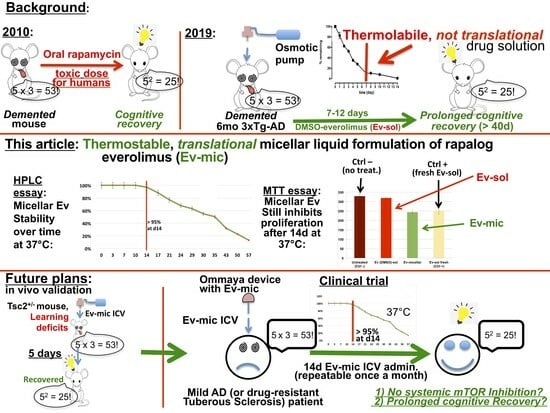
A Stable Micellar Formulation of RAD001 for Intracerebroventricular Delivery and the Treatment of Alzheimer’s Disease and Other Neurological Disorders
Abstract
:1. Introduction
2. Results
2.1. Validation of the HPLC Method
2.2. Characterisation of RAD001-Loaded Micelles
2.3. Evaluation of Ev-Sol and Ev-Mic Stability
2.4. Toxicity of Empty Micelles
2.5. Evaluation of Ev-Sol and Ev-Mic Stability Using Cell Cultures
3. Discussion
4. Materials and Methods
4.1. Preparation of RAD001 Stock, Standard, and Working Solutions
4.2. Preparation of RAD001-Loaded Micelles
4.3. HPLC Method Validation and RAD001 Quantification
4.3.1. HPLC Method Validation
4.3.2. RAD001 Quantification
4.3.3. RAD001 Solubility and Micelle Loading Efficiency
4.4. Micelle Size
4.5. Micelle Stability
4.6. Cell Cultures
4.7. Micelle Cytotoxicity Test
4.8. Functional Stability of Incubated RAD001 Micelles In Vitro
4.9. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nguyen, L.S.; Vautier, M.; Allenbach, Y.; Zahr, N.; Benveniste, O.; Funck-Brentano, C.; Salem, J.-E. Sirolimus and mTOR Inhibitors: A Review of Side Effects and Specific Management in Solid Organ Transplantation. Drug Saf. 2019, 42, 813–825. [Google Scholar] [CrossRef]
- Dumont, F.J.; Su, Q. Mechanism of Action of the Immunosuppressant Rapamycin. Life Sci. 1995, 58, 373–395. [Google Scholar] [CrossRef] [PubMed]
- Curatolo, P.; Bombardieri, R.; Jozwiak, S. Tuberous Sclerosis. Lancet Lond. Engl. 2008, 372, 657–668. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Shrikhande, G.; Xu, J.; McKay, R.M.; Burns, D.K.; Johnson, J.E.; Parada, L.F. Tsc1 Mutant Neural Stem/Progenitor Cells Exhibit Migration Deficits and Give Rise to Subependymal Lesions in the Lateral Ventricle. Genes Dev. 2011, 25, 1595–1600. [Google Scholar] [CrossRef] [PubMed]
- Franz, D.N.; Leonard, J.; Tudor, C.; Chuck, G.; Care, M.; Sethuraman, G.; Dinopoulos, A.; Thomas, G.; Crone, K.R. Rapamycin Causes Regression of Astrocytomas in Tuberous Sclerosis Complex. Ann. Neurol. 2006, 59, 490–498. [Google Scholar] [CrossRef] [PubMed]
- Franz, D.N.; Belousova, E.; Sparagana, S.; Bebin, E.M.; Frost, M.; Kuperman, R.; Witt, O.; Kohrman, M.H.; Flamini, J.R.; Wu, J.Y.; et al. Efficacy and Safety of Everolimus for Subependymal Giant Cell Astrocytomas Associated with Tuberous Sclerosis Complex (EXIST-1): A Multicentre, Randomised, Placebo-Controlled Phase 3 Trial. Lancet Lond. Engl. 2013, 381, 125–132. [Google Scholar] [CrossRef]
- French, J.A.; Lawson, J.A.; Yapici, Z.; Ikeda, H.; Polster, T.; Nabbout, R.; Curatolo, P.; de Vries, P.J.; Dlugos, D.J.; Berkowitz, N.; et al. Adjunctive Everolimus Therapy for Treatment-Resistant Focal-Onset Seizures Associated with Tuberous Sclerosis (EXIST-3): A Phase 3, Randomised, Double-Blind, Placebo-Controlled Study. Lancet Lond. Engl. 2016, 388, 2153–2163. [Google Scholar] [CrossRef]
