Emerging Role of Hydrogels in Drug Delivery Systems, Tissue Engineering and Wound Management
Abstract
:1. Introduction
2. Stimuli-Sensitive Hydrogels
2.1. Thermoresponsive
2.2. pH Sensitive
2.3. Photoresponsive
2.4. Electroresponsive
2.5. Ultrasonically Responsive
2.6. Magnetoresponsive
2.7. Enzyme Responsive
2.8. Ionic Strength Responsive
2.9. Inflammation Responsive
3. Functionalization Strategies of Hydrogels
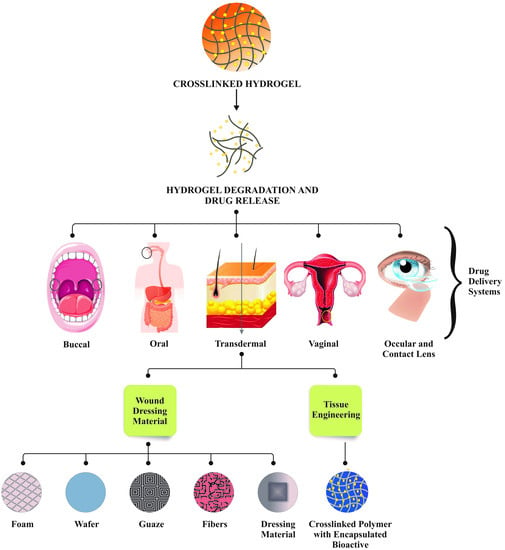
4. Role of Hydrogels in Drug Delivery Systems, Tissue Engineering, and Wound Healing
5. Drug Delivery Systems
5.1. Self-Assembled Nanoparticle System
5.2. Buccal Delivery
5.3. Oral Delivery
5.4. Transdermal Drug Delivery
5.5. Vaginal Drug Delivery
5.6. Ocular Delivery
5.7. Contact Lens
5.8. Wound Dressing
6. Tissue Engineering
6.1. Polymers
6.1.1. Collagen
6.1.2. Alginates
6.1.3. Hyaluronic Acid
7. Future Perspectives and Challenges
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tavakoli, J.; Wang, J.; Chuah, C.; Tang, Y. Natural-based Hydrogels: A Journey from Simple to Smart Networks for Medical Examination. Curr. Med. Chem. 2020, 27, 2704–2733. [Google Scholar] [CrossRef] [PubMed]
- Caló, E.; Khutoryanskiy, V.V. Biomedical applications of hydrogels: A review of patents and commercial products. Eur. Polym. J. 2015, 65, 252–267. [Google Scholar] [CrossRef] [Green Version]
- Okay, O. Self-Healing Hydrogels Formed via Hydrophobic Interactions. In Supramolecular Polymer Networks and Gels; Seiffert, S., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 101–142. [Google Scholar] [CrossRef]
- Lin, J.; Zheng, S.Y.; Xiao, R.; Yin, J.; Wu, Z.L.; Zheng, Q.; Qian, J. Constitutive behaviors of tough physical hydrogels with dynamic metal-coordinated bonds. J. Mech. Phys. Solids 2020, 139, 103935. [Google Scholar] [CrossRef]
- Hennink, W.E.; van Nostrum, C.F. Novel crosslinking methods to design hydrogels. Adv. Drug Deliv. Rev. 2002, 54, 13–36. [Google Scholar] [CrossRef]
- Ahmed, E.M. Hydrogel: Preparation, characterization, and applications: A review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiu, X.; Hu, S. “Smart” Materials Based on Cellulose: A Review of the Preparations, Properties, and Applications. Materials 2013, 6, 738–781. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, S. Encyclopedia of Polymeric Nanomaterials; Springer: Berlin/Heidelberg, Germany, 2015. [Google Scholar]
- Said, H.M.; Abd Alla, S.G.; El-Naggar, A.W.M. Synthesis and characterization of novel gels based on carboxymethyl cellulose/acrylic acid prepared by electron beam irradiation. React. Funct. Polym. 2004, 61, 397–404. [Google Scholar] [CrossRef]
- Zhang, L.; Ren, X.; Zhang, Y.; Zhang, K. Step-Growth Polymerization Method for Ultrahigh Molecular Weight Polymers. ACS Macro Lett. 2019, 8, 948–954. [Google Scholar] [CrossRef]
- Bajpai, A.K.; Shukla, S.K.; Bhanu, S.; Kankane, S. Responsive polymers in controlled drug delivery. Prog. Polym. Sci. 2008, 33, 1088–1118. [Google Scholar] [CrossRef]
- Schmaljohann, D. Thermo- and pH-responsive polymers in drug delivery. Adv. Drug Deliv. Rev. 2006, 58, 1655–1670. [Google Scholar] [CrossRef] [PubMed]
- Wells, L.A.; Lasowski, F.; Fitzpatrick, S.D.; Sheardown, H. Responding to change: Thermo- and photo-responsive polymers as unique biomaterials. Crit. Rev. Biomed. Eng. 2010, 38, 487–509. [Google Scholar] [CrossRef]
- Murdan, S. Electro-responsive drug delivery from hydrogels. J. Control. Release 2003, 92, 1–17. [Google Scholar] [CrossRef]
- Zardad, A.Z.; Choonara, Y.E.; Du Toit, L.C.; Kumar, P.; Mabrouk, M.; Kondiah, P.P.D.; Pillay, V. A Review of Thermo- and Ultrasound-Responsive Polymeric Systems for Delivery of Chemotherapeutic Agents. Polymers 2016, 8, 359. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Huang, G.; Zhang, X.; Li, B.; Chen, Y.; Lu, T.; Lu, T.J.; Xu, F. Magnetic Hydrogels and Their Potential Biomedical Applications. Adv. Funct. Mater. 2013, 23, 660–672. [Google Scholar] [CrossRef]
- Sharifzadeh, G.; Hosseinkhani, H. Biomolecule-Responsive Hydrogels in Medicine. Adv. Healthc. Mater. 2017, 6. [Google Scholar] [CrossRef]
- Erol, O.; Pantula, A.; Liu, W.; Gracias, D.H. Transformer Hydrogels: A Review. Adv. Mater. Technol. 2019, 4, 1900043. [Google Scholar] [CrossRef] [Green Version]
- Koh, R.H.; Jin, Y.; Kim, J.; Hwang, N.S. Inflammation-Modulating Hydrogels for Osteoarthritis Cartilage Tissue Engineering. Cells 2020, 9, 419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Overstreet, D.J.; McLemore, R.Y.; Doan, B.D.; Farag, A.; Vernon, B.L. Temperature-Responsive Graft Copolymer Hydrogels for Controlled Swelling and Drug Delivery. Soft Mater. 2013, 11, 294–304. [Google Scholar] [CrossRef]
- Park, M.H.; Joo, M.K.; Choi, B.G.; Jeong, B. Biodegradable thermogels. Acc. Chem. Res. 2012, 45, 424–433. [Google Scholar] [CrossRef]
- Klouda, L. Thermoresponsive hydrogels in biomedical applications: A seven-year update. Eur. J. Pharm. Biopharm. 2015, 97, 338–349. [Google Scholar] [CrossRef]
- Bawa, P.; Pillay, V.; Choonara, Y.E.; du Toit, L.C. Stimuli-responsive polymers and their applications in drug delivery. Biomed. Mater. 2009, 4, 022001. [Google Scholar] [CrossRef] [PubMed]
- Schoener, C.A.; Hutson, H.N.; Peppas, N.A. pH-responsive hydrogels with dispersed hydrophobic nanoparticles for the oral delivery of chemotherapeutics. J. Biomed. Mater. Res. A 2013, 101, 2229–2236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hendi, A.; Umair Hassan, M.; Elsherif, M.; Alqattan, B.; Park, S.; Yetisen, A.K.; Butt, H. Healthcare Applications of pH-Sensitive Hydrogel-Based Devices: A Review. Int. J. Nanomed. 2020, 15, 3887–3901. [Google Scholar] [CrossRef]
- Ho, D.K.; Nguyen, D.T.; Thambi, T.; Lee, D.S.; Huynh, D.P. Polyamide-based pH and temperature-responsive hydrogels: Synthesis and physicochemical characterization. J. Polym. Res. 2018, 26, 7. [Google Scholar] [CrossRef]
- Nguyen, D.T.; Phan, V.H.G.; Lee, D.S.; Thambi, T.; Huynh, D.P. Bioresorbable pH- and temperature-responsive injectable hydrogels-incorporating electrosprayed particles for the sustained release of insulin. Polym. Degrad. Stab. 2019, 162, 36–46. [Google Scholar] [CrossRef]
- Peng, K.; Tomatsu, I.; Kros, A. Light controlled protein release from a supramolecular hydrogel. Chem. Commun. 2010, 46, 4094–4096. [Google Scholar] [CrossRef]
- Mantha, S.; Pillai, S.; Khayambashi, P.; Upadhyay, A.; Zhang, Y.; Tao, O.; Pham, H.M.; Tran, S.D. Smart Hydrogels in Tissue Engineering and Regenerative Medicine. Materials 2019, 12, 3323. [Google Scholar] [CrossRef] [Green Version]
- Lo, C.-W.; Zhu, D.; Jiang, H. An infrared-light responsive graphene-oxide incorporated poly(N-isopropylacrylamide) hydrogel nanocomposite. Soft Matter 2011, 7, 5604–5609. [Google Scholar] [CrossRef]
- Servant, A.; Methven, L.; Williams, R.P.; Kostarelos, K. Electroresponsive polymer-carbon nanotube hydrogel hybrids for pulsatile drug delivery in vivo. Adv. Healthc. Mater. 2013, 2, 806–811. [Google Scholar] [CrossRef]
- Rahimi, N.; Swennen, G.; Verbruggen, S.; Scibiorek, M.; Molin, D.G.; Post, M.J. Short stimulation of electro-responsive PAA/fibrin hydrogel induces collagen production. Tissue Eng. Part C Methods 2014, 20, 703–713. [Google Scholar] [CrossRef] [Green Version]
- Miller, D.L.; Smith, N.B.; Bailey, M.R.; Czarnota, G.J.; Hynynen, K.; Makin, I.R. Overview of therapeutic ultrasound applications and safety considerations. J. Ultrasound Med. 2012, 31, 623–634. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Kohane, D.S. External triggering and triggered targeting strategies for drug delivery. Nat. Rev. Mater. 2017, 2, 17020. [Google Scholar] [CrossRef]
