Cytotoxic and Antileishmanial Effects of the Monoterpene β-Ocimene
Abstract
:1. Introduction
2. Results
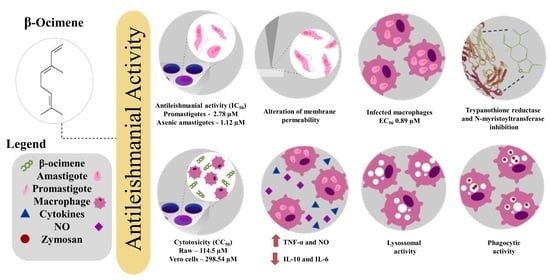
2.1. Antileishmanial Activity and Cytotoxic Effects of β-Ocimene
2.2. Membrane Integrity Assay
2.3. Morphological Alterations in β-Ocimene Treated Promastigote Forms
2.4. In Vitro Efficacy of β-Ocimene against Intra-Macrophage Amastigote Forms
2.5. Immunomodulatory Activity
3. Discussion
4. Materials and Methods
4.1. Chemicals and Pharmaceuticals
4.2. Drug Preparation
4.3. Maintenance of the Parasites and Macrophages
4.4. In Vitro Inhibition Assay of β-Ocimene on Promastigotes and Axenic Amastigotes
4.5. Membrane Integrity Assay
4.6. Parasite Morphological Evaluation Using Atomic Force Microscopy (AFM)
4.7. Cytotoxic Effect on Mammalian Cells and the Calculation of Selective Index (SI)
4.8. Treatment of Infected Macrophages
4.9. Lysosomal Activity
4.10. Phagocytosis Assay
4.11. Estimation of Th1/Th2 Cytokine and Nitric Oxide (NO) Production
4.12. Measurement of Reactive Oxygen Species (ROS)
4.13. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Burza, S.; Croft, S.L.; Boelaert, M. Leishmaniasis. Lancet 2018, 392, 951–970. [Google Scholar] [CrossRef] [PubMed]
- Mokni, M. Leishmanioses Cutanées. Ann. Dermatol. Venereol. 2019, 146, 232–246. [Google Scholar] [CrossRef]
- Bilgic-Temel, A.; Murrell, D.F.; Uzun, S. Cutaneous leishmaniasis: A neglected disfiguring disease for women. Int. J. Womens Dermatol. 2019, 5, 158–165. [Google Scholar] [CrossRef]
- Torres-Guerrero, E.; Quintanilla-Cedillo, M.R.; Ruiz-Esmenjaud, J.; Arenas, R. Leishmaniasis: A review. F1000Research 2017, 6, 750. [Google Scholar] [CrossRef]
- Ghorbani, M.; Farhoudi, R. Leishmaniasis in humans: Drug or vaccine therapy? Drug Des. Devel. Ther. 2017, 12, 25–40. [Google Scholar] [CrossRef] [Green Version]
- Chakravarty, J.; Sundar, S. Current and emerging medications for the treatment of leishmaniasis. Expert Opin. Pharmacother. 2019, 20, 1251–1265. [Google Scholar] [CrossRef]
- Luna, E.C.; Luna, I.S.; Scotti, L.; Monteiro, A.F.M.; Scotti, M.T.; de Moura, R.O.; de Araújo, R.S.A.; Monteiro, K.L.C.; de Aquino, T.M.; Ribeiro, F.F.; et al. Active essential oils and their components in use against neglected diseases and arboviruses. Oxid. Med. Cell Longev. 2019, 2019, 6587150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farré-Armengol, G.; Filella, I.; Llusià, J.; Peñuelas, J. β-Ocimene, a key floral and foliar volatile involved in multiple interactions between plants and other organisms. Molecules 2017, 22, 1148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiao, M.; Liu, R.; Long, C.; Ruan, Y.; Liu, C. Using β-Ocimene to increase the artemisinin content in juvenile plants of Artemisia annua L. Biotechnol. Lett. 2020, 42, 1161–1167. [Google Scholar] [CrossRef]
- Perring, T.M.; Battaglia, D.; Walling, L.L.; Toma, I.; Fanti, P. Aphids. In Sustainable Management of Arthropod Pests of Tomato; Elsevier: Amsterdam, The Netherlands, 2018; pp. 15–48. [Google Scholar]