- Chapman, N.M.; Chi, H. mTOR Signaling, Tregs and Immune Modulation. Immunotherapy 2014, 6, 1295–1311. [Google Scholar] [CrossRef] [PubMed]
- Silk, K.M.; Leishman, A.J.; Nishimoto, K.P.; Reddy, A.; Fairchild, P.J. Rapamycin Conditioning of Dendritic Cells Differentiated from Human ES Cells Promotes a Tolerogenic Phenotype. J. Biomed. Biotechnol. 2012, 2012, 172420. [Google Scholar] [CrossRef]
- Stallone, G.; Infante, B.; Di Lorenzo, A.; Rascio, F.; Zaza, G.; Grandaliano, G. mTOR Inhibitors Effects on Regulatory T Cells and on Dendritic Cells. J. Transl. Med. 2016, 14, 152. [Google Scholar] [CrossRef] [PubMed]
- Turnquist, H.R.; Raimondi, G.; Zahorchak, A.F.; Fischer, R.T.; Wang, Z.; Thomson, A.W. Rapamycin-Conditioned Dendritic Cells Are Poor Stimulators of Allogeneic CD4+ T Cells, but Enrich for Antigen-Specific Foxp3+ T Regulatory Cells and Promote Organ Transplant Tolerance. J. Immunol. Baltim. Md 1950 2007, 178, 7018–7031. [Google Scholar] [CrossRef] [PubMed]
- Fischer, R.; Turnquist, H.R.; Taner, T.; Thomson, A.W. Use of Rapamycin in the Induction of Tolerogenic Dendritic Cells. In Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2009; pp. 215–232. [Google Scholar] [CrossRef]
- Macedo, C.; Turquist, H.; Metes, D.; Thomson, A.W. Immunoregulatory Properties of Rapamycin-Conditioned Monocyte-Derived Dendritic Cells and Their Role in Transplantation. Transplant. Res. 2012, 1, 16. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Ruffini, F.; Bellone, M.; Gagliani, N.; Battaglia, M.; Martino, G.; Furlan, R. Rapamycin Inhibits Relapsing Experimental Autoimmune Encephalomyelitis by Both Effector and Regulatory T Cells Modulation. J. Neuroimmunol. 2010, 220, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Hou, H.; Miao, J.; Cao, R.; Han, M.; Sun, Y.; Liu, X.; Guo, L. Rapamycin Ameliorates Experimental Autoimmune Encephalomyelitis by Suppressing the mTOR-STAT3 Pathway. Neurochem. Res. 2017, 42, 2831–2840. [Google Scholar] [CrossRef] [PubMed]
- Dello Russo, C.; Lisi, L.; Feinstein, D.L.; Navarra, P. mTOR Kinase, a Key Player in the Regulation of Glial Functions: Relevance for the Therapy of Multiple Sclerosis. Glia 2013, 61, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Nie, L.; Chen, L.; Sun, Y.; Li, G. Rapamycin Relieves Inflammation of Experimental Autoimmune Encephalomyelitis by Altering the Balance of Treg/Th17 in a Mouse Model. Neurosci. Lett. 2019, 705, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-L.; Zhang, B.; Liu, W.; Sun, M.-J.; Zhang, Y.-L.; Liu, H.; Wang, M.-X. Rapamycin Alleviates the Symptoms of Multiple Sclerosis in Experimental Autoimmune Encephalomyelitis (EAE) Through Mediating the TAM-TLRs-SOCS Pathway. Front. Neurol. 2020, 11, 590884. [Google Scholar] [CrossRef] [PubMed]
