalcium Phosphate Solubilization by Root Nodule Bacteria of Sesbania cannabina and Crotalaria juncea. Plant Prod. Sci. 2006, 9, 388–389. [Google Scholar] [CrossRef]
- Das, K.; Prasanna, R.; Saxena, A.K. Rhizobia: A potential biocontrol agent for soil borne fungal pathogens. Folia Microbiol. (Praha) 2017, 62, 425–435. [Google Scholar] [CrossRef]
- Deryło, M.; Choma, A.; Puchalski, B.; Suchanek, W. Siderophore activity in Rhizobium species isolated from different legumes. Acta Biochim. Pol. 1994, 41, 7–11. [Google Scholar] [CrossRef] [Green Version]
- Afzal, A.; Bano, A. Rhizobium and Phosphate Solubilizing Bacteria Improve the Yield and Phosphorus Uptake in Wheat (Triticum aestivum). Int. J. Agric. Biol. 2008, 10, 1560–8530. [Google Scholar]
- Ahmad, M.; Zahir, Z.A.; Khalid, M.; Nazli, F.; Arshad, M. Efficacy of Rhizobium and Pseudomonas strains to improve physiology, ionic balance and quality of mung bean under salt-affected conditions on farmer’s fields. Plant Physiol. Biochem. 2013, 63, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Egamberdieva, D.; Berg, G.; Lindström, K.; Räsänen, L.A. Co-inoculation of Pseudomonas spp. with Rhizobium improves growth and symbiotic performance of fodder galega (Galega orientalis Lam.). Eur. J. Soil Biol. 2010, 46, 269–272. [Google Scholar] [CrossRef]
- Videira e Castro, I.; de Castro Silva, M.; Fernandez, C.; Colavolpe, B.; Machado, H. The Potential of Nitrogen-Fixing Bacteria in the Sustainability of Agro-Forestry Ecosystems. In Microbial Probiotics for Agricultural Systems: Advances in Agronomic Use; Zúñiga-Dávila, D., González-Andrés, F., Ormeño-Orrillo, E., Eds.; Sustainability in Plant and Crop Protection; Springer International Publishing: Cham, Switzerland, 2019; pp. 71–82. ISBN 978-3-030-17597-9. [Google Scholar]
- Pandya, M.; Rajput, M.; Rajkumar, S. Exploring plant growth promoting potential of non rhizobial root nodules endophytes of Vigna radiata. Microbiology 2015, 84, 80–89. [Google Scholar] [CrossRef]
- Saini, R.; Dudeja, S.S.; Giri, R.; Kumar, V. Isolation, characterization, and evaluation of bacterial root and nodule endophytes from chickpea cultivated in Northern India. J. Basic Microbiol. 2015, 55, 74–81. [Google Scholar] [CrossRef]
- Zhao, L.; Xu, Y.; Lai, X. Antagonistic endophytic bacteria associated with nodules of soybean (Glycine max L.) and plant growth-promoting properties. Braz. J. Microbiol. 2017, 49, 269–278. [Google Scholar] [CrossRef]
- Cardoso, P.; Alves, A.; Silveira, P.; Sá, C.; Fidalgo, C.; Freitas, R.; Figueira, E. Bacteria from nodules of wild legume species: Phylogenetic diversity, plant growth promotion abilities and osmotolerance. Sci. Total Environ. 2018, 645, 1094–1102. [Google Scholar] [CrossRef]
- Ampomah, O.Y.; Huss-Danell, K. Genetic diversity of root nodule bacteria nodulating Lotus corniculatus and Anthyllis vulneraria in Sweden. Syst. Appl. Microbiol. 2011, 34, 267–275. [Google Scholar] [CrossRef]
- Sánchez, M.; Ramírez-Bahena, M.-H.; Peix, A.; Lorite, M.J.; Sanjuán, J.; Velázquez, E.; Monza, J. Phyllobacterium loti sp. nov. isolated from nodules of Lotus corniculatus. Int. J. Syst. Evol. Microbiol. 2014, 64, 781–786. [Google Scholar] [CrossRef]





