Imbalance in Sirt1 Alternative Splicing in Response to Chronic Stress during the Adolescence Period in Female Mice
Abstract
:1. Introduction
2. Results
2.1. Exposure to UCMS during Adolescence Shows Long-Term Effect on Anxiety-like Behavior in Both Males and Females
2.2. Exposure to UCMS during Adolescence Impaired Cognition Later in Life
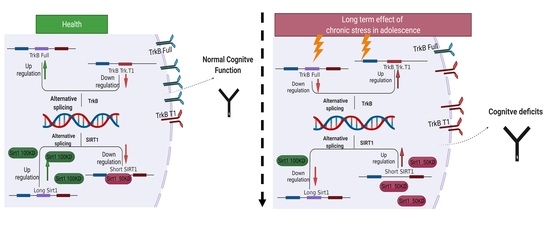
2.3. Imbalance in SIRT1 Alternative Splicing in Response to UCMS
2.4. Imbalance in TrkB Receptor Alternative Splicing in Response to UCMS
3. Discussion
4. Materials and Methods
4.1. Experimental Design
4.2. Animals
4.3. Unpredictable Chronic Mild Stress (UCMS) Manipulation
4.4. Open Field Test (OFT)
4.5. Elevated plus Maze (EPM)
4.6. Y-Maze
4.7. Western Blot (WB)
4.8. Real-Time PCR
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- McEwen, B.S. Physiology and Neurobiology of Stress and Adaptation: Central Role of the Brain. Physiol. Rev. 2007, 87, 873–904. [Google Scholar] [CrossRef] [Green Version]
- Kivimäki, M.; Virtanen, M.; Elovainio, M.; Kouvonen, A.; Väänänen, A.; Vahtera, J. Work Stress in the Etiology of Coronary Heart Disease—A Meta-Analysis. Scand. J. Work. Env. Health 2006, 32, 431–442. [Google Scholar] [CrossRef]
- Bower, J.E.; Crosswell, A.D.; Slavich, G.M. Childhood Adversity and Cumulative Life Stress: Risk Factors for Cancer-Related Fatigue. Clin. Psychol. Sci. USA 2014, 2, 108–115. [Google Scholar] [CrossRef] [Green Version]
- Baglietto-Vargas, D.; Chen, Y.; Suh, D.; Ager, R.R.; Rodriguez-Ortiz, C.J.; Medeiros, R.; Myczek, K.; Green, K.N.; Baram, T.Z.; LaFerla, F.M. Short-Term Modern Life-like Stress Exacerbates Aβ-Pathology and Synapse Loss in 3xTg-AD Mice. J. Neurochem. 2015, 134, 915–926. [Google Scholar] [CrossRef] [Green Version]
- Mineur, Y.S.; Belzung, C.; Crusio, W.E. Functional Implications of Decreases in Neurogenesis Following Chronic Mild Stress in Mice. Neuroscience 2007, 150, 251–259. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, Q.; Gu, J.; Wang, X.; Xie, K.; Xian, X.; Wang, J.; Jiang, H.; Wang, Z. Resveratrol Prevents Impaired Cognition Induced by Chronic Unpredictable Mild Stress in Rats. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2014, 49, 21–29. [Google Scholar] [CrossRef]
- Song, L.; Che, W.; Min-Wei, W.; Murakami, Y.; Matsumoto, K. Impairment of the Spatial Learning and Memory Induced by Learned Helplessness and Chronic Mild Stress. Pharmacol. Biochem. Behav. 2006, 83, 186–193. [Google Scholar] [CrossRef]
- Sakthivel, G.; Karunya, M.D.; Prajisha, P.; Keerthipriya, C.S.; Ravindran, R. Chronic unpredictable stress-induced behavioral and electrophysiological alterations and its ameliorative effect by myricetin microemulsion. Asian J. Pharm. Clin. Res. 2018, 11, 124. [Google Scholar] [CrossRef]
- Duman, R.S.; Malberg, J.; Thome, J. Neural Plasticity to Stress and Antidepressant Treatment. Biol. Psychiatry 1999, 46, 1181–1191. [Google Scholar] [CrossRef]