- Li, J.; Mooney, D.J. Designing hydrogels for controlled drug delivery. Nat. Rev. Mater. 2016, 1, 16071. [Google Scholar] [CrossRef]
- Adedoyin, A.A.; Ekenseair, A.K. Biomedical applications of magneto-responsive scaffolds. Nano Res. 2018, 11, 5049–5064. [Google Scholar] [CrossRef]
- Wang, C.; Fischer, A.; Ehrlich, A.; Nahmias, Y.; Willner, I. Biocatalytic reversible control of the stiffness of DNA-modified responsive hydrogels: Applications in shape-memory, self-healing and autonomous controlled release of insulin. Chem. Sci. 2020, 11, 4516–4524. [Google Scholar] [CrossRef] [Green Version]
- Stealey, S.; Guo, X.; Majewski, R.; Dyble, A.; Lehman, K.; Wedemeyer, M.; Steeber, D.A.; Kaltchev, M.G.; Chen, J.; Zhang, W. Calcium-oligochitosan-pectin microcarrier for colonic drug delivery. Pharm. Dev. Technol. 2020, 25, 260–265. [Google Scholar] [CrossRef]
- Palena, M.C.; García, M.C.; Manzo, R.H.; Jimenez-Kairuz, A.F. Self-organized drug-interpolyelectrolyte nanocomplexes loaded with anionic drugs. Characterization and in vitro release evaluation. J. Drug Deliv. Sci. Technol. 2015, 30, 45–53. [Google Scholar] [CrossRef]
- García, M.C.; Martinelli, M.; Ponce, N.E.; Sanmarco, L.M.; Aoki, M.P.; Manzo, R.H.; Jimenez-Kairuz, A.F. Multi-kinetic release of benznidazole-loaded multiparticulate drug delivery systems based on polymethacrylate interpolyelectrolyte complexes. Eur. J. Pharm. Sci. 2018, 120, 107–122. [Google Scholar] [CrossRef]
- Hu, X.; Zhang, Y.; Xie, Z.; Jing, X.; Bellotti, A.; Gu, Z. Stimuli-Responsive Polymersomes for Biomedical Applications. Biomacromolecules 2017, 18, 649–673. [Google Scholar] [CrossRef]
- Chang, P.C.; Chao, Y.C.; Hsiao, M.H.; Chou, H.S.; Jheng, Y.H.; Yu, X.H.; Lee, N.; Yang, C.; Liu, D.M. Inhibition of Periodontitis Induction Using a Stimuli-Responsive Hydrogel Carrying Naringin. J. Periodontol. 2017, 88, 190–196. [Google Scholar] [CrossRef]
- Murakami, T.; Schmidt, B.V.K.J.; Brown, H.R.; Hawker, C.J. One-Pot “Click” Fabrication of Slide-Ring Gels. Macromolecules 2015, 48, 7774–7781. [Google Scholar] [CrossRef]
- Kim, S.J.; Yoon, S.G.; Lee, Y.H.; Kim, S.I. Bending behavior of hydrogels composed of poly(methacrylic acid) and alginate by electrical stimulus. Polym. Int. 2004, 53, 1456–1460. [Google Scholar] [CrossRef]
- Lee, S.B.; Park, E.K.; Lim, Y.M.; Cho, S.K.; Kim, S.Y.; Lee, Y.M.; Nho, Y.C. Preparation of alginate/poly(N-isopropylacrylamide) semi-interpenetrating and fully interpenetrating polymer network hydrogels with γ-ray irradiation and their swelling behaviors. J. Appl. Polym. Sci. 2006, 100, 4439–4446. [Google Scholar] [CrossRef]
- Holten-Andersen, N. Bio-Inspired Composite Interfaces: Controlling Hydrogel Mechanics via Polymer-Nanoparticle Coordination Bond Dynamics. In Proceedings of the APS March Meeting Abstracts, Austin, TX, USA, 2–6 March 2015; p. D48.006. [Google Scholar]
- Anroop, B.N.; Mohamed, A.M.; Pottathil, S.; Sabna, K.; Mithra, C.; Abu, T. Advances of Non-iron Metal Nanoparticles in Biomedicine. J. Pharm. Pharm. Sci. 2021, 24, 41–61. [Google Scholar] [CrossRef]
- Yadollahi, M.; Gholamali, I.; Namazi, H.; Aghazadeh, M. Synthesis and characterization of antibacterial carboxymethyl cellulose/ZnO nanocomposite hydrogels. Int. J. Biol. Macromol. 2015, 74, 136–141. [Google Scholar] [CrossRef]
- Cui, Z.K.; Kim, S.; Baljon, J.J.; Wu, B.M.; Aghaloo, T.; Lee, M. Microporous methacrylated glycol chitosan-montmorillonite nanocomposite hydrogel for bone tissue engineering. Nat. Commun. 2019, 10, 3523. [Google Scholar] [CrossRef] [Green Version]
- Zheng, H.; Yoshitomi, T.; Yoshimoto, K. Analysis of Chirality Effects on Stem Cell Fate Using Three-dimensional Fibrous Peptide Hydrogels. ACS Appl. Bio Mater. 2018, 1, 538–543. [Google Scholar] [CrossRef]
- Motealleh, A.; Hermes, H.; Jose, J.; Kehr, N.S. Chirality-dependent cell adhesion and enrichment in Janus nanocomposite hydrogels. Nanomedicine 2018, 14, 247–256. [Google Scholar] [CrossRef]
- Xiao, G.; Wang, Y.; Zhang, H.; Chen, L.; Fu, S. Facile strategy to construct a self-healing and biocompatible cellulose nanocomposite hydrogel via reversible acylhydrazone. Carbohydr. Polym. 2019, 218, 68–77. [Google Scholar] [CrossRef]
- Kaur, G.; Adhikari, R.; Cass, P.; Bown, M.; Gunatillake, P. Electrically conductive polymers and composites for biomedical applications. RSC Adv. 2015, 5, 37553–37567. [Google Scholar] [CrossRef]
- Yom-Tov, O.; Neufeld, L.; Seliktar, D.; Bianco-Peled, H. A novel design of injectable porous hydrogels with in situ pore formation. Acta Biomater. 2014, 10, 4236–4246. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, J.; Zhang, J.; Li, S.; Zheng, L.; Zhang, Y.; Meng, H.; Zhang, X.; Wu, Z. A bio-inspired injectable hydrogel as a cell platform for real-time glycaemic regulation. J. Mater. Chem. B 2020, 8, 4627–4641. [Google Scholar] [CrossRef]
- Barker, K.; Rastogi, S.K.; Dominguez, J.; Cantu, T.; Brittain, W.; Irvin, J.; Betancourt, T. Biodegradable DNA-enabled poly(ethylene glycol) hydrogels prepared by copper-free click chemistry. J. Biomater. Sci. Polym. Ed. 2016, 27, 22–39. [Google Scholar] [CrossRef]
- Tóth, I.Y.; Veress, G.; Szekeres, M.; Illés, E.; Tombácz, E. Magnetic hyaluronate hydrogels: Preparation and characterization. J. Magn. Magn. Mater. 2015, 380, 175–180. [Google Scholar] [CrossRef] [Green Version]
- Song, J.; Hwang, S.; Im, K.; Hur, J.; Nam, J.; Hwang, S.; Ahn, G.O.; Kim, S.; Park, N. Light-responsible DNA hydrogel-gold nanoparticle assembly for synergistic cancer therapy. J. Mater. Chem. B 2015, 3, 1537–1543. [Google Scholar] [CrossRef]
- Um, S.H.; Lee, J.B.; Park, N.; Kwon, S.Y.; Umbach, C.C.; Luo, D. Enzyme-catalysed assembly of DNA hydrogel. Nat. Mater. 2006, 5, 797–801. [Google Scholar] [CrossRef]
- Gačanin, J.; Synatschke, C.V.; Weil, T. Biomedical Applications of DNA-Based Hydrogels. Adv. Funct. Mater. 2020, 30, 1906253. [Google Scholar] [CrossRef] [Green Version]
- Ankareddi, I.; Brazel, C.S. Synthesis and characterization of grafted thermosensitive hydrogels for heating activated controlled release. Int. J. Pharm. 2007, 336, 241–247. [Google Scholar] [CrossRef]
- Zahedi, P.; Lee, P.I. Solid molecular dispersions of poorly water-soluble drugs in poly(2-hydroxyethyl methacrylate) hydrogels. Eur. J. Pharm. Biopharm. 2007, 65, 320–328. [Google Scholar] [CrossRef]
- Lin, Y.; Sun, J.; Jiang, G.; Zan, J.; Ding, F. In vitro Evaluation of Lysozyme-loaded Microspheres in Thermosensitive Methylcellulose-based Hydrogel1 1Supported by the National Natural Science Foundation of China (No.20576057) and Fundamental Research Foundation of Tsinghua University (JCqn2005033). Chin. J. Chem. Eng. 2007, 15, 566–572. [Google Scholar] [CrossRef]
- Lalatsa, A.; Statts, L.; Adriana de Jesus, J.; Adewusi, O.; Auxiliadora Dea-Ayuela, M.; Bolas-Fernandez, F.; Dalastra Laurenti, M.; Felipe Domingues Passero, L.; Serrano, D.R. Topical buparvaquone nano-enabled hydrogels for cutaneous leishmaniasis. Int. J. Pharm. 2020, 588, 119734. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.; Sharma, A.K.; Kumar, P. Nanoscale Self-Assembly for Therapeutic Delivery. Front. Bioeng. Biotechnol. 2020, 8. [Google Scholar] [CrossRef]
- Dunn, S.S.; Tian, S.; Blake, S.; Wang, J.; Galloway, A.L.; Murphy, A.; Pohlhaus, P.D.; Rolland, J.P.; Napier, M.E.; DeSimone, J.M. Reductively responsive siRNA-conjugated hydrogel nanoparticles for gene silencing. J. Am. Chem. Soc. 2012, 134, 7423–7430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elzoghby, A.O. Gelatin-based nanoparticles as drug and gene delivery systems: Reviewing three decades of research. J. Control. Release 2013, 172, 1075–1091. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Yang, W.; Wang, C.; Hu, J.; Fu, S. Chitosan nanoparticles as a novel delivery system for ammonium glycyrrhizinate. Int. J. Pharm. 2005, 295, 235–245. [Google Scholar] [CrossRef]
