Photoprocesses in Derivatives of 1,4- and 1,3-Diazadistyryldibenzenes
Abstract
:1. Introduction
2. Results and Discussion
2.1. Absorption and Fluorescence Spectra
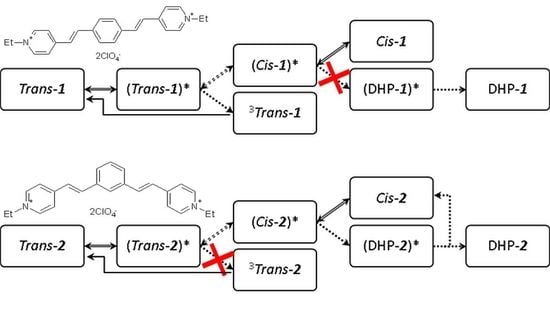
2.2. Kinetic Spectroscopy
2.3. Quantum Chemical Calculations
2.3.1. Model
2.3.2. Results
3. Materials and Methods
Calculation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kim, M.; Whang, D.R.; Gierschnerb, J.; Park, S.Y. A distyrylbenzene based highly efficient deep red/near-infrared emitting organic solid. J. Mater. Chem. C 2015, 3, 231–234. [Google Scholar] [CrossRef]
- Cavazzini, M.; Quici, S.; Orlandi, S.; Sissa, C.; Terenziani, F.; Painelli, A. Intimately bound coumarin and bis(alkylaminostyryl)benzene fragments: Synthesis and energy transfer. Tetrahedron 2013, 69, 2827–2833. [Google Scholar] [CrossRef]
- Chaieb, A.; Khoukh, A.; Brown, R.; Francois, J.; Dagron-Lartigau, C. Characterization of model luminescent PPV analogues with, donating or withdrawing groups. Opt. Mater. 2007, 30, 318–327. [Google Scholar] [CrossRef]
- Motoyoshiya, J.; Fengqiang, Z.; Nishii, Y.; Aoyama, H. Fluorescence quenching of versatile fluorescent probes based on strongly electron-donating distyrylbenzenes responsive to aromatic chlorinated and nitro compounds, boronic acid and Ca2+, Spectrochim. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2008, 69, 167–173. [Google Scholar] [CrossRef] [Green Version]
- Pond, S.J.K.; Tsutsumi, O.; Rumi, M.; Kwon, O.; Zojer, E.; Bredas, J.L.; Marder, S.R.; Perry, J.W. Metal-Ion Sensing Fluorophores with Large Two-Photon Absorption Cross Sections: Aza-Crown Ether Substituted Donor-Acceptor-Donor Distyrylbenzenes. J. Am. Chem. Soc. 2004, 126, 9291–9306. [Google Scholar] [CrossRef]
- Morgan, D.D.; Horgan, S.W.; Orchln, M. Photocyclization of stilbene analogs I. The oxidative photocyclization of 1,3-distyrylbenzene. Tetrahedron Lett. 1970, 11, 4347–4350. [Google Scholar] [CrossRef]
- Laarhoven, W.H.; Cuppen, T.J.H.M.; Nivard, R.J.F. Photodehydrocyclizations in stilbene-like compounds—II: Photochemistry of distyrylbenzenes. Tetrahedron 1970, 11, 1069–1083. [Google Scholar] [CrossRef]
- Zertani, R.; Meier, H. Photochemie des 1,3-Distyrylbenzols—Ein neuer Weg zu syn-[2.2](1,3)Cyclophanen. Chem. Ber. 1986, 119, 1704–1715. [Google Scholar] [CrossRef]
- Marri, E.; Galiazzo, G.; Mazzucato, U.; Spalletti, A. Excited state properties of cross-conjugated 1,2- and 1,3-distyrylbenzene and some aza-analogues. Chem. Phys. 2005, 312, 205–211. [Google Scholar] [CrossRef]
- Marri, E.; Elisei, F.; Mazzucato, U.; Pannacci, D.; Spalletti, A. Triplet-sensitized photobehaviour of the three stereoisomers of 1,4-distyrylbenzene and some aza-analogues. J. Photochem. Photobiol. A Chem. 2006, 177, 307–313. [Google Scholar] [CrossRef]
- Ginocchietti, G.; Galiazzo, G.; Pannacci, D.; Mazzucato, U.; Spalletti, A. Effect of stereoisomerism on the radiative and reactive relaxation channels of two thio-analogues of distyrylbenzene. Chem. Phys. 2006, 331, 164–172. [Google Scholar] [CrossRef]