- McEwen, B.S. Stress and hippocampal plasticity. Annu. Rev. Neurosci. 1999, 22, 105–122. [Google Scholar] [CrossRef] [Green Version]
- Rajkowska, G.; Legutko, B.; Moulana, M.; Syed, M.; Romero, D.G.; Stockmeier, C.A.; Miguel-Hidalgo, J.J. Astrocyte Pathology in the Ventral Prefrontal White Matter in Depression. J. Psychiatr. Res. 2018, 102, 150–158. [Google Scholar] [CrossRef]
- Epel, E.S.; Blackburn, E.H.; Lin, J.; Dhabhar, F.S.; Adler, N.E.; Morrow, J.D.; Cawthon, R.M. Accelerated Telomere Shortening in Response to Life Stress. Proc. Natl. Acad. Sci. USA 2004, 101, 17312–17315. [Google Scholar] [CrossRef] [Green Version]
- Mengin, A.; Allé, M.C.; Rolling, J.; Ligier, F.; Schroder, C.; Lalanne, L.; Berna, F.; Jardri, R.; Vaiva, G.; Geoffroy, P.A.; et al. Conséquences psychopathologiques du confinement. L’Encéphale 2020, 46, S43–S52. [Google Scholar] [CrossRef]
- Cavaliere, G.; Trinchese, G.; Penna, E.; Cimmino, F.; Pirozzi, C.; Lama, A.; Annunziata, C.; Catapano, A.; Mattace Raso, G.; Meli, R.; et al. High-Fat Diet Induces Neuroinflammation and Mitochondrial Impairment in Mice Cerebral Cortex and Synaptic Fraction. Front. Cell. Neurosci. 2019, 13, 509. [Google Scholar] [CrossRef]
- de Figueiredo, C.S.; Sandre, P.C.; Portugal, L.C.L.; Mázala-de-Oliveira, T.; da Silva Chagas, L.; Raony, Í.; Ferreira, E.S.; Giestal-de-Araujo, E.; dos Santos, A.A.; Bomfim, P.O.-S. COVID-19 Pandemic Impact on Children and Adolescents’ Mental Health: Biological, Environmental, and Social Factors. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2021, 106, 110171. [Google Scholar] [CrossRef]
- Lupien, S.J.; McEwen, B.S.; Gunnar, M.R.; Heim, C. Effects of Stress throughout the Lifespan on the Brain, Behaviour and Cognition. Nat. Rev. Neurosci. 2009, 10, 434–445. [Google Scholar] [CrossRef]
- McCrory, E.; De Brito, S.A.; Viding, E. The Link between Child Abuse and Psychopathology: A Review of Neurobiological and Genetic Research. J. R. Soc. Med. 2012, 105, 151–156. [Google Scholar] [CrossRef]
- Elwenspoek, M.M.C.; Kuehn, A.; Muller, C.P.; Turner, J.D. The Effects of Early Life Adversity on the Immune System. Psychoneuroendocrinology 2017, 82, 140–154. [Google Scholar] [CrossRef]
- Chen, Y.; Baram, T.Z. Toward Understanding How Early-Life Stress Reprograms Cognitive and Emotional Brain Networks. Neuropsychopharmacology 2016, 41, 197–206. [Google Scholar] [CrossRef] [Green Version]
- Naninck, E.F.G.; Hoeijmakers, L.; Kakava-Georgiadou, N.; Meesters, A.; Lazic, S.E.; Lucassen, P.J.; Korosi, A. Chronic Early Life Stress Alters Developmental and Adult Neurogenesis and Impairs Cognitive Function in Mice: Chronic Early Life Stress Alters Neurogenesis and Cognition. Hippocampus 2015, 25, 309–328. [Google Scholar] [CrossRef]
- Bondar, N.P.; Lepeshko, A.A.; Reshetnikov, V.V. Effects of Early-Life Stress on Social and Anxiety-Like Behaviors in Adult Mice: Sex-Specific Effects. Behav. Neurol. 2018, 2018, e1538931. [Google Scholar] [CrossRef] [Green Version]