- Sarmento, B.; Ribeiro, A.; Veiga, F.; Sampaio, P.; Neufeld, R.; Ferreira, D. Alginate/chitosan nanoparticles are effective for oral insulin delivery. Pharm. Res. 2007, 24, 2198–2206. [Google Scholar] [CrossRef] [Green Version]
- Kaul, G.; Amiji, M. Long-circulating poly(ethylene glycol)-modified gelatin nanoparticles for intracellular delivery. Pharm. Res. 2002, 19, 1061–1067. [Google Scholar] [CrossRef]
- Ding, D.; Zhu, Z.; Liu, Q.; Wang, J.; Hu, Y.; Jiang, X.; Liu, B. Cisplatin-loaded gelatin-poly(acrylic acid) nanoparticles: Synthesis, antitumor efficiency in vivo and penetration in tumors. Eur. J. Pharm. Biopharm. 2011, 79, 142–149. [Google Scholar] [CrossRef]
- Jain, S.; Valvi, P.U.; Swarnakar, N.K.; Thanki, K. Gelatin coated hybrid lipid nanoparticles for oral delivery of amphotericin B. Mol. Pharm. 2012, 9, 2542–2553. [Google Scholar] [CrossRef]
- Yılmaz, H.; Sanlıer, S.H. Preparation of magnetic gelatin nanoparticles and investigating the possible use as chemotherapeutic agent. Artif. Cells Nanomed. Biotechnol. 2013, 41, 69–77. [Google Scholar] [CrossRef]
- Vashist, A.; Kaushik, A.; Vashist, A.; Bala, J.; Nikkhah-Moshaie, R.; Sagar, V.; Nair, M. Nanogels as potential drug nanocarriers for CNS drug delivery. Drug Discov. Today 2018, 23, 1436–1443. [Google Scholar] [CrossRef]
- Lv, Q.; He, C.; Quan, F.; Yu, S.; Chen, X. DOX/IL-2/IFN-γ co-loaded thermo-sensitive polypeptide hydrogel for efficient melanoma treatment. Bioact. Mater. 2017, 3, 118–128. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.B.; Shah, J.; Al-Dhubiab, B.E.; Patel, S.S.; Morsy, M.A.; Patel, V.; Chavda, V.; Jacob, S.; Sreeharsha, N.; Shinu, P.; et al. Development of Asialoglycoprotein Receptor-Targeted Nanoparticles for Selective Delivery of Gemcitabine to Hepatocellular Carcinoma. Molecules 2019, 24, 4566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goodall, S.; Jones, M.L.; Mahler, S. Monoclonal antibody-targeted polymeric nanoparticles for cancer therapy—Future prospects. J. Chem. Technol. Biotechnol. 2015, 90, 1169–1176. [Google Scholar] [CrossRef]
- Zhang, Y.; Fan, W.; Wang, K.; Wei, H.; Zhang, R.; Wu, Y. Novel preparation of Au nanoparticles loaded Laponite nanoparticles/ECM injectable hydrogel on cardiac differentiation of resident cardiac stem cells to cardiomyocytes. J. Photochem. Photobiol. B 2019, 192, 49–54. [Google Scholar] [CrossRef]
- Bakarich, S.E.; Gorkin, R., 3rd; in het Panhuis, M.; Spinks, G.M. 4D Printing with Mechanically Robust, Thermally Actuating Hydrogels. Macromol. Rapid Commun. 2015, 36, 1211–1217. [Google Scholar] [CrossRef]
- Nair, A.B.; Al-Dhubiab, B.E.; Shah, J.; Jacob, S.; Saraiya, V.; Attimarad, M.; SreeHarsha, N.; Akrawi, S.H.; Shehata, T.M. Mucoadhesive buccal film of almotriptan improved therapeutic delivery in rabbit model. Saudi Pharm. J. 2020, 28, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Laffleur, F. Mucoadhesive polymers for buccal drug delivery. Drug Dev. Ind. Pharm. 2014, 40, 591–598. [Google Scholar] [CrossRef]
- Fonseca-Santos, B.; Chorilli, M. An overview of polymeric dosage forms in buccal drug delivery: State of art, design of formulations and their in vivo performance evaluation. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 86, 129–143. [Google Scholar] [CrossRef] [Green Version]
- Al-Dhubiab, B.E.; Nair, A.B.; Kumria, R.; Attimarad, M.; Harsha, S. Development and evaluation of buccal films impregnated with selegiline-loaded nanospheres. Drug Deliv. 2016, 23, 2154–2162. [Google Scholar] [CrossRef] [Green Version]
- Kumria, R.; Al-Dhubiab, B.E.; Shah, J.; Nair, A.B. Formulation and Evaluation of Chitosan-Based Buccal Bioadhesive Films of Zolmitriptan. J. Pharm. Innov. 2018, 13, 133–143. [Google Scholar] [CrossRef]
- Kumria, R.; Nair, A.B.; Goomber, G.; Gupta, S. Buccal films of prednisolone with enhanced bioavailability. Drug Deliv. 2016, 23, 471–478. [Google Scholar] [CrossRef] [Green Version]
- Macedo, A.S.; Castro, P.M.; Roque, L.; Thomé, N.G.; Reis, C.P.; Pintado, M.E.; Fonte, P. Novel and revisited approaches in nanoparticle systems for buccal drug delivery. J. Control. Release 2020, 320, 125–141. [Google Scholar] [CrossRef] [PubMed]
- Laffleur, F.; Egeling, M. Evaluation of cellulose based patches for oral mucosal impairment. J. Drug Deliv. Sci. Technol. 2020, 58, 101839. [Google Scholar] [CrossRef]
- Sani, E.S.; Lara, R.P.; Aldawood, Z.; Bassir, S.H.; Nguyen, D.; Kantarci, A.; Intini, G.; Annabi, N. An Antimicrobial Dental Light Curable Bioadhesive Hydrogel for Treatment of Peri-Implant Diseases. Matter 2019, 1, 926–944. [Google Scholar] [CrossRef] [Green Version]
- Sharpe, L.A.; Daily, A.M.; Horava, S.D.; Peppas, N.A. Therapeutic applications of hydrogels in oral drug delivery. Expert Opin. Drug Deliv. 2014, 11, 901–915. [Google Scholar] [CrossRef] [Green Version]
- Xu, J.; Strandman, S.; Zhu, J.X.; Barralet, J.; Cerruti, M. Genipin-crosslinked catechol-chitosan mucoadhesive hydrogels for buccal drug delivery. Biomaterials 2015, 37, 395–404. [Google Scholar] [CrossRef]
- Liu, Z.; Wei, J.; Faraj, Y.; Ju, X.J.; Xie, R.; Wang, W.; Chu, L.Y. Smart hydrogels: Network design and emerging applications. Can. J. Chem. Eng. 2018, 96, 2100–2114. [Google Scholar] [CrossRef]
- Liu, L.; Yao, W.; Rao, Y.; Lu, X.; Gao, J. pH-Responsive carriers for oral drug delivery: Challenges and opportunities of current platforms. Drug Deliv. 2017, 24, 569–581. [Google Scholar] [CrossRef] [Green Version]
- Liechty, W.B.; Caldorera-Moore, M.; Phillips, M.A.; Schoener, C.; Peppas, N.A. Advanced molecular design of biopolymers for transmucosal and intracellular delivery of chemotherapeutic agents and biological therapeutics. J. Control. Release 2011, 155, 119–127. [Google Scholar] [CrossRef] [Green Version]
- Mastropietro, D.J.; Omidian, H.; Park, K. Drug delivery applications for superporous hydrogels. Expert Opin. Drug Deliv. 2012, 9, 71–89. [Google Scholar] [CrossRef]
- Dalmoro, A.; Sitenkov, A.Y.; Cascone, S.; Lamberti, G.; Barba, A.A.; Moustafine, R.I. Hydrophilic drug encapsulation in shell-core microcarriers by two stage polyelectrolyte complexation method. Int. J. Pharm. 2017, 518, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Schoener, C.A.; Hutson, H.N.; Peppas, N.A. pH-Responsive Hydrogels with Dispersed Hydrophobic Nanoparticles for the Delivery of Hydrophobic Therapeutic Agents. Polym. Int. 2012, 61, 874–879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harsha, S.N.; Aldhubiab, B.E.; Nair, A.B.; Alhaider, I.A.; Attimarad, M.; Venugopala, K.N.; Srinivasan, S.; Gangadhar, N.; Asif, A.H. Nanoparticle formulation by Büchi b-90 nano spray dryer for oral mucoadhesion. Drug Des. Dev. Ther. 2015, 9, 273–282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, X.; Miao, L.; Wang, X.; Tian, H. Design and evaluation of pH-responsive hydrogel for oral delivery of amifostine and study on its radioprotective effects. Colloids Surf. B Biointerfaces 2020, 195, 111200. [Google Scholar] [CrossRef]
- Leong, K.H.; Chung, L.Y.; Noordin, M.I.; Onuki, Y.; Morishita, M.; Takayama, K. Lectin-functionalized carboxymethylated kappa-carrageenan microparticles for oral insulin delivery. Carbohydr. Polym. 2011, 86, 555–565. [Google Scholar] [CrossRef]