- Russo, E.B.; Marcu, J. Cannabis pharmacology: The usual suspects and a few promising leads. Adv. Pharmacol. 2017, 80, 67–134. [Google Scholar]
- Bouyahya, A.; Chadon Assemian, I.C.; Mouzount, H.; Bourais, I.; Et-Touys, A.; Fellah, H.; Benjouad, A.; Dakka, N.; Bakri, Y. Could volatile compounds from leaves and fruits of Pistacia lentiscus constitute a novel source of anticancer, antioxidant, antiparasitic and antibacterial drugs? Ind. Crops Prod. 2019, 128, 62–69. [Google Scholar] [CrossRef]
- Macêdo, C.G.; Fonseca, M.Y.N.; Caldeira, A.D.; Castro, S.P.; Pacienza-Lima, W.; Borsodi, M.P.G.; Sartoratto, A.; da Silva, M.N.; Salgado, C.G.; Rossi-Bergmann, B.; et al. Leishmanicidal activity of Piper marginatum Jacq. from Santarém-PA against Leishmania amazonensis. Exp. Parasitol. 2020, 210, 107847. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, K.A.D.F.; Amorim, L.V.; Dias, C.N.; Moraes, D.F.C.; Carneiro, S.M.P.; Carvalho, F.A.D.A. Syzygium cumini (L.) Skeels essential oil and its major constituent α-pinene exhibit anti-leishmania activity through immunomodulation in vitro. J. Ethnopharmacol. 2015, 160, 32–40. [Google Scholar] [CrossRef] [Green Version]
- Dias, C.N.; Rodrigues, K.A.F.; Carvalho, F.A.A.; Carneiro, S.M.P.; Maia, J.G.S.; Andrade, E.H.A.; Moraes, D.F.C. Molluscicidal and leishmanicidal activity of the leaf essential oil of Syzygium cumini (L.) Skeels from Brazil. Chem. Biodivers. 2013, 10, 1133–1141. [Google Scholar] [CrossRef]
- Youssefi, M.R.; Moghaddas, E.; Tabari, M.A.; Moghadamnia, A.A.; Hosseini, S.M.; Farash, B.R.H.; Ebrahimi, M.A.; Mousavi, N.N.; Fata, A.; Maggi, F.; et al. In Vitro and in vivo effectiveness of carvacrol, thymol and linalool against Leishmania infantum. Molecules 2019, 24, 2072. [Google Scholar] [CrossRef] [Green Version]
- Arruda, D.C.; Miguel, D.C.; Yokoyama-Yasunaka, J.K.U.; Katzin, A.M.; Uliana, S.R.B. Inhibitory Activity of Limonene against Leishmania Parasites in Vitro and in Vivo. Biomed. Pharmacother. 2009, 63, 643–649. [Google Scholar] [CrossRef]
- Takahashi, H.T.; Britta, E.A.; Longhini, R.; Ueda-Nakamura, T.; Palazzo De Mello, J.C.; Nakamura, C.V. Antileishmanial Activity of 5-Methyl-2,2′: 5′,2′- Terthiophene isolated from Porophyllum ruderale is related to mitochondrial dysfunction in Leishmania amazonensis. Planta Med. 2013, 79, 330–333. [Google Scholar] [CrossRef] [Green Version]
- Ramírez-Macías, I.; Maldonado, C.R.; Marín, C.; Olmo, F.; Gutiérrez-Sánchez, R.; Rosales, M.J.; Quirós, M.; Salas, J.M.; Sánchez-Moreno, M. In Vitro Anti-leishmania evaluation of nickel complexes with a triazolopyrimidine derivative against Leishmania infantum and Leishmania braziliensis. J. Inorg. Biochem. 2012, 112, 1–9. [Google Scholar] [CrossRef]
- Podinovskaia, M.; Descoteaux, A. Leishmania and the macrophage: A multifaceted interaction. Future Microbiol. 2015, 10, 111–129. [Google Scholar] [CrossRef]
- Mesquita, J.T.; da Costa-Silva, T.A.; Borborema, S.E.T.; Tempone, A.G. activity of imidazole compounds on Leishmania (L.) infantum chagasi: Reactive oxygen species induced by econazole. Mol. Cell Biochem. 2014, 389, 293–300. [Google Scholar] [CrossRef]