- van Eden, C.G.; Kros, J.M.; Uylings, H.B.M. Chapter 8 The Development of the Rat Prefrontal Cortex: Its Size and Development of Connections with Thalamus, Spinal Cord and Other Cortical Areas. In Progress in Brain Research; Elsevier: Amsterdam, The Netherlands, 1991; Volume 85, pp. 169–183. ISBN 978-0-444-81124-0. [Google Scholar]
- McCormick, C.M.; Mathews, I.Z. Adolescent Development, Hypothalamic-Pituitary-Adrenal Function, and Programming of Adult Learning and Memory. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2010, 34, 756–765. [Google Scholar] [CrossRef]
- Thompson, J.L.; Pogue-Geile, M.F.; Grace, A.A. Developmental Pathology, Dopamine, and Stress: A Model for the Age of Onset of Schizophrenia Symptoms. Schizophr. Bull. 2004, 30, 875–900. [Google Scholar] [CrossRef]
- Leussis, M.P.; Andersen, S.L. Is Adolescence a Sensitive Period for Depression? Behavioral and Neuroanatomical Findings from a Social Stress Model. Synapse 2008, 62, 22–30. [Google Scholar] [CrossRef]
- Kim, S.; Gacek, S.A.; Mocchi, M.M.; Redei, E.E. Sex-Specific Behavioral Response to Early Adolescent Stress in the Genetically More Stress-Reactive Wistar Kyoto More Immobile, and Its Nearly Isogenic Wistar Kyoto Less Immobile Control Strain. Front. Behav. Neurosci. 2021, 15, 779036. [Google Scholar] [CrossRef]
- Ji, M.T.; Pashnkar, N.; Pezybl, K.J.; Redei, E.E. Sexually Dimorphic Effects of Adolescent Stress in an X-Linked Congenic Strain Showing Passive Coping and Increased Anxiety; Society for Neuroscience: Chicago, IL, USA, 2021. [Google Scholar]
- Ducottet, C.; Belzung, C. Behaviour in the Elevated Plus-Maze Predicts Coping after Subchronic Mild Stress in Mice. Physiol. Behav. 2004, 81, 417–426. [Google Scholar] [CrossRef]
- Burstein, O.; Franko, M.; Gale, E.; Handelsman, A.; Barak, S.; Motsan, S.; Shamir, A.; Toledano, R.; Simhon, O.; Hirshler, Y.; et al. Escitalopram and NHT Normalized Stress-Induced Anhedonia and Molecular Neuroadaptations in a Mouse Model of Depression. PLoS ONE 2017, 12, e0188043. [Google Scholar] [CrossRef] [Green Version]
- Dincheva, I.; Glatt, C.E.; Lee, F.S. Impact of the BDNF Val66Met Polymorphism on Cognition: Implications for Behavioral Genetics. Neuroscientist 2012, 18, 439–451. [Google Scholar] [CrossRef]
- Eide, F.F.; Vining, E.R.; Eide, B.L.; Zang, K.; Wang, X.-Y.; Reichardt, L.F. Naturally Occurring Truncated TrkB Receptors Have Dominant Inhibitory Effects on Brain-Derived Neurotrophic Factor Signaling. J. Neurosci. 1996, 16, 3123–3129. [Google Scholar] [CrossRef]
- Vidaurre, O.G.; Gascón, S.; Deogracias, R.; Sobrado, M.; Cuadrado, E.; Montaner, J.; Rodríguez-Peña, A.; Díaz-Guerra, M. Imbalance of Neurotrophin Receptor Isoforms TrkB-FL/TrkB-T1 Induces Neuronal Death in Excitotoxicity. Cell Death Dis. 2012, 3, e256. [Google Scholar] [CrossRef] [Green Version]