- Fiorica, C.; Pitarresi, G.; Palumbo, F.S.; Di Stefano, M.; Calascibetta, F.; Giammona, G. A new hyaluronic acid pH sensitive derivative obtained by ATRP for potential oral administration of proteins. Int. J. Pharm. 2013, 457, 150–157. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, M.; Narain, R. Intracellular delivery of DNA and enzyme in active form using degradable carbohydrate-based nanogels. Mol. Pharm. 2012, 9, 3160–3170. [Google Scholar] [CrossRef]
- Jalababu, R.; Rao, K.S.V.K.; Rao, B.S.; Reddy, K.V.N.S. Dual responsive GG-g-PNPA/PIPAM based novel hydrogels for the controlled release of anti- cancer agent and their swelling and release kinetics. J. Polym. Res. 2020, 27, 83. [Google Scholar] [CrossRef]
- Brown, T.D.; Whitehead, K.A.; Mitragotri, S. Materials for oral delivery of proteins and peptides. Nat. Rev. Mater. 2020, 5, 127–148. [Google Scholar] [CrossRef]
- Jacob, S.; Nair, A.B.; Shah, J. Emerging role of nanosuspensions in drug delivery systems. Biomater. Res. 2020, 24, 3. [Google Scholar] [CrossRef] [Green Version]
- Nair, A.; Reddy, C.; Jacob, S. Delivery of a classical antihypertensive agent through the skin by chemical enhancers and iontophoresis. Ski. Res. Technol. 2009, 15, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.B.; Kiran Vaka, S.R.; Gupta, S.; Repka, M.A.; Murthy, S.N. In vitro and in vivo evaluation of a hydrogel-based prototype transdermal patch system of alfuzosin hydrochloride. Pharm. Dev. Technol. 2012, 17, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Jacob, S.; Nair, A.B.; Al-Dhubiab, B.E. Preparation and evaluation of niosome gel containing acyclovir for enhanced dermal deposition. J. Liposome Res. 2017, 27, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, R.F.; Singh, T.R.; Garland, M.J.; Migalska, K.; Majithiya, R.; McCrudden, C.M.; Kole, P.L.; Mahmood, T.M.; McCarthy, H.O.; Woolfson, A.D. Hydrogel-Forming Microneedle Arrays for Enhanced Transdermal Drug Delivery. Adv. Funct. Mater. 2012, 22, 4879–4890. [Google Scholar] [CrossRef] [Green Version]
- Migdadi, E.M.; Courtenay, A.J.; Tekko, I.A.; McCrudden, M.T.C.; Kearney, M.C.; McAlister, E.; McCarthy, H.O.; Donnelly, R.F. Hydrogel-forming microneedles enhance transdermal delivery of metformin hydrochloride. J. Control. Release 2018, 285, 142–151. [Google Scholar] [CrossRef]
- Alvarez-Figueroa, M.J.; Blanco-Méndez, J. Transdermal delivery of methotrexate: Iontophoretic delivery from hydrogels and passive delivery from microemulsions. Int. J. Pharm. 2001, 215, 57–65. [Google Scholar] [CrossRef]
- Fang, J.Y.; Sung, K.C.; Wang, J.J.; Chu, C.C.; Chen, K.T. The effects of iontophoresis and electroporation on transdermal delivery of buprenorphine from solutions and hydrogels. J. Pharm. Pharmacol. 2002, 54, 1329–1337. [Google Scholar] [CrossRef]
- An, Y.H.; Lee, J.; Son, D.U.; Kang, D.H.; Park, M.J.; Cho, K.W.; Kim, S.; Kim, S.H.; Ko, J.; Jang, M.H.; et al. Facilitated Transdermal Drug Delivery Using Nanocarriers-Embedded Electroconductive Hydrogel Coupled with Reverse Electrodialysis-Driven Iontophoresis. ACS Nano 2020, 14, 4523–4535. [Google Scholar] [CrossRef]
- Nair, A.B.; Shah, J.; Aljaeid, B.M.; Al-Dhubiab, B.E.; Jacob, S. Gellan Gum-Based Hydrogel for the Transdermal Delivery of Nebivolol: Optimization and Evaluation. Polymers 2019, 11, 1699. [Google Scholar] [CrossRef] [Green Version]
- Jiang, T.; Wang, T.; Li, T.; Ma, Y.; Shen, S.; He, B.; Mo, R. Enhanced Transdermal Drug Delivery by Transfersome-Embedded Oligopeptide Hydrogel for Topical Chemotherapy of Melanoma. ACS Nano 2018, 12, 9693–9701. [Google Scholar] [CrossRef]
- Ibrahim, M.M.; Nair, A.B.; Aldhubiab, B.E.; Shehata, T.M. Hydrogels and Their Combination with Liposomes, Niosomes, or Transfersomes for Dermal and Transdermal Drug Delivery. In Liposomes; InTech: London, UK, 2017; pp. 155–186. [Google Scholar]
- Lalatsa, A.; Emeriewen, K.; Protopsalti, V.; Skelton, G.; Saleh, G.M. Developing transcutaneous nanoenabled anaesthetics for eyelid surgery. Br. J. Ophthalmol. 2016, 100, 871–876. [Google Scholar] [CrossRef] [Green Version]
- Yuan, L.; Pan, M.; Lei, M.; Zhou, X.; Hu, D.; Liu, Q.; Chen, Y.; Li, W.; Qian, Z. A novel composite of micelles and hydrogel for improving skin delivery of hydrocortisone and application in atopic dermatitis therapy. Appl. Mater. Today 2020, 19, 100593. [Google Scholar] [CrossRef]
- Qindeel, M.; Ahmed, N.; Sabir, F.; Khan, S.; Ur-Rehman, A. Development of novel pH-sensitive nanoparticles loaded hydrogel for transdermal drug delivery. Drug Dev. Ind. Pharm. 2019, 45, 629–641. [Google Scholar] [CrossRef]
- Dos Santos, A.M.; Carvalho, S.G.; Araujo, V.H.S.; Carvalho, G.C.; Gremião, M.P.D.; Chorilli, M. Recent advances in hydrogels as strategy for drug delivery intended to vaginal infections. Int. J. Pharm. 2020, 590, 119867. [Google Scholar] [CrossRef] [PubMed]
- Abruzzo, A.; Bigucci, F.; Cerchiara, T.; Saladini, B.; Gallucci, M.C.; Cruciani, F.; Vitali, B.; Luppi, B. Chitosan/alginate complexes for vaginal delivery of chlorhexidine digluconate. Carbohydr. Polym. 2013, 91, 651–658. [Google Scholar] [CrossRef]
- Singh, V.K.; Anis, A.; Banerjee, I.; Pramanik, K.; Bhattacharya, M.K.; Pal, K. Preparation and characterization of novel carbopol based bigels for topical delivery of metronidazole for the treatment of bacterial vaginosis. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 44, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Vanić, Ž.; Hurler, J.; Ferderber, K.; Golja Gašparović, P.; Škalko-Basnet, N.; Filipović-Grčić, J. Novel vaginal drug delivery system: Deformable propylene glycol liposomes-in-hydrogel. J. Liposome Res. 2014, 24, 27–36. [Google Scholar] [CrossRef]
- Bouchemal, K.; Aka-Any-Grah, A.; Dereuddre-Bosquet, N.; Martin, L.; Lievin-Le-Moal, V.; Le Grand, R.; Nicolas, V.; Gibellini, D.; Lembo, D.; Poüs, C.; et al. Thermosensitive and mucoadhesive pluronic-hydroxypropylmethylcellulose hydrogel containing the mini-CD4 M48U1 is a promising efficient barrier against HIV diffusion through macaque cervicovaginal mucus. Antimicrob. Agents Chemother. 2015, 59, 2215–2222. [Google Scholar] [CrossRef] [Green Version]
- Perinelli, D.R.; Campana, R.; Skouras, A.; Bonacucina, G.; Cespi, M.; Mastrotto, F.; Baffone, W.; Casettari, L. Chitosan Loaded into a Hydrogel Delivery System as a Strategy to Treat Vaginal Co-Infection. Pharmaceutics 2018, 10, 23. [Google Scholar] [CrossRef] [Green Version]
- Vera-González, N.; Shukla, A. Advances in Biomaterials for the Prevention and Disruption of Candida Biofilms. Front. Microbiol. 2020, 11, 538602. [Google Scholar] [CrossRef] [PubMed]
- Osmari, B.F.; Giuliani, L.M.; Reolon, J.B.; Rigo, G.V.; Tasca, T.; Cruz, L. Gellan gum-based hydrogel containing nanocapsules for vaginal indole-3-carbinol delivery in trichomoniasis treatment. Eur. J. Pharm. Sci. 2020, 151, 105379. [Google Scholar] [CrossRef] [PubMed]
- Li, W.Z.; Zhao, N.; Zhou, Y.Q.; Yang, L.B.; Xiao-Ning, W.; Bao-Hua, H.; Peng, K.; Chun-Feng, Z. Post-expansile hydrogel foam aerosol of PG-liposomes: A novel delivery system for vaginal drug delivery applications. Eur. J. Pharm. Sci. 2012, 47, 162–169. [Google Scholar] [CrossRef]
- Jøraholmen, M.W.; Basnet, P.; Tostrup, M.J.; Moueffaq, S.; Škalko-Basnet, N. Localized Therapy of Vaginal Infections and Inflammation: Liposomes-In-Hydrogel Delivery System for Polyphenols. Pharmaceutics 2019, 11, 53. [Google Scholar] [CrossRef] [Green Version]
- Kirchhof, S.; Goepferich, A.M.; Brandl, F.P. Hydrogels in ophthalmic applications. Eur. J. Pharm. Biopharm. 2015, 95, 227–238. [Google Scholar] [CrossRef]
