The Use of Liquids Ionic Fluids as Pharmaceutically Active Substances Helpful in Combating Nosocomial Infections Induced by Klebsiella Pneumoniae New Delhi Strain, Acinetobacter Baumannii and Enterococcus Species
Abstract
:1. Introduction
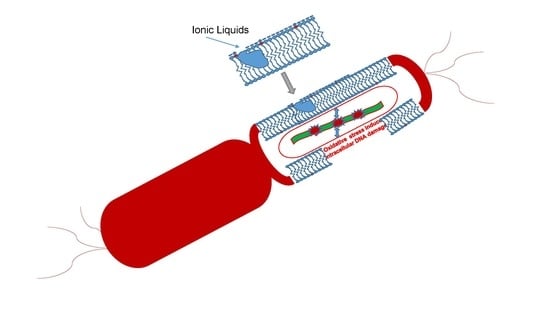
1.1. Physico-Chemical Properties of ILs in Antimicrobial Activity
1.2. Divisions of ILs
1.3. Synthesis of ILs
1.4. Pollution of ILs
1.5. Toxicology
2. Antimicrobial Properties of a Quaternary Ammonium Halide
Interaction Quaternary Ammonium Halides with Different Pathogenic Bacterial Strains Often Occurring in Nosocomial Infections
3. The Use of Ionic Liquids in Microbiology and Medicine
4. Polymorphism of Ionic Liquids as New Solvents in the Synthesis of Pharmacologically Active Compounds
5. Summary and Future Outlook
Author Contributions
Conflicts of Interest
Abbreviations
| A. baumannii | Acinetobacter baumannii |
| Ag+C3H5N2 – np | imidazolium-based silver nanoparticles |
| AIL | aprotic ionic liquid |
| APIs | active pharmacological ingredients |
| BF4 | tetrafluoroborate |
| A. baumannii | Acinetobacter baumannii |
| Ag+C3H5N2 - np | imidazolium-based silver nanoparticles |
| AIL | aprotic ionic liquid |
| APIs | active pharmacological ingredients |
| BF4 | tetrafluoroborate |
| BIOCONTAM | EFSA unit on biological hazards and contaminants |
| BIOHAZ | the panel on biological hazards EFSA |
| BIZ | growth inhibitory zone |
| blaNDM-1 | plasmid containing the metallo-β-lactamase type 1 gene sequence |
| BMP-NTf2 | 1-butyl-1-methylpyrrolidinumbistriflimide |
| BORSA | mecA-positive oxacillin resistant Staphylococcus aureus |
| BTFLA | bis trifluoromethylsulfonyl amide [(CF3SO2)2N]− |
| CA-MRSA | community-associated methicillin-resistant Staphylococcus aureus |
| Di-But C10 | phosphonium, 1,1′-(1,10-decanediyl)bis[1,1,1-tributyl] |
| Di-But C6 | phosphonium, 1,1′-(1,6-hexanediyl)bis[1,1,1-tributyl] |
| Di-Hex C10 | phosphonium, 1,1′-(1,10-decanediyl)bis[1,1,1-trihexyl] |
| Di-Hex C6 | phosphonium, 1,1′-(1,6-hexanediyl)bis[1,1,1-trihexyl] |
| E. coli | Escherichia coli |
| E. faecium | Enterococcus faecium |
| EARS-Net | European Antimicrobial Resistance Surveillance Network |
| ECDC | European Centre for Disease Prevention and Control |
| EFSA | European Food Safety Authority |
| ESBL+ | extended spectrum β-lactamases |
| ESKAPE | acronym from Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii and Pseudomonas aeruginosa infections |
| EU | European Union |
| FtsZ | prokaryotic homologue to the eukaryotic protein tubulin |
| HA-MRSA | hospital-associated methicillin-resistant Staphylococcus aureus |
| HILs | herbicidal ionic liquids |
| hlα-γ+ | α-γ haemolysins encoding genes |
| HMIM-Cl | 1-hexyl-3-methylimidazolium chloride |
| icaD+- | gene relevant to bio-film formation |
| ICU | intensive care unit |
| ILs | ionic liquids |
| K. pneumoniae | Klebsiella pneumoniae |
| KPC | Klebsiella pneumoniae carbapenemase |
| MBC | minimal bactericidal concentration |