- Cao, T.; Matyas, J.J.; Renn, C.L.; Faden, A.I.; Dorsey, S.G.; Wu, J. Function and Mechanisms of Truncated BDNF Receptor TrkB.T1 in Neuropathic Pain. Cells 2020, 9, 1194. [Google Scholar] [CrossRef]
- Ferrer, I.; Marín, C.; Rey, M.J.; Ribalta, T.; Goutan, E.; Blanco, R.; Tolosa, E.; Martí, E. BDNF and Full-Length and Truncated TrkB Expression in Alzheimer Disease. Implications in Therapeutic Strategies. J. Neuropathol. Exp. Neurol. 1999, 58, 729–739. [Google Scholar] [CrossRef]
- Weissleder, C.; Kondo, M.A.; Yang, C.; Fung, S.J.; Rothmond, D.A.; Wong, M.W.; Halliday, G.M.; Herman, M.M.; Kleinman, J.E.; Webster, M.J.; et al. Early-Life Decline in Neurogenesis Markers and Age-Related Changes of TrkB Splice Variant Expression in the Human Subependymal Zone. Eur. J. Neurosci. 2017, 46, 1768–1778. [Google Scholar] [CrossRef]
- Quintas, A.; de Solís, A.J.; Díez-Guerra, F.J.; Carrascosa, J.M.; Bogónez, E. Age-Associated Decrease of SIRT1 Expression in Rat Hippocampus. Exp. Gerontol. 2012, 47, 198–201. [Google Scholar] [CrossRef]
- Satoh, A.; Brace, C.S.; Rensing, N.; Cliften, P.; Wozniak, D.F.; Herzog, E.D.; Yamada, K.A.; Imai, S. Sirt1 Extends Life Span and Delays Aging in Mice through the Regulation of Nk2 Homeobox 1 in the DMH and LH. Cell Metab. 2013, 18, 416–430. [Google Scholar] [CrossRef] [Green Version]
- Sarne, Y.; Toledano, R.; Rachmany, L.; Sasson, E.; Doron, R. Reversal of Age-Related Cognitive Impairments in Mice by an Extremely Low Dose of Tetrahydrocannabinol. Neurobiol. Aging 2018, 61, 177–186. [Google Scholar] [CrossRef]
- Xu, C.; Wang, L.; Fozouni, P.; Evjen, G.; Chandra, V.; Jiang, J.; Lu, C.; Nicastri, M.; Bretz, C.; Winkler, J.D.; et al. SIRT1 Is Downregulated by Autophagy in Senescence and Ageing. Nat. Cell Biol. 2020, 22, 1170–1179. [Google Scholar] [CrossRef]
- Hayek, L.E.; Khalifeh, M.; Zibara, V.; Assaad, R.A.; Emmanuel, N.; Karnib, N.; El-Ghandour, R.; Nasrallah, P.; Bilen, M.; Ibrahim, P.; et al. Lactate Mediates the Effects of Exercise on Learning and Memory through SIRT1-Dependent Activation of Hippocampal Brain-Derived Neurotrophic Factor (BDNF). J. Neurosci. 2019, 39, 2369–2382. [Google Scholar] [CrossRef] [Green Version]
- Jiang, M.; Wang, J.; Fu, J.; Du, L.; Jeong, H.; West, T.; Xiang, L.; Peng, Q.; Hou, Z.; Cai, H.; et al. Neuroprotective Role of Sirt1 in Mammalian Models of Huntington’s Disease through Activation of Multiple Sirt1 Targets. Nat. Med. 2012, 18, 153–158. [Google Scholar] [CrossRef]
- Burstein, O.; Doron, R. The Unpredictable Chronic Mild Stress Protocol for Inducing Anhedonia in Mice. JoVE 2018, 140, 58184. [Google Scholar] [CrossRef] [Green Version]
- Freeston, M.H.; Tiplady, A.; Mawn, L.; Bottesi, G.; Thwaites, S. Towards a Model of Uncertainty Distress in the Context of Coronavirus (COVID-19); PsyArXiv: London, UK, 2020. [Google Scholar]
- Zhu, S.; Shi, R.; Wang, J.; Wang, J.-F.; Li, X.-M. Unpredictable Chronic Mild Stress Not Chronic Restraint Stress Induces Depressive Behaviours in Mice. NeuroReport 2014, 25, 1151–1155. [Google Scholar] [CrossRef]