- Ginocchietti, G.; Mazzucato, U.; Spalletti, A. Excited state behaviour of some thio-analogues of 1,3-distyrylbenzene. J. Photochem. Photobiol. A 2008, 196, 233–238. [Google Scholar] [CrossRef]
- Kalanoor, B.S.; Bisht, P.B.; Annamalai, S.; Aidhen, I.S. A new distyrylbenzene derivative with Weinreb amide functionality: An efficient laser dye and nonlinear optical material. J. Lumin. 2009, 129, 1094–1098. [Google Scholar] [CrossRef]
- Toyoshima, T.; Yoshida, S.; Watanabe, S. Synthesis of an alkylthio-substituted dibenz[a,j]anthracene with improved solubility via the oxidative photocyclization of 1,3-distyrylbenzene derivatives. Tetrahedron 2013, 69, 1904–1911. [Google Scholar] [CrossRef]
- Atabekyan, L.S.; Avakyan, V.G.; Nuriev, V.N.; Vatsadze, S.Z.; Gromov, S.P. Photonics of tetramethoxy-1,4-distyrylbenzene. Russ. Chem. Bull. 2018, 67, 2016–2024. [Google Scholar] [CrossRef]
- Vedernikov, A.I.; Nuriev, V.N.; Fedorov, O.V.; Moiseeva, A.A.; Kurchavov, N.A.; Kuz’mina, L.G.; Freidzon, A.Y.; Pod’yacheva, E.S.; Medved’ko, A.V.; Vatsadze, S.Z.; et al. Synthesis, structure and complexation of biscrowncontaining 1,4-distyrylbenzenes. Russ. Chem. Bull. 2016, 11, 2686–2703. [Google Scholar] [CrossRef]
- Nuriev, V.N.; Fedorov, O.V.; Moiseeva, A.A.; Freidzon, A.Y.; Kurchavov, N.A.; Vedernikov, A.I.; Medved’ko, A.V.; Pod’yacheva, E.S.; Vatsadze, S.Z.; Gromov, S.P. Synthesis, Structure, Spectral Properties, and Electrochemistry of Bis(crown ether) Containing 1,3-Distyrylbenzenes. Russ. J. Org. Chem. 2017, 53, 1726–1737. [Google Scholar] [CrossRef]
- Atabekyan, L.S.; Avakyan, V.G.; Chibisov, A.K.; Gromov, S.P.; Vatsadze, S.Z.; Nuriev, V.N.; Medved´ko, A.V. Bis(15-crown-5)-1,4-distyrylbenzene and its complexes with metal perchlorates: Photonics and structure. Russ. Chem. Bull. 2019, 68, 2053–2064. [Google Scholar] [CrossRef]
- Atabekyan, L.S.; Avakyan, V.G.; Chibisov, A.K.; Gromov, S.P.; Vatsadze, S.Z.; Nuriev, V.N.; Medved’ko, A.V. Photonics of bis(18-Crown-6)-1,4-Distyrylbenzene and Its Complexes with Metal Perchlorates. High Energy Chem. 2019, 53, 115–124. [Google Scholar] [CrossRef]
- Atabekyan, L.S.; Freidzon, A.Y.; Chibisov, A.K.; Gromov, S.P.; Vatsadze, S.Z.; Nuriev, V.N.; Medvedko, A.V. Photoprocesses in bis(18-crown-6)-1,3-distyrylbenzene and its complexes with metal perchlorates. High Energy Chem. 2021, 184, 108773–108782. [Google Scholar] [CrossRef]
- Atabekyan, L.S.; Avakyan, V.G.; Chibisov, A.K.; Nuriev, V.N.; Medvedko, A.V.; Koshkin, A.V.; Gromov, S.P. Photoprocesses in bis(15-crown-5)-1,3-distyrylbenzene and its complexes with metal perchlorates. J. Photochem. Photobiol. A 2021, 414, 113293–113302. [Google Scholar] [CrossRef]
- Atabekyan, L.S.; Chibisov, A.K. Photoprocesses in Aqueous Solutions of 9-Ethylthiacarbocyanine Dyes in the Presence of Surfactants. High Energy Chem. 2007, 41, 91–96. [Google Scholar] [CrossRef]
- Montalti, M.; Credi, A.; Prodi, L.; Gandolfi, M.T. Handbook of Phochemistry, 3rd ed.; Taylor and Fransis: London, UK, 2006; p. 574. [Google Scholar]
- Granovsky, A.A. Firefly, version 8.2.4; 2017. Available online: http://classic.chem.msu.su/gran/firefly/index.html (accessed on 1 September 2022).