- Basmaciyan, L.; Casanova, M. Cell Death in Leishmania. Parasite 2019, 26, 71. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Rodeheaver, D.P.; White, J.C.; Wright, A.M.; Walker, L.M.; Zhang, F.; Shannon, S. A Comparison of in Vitro Cytotoxicity Assays in Medical Device Regulatory Studies. Regul. Toxicol. Pharmacol. 2018, 97, 24–32. [Google Scholar] [CrossRef]
- Underhill, D.M. Macrophage Recognition of Zymosan Particles. J. Endotoxin. Res. 2003, 9, 176–180. [Google Scholar] [CrossRef]
- Jacob, P.L.; Leite, J.A.; Alves, A.K.A.; Rodrigues, Y.K.S.; Amorim, F.M.; Néris, P.L.N.; Oliveira, M.R.; Rodrigues-Mascarenhas, S. Immunomodulatory Activity of Ouabain in Leishmania Leishmania Amazonensis-Infected Swiss Mice. Parasitol. Res. 2013, 112, 1313–1321. [Google Scholar] [CrossRef] [PubMed]
- Mosser, D.M.; Zhang, X. Activation of Murine Macrophages. Curr. Protoc. Immunol. 2008, 83, 14.2.1–14.2.8. [Google Scholar] [CrossRef]
- Gabriel, Á.; Valério-Bolas, A.; Palma-Marques, J.; Mourata-Gonçalves, P.; Ruas, P.; Dias-Guerreiro, T.; Santos-Gomes, G. Cutaneous Leishmaniasis: The Complexity of Host’s Effective Immune Response against a Polymorphic Parasitic Disease. J. Immunol. Res. 2019, 2019, 2603730. [Google Scholar] [CrossRef] [Green Version]
- Vouldoukis, I.; Bécherel, P.-A.; Riveros-Moreno, V.; Arock, M.; Da Silva, O.; Debré, P.; Mazier, D.; Mossalayi, M.D. Interleukin-10 and Interleukin-4 Inhibit Intracellular Killing of Leishmania infantum and Leishmania major by Human Macrophages by Decreasing Nitric Oxide Generation. Eur. J. Immunol. 1997, 27, 860–865. [Google Scholar] [CrossRef]
- Noleto Dias, C.; de Nunes, T.A.L.; de Sousa, J.M.S.; Costa, L.H.; Rodrigues, R.R.L.; Araújo, A.J.; Marinho Filho, J.D.B.; da Silva, M.V.; Oliveira, M.R.; de Carvalho, F.A.A.; et al. Methyl gallate: Selective antileishmanial activity correlates with host-cell directed effects. Chem. Biol. Interact. 2020, 320, 109026. [Google Scholar] [CrossRef]
- de Carvalho, R.D.C.V.; de Sousa, V.C.; Santos, L.P.; Dos Santos, I.L.; Diniz, R.C.; Rodrigues, R.R.L.; de Medeiros, M.D.G.F.; da Franca Rodrigues, K.A.; de Moraes Alves, M.M.; Arcanjo, D.D.R.; et al. Limonene-carvacrol: A combination of monoterpenes with enhanced antileishmanial activity. Toxicol. In Vitro 2021, 74, 105158. [Google Scholar] [CrossRef]
- Eaton, P.; Bittencourt, C.R.; Costa Silva, V.; Véras, L.M.C.; Costa, C.H.N.; Feio, M.J.; Leite, J.R.S.A. Anti-leishmanial activity of the antimicrobial peptide drs 01 observed in Leishmania infantum (Syn. Leishmania chagasi) cells. Nanomedicine 2014, 10, 483–490. [Google Scholar] [CrossRef]
- de Lima Nunes, T.A.; Costa, L.H.; De Sousa, J.M.S.; De Souza, V.M.R.; Rodrigues, R.R.L.; Val, M.D.C.A.; da Cunha Pereira, A.C.T.; Ferreira, G.P.; Da Silva, M.V.; Da Costa, J.M.A.R.; et al. Eugenia piauhiensis Vellaff. essential oil and γ-elemene its major constituent exhibit antileishmanial activity, promoting cell membrane damage and in vitro immunomodulation. Chem. Biol. Interact. 2021, 339, 109429. [Google Scholar] [PubMed]