- Glowacz, F.; Schmits, E. Uncertainty and Psychological Distress during Lockdown during the COVID-19 Pandemic: The Young Adults Most at Risk. Psychiatry Res. 2020, 293, 113486. [Google Scholar] [CrossRef]
- Insel, T.R.; Miller, L.P.; Gelhard, R.E. The Ontogeny of Excitatory Amino Acid Receptors in Rat Forebrain—I.N-Methyl-d-Aspartate and Quisqualate Receptors. Neuroscience 1990, 35, 31–43. [Google Scholar] [CrossRef]
- Sterlemann, V.; Rammes, G.; Wolf, M.; Liebl, C.; Ganea, K.; Müller, M.B.; Schmidt, M.V. Chronic Social Stress during Adolescence Induces Cognitive Impairment in Aged Mice. Hippocampus 2010, 20, 540–549. [Google Scholar] [CrossRef]
- Sterlemann, V.; Ganea, K.; Liebl, C.; Harbich, D.; Alam, S.; Holsboer, F.; Müller, M.B.; Schmidt, M.V. Long-Term Behavioral and Neuroendocrine Alterations Following Chronic Social Stress in Mice: Implications for Stress-Related Disorders. Horm. Behav. 2008, 53, 386–394. [Google Scholar] [CrossRef]
- Jiang, W.; Chen, L. Alternative Splicing: Human Disease and Quantitative Analysis from High-Throughput Sequencing. Comput. Struct. Biotechnol. J. 2020, 19, 183–195. [Google Scholar] [CrossRef]
- Su, C.-H.; D., D.; Tarn, W.-Y. Alternative Splicing in Neurogenesis and Brain Development. Front. Mol. Biosci. 2018, 5, 12. [Google Scholar] [CrossRef] [Green Version]
- Reshetnikov, V.V.; Kisaretova, P.E.; Ershov, N.I.; Merkulova, T.I.; Bondar, N.P. Social Defeat Stress in Adult Mice Causes Alterations in Gene Expression, Alternative Splicing, and the Epigenetic Landscape of H3K4me3 in the Prefrontal Cortex: An Impact of Early-Life Stress. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2021, 106, 110068. [Google Scholar] [CrossRef]
- Angarola, B.L.; Anczuków, O. Splicing Alterations in Healthy Aging and Disease. WIREs RNA 2021, 12, e1643. [Google Scholar] [CrossRef]
- Deschênes, M.; Chabot, B. The Emerging Role of Alternative Splicing in Senescence and Aging. Aging Cell 2017, 16, 918–933. [Google Scholar] [CrossRef]
- Puigoriol-Illamola, D.; Martínez-Damas, M.; Griñán-Ferré, C.; Pallàs, M. Chronic Mild Stress Modified Epigenetic Mechanisms Leading to Accelerated Senescence and Impaired Cognitive Performance in Mice. Int. J. Mol. Sci. 2020, 21, 1154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lynch, C.J.; Shah, Z.H.; Allison, S.J.; Ahmed, S.U.; Ford, J.; Warnock, L.J.; Li, H.; Serrano, M.; Milner, J. SIRT1 Undergoes Alternative Splicing in a Novel Auto-Regulatory Loop with P53. PLoS ONE 2010, 5, e13502. [Google Scholar] [CrossRef]
- Gao, J.; Wang, W.-Y.; Mao, Y.-W.; Gräff, J.; Guan, J.-S.; Pan, L.; Mak, G.; Kim, D.; Su, S.C.; Tsai, L.-H. A Novel Pathway Regulates Memory and Plasticity via SIRT1 and MiR-134. Nature 2010, 466, 1105–1109. [Google Scholar] [CrossRef] [Green Version]