- Vakrakou, A.G.; Alexaki, A.; Brinia, M.-E.; Anagnostouli, M.; Stefanis, L.; Stathopoulos, P. The mTOR Signaling Pathway in Multiple Sclerosis; from Animal Models to Human Data. Int. J. Mol. Sci. 2022, 23, 8077. [Google Scholar] [CrossRef] [PubMed]
- Bagherpour, B.; Salehi, M.; Jafari, R.; Bagheri, A.; Kiani-Esfahani, A.; Edalati, M.; Kardi, M.T.; Shaygannejad, V. Promising Effect of Rapamycin on Multiple Sclerosis. Mult. Scler. Relat. Disord. 2018, 26, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Monti, P.; Scirpoli, M.; Maffi, P.; Piemonti, L.; Secchi, A.; Bonifacio, E.; Roncarolo, M.-G.; Battaglia, M. Rapamycin Monotherapy in Patients with Type 1 Diabetes Modifies CD4+CD25+FOXP3+ Regulatory T-Cells. Diabetes 2008, 57, 2341–2347. [Google Scholar] [CrossRef] [PubMed]
- Caccamo, A.; Majumder, S.; Richardson, A.; Strong, R.; Oddo, S. Molecular Interplay between Mammalian Target of Rapamycin (mTOR), Amyloid-Beta, and Tau: Effects on Cognitive Impairments. J. Biol. Chem. 2010, 285, 13107–13120. [Google Scholar] [CrossRef]
- Oddo, S.; Caccamo, A.; Shepherd, J.D.; Murphy, M.P.; Golde, T.E.; Kayed, R.; Metherate, R.; Mattson, M.P.; Akbari, Y.; LaFerla, F.M. Triple-Transgenic Model of Alzheimer’s Disease with Plaques and Tangles: Intracellular Abeta and Synaptic Dysfunction. Neuron 2003, 39, 409–421. [Google Scholar] [CrossRef] [PubMed]
- Haage, V.; De Jager, P.L. Neuroimmune Contributions to Alzheimer’s Disease: A Focus on Human Data. Mol. Psychiatry 2022, 27, 3164–3181. [Google Scholar] [CrossRef] [PubMed]
- Spilman, P.; Podlutskaya, N.; Hart, M.J.; Debnath, J.; Gorostiza, O.; Bredesen, D.; Richardson, A.; Strong, R.; Galvan, V. Inhibition of mTOR by Rapamycin Abolishes Cognitive Deficits and Reduces Amyloid-Beta Levels in a Mouse Model of Alzheimer’s Disease. PLoS ONE 2010, 5, e9979. [Google Scholar] [CrossRef]
- Lin, A.-L.; Zheng, W.; Halloran, J.J.; Burbank, R.R.; Hussong, S.A.; Hart, M.J.; Javors, M.; Shih, Y.-Y.I.; Muir, E.; Fonseca, R.S.; et al. Chronic Rapamycin Restores Brain Vascular Integrity and Function Through NO Synthase Activation and Improves Memory in Symptomatic Mice Modeling Alzheimer’s Disease. J. Cereb. Blood Flow Metab. 2013, 33, 1412–1421. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.-L.; Jahrling, J.B.; Zhang, W.; DeRosa, N.; Bakshi, V.; Romero, P.; Galvan, V.; Richardson, A. Rapamycin Rescues Vascular, Metabolic and Learning Deficits in Apolipoprotein E4 Transgenic Mice with Pre-Symptomatic Alzheimer’s Disease. J. Cereb. Blood Flow Metab. Off. J. Int. Soc. Cereb. Blood Flow Metab. 2017, 37, 217–226. [Google Scholar] [CrossRef]
- Harrison, D.E.; Strong, R.; Sharp, Z.D.; Nelson, J.F.; Astle, C.M.; Flurkey, K.; Nadon, N.L.; Wilkinson, J.E.; Frenkel, K.; Carter, C.S.; et al. Rapamycin Fed Late in Life Extends Lifespan in Genetically Heterogeneous Mice. Nature 2009, 460, 392–395. [Google Scholar] [CrossRef]
