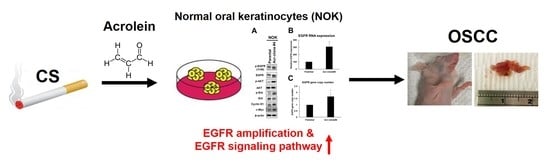
Cigarette Smoke Containing Acrolein Upregulates EGFR Signaling Contributing to Oral Tumorigenesis In Vitro and In Vivo
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture and Acrolein Treatment
2.2. Cell Viability and Cell Proliferation Assay
2.3. Flow Cytometry Analysis of Cell Cycle Phases
2.4. Soft Agar Colony Formation Assay
2.5. Cell Migration Assay
2.6. Quantitative Real-Time PCR
2.7. Western Blot Analysis
2.8. Xenograft Mouse Model
2.9. Collection of Buccal Cells
2.10. Slot Blot Assay for Acr-dG Detection
2.11. Statistical Analyses
3. Results
3.1. Acrolein Increased Cell Proliferation, Anchorage-Independent Activity, and Cell Migration Activity in Normal Human Keratinocytes (NOK)
3.2. NOK Acr-Clone #4 Formed Tumors in Xenografts Nude Mice
3.3. Acrolein Induced EGFR Amplification and Activated EGFR Signaling Pathway in NOK
3.4. EGFR Amplification Was Associated with Acrolein-Induced DNA Damages in OSCC Patients
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Grandis, J.R.; Melhem, M.F.; Gooding, W.E.; Day, R.S.; Holst, V.A.; Wagener, M.M.; Drenning, S.D.; Tweardy, D.J. Levels of TGF-α and EGFR Protein in Head and Neck Squamous Cell Carcinoma and Patient Survival. J. Natl. Cancer Inst. 1998, 90, 824–832. [Google Scholar] [CrossRef] [Green Version]
- Sasco, A.; Lauby-Secretan, B.; Straif, K. Tobacco smoking and cancer: A brief review of recent epidemiological evidence. Lung Cancer 2004, 45 (Suppl. 2), S3–S9. [Google Scholar] [CrossRef]
- Vineis, P.; Alavanja, M.; Buffler, P.; Fontham, E.; Franceschi, S.; Gao, Y.T.; Gupta, P.C.; Hackshaw, A.; Matos, E.; Samet, J.; et al. Tobacco and Cancer: Recent Epidemiological Evidence. J. Natl. Cancer Inst. 2004, 96, 99–106. [Google Scholar] [CrossRef]
- Saxena, S.; Sankhla, B.; Sundaragiri, K.S.; Bhargava, A. A Review of Salivary Biomarker: A Tool for Early Oral Cancer Diagnosis. Adv. Biomed. Res. 2017, 6, 90. [Google Scholar] [CrossRef]
- Lai, S.Y.; Koppikar, P.; Thomas, S.M.; Childs, E.E.; Egloff, A.M.; Seethala, R.R.; Branstetter, B.F.; Gooding, W.E.; Muthukrishnan, A.; Mountz, J.M.; et al. Intratumoral Epidermal Growth Factor Receptor Antisense DNA Therapy in Head and Neck Cancer: First Human Application and Potential Antitumor Mechanisms. J. Clin. Oncol. 2009, 27, 1235–1242. [Google Scholar] [CrossRef] [Green Version]
- Rogers, S.J.; Box, C.; Chambers, P.; Barbachano, Y.; Nutting, C.M.; Rhŷs-Evans, P.; Workman, P.; Harrington, K.; Eccles, S.A. Determinants of response to epidermal growth factor receptor tyrosine kinase inhibition in squamous cell carcinoma of the head and neck. J. Pathol. 2009, 218, 122–130. [Google Scholar] [CrossRef]