- Cooper, R.C.; Yang, H. Hydrogel-based ocular drug delivery systems: Emerging fabrication strategies, applications, and bench-to-bedside manufacturing considerations. J. Control. Release 2019, 306, 29–39. [Google Scholar] [CrossRef]
- Xu, X.; Weng, Y.; Xu, L.; Chen, H. Sustained release of Avastin® from polysaccharides cross-linked hydrogels for ocular drug delivery. Int. J. Biol. Macromol. 2013, 60, 272–276. [Google Scholar] [CrossRef]
- Cheng, Y.H.; Hung, K.H.; Tsai, T.H.; Lee, C.J.; Ku, R.Y.; Chiu, A.W.; Chiou, S.H.; Liu, C.J. Sustained delivery of latanoprost by thermosensitive chitosan-gelatin-based hydrogel for controlling ocular hypertension. Acta Biomater. 2014, 10, 4360–4366. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Sun, Y.; Ren, F.; Gao, S. PLGA-PEG-PLGA hydrogel for ocular drug delivery of dexamethasone acetate. Drug Dev. Ind. Pharm. 2010, 36, 1131–1138. [Google Scholar] [CrossRef]
- Zhang, X.-Z.; Yang, Y.-Y.; Chung, T.-S.; Ma, K.-X. Preparation and Characterization of Fast Response Macroporous Poly(N-isopropylacrylamide) Hydrogels. Langmuir 2001, 17, 6094–6099. [Google Scholar] [CrossRef]
- Kang Derwent, J.J.; Mieler, W.F. Thermoresponsive hydrogels as a new ocular drug delivery platform to the posterior segment of the eye. Trans. Am. Ophthalmol. Soc. 2008, 106, 206–213. [Google Scholar]
- Eljarrat-Binstock, E.; Domb, A.J.; Orucov, F.; Frucht-Pery, J.; Pe’er, J. Methotrexate delivery to the eye using transscleral hydrogel iontophoresis. Curr. Eye Res. 2007, 32, 639–646. [Google Scholar] [CrossRef] [PubMed]
- Xie, B.; Jin, L.; Luo, Z.; Yu, J.; Shi, S.; Zhang, Z.; Shen, M.; Chen, H.; Li, X.; Song, Z. An injectable thermosensitive polymeric hydrogel for sustained release of Avastin® to treat posterior segment disease. Int. J. Pharm. 2015, 490, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.Y.; Hsieh, A.C. A gelatin-g-poly(N-isopropylacrylamide) biodegradable in situ gelling delivery system for the intracameral administration of pilocarpine. Biomaterials 2012, 33, 2372–2387. [Google Scholar] [CrossRef]
- Liu, W.; Lee, B.S.; Mieler, W.F.; Kang-Mieler, J.J. Biodegradable Microsphere-Hydrogel Ocular Drug Delivery System for Controlled and Extended Release of Bioactive Aflibercept In Vitro. Curr. Eye Res. 2019, 44, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Oelker, A.M.; Grinstaff, M.W. Ophthalmic adhesives: A materials chemistry perspective. J. Mater. Chem. 2008, 18, 2521–2536. [Google Scholar] [CrossRef]
- Trujillo-de Santiago, G.; Sharifi, R.; Yue, K.; Sani, E.S.; Kashaf, S.S.; Alvarez, M.M.; Leijten, J.; Khademhosseini, A.; Dana, R.; Annabi, N. Ocular adhesives: Design, chemistry, crosslinking mechanisms, and applications. Biomaterials 2019, 197, 345–367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Su, X.; Tan, M.J.; Li, Z.; Wong, M.; Rajamani, L.; Lingam, G.; Loh, X.J. Recent Progress in Using Biomaterials as Vitreous Substitutes. Biomacromolecules 2015, 16, 3093–3102. [Google Scholar] [CrossRef] [PubMed]
- Xinming, L.; Yingde, C.; Lloyd, A.W.; Mikhalovsky, S.V.; Sandeman, S.R.; Howel, C.A.; Liewen, L. Polymeric hydrogels for novel contact lens-based ophthalmic drug delivery systems: A review. Cont. Lens Anterior Eye 2008, 31, 57–64. [Google Scholar] [CrossRef]
- Singh, K.; Nair, A.B.; Kumar, A.; Kumria, R. Novel approaches in formulation and drug delivery using contact lenses. J. Basic Clin. Pharm. 2011, 2, 87. [Google Scholar]
- Andrade-Vivero, P.; Fernandez-Gabriel, E.; Alvarez-Lorenzo, C.; Concheiro, A. Improving the loading and release of NSAIDs from pHEMA hydrogels by copolymerization with functionalized monomers. J. Pharm. Sci. 2007, 96, 802–813. [Google Scholar] [CrossRef]
- Gulsen, D.; Chauhan, A. Dispersion of microemulsion drops in HEMA hydrogel: A potential ophthalmic drug delivery vehicle. Int. J. Pharm. 2005, 292, 95–117. [Google Scholar] [CrossRef]
- Maulvi, F.A.; Lakdawala, D.H.; Shaikh, A.A.; Desai, A.R.; Choksi, H.H.; Vaidya, R.J.; Ranch, K.M.; Koli, A.R.; Vyas, B.A.; Shah, D.O. In vitro and in vivo evaluation of novel implantation technology in hydrogel contact lenses for controlled drug delivery. J. Control. Release 2016, 226, 47–56. [Google Scholar] [CrossRef]
- Lin, Q.; Xu, X.; Wang, B.; Shen, C.; Tang, J.; Han, Y.; Chen, H. Hydrated polysaccharide multilayer as an intraocular lens surface coating for biocompatibility improvements. J. Mater. Chem. B 2015, 3, 3695–3703. [Google Scholar] [CrossRef]
- Uchida, R.; Sato, T.; Tanigawa, H.; Uno, K. Azulene incorporation and release by hydrogel containing methacrylamide propyltrimenthylammonium chloride, and its application to soft contact lens. J. Control. Release 2003, 92, 259–264. [Google Scholar] [CrossRef]
- Jacob, S.; Nair, A.B. Cyclodextrin complexes: Perspective from drug delivery and formulation. Drug Dev. Res. 2018, 79, 201–217. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, J.F.; Alvarez-Lorenzo, C.; Silva, M.; Balsa, L.; Couceiro, J.; Torres-Labandeira, J.J.; Concheiro, A. Soft contact lenses functionalized with pendant cyclodextrins for controlled drug delivery. Biomaterials 2009, 30, 1348–1355. [Google Scholar] [CrossRef]
- Alvarez-Lorenzo, C.; Hiratani, H.; Gómez-Amoza, J.L.; Martínez-Pacheco, R.; Souto, C.; Concheiro, A. Soft contact lenses capable of sustained delivery of timolol. J. Pharm. Sci. 2002, 91, 2182–2192. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Lorenzo, C.; Yañez, F.; Barreiro-Iglesias, R.; Concheiro, A. Imprinted soft contact lenses as norfloxacin delivery systems. J. Control. Release 2006, 113, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Rossos, A.K.; Banti, C.N.; Kalampounias, A.G.; Papachristodoulou, C.; Kordatos, K.; Zoumpoulakis, P.; Mavromoustakos, T.; Kourkoumelis, N.; Hadjikakou, S.K. pHEMA@AGMNA-1: A novel material for the development of antibacterial contact lens. Mater. Sci. Eng. C 2020, 111, 110770. [Google Scholar] [CrossRef] [PubMed]
- Silva, D.; de Sousa, H.C.; Gil, M.H.; Santos, L.F.; Moutinho, G.M.; Salema-Oom, M.; Alvarez-Lorenzo, C.; Serro, A.P.; Saramago, B. Diclofenac sustained release from sterilised soft contact lens materials using an optimised layer-by-layer coating. Int. J. Pharm. 2020, 585, 119506. [Google Scholar] [CrossRef]
- Kamoun, E.A.; Kenawy, E.S.; Chen, X. A review on polymeric hydrogel membranes for wound dressing applications: PVA-based hydrogel dressings. J. Adv. Res. 2017, 8, 217–233. [Google Scholar] [CrossRef]
- Tavakoli, S.; Klar, A.S. Advanced Hydrogels as Wound Dressings. Biomolecules 2020, 10, 1169. [Google Scholar] [CrossRef]
- Rosiak, J.; Rucinska-Rybus, A.; Pekala, W. Method of Manufacturing Hydrogel Dressings. U.S. Patent 4871490A, 3 October 1989. [Google Scholar]
- Khutoryanskiy, V.; Khutoryanskaya, O.; Cook, J.P.; Goodall, G.W. Hydrogel Synthesis. Google Patents US20130018110A1, 17 January 2013. [Google Scholar]
- Liang, Y.; Zhao, X.; Hu, T.; Han, Y.; Guo, B. Mussel-inspired, antibacterial, conductive, antioxidant, injectable composite hydrogel wound dressing to promote the regeneration of infected skin. J. Colloid Interface Sci. 2019, 556, 514–528. [Google Scholar] [CrossRef]
- Zhang, Y.; Jiang, M.; Zhang, Y.; Cao, Q.; Wang, X.; Han, Y.; Sun, G.; Li, Y.; Zhou, J. Novel lignin-chitosan-PVA composite hydrogel for wound dressing. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 104, 110002. [Google Scholar] [CrossRef]
- Li, X.; Chen, S.; Zhang, B.; Li, M.; Diao, K.; Zhang, Z.; Li, J.; Xu, Y.; Wang, X.; Chen, H. In situ injectable nano-composite hydrogel composed of curcumin, N,O-carboxymethyl chitosan and oxidized alginate for wound healing application. Int. J. Pharm. 2012, 437, 110–119. [Google Scholar] [CrossRef]