- Quesseveur, G.; David, D.J.; Gaillard, M.C.; Pla, P.; Wu, M.V.; Nguyen, H.T.; Nicolas, V.; Auregan, G.; David, I.; Dranovsky, A.; et al. BDNF Overexpression in Mouse Hippocampal Astrocytes Promotes Local Neurogenesis and Elicits Anxiolytic-like Activities. Transl. Psychiatry 2013, 3, e253. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Siao, C.-J.; Nagappan, G.; Marinic, T.; Jing, D.; McGrath, K.; Chen, Z.-Y.; Mark, W.; Tessarollo, L.; Lee, F.S.; et al. Neuronal Release of ProBDNF. Nat. Neurosci. 2009, 12, 113–115. [Google Scholar] [CrossRef] [PubMed]
- Bramham, C.R.; Messaoudi, E. BDNF Function in Adult Synaptic Plasticity: The Synaptic Consolidation Hypothesis. Prog. Neurobiol. 2005, 76, 99–125. [Google Scholar] [CrossRef]
- Rossi, C.; Angelucci, A.; Costantin, L.; Braschi, C.; Mazzantini, M.; Babbini, F.; Fabbri, M.E.; Tessarollo, L.; Maffei, L.; Berardi, N.; et al. Brain-Derived Neurotrophic Factor (BDNF) Is Required for the Enhancement of Hippocampal Neurogenesis Following Environmental Enrichment. Eur. J. Neurosci. 2006, 24, 1850–1856. [Google Scholar] [CrossRef]
- Huang, E.J.; Reichardt, L.F. Neurotrophins: Roles in Neuronal Development and Function. Annu. Rev. Neurosci. 2001, 24, 677–736. [Google Scholar] [CrossRef] [Green Version]
- Dawood, T.; Anderson, J.; Barton, D.; Lambert, E.; Esler, M.; Hotchkin, E.; Haikerwal, D.; Kaye, D.; Lambert, G. Reduced Overflow of BDNF from the Brain Is Linked with Suicide Risk in Depressive Illness. Mol. Psychiatry 2007, 12, 981–983. [Google Scholar] [CrossRef]
- Middeldorp, C.M.; Slof-Op’t Landt, M.C.T.; Medland, S.E.; van Beijsterveldt, C.E.M.; Bartels, M.; Willemsen, G.; Hottenga, J.-J.; de Geus, E.J.C.; Suchiman, H.E.D.; Dolan, C.V.; et al. Anxiety and Depression in Children and Adults: Influence of Serotonergic and Neurotrophic Genes? Genes Brain Behav. 2010, 9, 808–816. [Google Scholar] [CrossRef] [Green Version]
- Akirav, I.; Richter-Levin, G. Biphasic Modulation of Hippocampal Plasticity by Behavioral Stress and Basolateral Amygdala Stimulation in the Rat. J. Neurosci. 1999, 19, 10530–10535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doron, R.; Lotan, D.; Einat, N.; Yaffe, R.; Winer, A.; Marom, I.; Meron, G.; Kately, N.; Rehavi, M. A Novel Herbal Treatment Reduces Depressive-like Behaviors and Increases BDNF Levels in the Brain of Stressed Mice. Life Sci. 2014, 94, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Tao, W.; Dong, Y.; Su, Q.; Wang, H.; Chen, Y.; Xue, W.; Chen, C.; Xia, B.; Duan, J.; Chen, G. Liquiritigenin Reverses Depression-like Behavior in Unpredictable Chronic Mild Stress-Induced Mice by Regulating PI3K/Akt/MTOR Mediated BDNF/TrkB Pathway. Behav. Brain Res. 2016, 308, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Gu, F.; Chen, J.; Dong, W. Chronic Antidepressant Administration Alleviates Frontal and Hippocampal BDNF Deficits in CUMS Rat. Brain Res. 2010, 1366, 141–148. [Google Scholar] [CrossRef]
- Simchon-Tenenbaum, Y.; Weizman, A.; Rehavi, M. Alterations in Brain Neurotrophic and Glial Factors Following Early Age Chronic Methylphenidate and Cocaine Administration. Behav. Brain Res. 2015, 282, 125–132. [Google Scholar] [CrossRef]