- Sheu, J.J.-C.; Hua, C.-H.; Wan, L.; Lin, Y.-J.; Lai, M.-T.; Tseng, H.-C.; Jinawath, N.; Tsai, M.-H.; Chang, N.-W.; Lin, C.-F.; et al. Functional Genomic Analysis Identified Epidermal Growth Factor Receptor Activation as the Most Common Genetic Event in Oral Squamous Cell Carcinoma. Cancer Res. 2009, 69, 2568–2576. [Google Scholar] [CrossRef] [Green Version]
- Baumeister, P.; Märte, M.; Reiter, M.; Welz, C.; Schwenk-Zieger, S.; Harréus, U. EGFR and P-GP expression in oropharyngeal mucosa in relation to smoking. Anticancer. Res. 2012, 32, 1639–1648. [Google Scholar]
- Chung, C.H.; Ely, K.; McGavran, L.; Varella-Garcia, M.; Parker, J.; Parker, N.; Jarrett, C.; Carter, J.; Murphy, B.A.; Netterville, J.; et al. Increased Epidermal Growth Factor Receptor Gene Copy Number Is Associated With Poor Prognosis in Head and Neck Squamous Cell Carcinomas. J. Clin. Oncol. 2006, 24, 4170–4176. [Google Scholar] [CrossRef]
- Laimer, K.; Spizzo, G.; Gastl, G.; Obrist, P.; Brunhuber, T.; Fong, D.; Barbieri, V.; Jank, S.; Doppler, W.; Rasse, M.; et al. High EGFR expression predicts poor prognosis in patients with squamous cell carcinoma of the oral cavity and oropharynx: A TMA-based immunohistochemical analysis. Oral Oncol. 2007, 43, 193–198. [Google Scholar] [CrossRef]
- Cohen, E.R.; Reis, I.M.; Gomez, C.; Pereira, L.; Freiser, M.E.; Hoosien, G.; Franzmann, E.J. Immunohistochemistry Analysis of CD44, EGFR, and p16 in Oral Cavity and Oropharyngeal Squamous Cell Carcinoma. Otolaryngol. Neck Surg. 2017, 157, 239–251. [Google Scholar] [CrossRef]
- Vermorken, J.B.; Mesia, R.; Rivera, F.; Remenar, E.; Kawecki, A.; Rottey, S.; Erfan, J.; Zabolotnyy, D.; Kienzer, H.-R.; Cupissol, D.; et al. Platinum-Based Chemotherapy plus Cetuximab in Head and Neck Cancer. N. Engl. J. Med. 2008, 359, 1116–1127. [Google Scholar] [CrossRef] [Green Version]
- Griffin, S.; Walker, S.; Sculpher, M.; White, S.; Erhorn, S.; Brent, S.; Dyker, A.; Ferrie, L.; Gilfillan, C.; Horsley, W.; et al. Cetuximab plus radiotherapy for the treatment of locally advanced squamous cell carcinoma of the head and neck. Health Technol. Assess. 2009, 13 (Suppl. 1), 49–54. [Google Scholar] [CrossRef]
- De Mello, R.A.; Gerós, S.; Alves, M.P.; Moreira, F.; Avezedo, I.; Dinis, J. Cetuximab Plus Platinum-Based Chemotherapy in Head and Neck Squamous Cell Carcinoma: A Retrospective Study in a Single Comprehensive European Cancer Institution. PLoS ONE 2014, 9, e86697. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, F.A.P.; Noguti, J.; Oshima, C.T.F.; Ribeiro, D.A. Effective targeting of the epidermal growth factor receptor (EGFR) for treating oral cancer: A promising approach. Anticancer. Res. 2014, 34, 1547–1552. [Google Scholar]
- Gillison, M.L.; Zhang, Q.; Jordan, R.; Xiao, W.; Westra, W.H.; Trotti, A.; Spencer, S.; Harris, J.; Chung, C.H.; Ang, K.K. Tobacco Smoking and Increased Risk of Death and Progression for Patients With p16-Positive and p16-Negative Oropharyngeal Cancer. J. Clin. Oncol. 2012, 30, 2102–2111. [Google Scholar] [CrossRef]