- Amirian, J.; Zeng, Y.; Shekh, M.I.; Sharma, G.; Stadler, F.J.; Song, J.; Du, B.; Zhu, Y. In-situ crosslinked hydrogel based on amidated pectin/oxidized chitosan as potential wound dressing for skin repairing. Carbohydr. Polym. 2021, 251, 117005. [Google Scholar] [CrossRef]
- Azadmanesh, F.; Pourmadadi, M.; Zavar Reza, J.; Yazdian, F.; Omidi, M.; Haghirosadat, B.F. Synthesis of a Novel Nanocomposite Containing Chitosan as a 3D Printed Wound Dressing Technique: Emphasis on Gene Expression. Biotechnol. Prog. 2021, e3132. [Google Scholar] [CrossRef]
- Schuhladen, K.; Raghu, S.N.V.; Liverani, L.; Neščáková, Z.; Boccaccini, A.R. Production of a novel poly(ɛ-caprolactone)-methylcellulose electrospun wound dressing by incorporating bioactive glass and Manuka honey. J. Biomed. Mater. Res. Part B Appl. Biomater. 2021, 109, 180–192. [Google Scholar] [CrossRef] [PubMed]
- Fleck, C.A.; Simman, R. Modern collagen wound dressings: Function and purpose. J. Am. Coll. Certif. Wound Spec. 2011, 2, 50–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tao, G.; Cai, R.; Wang, Y.; Zuo, H.; He, H. Fabrication of antibacterial sericin based hydrogel as an injectable and mouldable wound dressing. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 119, 111597. [Google Scholar] [CrossRef] [PubMed]
- Varaprasad, K.; Jayaramudu, T.; Kanikireddy, V.; Toro, C.; Sadiku, E.R. Alginate-based composite materials for wound dressing application:A mini review. Carbohydr. Polym. 2020, 236, 116025. [Google Scholar] [CrossRef]
- Hsu, L.C.; Peng, B.Y.; Chen, M.S.; Thalib, B.; Ruslin, M.; Tung, T.D.X.; Chou, H.H.; Ou, K.L. The potential of the stem cells composite hydrogel wound dressings for promoting wound healing and skin regeneration: In vitro and in vivo evaluation. J. Biomed. Mater. Res. B Appl. Biomater. 2019, 107, 278–285. [Google Scholar] [CrossRef]
- Jeon, M.S.; Jeon, Y.; Hwang, J.H.; Heu, C.S.; Jin, S.; Shin, J.; Song, Y.; Chang Kim, S.; Cho, B.-K.; Lee, J.-K.; et al. Fabrication of three-dimensional porous carbon scaffolds with tunable pore sizes for effective cell confinement. Carbon 2018, 130, 814–821. [Google Scholar] [CrossRef]
- Wang, X.; Salick, M.R.; Gao, Y.; Jiang, J.; Li, X.; Liu, F.; Cordie, T.; Li, Q.; Turng, L.-S. Interconnected porous poly(ɛ-caprolactone) tissue engineering scaffolds fabricated by microcellular injection molding. J. Cell. Plast. 2018, 54, 379–397. [Google Scholar] [CrossRef]
- Yamamoto, T.; Inoue, K.; Tezuka, Y. Hydrogel formation by the ‘topological conversion’ of cyclic PLA–PEO block copolymers. Polym. J. 2016, 48, 391–398. [Google Scholar] [CrossRef] [Green Version]
- Santoro, M.; Shah, S.R.; Walker, J.L.; Mikos, A.G. Poly(lactic acid) nanofibrous scaffolds for tissue engineering. Adv. Drug Deliv. Rev. 2016, 107, 206–212. [Google Scholar] [CrossRef] [Green Version]
- Vidović, E.; Klee, D.; Höcker, H. Evaluation of water uptake and mechanical properties of biomedical polymers. J. Appl. Polym. Sci. 2013, 130, 3682–3688. [Google Scholar] [CrossRef]
- Jacob, S.; Nair, A.B.; Patel, V.; Shah, J. 3D Printing Technologies: Recent Development and Emerging Applications in Various Drug Delivery Systems. AAPS PharmSciTech 2020, 21. [Google Scholar] [CrossRef] [PubMed]
- Fetah, K.; Tebon, P.; Goudie, M.J.; Eichenbaum, J.; Ren, L.; Barros, N.; Nasiri, R.; Ahadian, S.; Ashammakhi, N.; Dokmeci, M.R.; et al. The emergence of 3D bioprinting in organ-on-chip systems. Prog. Biomed. Eng. 2019, 1, 012001. [Google Scholar] [CrossRef]
- Xu, S.; An, X. Preparation, microstructure and function for injectable liposome-hydrogels. Colloids Surf. A Physicochem. Eng. Asp. 2019, 560, 20–25. [Google Scholar] [CrossRef]
- Cunniffe, G.M.; Dickson, G.R.; Partap, S.; Stanton, K.T.; O’Brien, F.J. Development and characterisation of a collagen nano-hydroxyapatite composite scaffold for bone tissue engineering. J. Mater. Sci. Mater. Med. 2010, 21, 2293–2298. [Google Scholar] [CrossRef]
- Lyons, F.G.; Al-Munajjed, A.A.; Kieran, S.M.; Toner, M.E.; Murphy, C.M.; Duffy, G.P.; O’Brien, F.J. The healing of bony defects by cell-free collagen-based scaffolds compared to stem cell-seeded tissue engineered constructs. Biomaterials 2010, 31, 9232–9243. [Google Scholar] [CrossRef]
- Wang, G.; Hu, X.; Lin, W.; Dong, C.; Wu, H. Electrospun PLGA-silk fibroin-collagen nanofibrous scaffolds for nerve tissue engineering. In Vitro Cell. Dev. Biol. Anim. 2011, 47, 234–240. [Google Scholar] [CrossRef]
- Kijeńska, E.; Prabhakaran, M.P.; Swieszkowski, W.; Kurzydlowski, K.J.; Ramakrishna, S. Electrospun bio-composite P(LLA-CL)/collagen I/collagen III scaffolds for nerve tissue engineering. J. Biomed. Mater. Res. B Appl. Biomater. 2012, 100, 1093–1102. [Google Scholar] [CrossRef]
- Prabhakaran, M.P.; Vatankhah, E.; Ramakrishna, S. Electrospun aligned PHBV/collagen nanofibers as substrates for nerve tissue engineering. Biotechnol. Bioeng. 2013, 110, 2775–2784. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Huang, D.; Yan, Z.; Wang, C. Heparin/collagen encapsulating nerve growth factor multilayers coated aligned PLLA nanofibrous scaffolds for nerve tissue engineering. J. Biomed. Mater. Res. A 2017, 105, 1900–1910. [Google Scholar] [CrossRef] [PubMed]
- Pupkaite, J.; Rosenquist, J.; Hilborn, J.; Samanta, A. Injectable Shape-Holding Collagen Hydrogel for Cell Encapsulation and Delivery Cross-linked Using Thiol-Michael Addition Click Reaction. Biomacromolecules 2019, 20, 3475–3484. [Google Scholar] [CrossRef]
- Kim, W.S.; Mooney, D.J.; Arany, P.R.; Lee, K.; Huebsch, N.; Kim, J. Adipose tissue engineering using injectable, oxidized alginate hydrogels. Tissue Eng. Part A 2012, 18, 737–743. [Google Scholar] [CrossRef]
- Jia, J.; Richards, D.J.; Pollard, S.; Tan, Y.; Rodriguez, J.; Visconti, R.P.; Trusk, T.C.; Yost, M.J.; Yao, H.; Markwald, R.R.; et al. Engineering alginate as bioink for bioprinting. Acta Biomater. 2014, 10, 4323–4331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, F.; Tian, M.; Zhang, D.; Wang, J.; Wang, Q.; Yu, X.; Zhang, X.; Wan, C. Preparation and characterization of oxidized alginate covalently cross-linked galactosylated chitosan scaffold for liver tissue engineering. Mater. Sci. Eng. C 2012, 32, 310–320. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, Y.; Chen, H.; Ozbolat, I.T. Characterization of printable cellular micro-fluidic channels for tissue engineering. Biofabrication 2013, 5, 025004. [Google Scholar] [CrossRef]
- Wüst, S.; Godla, M.E.; Müller, R.; Hofmann, S. Tunable hydrogel composite with two-step processing in combination with innovative hardware upgrade for cell-based three-dimensional bioprinting. Acta Biomater. 2014, 10, 630–640. [Google Scholar] [CrossRef] [PubMed]
- Poldervaart, M.T.; Wang, H.; van der Stok, J.; Weinans, H.; Leeuwenburgh, S.C.; Öner, F.C.; Dhert, W.J.; Alblas, J. Sustained release of BMP-2 in bioprinted alginate for osteogenicity in mice and rats. PLoS ONE 2013, 8, e72610. [Google Scholar] [CrossRef] [PubMed]
- Thomas, L.V.; Rahul, V.G.; Nair, P.D. Effect of stiffness of chitosan-hyaluronic acid dialdehyde hydrogels on the viability and growth of encapsulated chondrocytes. Int. J. Biol. Macromol. 2017, 104, 1925–1935. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.J.; Arai, Y.; Choi, B.; Park, S.; Ahn, J.; Han, I.-B.; Lee, S.-H. Restoration of articular osteochondral defects in rat by a bi-layered hyaluronic acid hydrogel plug with TUDCA-PLGA microsphere. J. Ind. Eng. Chem. 2018, 61, 295–303. [Google Scholar] [CrossRef]