| MDEP | mucoid exopolysaccharide strain of Pseudomonas aeruginosa |
| MDR+ | multi-drug-resistant Acinetobacter baumannii |
| MDRSA | multi-drug-resistant and methicillin-resistant Staphylococcus aureus |
| MGEs+ | mobile genetic elements encoding methicillin resistance |
| MIC | minimal inhibitory concentration |
| MS 2 virus | Enterobacteriophage type 2 |
| NorA | Norfloxacin efflux pump gene |
| NSAIDs | non-steroidal anti-inflammatory drugs |
| OTBDMS | O-tert-butyldimethylsilyl |
| p100 virus | Listeria phage 100 |
| PAPI+ | Pseudomonas aeruginosa pathogenicity islands of strain 14 |
| PBP | penicillin binding protein |
| PF6 | Hexafluorophosphate |
| PILs | phosphonium ionic liquids |
| ppm | parts per million |
| PrILs | protic ionic liquids |
| PTLFS | p-toluenesulfonate [4MePhSO3]- |
| pvl+ | Panton-Valentine gene encoding leucocidin |
| RTIL | room-temperature ionic liquids |
| S. aureus | Staphylococcus aureus |
| SCCmec+IV-V | staphylococcal mec chromosome cassette type IV and V |
| TBAF | tetra-n-butylammonium fluoride |
| TGA | thermal gravimetric analysis |
| TPA | triphenylamine |
| tsst-1 | toxic shock syndrome toxin 1 gene |
| USA | United States of America |
| USD | United States dollar |
| VRE | vancomycin-resistant enterococci |
| [(C2)2(C1)2(C1)23gu][C2OSO3] | 2-ethyl-1,1,3,3-tetramethylguanidinium ethyl sulfate |
| [(C2)22(C1)2(C1)23gu][C2OSO3] | 2,2-diethyl-1,1,3,3-tetramethylguanidinium ethyl sulfate |
| [BMIm+Cl-] | butylmethylimmidazolium chloride |
| [C12Im] | C12 alkyl imidazolium |
| [C12Py] | C12 alkyl pyridinium |
| [C16M1Im][Br] | 1-hexadecyl-3-methylimidazolium bromide |
| [C16M2Im][Br] | 1-hexadecyl-2,3-dimethylimidazolium bromide |
| [C18Im] | C18 alkyl imidazolium |
| [C18Py] | C18 alkyl pyridinium |
| [C1C14pi][BF4]2 | 1,4-dimethylpiperazinum bis(tetrafluoroborate) |
| [C2C1C14pi][I] | 1-ethyl-1,4-dimethylpiperazinium iodide |
| [C2mim][Cl] | 1-ethyl-3-methyl imidazolium chloride |
| [C2pi][BF4] | 1-ethylpiperazinium tetrafluoroborate |
| [C8mim][Cl] | 1-octyl-3-methyl imidazolinum chloride |
| [Chol][Cl] | choline chloride |
| [CPB] | cetylpyridinum bromide |
| [DODMA][Cl] | 1,2-dioleyloxy-3-dimethylaminopropane chloride |
| [EMIm+Tf2N-] | ethylmethylimmidazolium bis(trifluoromethylsulfonyl)imide |
| [P(C14H29)(C6H13)3]+ | tetralkylophosphonium oleate |
| [Phpi][BF4] | 1-phenylpiperazinum tetrafluoroborate |
| [TMC8A][Cl] | trimethyloctylammonium |
| (ZnCl2)2 | BZBN benzethonium chloride |
| * | ampicillin-based ILs |
| ** | The MIC value was equal to the MBC of ampicillin-based ILs. |
| *[C16M1Im][Amp] | 1-hexadecyl-3-methylimidazolium ampicillin |
| *[C16M2Im][AMP] | 1-hexadecyl-2,3-dimethylimidazolium ampicillin |
| *[CPB][AMP] | ampicillin-based ILs cetylpyridinum |
References
- De la Fuente-Nunez, C.; Brown, P.; Torres, M.D.T.; Cao, J.; Lu, T.K. Magnetic surfactant ionic liquids and polymers with tetrahaloferrate (iii) anions as antimicrobial agents with low cytotoxicity. Colloid Interface Sci. Commun. 2018, 22, 11–13. [Google Scholar] [CrossRef]