- Kumar, B.; Cordell, K.G.; Lee, J.S.; Prince, M.E.; Tran, H.H.; Wolf, G.T.; Urba, S.G.; Worden, F.P.; Chepeha, D.B.; Teknos, T.N.; et al. Response to Therapy and Outcomes in Oropharyngeal Cancer Are Associated With Biomarkers Including Human Papillomavirus, Epidermal Growth Factor Receptor, Gender, and Smoking. Int. J. Radiat. Oncol. 2007, 69, S109–S111. [Google Scholar] [CrossRef] [Green Version]
- Sivarajah, S.; Kostiuk, M.; Lindsay, C.; Puttagunta, L.; O’Connell, D.A.; Harris, J.; Seikaly, H.; Biron, V.L. EGFR as a biomarker of smoking status and survival in oropharyngeal squamous cell carcinoma. J. Otolaryngol. Head Neck Surg. 2019, 48, 1. [Google Scholar] [CrossRef]
- Flesher, J.W.; Lehner, A.F. Structure, function and carcinogenicity of metabolites of methylated and non-methylated polycyclic aromatic hydrocarbons: A comprehensive review. Toxicol. Mech. Methods 2016, 26, 1–29. [Google Scholar] [CrossRef]
- Comes, R.M.M.; Eggleton, M. Concise International Chemical Assessment Document No 43; World Health Organization: Geneva, Switzerland, 2002. [Google Scholar]
- Hoffman, D.H.S. The Handbook of Experimental Pharmacology; Copper, C.S., Grover, P.L., Eds.; Springer: Berlin/Heidelberg, Germany, 1990; pp. 70–74. [Google Scholar]
- Fujioka, K.; Shibamoto, T. Determination of toxic carbonyl compounds in cigarette smoke. Environ. Toxicol. 2006, 21, 47–54. [Google Scholar] [CrossRef]
- Moghe, A.; Ghare, S.; Lamoreau, B.; Mohammad, M.; Barve, S.; McClain, C.; Joshi-Barve, S. Molecular Mechanisms of Acrolein Toxicity: Relevance to Human Disease. Toxicol. Sci. 2015, 143, 242–255. [Google Scholar] [CrossRef]
- Marques, M.M.; Beland, F.; Lachenmeier, D.; Phillips, D.; Chung, F.L.; Dorman, D.; Elmore, S.; Hammond, S.S.; Krstev, S.; Linhart, I.; et al. Carcinogenicity of acrolein, crotonaldehyde, and arecoline (IARC Monographs Vol 128 group). Lancet Oncol. 2021, 22, 19–20. [Google Scholar] [CrossRef]
- Chung, F.L.; Young, R.; Hecht, S.; Chung, F.L.; Young, R.; Hecht, S. Formation of cyclic 1,N2-propanodeoxyguanosine adducts in DNA upon reaction with acrolein or crotonaldehyde. Cancer Res. 1984, 44, 990–995. [Google Scholar] [PubMed]
- Wang, H.-T. Effect of Acrolein in DNA Damage, DNA Repair and Lung Carcinogenesis. Ph.D. Thesis, New York University, New York, NY, USA, 2012. [Google Scholar]
