The Role of Calcium and Strontium as the Most Dominant Elements during Combinations of Different Alkaline Earth Metals in the Synthesis of Crystalline Silica-Carbonate Biomorphs
Abstract
:1. Introduction
2. Materials and Methods
2.1. Strains Used for Nucleic Acids Extraction
2.2. Extraction of Genomic DNA
2.3. RNA Extraction
2.4. Biomorphs Formation
2.5. Characterization of Biomorphs
3. Results and Discussion
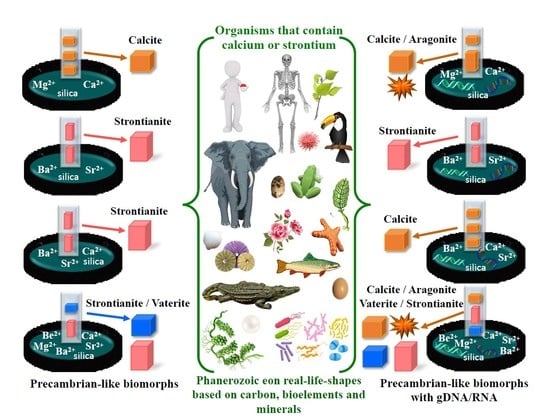
3.1. Composition of Biomorphs Depends on the Nature of the Chemical Elements, Whereas the Shape Depends on the Biomolecules
3.2. Strontium is the Dominant Element in the Biomorphs Synthesized in a Combination of Ba2+ and Sr2+
3.3. In the Presence of Nucleic Acids, Calcium is the Element that Controls the Synthesis of Biomorphs
3.4. Calcium and Strontium in the Presence of Nucleic Acids are Part of Solid Structures Obtained from a Mixture with Multiple Chemical Elements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Oparin, A.I. Modern Views on the Origin of Life. Scientia. 1960, 95, 322–326. [Google Scholar]
- Cech, T.R.; Grabowski, P.J.; Zaug, A.J. Splicing of the Ribosomal-RNA Precursor and Cyclization of the Excised Intervening Sequence Rna in Nuclei of Tetrahymena. Fed. Proc. 1981, 40, 1651. [Google Scholar]
- Cech, T.R. Rna as an Enzyme. Biochem. Int. 1989, 18, 7–14. [Google Scholar] [CrossRef]
- Larsen, T.; Sandstrom, B. Effect of Calcium, Copper, and Zinc Levels in a Rapeseed Meal Diet on Mineral and Trace-Element Utilization in the Rat. Biol. Trace. Elem. Res. 1992, 35, 167–184. [Google Scholar] [CrossRef]
- Williams, R.J.P.; Fraústo da Silva, J.J.R. Bringing Chemistry to Life: From Matter to Man; Oxford University Press: New York, NY, USA, 1999; pp. 293–298. [Google Scholar]
- Fraústo da Silva, J.J.R.; Williams, R.J.P. The Biological Chemistry of the Elements: The Inorganic Chemistry of Life, 2nd ed.; Oxford University Press: New York, NY, USA, 1991; pp. 7–10. [Google Scholar]
- Turner, D.R.; Whitfield, M.; Dickson, A.G. The Equilibrium Speciation of Dissolved Components in Fresh-Water and Seawater at 25-Degrees-C and 1 Atm Pressure. Geochim. Cosmochim. Ac 1981, 45, 855–881. [Google Scholar] [CrossRef]
- Schopf, J.W. Microfossils of the Early Archean Apex Chert-New Evidence of the Antiquity of Life. Science 1993, 260, 640–646. [Google Scholar] [CrossRef]
- Pearce, B.K.D.; Pudritz, R.E.; Semenov, D.A.; Henning, T.K. Origin of the RNA world: The fate of nucleobases in warm little ponds. Proc. Natl. Acad. Sci. USA 2017, 114, 11327–11332. [Google Scholar] [CrossRef] [Green Version]
- Zhang, G.; Morales, J.; Garcia-Ruiz, J.M. Growth behaviour of silica/carbonate nanocrystalline composites of calcite and aragonite. J. Mater. Chem. B 2017, 5, 1658–1663. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Ruiz, J.M.; Hyde, S.T.; Carnerup, A.M.; Christy, A.G.; Van Kranendonk, M.J.; Welham, N.J. Self-assembled silica-carbonate structures and detection of ancient microfossils. Science 2003, 302, 1194–1197. [Google Scholar] [CrossRef]