- Dolcetta, D.; Dominici, R. The Local Mammalian Target of Rapamycin (mTOR) Modulation: A Promising Strategy to Counteract Neurodegeneration. Neural Regen. Res. 2019, 14, 1711–1712. [Google Scholar] [CrossRef]
- Cassano, T.; Magini, A.; Giovagnoli, S.; Polchi, A.; Calcagnini, S.; Pace, L.; Lavecchia, M.A.; Scuderi, C.; Bronzuoli, M.R.; Ruggeri, L.; et al. Early Intrathecal Infusion of Everolimus Restores Cognitive Function and Mood in a Murine Model of Alzheimer’s Disease. Exp. Neurol. 2019, 311, 88–105. [Google Scholar] [CrossRef]
- Gill, K.K.; Kaddoumi, A.; Nazzal, S. Mixed Micelles of PEG(2000)-DSPE and Vitamin-E TPGS for Concurrent Delivery of Paclitaxel and Parthenolide: Enhanced Chemosenstization and Antitumor Efficacy against Non-Small Cell Lung Cancer (NSCLC) Cell Lines. Eur. J. Pharm. Sci. Off. J. Eur. Fed. Pharm. Sci. 2012, 46, 64–71. [Google Scholar] [CrossRef]
- Gill, K.K.; Kaddoumi, A.; Nazzal, S. PEG-Lipid Micelles as Drug Carriers: Physiochemical Attributes, Formulation Principles and Biological Implication. J. Drug Target. 2015, 23, 222–231. [Google Scholar] [CrossRef]
- Meguri, Y.; Asano, T.; Yoshioka, T.; Iwamoto, M.; Ikegawa, S.; Sugiura, H.; Kishi, Y.; Nakamura, M.; Sando, Y.; Kondo, T.; et al. Responses of Regulatory and Effector T-Cells to Low-Dose Interleukin-2 Differ Depending on the Immune Environment after Allogeneic Stem Cell Transplantation. Front. Immunol. 2022, 13, 891925. [Google Scholar] [CrossRef]
- Kastantin, M.; Ananthanarayanan, B.; Karmali, P.; Ruoslahti, E.; Tirrell, M. Effect of the Lipid Chain Melting Transition on the Stability of DSPE-PEG(2000) Micelles. Langmuir ACS J. Surf. Colloids 2009, 25, 7279–7286. [Google Scholar] [CrossRef] [PubMed]
- Kastantin, M.; Missirlis, D.; Black, M.; Ananthanarayanan, B.; Peters, D.; Tirrell, M. Thermodynamic and Kinetic Stability of DSPE-PEG(2000) Micelles in the Presence of Bovine Serum Albumin. J. Phys. Chem. B 2010, 114, 12632–12640. [Google Scholar] [CrossRef] [PubMed]
- Vuković, L.; Khatib, F.A.; Drake, S.P.; Madriaga, A.; Brandenburg, K.S.; Král, P.; Onyuksel, H. Structure and Dynamics of Highly PEG-Ylated Sterically Stabilized Micelles in Aqueous Media. J. Am. Chem. Soc. 2011, 133, 13481–13488. [Google Scholar] [CrossRef] [PubMed]
- Remsberg, C.M.; Zhao, Y.; Takemoto, J.K.; Bertram, R.M.; Davies, N.M.; Forrest, M.L. Pharmacokinetic Evaluation of a DSPE-PEG2000 Micellar Formulation of Ridaforolimus in Rat. Pharmaceutics 2012, 5, 81–93. [Google Scholar] [CrossRef] [PubMed]
- EMEA CHMP Assessment Report for Afinitor. International Nonproprietary Name: Everolimus Procedure No. EMEA/H/C/001038. 2009. Available online: https://www.ema.europa.eu/en/documents/assessment-report/afinitor-epar-public-assessment-report_en.pdf (accessed on 29 May 2009).