- Tang, M.-S.; Wang, H.-T.; Hu, Y.; Chen, W.-S.; Akao, M.; Feng, Z.; Hu, W. Acrolein induced DNA damage, mutagenicity and effect on DNA repair. Mol. Nutr. Food Res. 2011, 55, 1291–1300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.-W.; Wang, H.-T.; Weng, M.-W.; Chin, C.; Huang, W.; Lepor, H.; Wu, X.-R.; Rom, W.; Chen, L.C.; Tang, M.-S. Cigarette side-stream smoke lung and bladder carcinogenesis: Inducing mutagenic acrolein-DNA adducts, inhibiting DNA repair and enhancing anchorage-independent-growth cell transformation. Oncotarget 2015, 6, 33226–33236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feng, Z.; Hu, W.; Hu, Y.; Tang, M.-S. Acrolein is a major cigarette-related lung cancer agent: Preferential binding at p53 mutational hotspots and inhibition of DNA repair. Proc. Natl. Acad. Sci. USA 2006, 103, 15404–15409. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.-T.; Tsou, H.-H.; Hu, C.-H.; Liu, J.-H.; Liu, C.-J.; Lee, C.-H.; Liu, T.-Y. Acrolein is involved in the synergistic potential of cigarette smoking- and betel quid chewing-related human oral cancer. Cancer Epidemiol. Biomark. Prev. 2019, 28, 954–962. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, S.-Y.; Tu, H.-F.; Yang, C.-C.; Wu, C.-H.; Liu, C.-J.; Chang, K.-W.; Lin, S.-C. miR-134targetsPDCD7to reduce E-cadherin expression and enhance oral cancer progression. Int. J. Cancer 2018, 143, 2892–2904. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.-T.; Chen, J.-J.; Wang, H.-T. Targeting RNA Polymerase I with Hernandonine Inhibits Ribosomal RNA Synthesis and Tumor Cell Growth. Mol. Cancer Res. 2019, 17, 2294–2305. [Google Scholar] [CrossRef] [PubMed]
- Vichai, V.; Kirtikara, K. Sulforhodamine B colorimetric assay for cytotoxicity screening. Nat. Protoc. 2006, 1, 1112–1116. [Google Scholar] [CrossRef]
- Borowicz, S.; Van Scoyk, M.; Avasarala, S.; Rathinam, M.K.K.; Tauler, J.; Bikkavilli, R.K.; Winn, R.A. The Soft Agar Colony Formation Assay. J. Vis. Exp. 2014, e51998. [Google Scholar] [CrossRef] [Green Version]
- Justus, C.R.; Leffler, N.; Ruiz-Echevarria, M.; Yang, L.V. In vitro Cell Migration and Invasion Assays. J. Vis. Exp. 2014, e51046. [Google Scholar] [CrossRef] [Green Version]
- Mizuuchi, H.; Suda, K.; Murakami, I.; Sakai, K.; Sato, K.; Kobayashi, Y.; Shimoji, M.; Chiba, M.; Sesumi, Y.; Tomizawa, K.; et al. Oncogene swap as a novel mechanism of acquired resistance to epidermal growth factor receptor-tyrosine kinase inhibitor in lung cancer. Cancer Sci. 2016, 107, 461–468. [Google Scholar] [CrossRef]
- Wang, H.-T.; Hu, Y.; Tong, D.; Huang, J.; Gu, L.; Wu, X.-R.; Chung, F.-L.; Li, G.-M.; Tang, M.-S. Effect of Carcinogenic Acrolein on DNA Repair and Mutagenic Susceptibility. J. Biol. Chem. 2012, 287, 12379–12386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.-T.; Weng, M.-W.; Chen, W.-C.; Yobin, M.; Pan, J.; Chung, F.-L.; Wu, X.-R.; Rom, W.; Tang, M.-S. Effect of CpG methylation at different sequence context on acrolein- and BPDE-DNA binding and mutagenesis. Carcinogenesis 2012, 34, 220–227. [Google Scholar] [CrossRef] [Green Version]