- Park, S.H.; Seo, J.Y.; Park, J.Y.; Ji, Y.B.; Kim, K.; Choi, H.S.; Choi, S.; Kim, J.H.; Min, B.H.; Kim, M.S. An injectable, click-crosslinked, cytomodulin-modified hyaluronic acid hydrogel for cartilage tissue engineering. NPG Asia Mater. 2019, 11, 30. [Google Scholar] [CrossRef]
- Cui, N.; Qian, J.; Xu, W.; Xu, M.; Zhao, N.; Liu, T.; Wang, H. Preparation, characterization, and biocompatibility evaluation of poly(Nɛ-acryloyl-L-lysine)/hyaluronic acid interpenetrating network hydrogels. Carbohydr. Polym. 2016, 136, 1017–1026. [Google Scholar] [CrossRef]
- Gwon, K.; Kim, E.; Tae, G. Heparin-hyaluronic acid hydrogel in support of cellular activities of 3D encapsulated adipose derived stem cells. Acta Biomater. 2017, 49, 284–295. [Google Scholar] [CrossRef]
- Lou, J.; Stowers, R.; Nam, S.; Xia, Y.; Chaudhuri, O. Stress relaxing hyaluronic acid-collagen hydrogels promote cell spreading, fiber remodeling, and focal adhesion formation in 3D cell culture. Biomaterials 2018, 154, 213–222. [Google Scholar] [CrossRef]
- Li, L.; Yu, F.; Zheng, L.; Wang, R.; Yan, W.; Wang, Z.; Xu, J.; Wu, J.; Shi, D.; Zhu, L.; et al. Natural hydrogels for cartilage regeneration: Modification, preparation and application. J. Orthop. Transl. 2019, 17, 26–41. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Ren, J.; Chen, G.; Li, G.; Wu, X.; Wang, G.; Gu, G.; Li, J. Injectable in situ cross-linking chitosan-hyaluronic acid based hydrogels for abdominal tissue regeneration. Sci. Rep. 2017, 7, 2699. [Google Scholar] [CrossRef] [Green Version]
- Kwarta, C.P.; Widiyanti, P.; Siswanto. Hyaluronic Acid (HA)-Polyethylene glycol (PEG) as injectable hydrogel for intervertebral disc degeneration patients therapy. J. Phys. Conf. Ser. 2017, 853, 012036. [Google Scholar] [CrossRef] [Green Version]
- Xu, X.; Zeng, Z.; Huang, Z.; Sun, Y.; Huang, Y.; Chen, J.; Ye, J.; Yang, H.; Yang, C.; Zhao, C. Near-infrared light-triggered degradable hyaluronic acid hydrogel for on-demand drug release and combined chemo-photodynamic therapy. Carbohydr. Polym. 2020, 229, 115394. [Google Scholar] [CrossRef]
- Tavakoli, J.; Tang, Y. Hydrogel Based Sensors for Biomedical Applications: An Updated Review. Polymers 2017, 9, 364. [Google Scholar] [CrossRef] [Green Version]
- Chimene, D.; Kaunas, R.; Gaharwar, A.K. Hydrogel Bioink Reinforcement for Additive Manufacturing: A Focused Review of Emerging Strategies. Adv. Mater. 2020, 32, e1902026. [Google Scholar] [CrossRef]







| Techniques | Types of Materials Used | Procedure | Advantages | Disadvantages | Reference |
|---|---|---|---|---|---|
| Hydrophobic interaction | Hydrophilic monomers and hydrophobic comonomers | Free radical copolymerization of a hydrophilic monomer with a hydrophobic co-monomer | Absence of cross-linking agents and relative ease of production | Poor mechanical characteristics | [3] |
| Ionic interaction | Polyelectrolyte solution and multivalent ions of opposite charge | Ionic interaction through simple ion exchange mechanisms and complex formation | Cross-linking takes place at room temperature and physiological pH Properties can be fine-tuned by cationic and anionic constituents | Limited to ionic polymers and sensitive to impurities | [4] |
| Hydrogen bond | Polymeric functional groups of high electron density with electron-deficient hydrogen atom | Self-assemble through secondary molecular interactions | Increase in polymer concentration can increase the stability of gel. | Influx of water can disperse/dissolve the gel within short duration. | [5] |
| Bulk polymerization | Monomers and monomer-soluble initiators | The polymerization reaction is initiated with radiation, ultraviolet, or chemical catalysts at low rate of conversions | A simple and versatile technique to prepare hydrogels with desired physical properties and forms | Increase in viscosity during high rate of polymerization reaction can generate heat Weak polymer structure | [6] |
| Solution polymerization/ | Ionic or neutral monomers with the multifunctional cross-linking agent | Reaction initiated thermally with UV irradiation or by redox initiator system | Control of temperature Performed in non-toxic aqueous medium at room temperature High polymerization rate | To be washed to eliminate reactants, the polymers, and other impurities | [7] |
| Suspension polymerization | Hydrophilic monomers, initiators, cross-linkers, and suspending agent | The monomers and initiator are dispersed in the organic phase as a homogenous mixture | Directly usable as powders, beads, or microspheres | Restricted to water insoluble polymer Cooling jacket required to dissipate heat Requirement of agitators and dispersant | [2] |
| Grafting | Viny polymers, initiators and cross-linking agents | Covalent bonding of monomers on free radicals generated on stronger support structures | Improve functional properties of the polymer | Difficulty of characterizing side chains | [8] |
| Irradiation | High energy gamma beams and electron beams as initiators | Irradiation of aqueous polymer solution results in the formation of radicals and macroradicals on the polymer chains | Pure, sterile, residue-free hydrogel Does not require catalyst and other additives Irradiation dose can control swelling capacity | Irradiation can cause polymer degradation via chain scission and cross-linking events | [9] |
| Step growth polymerization | Bi or multifunctional monomers and each with attest two sites for bonding | Multifunctional monomers react to form oligomers resulting in long chain polymers | No initiator is required to start the polymerization and termination reactions | Prolonged reaction times required to achieve a high degree of conversion and high molecular weights | [10] |
| Types | Description | Examples | References |
|---|---|---|---|
| Temperature responsive | Change in temperature disturbs the equilibrium exists between hydrophilic and hydrophobic segments of the polymer chain and induce the sol-gel transformation | Pluronic, poloxamer, poly(acrylic acid), poly(N-isopropylacrylamide), and tetronic | [11] |
| pH responsive | Change in pH results in swelling/deswelling behavior due to the changes in hydrophobicity of the polymer chain | Chitosan, guar gum succinate, kappa-carrageenan, poly (ethylene imine), poly(acrylamide), poly(acrylic acid), poly(diethylaminoethyl methacrylate), poly(dimethylaminoethyl methacrylate), poly(ethylacrylic acid), poly(hydroxyethyl methacrylate), poly(methacrylic acid), poly(propylacrylic acid), and poly(vinyl alcohol) | [12] |
| Photoresponsive | External stimulus of either visible or UV light initiates sol-gel transformation | Azo benzene-poly(2-hydroxyethyl methacrylate), azo benzene-bovine albumin, triphenylmethane leuco derivatives, and trisodium salt of copper chlorophyllin-poly(N-isopropylacrylamide) | [13] |
| Electroresponsive | Upon the application of an electric field, deswelling or bending takes place, based on the shape and position of the gel relative to the electrodes | Agarose, calcium alginate, carbomer, chondroitin sulphate, hyaluronic acid, partially hydrolyzed polyacrylamide, polydimethylaminopropyl acrylamide, and xanthan gum | [14] |
| Ultrasonically responsive | External application of ultrasonic waves modulates the drug release from the hydrogel matrix | Ethylene vinyl acetate, poly (2-hydroxyethyl methacrylate), poly(bis(p-carboxyphenoxy)alkane-anhydrides, poly(lactide-co-glycolide, polyglycolide and polylactide | [15] |
| Magneto responsive | Application of heating, mechanical deformation, or external magnetic field to magnetic nanoparticles, such as nanoparticles of magnetite, maghemite, and ferrite | Alginate-xanthan cross-linked with Ca2+ magnetic nanoparticles, hemicellulose hydrogels with magnetic iron oxide (Fe3O4), methacrylated chondroitin sulfate with magnetic nanoparticles, poly(N-isopropylacrylamide), and xanthan-bovine serum albumin-magnetic nanoparticles | [16] |