- Opel, J.; Wimmer, F.P.; Kellermeier, M.; Colfen, H. Functionalisation of silica-carbonate biomorphs. Nanoscale Horiz. 2016, 1, 144–149. [Google Scholar] [CrossRef]
- Eiblmeier, J.; Dankesreiter, S.; Ptitzner, A.; Schmalz, G.; Kunz, W.; Kellermeier, M. Crystallization of mixed alkaline-earth carbonates in silica solutions at high pH. Cryst. Growth Des. 2014, 14, 6177–6188. [Google Scholar] [CrossRef]
- Cuéllar-Cruz, M.; Islas, S.; González, G.; Moreno, A. The influence of nucleic acids on the synthesis of crystalline Ca(II), Ba(II) and Sr(II) silica-carbonate biomorphs: Implications to the chemical origin of life on primitive Earth. Cryst. Growth Des. 2019. Available online: https://pubs.acs.org/doi/10.1021/acs.cgd.9b00573. (accessed on 23 July 2019).
- Nakouzi, E.; Steinbock, O. Self-organization in precipitation reactions far from the equilibrium. Sci. Adv. 2016, 2, e160144. [Google Scholar] [CrossRef]
- Garcia-Ruiz, J.M.; Melero-Garcia, E.; Hyde, S.T. Morphogenesis of Self-Assembled Nanocrystalline Materials of Barium Carbonate and Silica. Science 2009, 323, 362–365. [Google Scholar] [CrossRef] [Green Version]
- Noorduin, W.L.; Grinthal, A.; Mahadevan, L.; Aizenberg, J. Rationally Designed Complex, Hierarchical Microarchitectures. Science 2013, 340, 832–837. [Google Scholar] [CrossRef] [Green Version]
- DeOliveira, D.B.; Laursen, R.A. Control of calcite crystal morphology by a peptide designed to bind to a specific surface. J. Am. Chem. Soc. 1997, 119, 10627–10631. [Google Scholar] [CrossRef]
- Carteret, C.; Dandeu, A.; Moussaoui, S.; Muhr, H.; Humbert, B.; Plasari, E. Polymorphism Studied by Lattice Phonon Raman Spectroscopy and Statistical Mixture Analysis Method. Application to Calcium Carbonate Polymorphs during Batch Crystallization. Cryst. Growth Des. 2009, 9, 807–812. [Google Scholar] [CrossRef]
- Vagenas, N.V.; Gatsouli, A.; Kontoyannis, C.G. Quantitative analysis of synthetic calcium carbonate polymorphs using FT-IR spectroscopy. Talanta 2003, 59, 831–836. [Google Scholar] [CrossRef]
- Agarwal, P.; Berglund, K.A. In situ monitoring of calcium carbonate polymorphs during batch crystallization in the presence of polymeric additives using Raman spectroscopy. Cryst. Growth Des. 2003, 3, 941–946. [Google Scholar] [CrossRef]
- Cuellar-Cruz, M. Synthesis of inorganic and organic crystals mediated by proteins in different biological organisms. A mechanism of biomineralization conserved throughout evolution in all living species. Prog. Cryst. Growth Charact. Mater. 2017, 63, 94–103. [Google Scholar] [CrossRef]
- Ruiz-Arellano, R.R.; Moreno, A. Obtainment of Spherical-Shaped Calcite Crystals Induced by Intramineral Proteins Isolated from Eggshells of Ostrich and Emu. Cryst. Growth Des. 2014, 14, 5137–5143. [Google Scholar] [CrossRef]