- Earle, M.J.; Esperança, J.M.S.S.; Gilea, M.A.; Canongia Lopes, J.N.; Rebelo, L.P.N.; Magee, J.W.; Seddon, K.R.; Widegren, J.A. The distillation and volatility of ionic liquids. Nature 2006, 439, 831. [Google Scholar] [CrossRef] [PubMed]
- Huddleston, J.G.; Visser, A.E.; Reichert, W.M.; Willauer, H.D.; Broker, G.A.; Rogers, R.D. Characterization and comparison of hydrophilic and hydrophobic room temperature ionic liquids incorporating the imidazolium cation. Green Chem. 2001, 3, 156–164. [Google Scholar] [CrossRef]
- Gehlot, P.S.; Gupta, H.; Kumar, A. Paramagnetic surface active ionic liquids: Interaction with DNA and mri application. Colloid Interface Sci. Commun. 2018, 26, 14–23. [Google Scholar] [CrossRef]
- Pernak, J.; Goc, I. New ionic liquids with organic anions. Pol. J. Chem. 2003, 77, 975–984. [Google Scholar] [CrossRef]
- Dai, L.; Yu, S.; Shan, Y.; He, M. Novel room temperature inorganic ionic liquids. Eur. J. Inorg. Chem. 2004, 2004, 237–241. [Google Scholar] [CrossRef]
- Vekariya, R.L. A review of ionic liquids: Applications towards catalytic organic transformations. J. Mol. Liquids 2017, 227, 44–60. [Google Scholar] [CrossRef]
- Soto, A. Ionic liquids for extraction processes in refinery-related applications. In Ionic Liquids for Better Separation Processes; Rodríguez, H., Ed.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 39–65. [Google Scholar]
- Hayes, R.; Warr, G.G.; Atkin, R. Structure and nanostructure in ionic liquids. Chem. Rev. 2015, 115, 6357–6426. [Google Scholar] [CrossRef] [PubMed]
- Claus, J.; Sommer, F.O.; Kragl, U. Ionic liquids in biotechnology and beyond. Solid State Ion. 2018, 314, 119–128. [Google Scholar] [CrossRef]
- Egorova, K.S.; Gordeev, E.G.; Ananikov, V.P. Biological activity of ionic liquids and their application in pharmaceutics and medicine. Chem. Rev. 2017, 117, 7132–7189. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Xia, S.; Wang, Q.; Yang, Z.; Ma, P. Predicting the melting points of ionic liquids by the quantitative structure property relationship method using a topological index. J. Chem. Thermodyn. 2013, 62, 196–200. [Google Scholar] [CrossRef]
- Marrucho, I.M.; Branco, L.C.; Rebelo, L.P.N. Ionic liquids in pharmaceutical applications. Annu. Rev. Chem. Biomol. Eng. 2014, 5, 527–546. [Google Scholar] [CrossRef] [PubMed]
- Kurnia, K.A.; Sintra, T.E.; Neves, C.M.; Shimizu, K.; Canongia Lopes, J.N.; Goncalves, F.; Ventura, S.P.; Freire, M.G.; Santos, L.M.; Coutinho, J.A. The effect of the cation alkyl chain branching on mutual solubilities with water and toxicities. Phys. Chem. Chem. Phys. 2014, 16, 19952–19963. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Zhao, D.; Wen, L.; Yang, S.; Chen, X. Viscosity of ionic liquids: Database, observation, and quantitative structure-property relationship analysis. AIChE J. 2012, 58, 2885–2899. [Google Scholar] [CrossRef]