- Martinez-Cengotitabengoa, M.; MacDowell, K.S.; Alberich, S.; Diaz, F.J.; Garcia-Bueno, B.; Rodriguez-Jimenez, R.; Bioque, M.; Berrocoso, E.; Parellada, M.; Lobo, A.; et al. BDNF and NGF Signalling in Early Phases of Psychosis: Relationship With Inflammation and Response to Antipsychotics After 1 Year. Schizophr. Bull. 2016, 42, 142–151. [Google Scholar] [CrossRef] [Green Version]
- Perna, G.; Iannone, G.; Alciati, A.; Caldirola, D. Are Anxiety Disorders Associated with Accelerated Aging? A Focus on Neuroprogression. Neural Plast. 2016, 2016, 8457612. [Google Scholar] [CrossRef] [Green Version]
- Amram, S.; Iram, T.; Lazdon, E.; Vassar, R.; Ben-Porath, I.; Frenkel, D. Astrocyte Senescence in an Alzheimer’s Disease Mouse Model Is Mediated by TGF-Β1 and Results in Neurotoxicity. Neuroscience 2019, 10, 700013. [Google Scholar]
- Doron, R.; Lotan, D.; Rak-Rabl, A.; Raskin-Ramot, A.; Lavi, K.; Rehavi, M. Anxiolytic Effects of a Novel Herbal Treatment in Mice Models of Anxiety. Life Sci. 2012, 90, 995–1000. [Google Scholar] [CrossRef]
- O’Donovan, A.; Tomiyama, A.J.; Lin, J.; Puterman, E.; Adler, N.E.; Kemeny, M.; Wolkowitz, O.M.; Blackburn, E.H.; Epel, E.S. Stress Appraisals and Cellular Aging: A Key Role for Anticipatory Threat in the Relationship between Psychological Stress and Telomere Length. Brain Behav. Immun. 2012, 26, 573–579. [Google Scholar] [CrossRef] [Green Version]
- Montaron, M.F.; Drapeau, E.; Dupret, D.; Kitchener, P.; Aurousseau, C.; Le Moal, M.; Piazza, P.V.; Abrous, D.N. Lifelong Corticosterone Level Determines Age-Related Decline in Neurogenesis and Memory. Neurobiol. Aging 2006, 27, 645–654. [Google Scholar] [CrossRef] [PubMed]
- Harman, D.; International Association of Biomedical Gerontology (Eds.) Towards Prolongation of the Healthy Life Span: Practical Approaches to Intervention; [Result of a Conference, ... Held in Adelaide, Australia, on August 15–18, 1997, by the Seventh Congress of the International Association of Biomedical Gerontology]; Annals of the New York Academy of Sciences: New York, NY, USA, 1998; ISBN 978-1-57331-109-0. [Google Scholar]
- Campisi, J. Senescent Cells, Tumor Suppression, and Organismal Aging: Good Citizens, Bad Neighbors. Cell 2005, 120, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Toussaint, O.; Dumont, P.; Remacle, J.; Dierick, J.-F.; Pascal, T.; Frippiat, C.; Magalhaes, J.P.; Zdanov, S.; Chainiaux, F. Stress-Induced Premature Senescence or Stress-Induced Senescence-Like Phenotype: One In Vivo Reality, Two Possible Definitions? Sci. World J. 2002, 2, 230–247. [Google Scholar] [CrossRef] [Green Version]