- Novartis FDA. Drug Approval Package Afinitor Disperz (Everolimus Tablets for Oral Suspension), 2 mg, 3 mg and 5 mg. 2013. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2012/203985_afinitor_toc.cfm (accessed on 12 November 2013).
- Takayama, R.; Inoue, Y.; Murata, I.; Kanamoto, I. Characterization of Nanoparticles Using DSPE-PEG2000 and Soluplus. Colloids Interfaces 2020, 4, 28. [Google Scholar] [CrossRef]
- Garcia-Alcantara, E.; Lopez-Callejas, R.; Serment-Guerrero, J.; Pena-Eguiluz, R.; Munoz-Castro, A.E.; Rodriguez-Mendez, B.G.; Mercado-Cabrera, A.; Valencia-Alvarado, R.; De-la-Piedad-Beneitez, A.; Contreras-Ortiz, J.M.E.; et al. Toxicity and Genotoxicity in HeLa and E. Coli Cells Caused by a Helium Plasma Needle. Appl. Phys. Res. 2013, 5, 21. [Google Scholar] [CrossRef]
- Bich-Loan, N.T.; Kien, K.T.; Thanh, N.L.; Kim-Thanh, N.T.; Huy, N.Q.; The-Hai, P.; Muller, M.; Nachtergael, A.; Duez, P.; Thang, N.D. Toxicity and Anti-Proliferative Properties of Anisomeles Indica Ethanol Extract on Cervical Cancer HeLa Cells and Zebrafish Embryos. Life 2021, 11, 257. [Google Scholar] [CrossRef]
- Xicoy, H.; Wieringa, B.; Martens, G.J.M. The SH-SY5Y Cell Line in Parkinson’s Disease Research: A Systematic Review. Mol. Neurodegener. 2017, 12, 10. [Google Scholar] [CrossRef]
- Lopez-Suarez, L.; Awabdh, S.A.; Coumoul, X.; Chauvet, C. The SH-SY5Y Human Neuroblastoma Cell Line, a Relevant in Vitro Cell Model for Investigating Neurotoxicology in Human: Focus on Organic Pollutants. NeuroToxicology 2022, 92, 131–155. [Google Scholar] [CrossRef] [PubMed]
- Sedrani, R.; Cottens, S.; Kallen, J.; Schuler, W. Chemical Modification of Rapamycin: The Discovery of SDZ RAD. Transplant. Proc. 1998, 30, 2192–2194. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, M.G.; Tsai, V.W.W.; Ruitenberg, M.J.; Hassanpour, M.; Li, H.; Hart, P.H.; Breit, S.N.; Sawchenko, P.E.; Brown, D.A. Immune Cell Trafficking from the Brain Maintains CNS Immune Tolerance. J. Clin. Investig. 2014, 124, 1228–1241. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Park, S.-Y.; Baek, H.; Lee, C.; Chung, G.; Liu, X.; Lee, J.H.; Kim, B.; Kwon, M.; Choi, H.; et al. Adoptive Therapy with Amyloid-β Specific Regulatory T Cells Alleviates Alzheimer’s Disease. Theranostics 2022, 12, 7668–7680. [Google Scholar] [CrossRef]
- Faridar, A.; Thome, A.D.; Zhao, W.; Thonhoff, J.R.; Beers, D.R.; Pascual, B.; Masdeu, J.C.; Appel, S.H. Restoring Regulatory T-Cell Dysfunction in Alzheimer’s Disease through Ex Vivo Expansion. Brain Commun. 2020, 2, fcaa112. [Google Scholar] [CrossRef] [PubMed]
- Weaver, D.F. Alzheimer’s Disease as an Innate Autoimmune Disease (AD2): A New Molecular Paradigm. Alzheimers Dement. J. Alzheimers Assoc. 2022, 19, 1086–1098. [Google Scholar] [CrossRef]
- Liston, A.; Dooley, J.; Yshii, L. Brain-Resident Regulatory T Cells and Their Role in Health and Disease. Immunol. Lett. 2022, 248, 26–30. [Google Scholar] [CrossRef]