- Yoo, B.; Jing, B.; Jones, S.E.; Lamberti, G.A.; Zhu, Y.; Shah, J.; Maginn, E. Molecular mechanisms of ionic liquid cytotoxicity probed by an integrated experimental and computational approach. Sci. Rep. 2016, 6, 19889. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eftekhari, A.; Saito, T. Synthesis and properties of polymerized ionic liquids. Eur. Polym. J. 2017, 90, 245–272. [Google Scholar] [CrossRef]
- Zhang, H.; Zhou, X.; Dong, J.; Zhang, G.; Wang, C. A novel family of green ionic liquids with surface activities. Sci. China Ser. B Chem. 2007, 50, 238–242. [Google Scholar] [CrossRef]
- Horike, S.; Ayano, M.; Tsuno, M.; Fukushima, T.; Koshiba, Y.; Misaki, M.; Ishida, K. Thermodynamics of ionic liquid evaporation under vacuum. Phys. Chem. Chem. Phys. 2018, 20, 21262–21268. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Zhao, Y.; Chen, G. Multiphase processes with ionic liquids in microreactors: Hydrodynamics, mass transfer and applications. Chem. Eng. Sci. 2018, 189, 340–359. [Google Scholar] [CrossRef]
- Pernak, J.; Skrzypczak, A.; Lota, G.; Frackowiak, E. Synthesis and properties of trigeminal tricationic ionic liquids. Chemistry 2007, 13, 3106–3112. [Google Scholar] [CrossRef] [PubMed]
- Pernak, J.; Syguda, A.; Janiszewska, D.; Materna, K.; Praczyk, T. Ionic liquids with herbicidal anions. Tetrahedron 2011, 67, 4838–4844. [Google Scholar] [CrossRef]
- Suresh; Sandhu, J.S. Recent advances in ionic liquids: Green unconventional solvents of this century: Part I. Green Chem. Lett. Rev. 2011, 4, 289–310. [Google Scholar]
- Earle, M.J.; Seddon, K.R. Ionic liquids: Green solvents for the future. In Clean Solvents; American Chemical Society: Washington, DC, USA, 2002; Volume 819, pp. 10–25. [Google Scholar]
- Kogelnig, D.; Stojanovic, A.; Galanski, M.; Groessl, M.; Jirsa, F.; Krachler, R.; Keppler, B.K. Greener synthesis of new ammonium ionic liquids and their potential as extracting agents. Tetrahedron Lett. 2008, 49, 2782–2785. [Google Scholar] [CrossRef]
- Yadav, J.S.; Reddy, B.V.S.; Basak, A.K.; Venkat Narsaiah, A. Recyclable 2nd generation ionic liquids as green solvents for the oxidation of alcohols with hypervalent iodine reagents. Tetrahedron 2004, 60, 2131–2135. [Google Scholar] [CrossRef]
- Dickinson, Q.; Bottoms, S.; Hinchman, L.; McIlwain, S.; Li, S.; Myers, C.L.; Boone, C.; Coon, J.J.; Hebert, A.; Sato, T.K.; et al. Mechanism of imidazolium ionic liquids toxicity in saccharomyces cerevisiae and rational engineering of a tolerant, xylose-fermenting strain. Microb. Cell Fact. 2016, 15, 17. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, G.; Giri, R.P.; Dubey, A.; Mitra, S.; Priyadarshini, R.; Gupta, A.; Mukhopadhyay, M.K.; Ghosh, S.K. Structural changes in cellular membranes induced by ionic liquids: From model to bacterial membranes. Chem. Phys. Lipids 2018, 215, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Petkovic, M.; Hartmann, D.O.; Adamová, G.; Seddon, K.R.; Rebelo, L.P.N.; Pereira, C.S. Unravelling the mechanism of toxicity of alkyltributylphosphonium chlorides in aspergillus nidulans conidia. N. J. Chem. 2012, 36, 56–63. [Google Scholar] [CrossRef]