- Bonatto, S.J.R.; Folador, A.; Aikawa, J.; Yamazaki, R.K.; Pizatto, N.; Oliveira, H.H.P.; Vecchi, R.; Curi, R.; Calder, P.C.; Fernandes, L.C. Lifelong exposure to dietary fish oil alters macrophage responses in walker 256 tumor-bearing rats. Cell Immunol. 2004, 231, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, G.A.; Cunha-Júnior, E.F.; Pinheiro, R.O.; da-Silva, S.A.G.; Canto-Cavalheiro, M.M.; da Silva, A.J.M.; Costa, P.R.R.; Netto, C.D.; Melo, R.C.N.; Almeida-Amaral, E.E.; et al. LQB-118, an orally active pterocarpanquinone, induces selective oxidative stress and apoptosis in Leishmania amazonensis. J. Antimicrob. Chemother. 2013, 68, 789–799. [Google Scholar] [CrossRef] [PubMed] [Green Version]






| Concentrations | % Cytotoxicity | |||
|---|---|---|---|---|
| Promastigotes | Axenic Amastigotes | RAW 264.7 Macrophages | Vero Cells | |
| 1.56 µM | 27.3 ± 2.16 b | 59.06 ± 2.4 c | - | - |
| 3.12 µM | 70.25 ± 2.2 c | 79.11 ± 3.7 c | - | - |
| 6.25 µM | 81.4 c | 100 c | - | - |
| 12.5 µM | 100 c | 100 c | - | - |
| 50 µM | - | - | 1.17 ± 0.09 | 1.14 ± 0.1 |
| 100 µM | - | - | 46.3 ± 4.11 b | 6.04 ± 0.41 |
| 200 µM | - | - | 56.58 ± 5.92 c | 36.2 ± 4.26 a |
| 400 µM | - | - | 81.7 ± 8.16 c | 69.18 ± 4.9 c |
| Compounds | RAW 264.7 | VERO Cells | Promastigotes | Axenic Amastigotes | Intramacrophagic Amastigotes | |||
|---|---|---|---|---|---|---|---|---|
| CC50 µM | CC50 µM | IC50 µM | SI | EC50 µM | SI | EC50 µM | SI | |
| β-ocimene | 114.5 ± 8.2 | 298.54 ± 7.17 | 2.78 ± 0.17 | 41.18 | 1.12 ± 0.05 | 102.23 | 0.89 ± 0.03 | 128.65 |
| Amphotericin B | 0.39 ± 0.03 | 2.46 ± 0.6 | 0.35 ± 0.09 | 1.11 | 0.51 ± 0.02 | 0.76 | 0.13 ± 0.4 | 3 |
| Meglumine antimoniate | 15,863 ± 95.71 | 22,831 ± 103.61 | 2150 ± 31 | 0.73 | 1730 ± 36.8 | 9.16 | 577.5 ± 19.1 | 0.274 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sousa, J.M.S.d.; Nunes, T.A.d.L.; Rodrigues, R.R.L.; Sousa, J.P.A.d.; Val, M.d.C.A.; Coelho, F.A.d.R.; Santos, A.L.S.d.; Maciel, N.B.; Souza, V.M.R.d.; Machado, Y.A.A.; et al. Cytotoxic and Antileishmanial Effects of the Monoterpene β-Ocimene. Pharmaceuticals 2023, 16, 183. https://doi.org/10.3390/ph16020183
Sousa JMSd, Nunes TAdL, Rodrigues RRL, Sousa JPAd, Val MdCA, Coelho FAdR, Santos ALSd, Maciel NB, Souza VMRd, Machado YAA, et al. Cytotoxic and Antileishmanial Effects of the Monoterpene β-Ocimene. Pharmaceuticals. 2023; 16(2):183. https://doi.org/10.3390/ph16020183
Chicago/Turabian StyleSousa, Julyanne Maria Saraiva de, Thaís Amanda de Lima Nunes, Raiza Raianne Luz Rodrigues, João Paulo Araújo de Sousa, Maria da Conceição Albuquerque Val, Francisco Alex da Rocha Coelho, Airton Lucas Sousa dos Santos, Nicolle Barreira Maciel, Vanessa Maria Rodrigues de Souza, Yasmim Alves Aires Machado, and et al. 2023. "Cytotoxic and Antileishmanial Effects of the Monoterpene β-Ocimene" Pharmaceuticals 16, no. 2: 183. https://doi.org/10.3390/ph16020183