- Pan, J.; Awoyemi, B.; Xuan, Z.; Vohra, P.; Wang, H.-T.; Dyba, M.; Greenspan, E.; Fu, Y.; Creswell, K.; Zhang, L.; et al. Detection of Acrolein-Derived Cyclic DNA Adducts in Human Cells by Monoclonal Antibodies. Chem. Res. Toxicol. 2012, 25, 2788–2795. [Google Scholar] [CrossRef] [Green Version]
- Nyati, M.K.; Morgan, M.A.; Feng, F.Y.; Lawrence, T.S. Integration of EGFR inhibitors with radiochemotherapy. Nat. Rev. Cancer 2006, 6, 876–885. [Google Scholar] [CrossRef]
- Wang, H.-T.; Zhang, S.; Hu, Y.; Tang, M.-S. Mutagenicity and Sequence Specificity of Acrolein-DNA Adducts. Chem. Res. Toxicol. 2009, 22, 511–517. [Google Scholar] [CrossRef] [Green Version]
- Kato, S.; Okamura, R.; Mareboina, M.; Lee, S.; Goodman, A.; Patel, S.P.; Fanta, P.T.; Schwab, R.B.; Vu, P.; Raymond, V.M.; et al. Revisiting Epidermal Growth Factor Receptor (EGFR) Amplification as a Target for Anti-EGFR Therapy: Analysis of Cell-Free Circulating Tumor DNA in Patients with Advanced Malignancies. JCO Precis. Oncol. 2019, 3, 1–14. [Google Scholar] [CrossRef]
- Temam, S.; Kawaguchi, H.; El-Naggar, A.K.; Jelinek, J.; Tang, H.; Liu, D.D.; Lang, W.; Issa, J.-P.; Lee, J.J.; Mao, L. Epidermal Growth Factor Receptor Copy Number Alterations Correlate with Poor Clinical Outcome in Patients With Head and Neck Squamous Cancer. J. Clin. Oncol. 2007, 25, 2164–2170. [Google Scholar] [CrossRef]





| Characteristic | Count (%) |
|---|---|
| Age (y) | |
| mean ± SD | 56.2 ± 11.7 |
| range | 42–89 |
| Sex (n (%)) | |
| Male | 17 (94.4) |
| Female | 1 (5.6) |
| Cancer location (n (%)) | |
| Tongue | 4 (22.2) |
| Buccal mucosa | 4 (22.2) |
| Gingiva | 4 (22.2) |
| Hard palate | 2 (11.1) |
| Retromolar trigone | 2 (11.1) |
| Oropharynx | 2 (11.1) |
| Clinical stage (n (%)) ^ | |
| Stage I | 2 (11.1) |
| Stage II | 4 (22.2) |
| Stage III | 1 (5.6) |
| Stage IV | 11 (61.1) |
| Cigarette smoker (n (%)) # | 17 (94.4) |
| Alcohol drinker(n (%))# | 16 (33.3) |
| Betel quid chewer (n (%))# | 17 (94.4) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsou, H.-H.; Tsai, H.-C.; Chu, C.-T.; Cheng, H.-W.; Liu, C.-J.; Lee, C.-H.; Liu, T.-Y.; Wang, H.-T. Cigarette Smoke Containing Acrolein Upregulates EGFR Signaling Contributing to Oral Tumorigenesis In Vitro and In Vivo. Cancers 2021, 13, 3544. https://doi.org/10.3390/cancers13143544
Tsou H-H, Tsai H-C, Chu C-T, Cheng H-W, Liu C-J, Lee C-H, Liu T-Y, Wang H-T. Cigarette Smoke Containing Acrolein Upregulates EGFR Signaling Contributing to Oral Tumorigenesis In Vitro and In Vivo. Cancers. 2021; 13(14):3544. https://doi.org/10.3390/cancers13143544
Chicago/Turabian StyleTsou, Han-Hsing, Hong-Chieh Tsai, Chiao-Ting Chu, Hsiao-Wei Cheng, Chung-Ji Liu, Chien-Hung Lee, Tsung-Yun Liu, and Hsiang-Tsui Wang. 2021. "Cigarette Smoke Containing Acrolein Upregulates EGFR Signaling Contributing to Oral Tumorigenesis In Vitro and In Vivo" Cancers 13, no. 14: 3544. https://doi.org/10.3390/cancers13143544