| Primer | Gene | Position * | Direction | Length | Sequence (5′–3′) |
|---|---|---|---|---|---|
| recA107 | recA | 107 | Forward | 23 bp | TTAGGTGATACTGCCRTBGARCC |
| recA593 | recA | 593 | Reverse | 24 bp | GGGTTWCCGAACATTACRCCRATT |
| atpD183 | atpD | 183 | Forward | 24 bp | CGTGTTCGTRCHATTGCBATGGAY |
| atpD872 | atpD | 872 | Reverse | 20 bp | GGCATACGGCCCAGCAGTGC |
| atpD133 | atpD | 133 | Forward | 23 bp | GACCCTGGAAGTTCAGCAGCAGC |
| atpD762 | atpD | 762 | Reverse | 20 bp | GGCATACGGCCCAGCAGTGC |
| 16SIR | 16SrRNA | 875 | Reverse | 21 bp | AAACACATGCTCCACCGCTTG |
| Isolates | Congo Red Absorption | Growing Velocity | |
|---|---|---|---|
| Sample Spot A | LpA5a, LpA6, LpA8, LpA9, LpA12, LpA13b, LpA14, LpA5b | − | Fast |
| LpA7a, LpA7b, LpA10, LpA13a | + | Moderate | |
| LpA11 | − | Slow | |
| Sample Spot B | LpB5b, LpB5c, LpB5e, LpB5f, LpB5g, LpB12a, LpB16a, LpB16b, LpB16c, LpB16d | − | Very fast |
| LpB10d | + | ||
| LpB9a, LpB9b, LpB12b, LpB13, LpB14a, LpB14b, LpB15b¸ LpB17b | − | Fast | |
| LpB5d, LpB10a LpB10b | + | ||
| LpB19a, LpB23b, LpB26 | − | Slow | |
| LpB10c, LpB23a | + |
| Isolates | Symbiotic Phenotype | ||||||
|---|---|---|---|---|---|---|---|
| Lotus Group I | Lotus Group II | Lupinus | Glycine | ||||
| Lc | Lt P | Lt E | Lu | Lp | Ll | Gm | |
| LpA11 | Fix− | Nod− | Fix− | Fix+ | Fix+ | Fix+ | Nod- |
| LpB19a, LpB23b, LpB26 | Fix− | Fix− | NT | Fix+ | Fix+ | Fix+ | Nod- |
| LpA5a, LpA5b, LpA6, LpA7a, LpA7b, LpA8, LpA9, LpA10, LpA12, LpA13a, LpA13b, LpA14 | Nod− | Nod− | Nod− | Nod− | Nod− | NT | NT |
| LpB5b, LpB5c, LpB5d, LpB5e, LpB5f, LpB5g, LpB9a, LpB9b, LpB10a, LpB10b, LpB10c, LpB10d, LpB12a, LpB12b, LpB13, LpB14a, LpB14b, LpB15b, LpB16a, LpB16b, LpB16c, LpB16d, LpB17b, LpB23a | Nod− | Nod− | NT | Nod− | Nod− | NT | NT |
| Isolates | P sol | Sid Production | Antagonistic Activity | Lytic Enzymes Production | |||
|---|---|---|---|---|---|---|---|
| P. cinammomi | B. corticola | Cel | Pect | ||||
| LpB16d | + | − | + | + | + | − | |
| LpA7a, LpA7b, LpB16a, LpB16b | − | − | + | + | + | − | |
| LpB23a | − | − | − | + | + | + | |
| LpB9b | − | − | − | − | + | + | |
| LpB5b, | − | + | − | − | + | − | |
| LpB12b, LpB10b, LpB5f | − | − | − | − | + | − | |
| LpA6, LpB12a, LpB5c | − | + | − | − | − | − | |
| LpB5e | − | − | − | − | − | − | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soares, R.; Trejo, J.; Lorite, M.J.; Figueira, E.; Sanjuán, J.; Videira e Castro, I. Diversity, Phylogeny and Plant Growth Promotion Traits of Nodule Associated Bacteria Isolated from Lotus parviflorus. Microorganisms 2020, 8, 499. https://doi.org/10.3390/microorganisms8040499
Soares R, Trejo J, Lorite MJ, Figueira E, Sanjuán J, Videira e Castro I. Diversity, Phylogeny and Plant Growth Promotion Traits of Nodule Associated Bacteria Isolated from Lotus parviflorus. Microorganisms. 2020; 8(4):499. https://doi.org/10.3390/microorganisms8040499
Chicago/Turabian StyleSoares, Ricardo, Jesús Trejo, Maria J. Lorite, Etelvina Figueira, Juan Sanjuán, and Isabel Videira e Castro. 2020. "Diversity, Phylogeny and Plant Growth Promotion Traits of Nodule Associated Bacteria Isolated from Lotus parviflorus" Microorganisms 8, no. 4: 499. https://doi.org/10.3390/microorganisms8040499