- Ruiz-Arellano, R.R.; Medrano, F.J.; Moreno, A.; Romero, A. Structure of struthiocalcin-1, an intramineral protein from Struthio camelus eggshell, in two crystal forms. Acta Crystallogr. D: Biol. Crystallogr. 2015, 71, 809–818. [Google Scholar] [CrossRef] [PubMed]
- Colfen, H. Biomineralization a crystal-clear view. Nat. Mater. 2010, 9, 960–961. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Su, J.T.; Zheng, G.L.; Liang, J.; Zhang, G.Y.; Wang, H.Z.; Xie, L.P.; Zhang, R.Q. Patterns of Expression in the Matrix Proteins Responsible for Nucleation and Growth of Aragonite Crystals in Flat Pearls of Pinctada fucata. PloS One 2013, 8, e66564. [Google Scholar] [CrossRef]
- Rudel, H.; Muller, J.; Jurling, H.; Bartel-Steinbach, M.; Koschorreck, J. Survey of patterns, levels, and trends of perfluorinated compounds in aquatic organisms and bird eggs from representative German ecosystems. Environ. Sci. Pollut. R 2011, 18, 1457–1470. [Google Scholar] [CrossRef] [PubMed]
- Demichelis, R.; Raiteri, P.; Gale, J.D.; Dovesi, R. The Multiple Structures of Vaterite. Cryst. Growth Des. 2013, 13, 2247–2251. [Google Scholar] [CrossRef]
- Lowenstam, H.A. Spicular Morphology and Mineralogy in Some Pyuridae (Ascidiacea). B Mar. Sci. 1989, 45, 243–252. [Google Scholar]
- Watabe, N. Crystal-Growth of Calcium-Carbonate in Biological-Systems. J. Cryst. Growth 1974, 24, 116–122. [Google Scholar] [CrossRef]
- Beniash, E.; Aizenberg, J.; Addadi, L.; Weiner, S. Amorphous calcium carbonate transforms into calcite during sea urchin larval spicule growth. Proc. Roy. Soc. B Biol. Sci. 1997, 264, 461–465. [Google Scholar] [CrossRef]
- Lowenstam, H.A.; Abbott, D.P. Vaterite - Mineralization Product of Hard Tissues of a Marine Organism (Ascidiacea). Science 1975, 188, 363–365. [Google Scholar] [CrossRef]
- Nehrke, G.; Nouet, J. Confocal Raman microscope mapping as a tool to describe different mineral and organic phases at high spatial resolution within marine biogenic carbonates: Case study on Nerita undata (Gastropoda, Neritopsina). Biogeosci. 2011, 8, 3761–3769. [Google Scholar] [CrossRef]
- Soldati, A.L.; Jacob, D.E.; Wehrmeister, U.; Hofmeister, W. Structural characterization and chemical composition of aragonite and vaterite in freshwater cultured pearls. Miner. Mag. 2008, 72, 579–592. [Google Scholar] [CrossRef]
- Spann, N.; Harper, E.M.; Aldridge, D.C. The unusual mineral vaterite in shells of the freshwater bivalve Corbicula fluminea from the UK. Naturwissenschaften 2010, 97, 743–751. [Google Scholar] [CrossRef] [PubMed]
- Falini, G.; Albeck, S.; Weiner, S.; Addadi, L. Control of aragonite or calcite polymorphism by mollusk shell macromolecules. Science 1996, 271, 67–69. [Google Scholar] [CrossRef]
- Veizer, J.; Ala, D.; Azmy, K.; Bruckschen, P.; Buhl, D.; Bruhn, F.; Carden, G.A.F.; Diener, A.; Ebneth, S.; Godderis, Y.; et al. Sr-87/Sr-86, delta C-13 and delta O-18 evolution of Phanerozoic seawater. Chem. Geol. 1999, 161, 59–88. [Google Scholar] [CrossRef]