- Schmidt, M.W.; Baldridge, K.K.; Boatz, J.A.; Elbert, S.T.; Gordon, M.S.; Jensen, J.H.; Koseki, S.; Matsunaga, N.; Nguyen, K.A.; Su, S.; et al. General Atomic and Molecular Electronic Structure System. J. Comput. Chem. 1993, 14, 1347–1363. [Google Scholar] [CrossRef]
- Tomasi, J.; Mennucci, B.; Cammi, R. Quantum Mechanical Continuum Solvation Models. Chem. Rev. 2005, 105, 2999–3093. [Google Scholar] [CrossRef] [PubMed]
- Peach, M.J.G.; Tozer, D. Overcoming Low Orbital Overlap and Triplet Instability Problems in TDDFT. J. Phys. Chem. A 2012, 116, 9783–9789. [Google Scholar] [CrossRef]
- Eyring, H.; Walter, J.; Kimball, G.E. Quantum Chemistry; Wiley: New York, NY, USA, 1944. [Google Scholar]
- Bazilevskii, M.V.; Faustov, V.I. Modern theories of chemical reactions in condensed phases. Russ. Chem. Rev. 1992, 61, 651–672. [Google Scholar] [CrossRef]
- Henriksen, N.E.; Hansen, F.Y. Theories of Molecular Reaction Dynamics: The Microscopic Foundation of Chemical Kinetics; Oxford University Press: Oxford, UK, 2008. [Google Scholar]
- Casida, M. Time-Dependent Density Functional Response Theory for Molecules in Recent Advances in Density Functional Methods; Chong, D.P., Ed.; World Scientific: Singapore, 1995; pp. 155–192. [Google Scholar] [CrossRef]
- Atchity, G.J.; Xantheas, S.S.; Ruedenberg, K. Potential energy surfaces near intersections. J. Chem. Phys. 1991, 95, 1862–1876. [Google Scholar] [CrossRef]
- Quentin, C.; Gerasimaitė, R.; Freidzon, A.; Atabekyan, L.S.; Lukinavičius, G.; Belov, V.N.; Mitronova, G.Y. Direct Visualization of Amlodipine Intervention into Living Cells by Means of Fluorescence Microscopy. Molecules 2021, 26, 2997. [Google Scholar] [CrossRef]














| Compound | Experiment | Calculation | ||
|---|---|---|---|---|
| λmax, nm | ε, L∙mol−1∙cm−1 | λmax, nm | f | |
| 1 | 390 | 5.4 × 104 | 420 | 2.07 |
| 2 | 350 | 5.0 × 104 | 372 1 350 1 348 1 314 1 | 0.59 1 0.58 1 0.75 1 0.23 1 |
| Compound | Experiment | Calculation | |||||||
|---|---|---|---|---|---|---|---|---|---|
| φ | λmax, nm | λ0-0, nm | λexc, nm | Δλ, nm | τfl, ps | λmax, nm | f | τr, ps | |
| 1 | 0.05 | 470 | 434 | 393 | 77 | 120 | 454 1 472 2 | 2.18 1.34 | 1420 2500 |
| 2 | 0.02 | 455 | 395 | 346 | 109 | 160 | 409 1 422 2 | 0.30 0.19 | 8350 13700 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atabekyan, L.S.; Freidzon, A.Y.; Ionov, D.S.; Nuriev, V.N.; Medved’ko, A.V.; Vatsadze, S.Z.; Gromov, S.P.; Chibisov, A.K. Photoprocesses in Derivatives of 1,4- and 1,3-Diazadistyryldibenzenes. Int. J. Mol. Sci. 2022, 23, 15346. https://doi.org/10.3390/ijms232315346
Atabekyan LS, Freidzon AY, Ionov DS, Nuriev VN, Medved’ko AV, Vatsadze SZ, Gromov SP, Chibisov AK. Photoprocesses in Derivatives of 1,4- and 1,3-Diazadistyryldibenzenes. International Journal of Molecular Sciences. 2022; 23(23):15346. https://doi.org/10.3390/ijms232315346
Chicago/Turabian StyleAtabekyan, Levon S., Alexandra Y. Freidzon, Dmitry S. Ionov, Vyacheslav N. Nuriev, Alexey V. Medved’ko, Sergey Z. Vatsadze, Sergey P. Gromov, and Alexander K. Chibisov. 2022. "Photoprocesses in Derivatives of 1,4- and 1,3-Diazadistyryldibenzenes" International Journal of Molecular Sciences 23, no. 23: 15346. https://doi.org/10.3390/ijms232315346