- Figueiras, A.; Domingues, C.; Jarak, I.; Santos, A.I.; Parra, A.; Pais, A.; Alvarez-Lorenzo, C.; Concheiro, A.; Kabanov, A.; Cabral, H.; et al. New Advances in Biomedical Application of Polymeric Micelles. Pharmaceutics 2022, 14, 1700. [Google Scholar] [CrossRef]
- Negut, I.; Bita, B. Polymeric Micellar Systems-A Special Emphasis on “Smart” Drug Delivery. Pharmaceutics 2023, 15, 976. [Google Scholar] [CrossRef]
- Wang, Q.; Atluri, K.; Tiwari, A.K.; Babu, R.J. Exploring the Application of Micellar Drug Delivery Systems in Cancer Nanomedicine. Pharmaceuticals 2023, 16, 433. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Huang, J.; Leng, D.; Yang, S.; Yao, Q.; Sun, J.; Hu, J. Gefitinib-Loaded DSPE-PEG2000 Nanomicelles with CD133 Aptamers Target Lung Cancer Stem Cells. World J. Surg. Oncol. 2017, 15, 167. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Feng, Q.; Ding, C.; Kang, W.; Xiao, X.; Yu, Y.; Zhou, Q. Controllable Drug Delivery by Na+/K+ ATPase A1 Targeting Peptide Conjugated DSPE-PEG Nanocarriers for Breast Cancer. Technol. Cancer Res. Treat. 2021, 20, 15330338211027898. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Wang, J.; Zhang, S.; Li, Y.; Wang, B.; Zhang, J.; Qiu, Y.; Zhang, Y.; Zhang, Y. Preparation of Amentoflavone-loaded DSPE-PEG 2000 Micelles with Improved Bioavailability and in Vitro Antitumor Efficacy. Biomed. Chromatogr. 2023, 37, e5690. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Bai, L.; Yang, Y.; Wang, Y.; Xu, P.; Zhao, Y.; Zhou, X.; Li, X.; Xue, M. Long-Circulation and Brain Targeted Isoliquiritigenin Micelle Nanoparticles: Formation, Characterization, Tissue Distribution, Pharmacokinetics and Effects for Ischemic Stroke. Int. J. Nanomedicine 2022, 17, 3655–3670. [Google Scholar] [CrossRef] [PubMed]
- Cox, A.; Tung, M.; Li, H.; Hallows, K.R.; Chung, E.J. In Vitro Delivery of mTOR Inhibitors by Kidney-Targeted Micelles for Autosomal Dominant Polycystic Kidney Disease. SLAS Technol. 2023, 28, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, A.J. Intracerebroventricular Drug Administration. Transl. Clin. Pharmacol. 2017, 25, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Chaichana, K.L.; Pinheiro, L.; Brem, H. Delivery of Local Therapeutics to the Brain: Working toward Advancing Treatment for Malignant Gliomas. Ther. Deliv. 2015, 6, 353–369. [Google Scholar] [CrossRef]
- Peyrl, A.; Chocholous, M.; Azizi, A.A.; Czech, T.; Dorfer, C.; Mitteregger, D.; Gojo, J.; Minichmayr, E.; Slavc, I. Safety of Ommaya Reservoirs in Children with Brain Tumors: A 20-Year Experience with 5472 Intraventricular Drug Administrations in 98 Patients. J. Neurooncol. 2014, 120, 139–145. [Google Scholar] [CrossRef]
- Giordan, E.; Palandri, G.; Lanzino, G.; Murad, M.H.; Elder, B.D. Outcomes and Complications of Different Surgical Treatments for Idiopathic Normal Pressure Hydrocephalus: A Systematic Review and Meta-Analysis. J. Neurosurg. 2018, 131, 1024–1036. [Google Scholar] [CrossRef]
- Kocak, M.; Ezazi Erdi, S.; Jorba, G.; Maestro, I.; Farrés, J.; Kirkin, V.; Martinez, A.; Pless, O. Targeting Autophagy in Disease: Established and New Strategies. Autophagy 2022, 18, 473–495. [Google Scholar] [CrossRef]