- Templeton, W.L.; Brown, V.M. The Relationship between the Concentrations of Calcium, Strontium and Strontium-90 in Wild Brown Trout, Salmo-Trutta L and the Concentrations of the Stable Elements in Some Waters of the United-Kingdom, and the Implications in Radiological Health Studies. Air Water Pollut. 1964, 8, 49–75. [Google Scholar]
- Rouillard, J.; García-Ruiz, J.M.; Gong, J.; van Zuilen, M.A. A morphogram for silica-witherite biomorphs and its application to microfossil identification in the early earth rock record. Geobiology 2018, 16, 279–296. [Google Scholar] [CrossRef] [Green Version]
- Bonales, L.J.; Munoz-Iglesias, V.; Santamaria-Perez, D.; Caceres, M.; Fernandez-Remolar, D.; Prieto-Ballesteros, O. Quantitative Raman spectroscopy as a tool to study the kinetics and formation mechanism of carbonates. Spectrochim. Acta A 2013, 116, 26–30. [Google Scholar] [CrossRef]
- Rieder, K.H.; Migoni, R.; Renker, B. Lattice-Dynamics of Strontium Oxide. Phys. Rev. B 1975, 12, 3374–3379. [Google Scholar] [CrossRef]
- Lin, C.C.; Liu, L.G. High-pressure Raman spectroscopic study of post-aragonite phase transition in witherite (BaCO3). Eur. J. Miner. 1997, 9, 785–792. [Google Scholar] [CrossRef]
- Bowen, H.J.M. Strontium and Barium in Sea Water and Marine Organisms. J. Mar. Biol. Assoc. U.K. 1956, 35, 451–460. [Google Scholar] [CrossRef]
- Niedermeier, M.; Gierlinger, N.; Lutz-Meindl, U. Biomineralization of strontium and barium contributes to detoxification in the freshwater alga Micrasterias. J. Plant. Physiol. 2018, 230, 80–91. [Google Scholar] [CrossRef] [PubMed]
- Isermann, K.; Glatzle, A. Discrimination of Calcium against Strontium in Plants with a Cryptating Agent. Naturwissenschaften 1975, 62, 238–239. [Google Scholar] [CrossRef]
- Gilbert, W. Origin of Life: The RNA World. Nature 1986, 319, 618. [Google Scholar] [CrossRef]
- Christy, A.G. A review of the structures of vaterite: The Impossible, the possible and the likely. Cryst. Growth Des. 2017, 17, 3567–3578. [Google Scholar] [CrossRef]
- Currey, J.; Taylor, J. The mechanical behaviour of some molluscan hard tissues. J. Zool. 1974, 173, 395–406. [Google Scholar] [CrossRef]
- Burgess, K.M.N.; Bryce, D.L. On the crystal structure of the vaterite polymorph of CaCO3: A calcium-43 solid-state NMR and computational assessment. Solid State Nucl. Mag. Res. 2015, 65, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Mann, K. The calcified eggshell matrix proteome of a songbird, the zebra finch (Taeniopygia guttata). Proteome. Sci. 2015, 13, 29. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Grajeda, J.P.; Moreno, A.; Romero, A. Crystal structure of ovocleidin-17, a major protein of the calcified Gallus gallus eggshell - Implications in the calcite mineral growth pattern. J. Biol. Chem. 2004, 279, 40876–40881. [Google Scholar] [CrossRef] [PubMed]