- Hueston, C.M.; Cryan, J.F.; Nolan, Y.M. Stress and Adolescent Hippocampal Neurogenesis: Diet and Exercise as Cognitive Modulators. Transl. Psychiatry 2017, 7, e1081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.J.; Yang, S.J. Aging-Related Correlation between Serum Sirtuin 1 Activities and Basal Metabolic Rate in Women, but Not in Men. Clin. Nutr. Res. 2017, 6, 18–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Opstad, T.B.; Kalstad, A.A.; Pettersen, A.Å.; Arnesen, H.; Seljeflot, I. Novel Biomolecules of Ageing, Sex Differences and Potential Underlying Mechanisms of Telomere Shortening in Coronary Artery Disease. Exp. Gerontol. 2019, 119, 53–60. [Google Scholar] [CrossRef]
- Zawada, I.; Masternak, M.M.; List, E.O.; Stout, M.B.; Berryman, D.E.; Lewinski, A.; Kopchick, J.J.; Bartke, A.; Karbownik-Lewinska, M.; Gesing, A. Gene Expression of Key Regulators of Mitochondrial Biogenesis Is Sex Dependent in Mice with Growth Hormone Receptor Deletion in Liver. Aging 2015, 7, 195–204. [Google Scholar] [CrossRef] [Green Version]
- Jiang, X.; Chen, Z.; Yu, X.; Chen, J.; Sun, C.; Jing, C.; Xu, L.; Liu, F.; Ni, W.; Chen, L. Lipopolysaccharide-Induced Depression Is Associated with Estrogen Receptor-α/SIRT1/NF-ΚB Signaling Pathway in Old Female Mice. Neurochem. Int. 2021, 148, 105097. [Google Scholar] [CrossRef]
- Smith, B.L.; Morano, R.L.; Ulrich-Lai, Y.M.; Myers, B.; Solomon, M.B.; Herman, J.P. Adolescent Environmental Enrichment Prevents Behavioral and Physiological Sequelae of Adolescent Chronic Stress in Female (but Not Male) Rats. Stress 2018, 21, 464–473. [Google Scholar] [CrossRef]
- Brancato, A.; Castelli, V.; Lavanco, G.; Cannizzaro, C. Environmental Enrichment During Adolescence Mitigates Cognitive Deficits and Alcohol Vulnerability Due to Continuous and Intermittent Perinatal Alcohol Exposure in Adult Rats. Front. Behav. Neurosci. 2020, 14, 583122. [Google Scholar] [CrossRef]
- Doron, R.; Sever, A.; Handelsman, A.; Toledano, R.; Franko, M.; Hirshler, Y.; Shamir, A.; Burstein, O.; Rehavi, M. GABAA Receptor Density Is Not Altered by a Novel Herbal Anxiolytic Treatment. J. Mol. Neurosci. 2018, 65, 110–117. [Google Scholar] [CrossRef] [PubMed]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shlomi, S.; Toledano, R.; Nitzan, K.; Shahaf, S.D.; Break, E.P.; Frenkel, D.; Doron, R. Imbalance in Sirt1 Alternative Splicing in Response to Chronic Stress during the Adolescence Period in Female Mice. Int. J. Mol. Sci. 2022, 23, 4945. https://doi.org/10.3390/ijms23094945
Shlomi S, Toledano R, Nitzan K, Shahaf SD, Break EP, Frenkel D, Doron R. Imbalance in Sirt1 Alternative Splicing in Response to Chronic Stress during the Adolescence Period in Female Mice. International Journal of Molecular Sciences. 2022; 23(9):4945. https://doi.org/10.3390/ijms23094945
Chicago/Turabian StyleShlomi, Shir, Roni Toledano, Keren Nitzan, Sigal Dror Shahaf, Emanuela P. Break, Dan Frenkel, and Ravid Doron. 2022. "Imbalance in Sirt1 Alternative Splicing in Response to Chronic Stress during the Adolescence Period in Female Mice" International Journal of Molecular Sciences 23, no. 9: 4945. https://doi.org/10.3390/ijms23094945