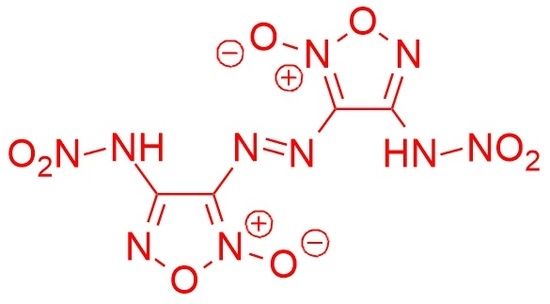
3,3′-(Diazene-1,2-diyl)bis[4-(nitroamino)-1,2,5-oxadiazole 2-oxide]
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. General Information
3.2. Synthesis of 3,3′-(Diazene-1,2-diyl)bis[4-(nitroamino)-1,2,5-oxadiazole 2-oxide] 1
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Wu, B.; Yang, H.; Lin, Q.; Wang, Z.; Lu, C.; Cheng, G. New thermally stable energetic materials: Synthesis and characterization of guanylhydrazone substituted furoxan energetic derivatives. New J. Chem. 2015, 39, 179–186. [Google Scholar] [CrossRef]
- Klapoetke, T.M.; Witkowski, T.G. Nitrogen-Rich Energetic 1,2,5-Oxadiazole-Tetrazole—Based Energetic Materials. Propellants Explos. Pyrotech. 2015, 40, 366–373. [Google Scholar] [CrossRef]
- Liang, L.; Wang, K.; Bian, C.; Ling, L.; Zhou, Z. 4-Nitro-3-(5-tetrazole)furoxan and Its Salts: Synthesis, Characterization, and Energetic Properties. Chem. Eur. J. 2013, 19, 14902–14910. [Google Scholar] [CrossRef] [PubMed]
- Fischer, D.; Klapoetke, T.M.; Stierstorfer, J. Synthesis and Characterization of Diaminobisfuroxane. Eur. J. Inorg. Chem. 2014, 2014, 5808–5811. [Google Scholar] [CrossRef]
- He, C.; Shreeve, J.M. Potassium 4,5-Bis(dinitromethyl)furoxanate: A Green Primary Explosive with a Positive Oxygen Balance. Angew. Chem. Int. Ed. 2016, 55, 772–775. [Google Scholar] [CrossRef] [PubMed]
- Ovchinnikov, I.V.; Makhova, N.N.; Khmel’nitskii, L.I.; Kuz’min, V.S.; Akimova, L.N.; Pepekin, V.I. Dinitrodiazenofuroxan as a new energetic explosive. Dokl. Chem. 1998, 359, 67–70. [Google Scholar]
- Stepanov, A.I.; Dashko, D.V.; Astrat’ev, A.A. 3,4-Bis(4′-nitrofurazan-3′-yl)furoxan: A Melt Cast Powerful Explosive and a Valuable Building Block in 1,2,5-Oxadiazole Chemistry. Cent. Eur. J. Energ. Mater. 2012, 9, 329–342. [Google Scholar]
- He, C.; Gao, H.; Imler, G.H.; Parrish, D.A.; Shreeve, J.M. Boosting energetic performance by trimerizing furoxan. J. Mater. Chem. A 2018, 6, 9391–9396. [Google Scholar] [CrossRef]
- He, C.; Tang, Y.; Mitchell, L.A.; Parrish, D.A.; Shreeve, J.M. N-Oxides light up energetic performances: Synthesis and characterization of dinitraminobisfuroxans and their salts. J. Mater. Chem. A 2016, 4, 8969–8973. [Google Scholar] [CrossRef]
- Fershtat, L.L.; Epishina, M.A.; Kulikov, A.S.; Ovchinnikov, I.V.; Ananyev, I.V.; Makhova, N.N. An efficient access to (1H-tetrazol-5-yl)furoxan ammonium salts via a two-step dehydration/[3+2]-cycloaddition strategy. Tetrahedron 2015, 71, 6764–6775. [Google Scholar] [CrossRef]
- Fershtat, L.L.; Makhova, N.N. Advances in the synthesis of non-annelated polynuclear heterocyclic systems comprising the 1,2,5-oxadiazole ring. Russ. Chem. Rev. 2016, 85, 1097–1145. [Google Scholar] [CrossRef]
- Zlotin, S.G.; Churakov, A.M.; Dalinger, I.L.; Luk’yanov, O.A.; Makhova, N.N.; Sukhorukov, A.Y.; Tartakovsky, V.A. Recent advances in synthesis of organic nitrogen–oxygen systems for medicine and material science. Mendeleev Commun. 2017, 27, 535–546. [Google Scholar] [CrossRef]
- Kuchurov, I.V.; Zharkov, M.N.; Fershtat, L.L.; Makhova, N.N.; Zlotin, S.G. Prospective Symbiosis of Green Chemistry and Energetic Materials. ChemSusChem 2017, 10, 3914–3946. [Google Scholar] [CrossRef] [PubMed]
- Fershtat, L.L.; Ovchinnikov, I.V.; Epishina, M.A.; Romanova, A.A.; Lempert, D.B.; Muravyev, N.V.; Makhova, N.N. Assembly of Nitrofurazan and Nitrofuroxan Frameworks for High-Performance Energetic Materials. ChemPlusChem 2017, 82, 1315–1319. [Google Scholar] [CrossRef]

| Entry | Nitrating Agent | Additive | Solvent | Temperature, °C | Time, h | Yield, a % |
|---|---|---|---|---|---|---|
| 1 | HNO3 b (145 equiv.) | - | - | 0 | 1 | - c |
| 2 | HNO3 b (72 equiv.) | (CF3CO)2O (22 equiv.) | - | −10 | 1 | - c |
| 3 | HNO3 b (15 equiv.) | Ac2O (3 equiv.) | CCl4 | 0, then 20 | 2 | 28 |
| 4 | HNO3 b (15 equiv.) | (CF3CO)2O (2.2 equiv.) | CCl4 | 0 | 1 | 57 |
| 5 | HNO3 b (15 equiv.) | (CF3CO)2 O(3 equiv.) | CCl4 | −5 | 0.5 | 80 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Larin, A.; Ovchinnikov, I.; Fershtat, L.; Makhova, N. 3,3′-(Diazene-1,2-diyl)bis[4-(nitroamino)-1,2,5-oxadiazole 2-oxide]. Molbank 2018, 2018, M1003. https://doi.org/10.3390/M1003
Larin A, Ovchinnikov I, Fershtat L, Makhova N. 3,3′-(Diazene-1,2-diyl)bis[4-(nitroamino)-1,2,5-oxadiazole 2-oxide]. Molbank. 2018; 2018(3):M1003. https://doi.org/10.3390/M1003
Chicago/Turabian StyleLarin, Alexander, Igor Ovchinnikov, Leonid Fershtat, and Nina Makhova. 2018. "3,3′-(Diazene-1,2-diyl)bis[4-(nitroamino)-1,2,5-oxadiazole 2-oxide]" Molbank 2018, no. 3: M1003. https://doi.org/10.3390/M1003