- Vanhoutte, P.M.; Collis, M.G.; Janssens, W.J.; Verbeuren, T.J. Calcium Dependence of Prejunctional Inhibitory Effects of Adenosine and Acetylcholine on Adrenergic Neurotransmission in Canine Saphenous Veins. Eur. J. Pharmacol. 1981, 72, 189–198. [Google Scholar] [CrossRef]
- Douglas, W.W.; Rubin, R.P. The Role of Calcium in Secretory Response of the Adrenal Medulla to Acetylcholine. J. Physiol. 1961, 159, 40–57. [Google Scholar] [CrossRef] [PubMed]
- Katz, B. Neural transmitter release: From quantal secretion to exocytosis and beyond. J. Neurocytol. 1996, 25, 677–686. [Google Scholar] [CrossRef] [PubMed]
- White, P.J.; Broadley, M.R. Calcium in plants. Ann. Bot. 2003, 92, 487–511. [Google Scholar] [CrossRef] [PubMed]
- Dominguez, D.C. Calcium signalling in bacteria. Mol. Microbiol. 2004, 54, 291–297. [Google Scholar] [CrossRef] [PubMed]









| Number of Combinations | Combinations |
|---|---|
| 1 | Ca2+ + Mg2+ |
| 2 | Ca2+ + Mg2+ + RNA |
| 3 | Ca2+ + Mg2+ + gDNA |
| 4 | Ba2+ + Sr2+ |
| 5 | Ba2+ + Sr2+ + RNA |
| 6 | Ba2+ + Sr2+ + gDNA |
| 7 | Ca2+ + Ba2+ + Sr2+ |
| 8 | Ca2+ + Ba2+ + Sr2+ + RNA |
| 9 | Ca2+ + Ba2+ + Sr2+ + gDNA |
| 10 | Be2+ + Mg2+ + Ca2+ + Ba2+ + Sr2+ |
| 11 | Be2+ + Mg2+ + Ca2+ + Ba2+ + Sr2+ + RNA |
| 12 | Be2+ + Mg2+ + Ca2+ + Ba2+ + Sr2+ + gDNA |
| Number | Combinations | Prevalent Polymorphs |
|---|---|---|
| 1 | Ca2+ + Mg2+ | Calcite |
| 2 | Ca2+ + Mg2+ + RNA | Calcite |
| 3 | Ca2+ + Mg2+ + gDNA | Aragonite |
| 4 | Ba2+ + Sr2+ | Strontianite |
| 5 | Ba2+ + Sr2+ + RNA | Strontianite |
| 6 | Ba2+ + Sr2+ + gDNA | Strontianite |
| 7 | Ca2+ + Ba2+ + Sr2+ | Strontianite |
| 8 | Ca2+ + Ba2+ + Sr2+ + RNA | Calcite, Vaterite |
| 9 | Ca2+ + Ba2+ + Sr2+ + gDNA | Calcite |
| 10 | Be2+ + Mg2+ + Ca2+ + Ba2+ + Sr2+ | Strontianite, Vaterite |
| 11 | Be2+ + Mg2+ + Ca2+ + Ba2+ + Sr2+ + RNA | Calcite, Strontianite, Aragonite, Vaterite |
| 12 | Be2+ + Mg2+ + Ca2+ + Ba2+ + Sr2+ + gDNA | Strontianite, Aragonite, Vaterite |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuéllar-Cruz, M.; Moreno, A. The Role of Calcium and Strontium as the Most Dominant Elements during Combinations of Different Alkaline Earth Metals in the Synthesis of Crystalline Silica-Carbonate Biomorphs. Crystals 2019, 9, 381. https://doi.org/10.3390/cryst9080381
Cuéllar-Cruz M, Moreno A. The Role of Calcium and Strontium as the Most Dominant Elements during Combinations of Different Alkaline Earth Metals in the Synthesis of Crystalline Silica-Carbonate Biomorphs. Crystals. 2019; 9(8):381. https://doi.org/10.3390/cryst9080381
Chicago/Turabian StyleCuéllar-Cruz, Mayra, and Abel Moreno. 2019. "The Role of Calcium and Strontium as the Most Dominant Elements during Combinations of Different Alkaline Earth Metals in the Synthesis of Crystalline Silica-Carbonate Biomorphs" Crystals 9, no. 8: 381. https://doi.org/10.3390/cryst9080381