- Zhang, Z.; Fan, Q.; Luo, X.; Lou, K.; Weiss, W.A.; Shokat, K.M. Brain-Restricted mTOR Inhibition with Binary Pharmacology. Nature 2022, 609, 822–828. [Google Scholar] [CrossRef] [PubMed]
- Alruwais, N.M.; Rusted, J.M.; Tabet, N.; Dowell, N.G. Evidence of Emerging BBB Changes in Mid-Age Apolipoprotein E Epsilon-4 Carriers. Brain Behav. 2022, 12, e2806. [Google Scholar] [CrossRef] [PubMed]
- Patil, R.; Galstyan, A.; Grodzinski, Z.B.; Shatalova, E.S.; Wagner, S.; Israel, L.L.; Ding, H.; Black, K.L.; Ljubimova, J.Y.; Holler, E. Single- and Multi-Arm Gadolinium MRI Contrast Agents for Targeted Imaging of Glioblastoma. Int. J. Nanomedicine 2020, 15, 3057–3070. [Google Scholar] [CrossRef] [PubMed]
- ICH Expert Working Group. Validation of Analytical Procedures: Text and Methodology Q2(R1). In ICH Harmonised Tripartite Guideline; ICH: Geneva, Switzerland, 2005. [Google Scholar]
- Prasad, S.S.; Krishna Mohan, G.V.; Naga Babu, A. Development of Simple and Robust RP-HPLC Method for Determination of Everolimus and Its Impurities in Oral Solid Dosage Form. Asian J. Chem. 2019, 31, 1002–1008. [Google Scholar] [CrossRef]
- Sharmila, D.; Rao, A.L.; Kalyani, L. Development and Validation of Stability-Indicating High Performance Liquid Chromatographic Method for the Estimation of Everolimus in Tablets. Indian J. Pharm. Sci. 2015, 77, 599–604. [Google Scholar] [CrossRef]
- Giovagnoli, S.; Cassano, T.; Pace, L.; Magini, A.; Polchi, A.; Tancini, B.; Perluigi, M.; De Marco, F.; Emiliani, C.; Dolcetta, D. Evaluation of a LC-MS Method for Everolimus Preclinical Determination in Brain by Using [13C2D4]RAD001 Internal Standard. J. Chromatogr. B Analyt. Technol. Biomed. Life. Sci. 2015, 985, 155–163. [Google Scholar] [CrossRef]







| RADD001/Lipid w/w Ratio | RADD001 Molar Fraction | LE (%w/w) ± S.D. (n = 3) |
|---|---|---|
| 1/10 | 0.29 | 56.4 ± 4.5 |
| 1/20 | 0.14 | 98.7 ± 2.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gianessi, L.; Magini, A.; Dominici, R.; Giovagnoli, S.; Dolcetta, D. A Stable Micellar Formulation of RAD001 for Intracerebroventricular Delivery and the Treatment of Alzheimer’s Disease and Other Neurological Disorders. Int. J. Mol. Sci. 2023, 24, 17478. https://doi.org/10.3390/ijms242417478
Gianessi L, Magini A, Dominici R, Giovagnoli S, Dolcetta D. A Stable Micellar Formulation of RAD001 for Intracerebroventricular Delivery and the Treatment of Alzheimer’s Disease and Other Neurological Disorders. International Journal of Molecular Sciences. 2023; 24(24):17478. https://doi.org/10.3390/ijms242417478
Chicago/Turabian StyleGianessi, Laura, Alessandro Magini, Roberto Dominici, Stefano Giovagnoli, and Diego Dolcetta. 2023. "A Stable Micellar Formulation of RAD001 for Intracerebroventricular Delivery and the Treatment of Alzheimer’s Disease and Other Neurological Disorders" International Journal of Molecular Sciences 24, no. 24: 17478. https://doi.org/10.3390/ijms242417478