| Glucose responsive | Hydrogel as a self-regulated, insulin-delivery system discretely switching release at normoglycemia | Catalase, insulin, phenylborate derivative {4-(1,6-dioxo-2,5-diaza-7-oxamyl) phenylboronic acid in combination with poly(N- isopropylmethacrylamide), and poly(2-hydroxyethyl methacrylate-co-N,N-dimethylaminoethyl methacrylate) in combination with glucose oxidase | [17] |
| Ionic strength | Varying ionic strength and pH can expand the polyelectrolytes resulting in dissociation of ionizable groups and subsequent drug release | Alginic acid, carboxymethyl cellulose, carboxymethyl starch, carrageenan, cellulose sulfate, chitosan, dextran sulfate, eudragit E, RL, RS, and hyaluronic acid | [18] |
| Inflammation responsive | pH changes at the inflammatory site resulted in drug release or pH-responsive hydrogel with inflammatory responsive characteristics and the capability to passively targeting macrophages | Aliphatic polyketals and pH-responsive polymers | [19] |
| Commercial Product | Polymers | Active Constituents | Dosage Form | Application | Manufacturer |
|---|---|---|---|---|---|
| Buccastem® M | Povidone K30, xanthan gum, locust bean gum | Prochlorperazine maleate | Tablet | Nausea and vomiting in migraine | Alliance |
| Biotene | Carbomer and hydroxyethyl cellulose | Nil | Gel | Oral moisturizing agent in dry mouth | GlaxoSmithKline |
| Gengigel® | Hyaluronan | Nil | Gel | Mouth and gum care-oral ulcers | Oral Science |
| Hydrogel 15% | Carbomer in ozonized sunflower oil | Ozone | Gel | Oral health | Honest 03 |
| Lubrajel™ BA | Glyceryl acrylate and glyceryl polyacrylate | Nil | Gel | Oral moisturizing agent | Prospector |
| Nicorette® | Hydroxypropyl methylcellulose | Nicotine | Chewing gum | Smoking cessation | GlaxoSmithKline |
| Nicotinell® | Xanthan gum and gelatin | Nicotine | Chewing gum | Smoking cessation | GlaxoSmithKline |
| Zilactin-B Gel® | Hydroxypropyl cellulose | Benzocaine | Gel | Local anesthetic in minor oral problems | Blairex laboratories Inc. |
| ZuplenzTM | Polyethylene glycol 1000, polyvinyl alcohol and rice starch | Ondansetron | Soluble oral film | Chemotherapy, radiation, surgery-induced nausea and vomiting | Aquestive Therapeutics |
| Product Name | Principal Components | Indications | Manufacturer |
|---|---|---|---|
| Biofinity® | Silicone hydrogel | Continuous wear up to 7 days. Corrects near sightedness and farsightedness | Cooper vision |
| Air optix® night and day®aqua | Lotrafilcon-A (fluoro-silicone hydrogel) | Continuous wear up to 7 days. Corrects near sightedness and farsightedness | Alcon |
| Retisert® | Fluocinolone acetonide, silicone elastomer and polyvinyl alcohol membrane | Deliver long term control of inflammation | Bausch and Lomb |
| Lacrisert® | Hydroxypropyl cellulose | Moderate to severe dry eyes | Bausch and Lomb |
| Systane® | Propylene glycol | For use as a lubricant to prevent further irritation or to relieve dryness of the eye | Alcon |
| Restasis® | Cyclosporine, carbomer copolymer Type A | Indicated to increase tear production | Allergan |
| Proclear (Omafilcon B) | 2-hydroxy-ethylmethacrylate and 2-methacryloxyethyl phosphorylcholine cross-linked with ethylene glycol dimethacrylate | Indicated for daily wear for the correction of visual acuity | Cooper vision |
| Clintas Hydrate® | Carbomer | Lubricating eye gel for occasional dry eye discomfort | Altacor |
| Dailies® AquaComfort | Nelfilcon A polymer (polyvinyl alcohol partially acetalized with N-formylmethyl acrylamide) | Optical correction of refractive ametropia | Ciba vision |
| Systane® gel drops | Polyethylene glycol 400, propylene glycol | For the temporary relief of burning and irritation due to dryness of the eye | Alcon |
| Hylo® gel | Sodium hyaluronate, citrate buffer, sorbitol | Long lasting dry eye relief | Candorvision |
| Iluvien® | Fluocinolone acetonide, polyvinyl alcohol, and silicone adhesives | Treatment of diabetic macular edema | Alimera Sciences |
| Yutiq™ | Fluocinolone acetonide, polyvinyl alcohol | Treatment of chronic non-infectious uveitis affecting the posterior segment of the eye | EyePoint Pharmaceuticals Inc. |
| Ozurdex® | Dexamethasone, poly (d,l-lactide-co-glycolide) | Macular edema, non-infectious uveitis | Allergan |
| Product Name | Principal Components | Indications | Manufacturer |
|---|---|---|---|
| Helix3-cm® | Type 1 native bovine collagen | Management of burns, sores, blisters, ulcers, and other wounds | Amerx Health Care Corp. |
| 3M™ Tegaderm™ hydrogel wound filler | Hydrocolloid dressing | Low to moderate draining wounds, partial- and full-thickness dermal ulcers | 3M Health Care Ltd. |
| AquaSite ®amorphous hydrogel dressing | Glycerin based hydrocolloid dressing | Provide moist heat healing environment and autolytic debridement | Integra Life Science Corp. |
| Algicell® Ag calcium Alginate dressing with antimicrobial silver | Calcium alginate ionic silver | Effective against a broad range of bacteria and more absorption of drainage | Integra Life Science Corp. |
| INTRASITE® gel hydrogel wound dressing | Modified carboxymethyl cellulose, propylene glycol | Re-hydrates necrotic tissue, facilitating autolytic debridement minor burns, superficial lacerations, cuts, and abrasions | Smith &Nephew Healthcare Limited |
| Microcyn® skin and wound hydrogel | Hypochlorous acid | All types of chronic and acute wounds as all types of burns | Microsafe group |
| Prontosan® wound gel | Polyhexanide and betaine | Cleansing and moisturizing of skin wounds and burns | Bbraun |
| Purilon® gel, Regenecare® wound gel | 2% lidocaine collagen, aloe and sodium alginate | Pressure ulcers, cuts, burns, and abrasions | MPM Medical |
| Cutimed® gel | Carbomer 940 | Supports autolytic debridement in necrotic and sloughy wounds | BSN Medical |
| Viniferamine® wound hydrogel Ag | Glycerin Metallic silver | Partial and full thickness wounds with signs of infection and little to no exudate | McKesson |
| HemCon® bandage PRO | Chitosan | Providing hemostasis, antibacterial barrier against wide range of microorganisms | TriCol biomedical Inc. |
| Hyalofill®-F and R | Hyaluronic acid in fleece and rope | Absorbs wound exudate, promotes granulation tissue formulation, supports healing process | Anika |
| CMC fiber dressing | Carboxymethyl cellulose | Absorptive dressing for moderate to heavy exudate | Gentell |
| Inadine™ (PVP-1) non-adherent dressing | Polyethylene glycol, povidone iodine | Ulcers deriving from different etiologies, chronic wounds | 3M Health Care Ltd. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jacob, S.; Nair, A.B.; Shah, J.; Sreeharsha, N.; Gupta, S.; Shinu, P. Emerging Role of Hydrogels in Drug Delivery Systems, Tissue Engineering and Wound Management. Pharmaceutics 2021, 13, 357. https://doi.org/10.3390/pharmaceutics13030357
Jacob S, Nair AB, Shah J, Sreeharsha N, Gupta S, Shinu P. Emerging Role of Hydrogels in Drug Delivery Systems, Tissue Engineering and Wound Management. Pharmaceutics. 2021; 13(3):357. https://doi.org/10.3390/pharmaceutics13030357
Chicago/Turabian StyleJacob, Shery, Anroop B. Nair, Jigar Shah, Nagaraja Sreeharsha, Sumeet Gupta, and Pottathil Shinu. 2021. "Emerging Role of Hydrogels in Drug Delivery Systems, Tissue Engineering and Wound Management" Pharmaceutics 13, no. 3: 357. https://doi.org/10.3390/pharmaceutics13030357