- Hartmann, D.O.; Silva Pereira, C. A molecular analysis of the toxicity of alkyltributylphosphonium chlorides in aspergillus nidulans. N. J. Chem. 2013, 37, 1569–1577. [Google Scholar] [CrossRef]
- Feder-Kubis, J.; Kubicki, M.; Pernak, J. 3-alkoxymethyl-1-(1r,2s,5r)-(−)-menthoxymethylimidazolium salts-based chiral ionic liquids. Tetrahedron Asymmetry 2010, 21, 2709–2718. [Google Scholar] [CrossRef]
- Filippov, A.; Taher, M.; Shah, F.U.; Glavatskih, S.; Antzutkin, O. Effect of cation alkyl chain length on density and diffusion in dialkylpyrrolidinium bis(mandelato)borate ionic liquids. Phys. Chem. Chem. Phys. 2014, 16, 26798–26805. [Google Scholar] [CrossRef] [PubMed]
- Suchodolski, J.; Feder-Kubis, J.; Krasowska, A. Antifungal activity of ionic liquids based on (−)-menthol: A mechanism study. Microbiol. Res. 2017, 197, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Bica, K.; Rijksen, C.; Nieuwenhuyzen, M.; Rogers, R.D. In search of pure liquid salt forms of aspirin: Ionic liquid approaches with acetylsalicylic acid and salicylic acid. Phys. Chem. Chem. Phys. 2010, 12, 2011–2017. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; V Malhotra, S. Study on the potential anti-cancer activity of phosphonium and ammonium-based ionic liquids. Bioorg. Med. Chem. Lett. 2009, 19, 4643–4646. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Malhotra, S.V. Synthesis of nucleoside-based antiviral drugs in ionic liquids. Bioorg. Med. Chem. Lett. 2008, 18, 5640–5642. [Google Scholar] [CrossRef] [PubMed]
- Shi, F.; Xi, J.; Hou, F.; Han, L.; Li, G.; Gong, S.; Chen, C.; Sun, W. Application of three-dimensional reduced graphene oxide-gold composite modified electrode for direct electrochemistry and electrocatalysis of myoglobin. Mater. Sci. Eng. C 2016, 58, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Prakash, P.; Rawat, K.; Solanki, P.R.; Bohidar, H.B. Antibacterial and antifungal activity of biopolymers modified with ionic liquid and laponite. Appl. Biochem. Biotechnol. 2015, 177, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Borkowski, A.; Kowalczyk, P.; Czerwonka, G.; Cieśla, J.; Cłapa, T.; Misiewicz, A.; Szala, M.; Drabik, M. Interaction of quaternary ammonium ionic liquids with bacterial membranes – studies with escherichia coli r1–r4-type lipopolysaccharides. J. Mol. Liquids 2017, 246, 282–289. [Google Scholar] [CrossRef]
- Kerpen, C.; Sprenger, J.A.; Herkert, L.; Schafer, M.; Bischoff, L.A.; Zeides, P.; Grune, M.; Bertermann, R.; Brede, F.A.; Muller-Buschbaum, K.; et al. Protonation versus oxonium salt formation: Basicity and stability tuning of cyanoborate anions. Angew. Chem. Int. Ed. Engl. 2017, 56, 2800–2804. [Google Scholar] [CrossRef] [PubMed]
- Howarth, J.; Hanlon, K.; Fayne, D.; McCormac, P. Moisture stable dialkylimidazolium salts as heterogeneous and homogeneous lewis acids in the diels-alder reaction. Tetrahedron Lett. 1997, 38, 3097–3100. [Google Scholar] [CrossRef]
- Mu, T.; Han, B. Structures and thermodynamic properties of ionic liquids. In Structures and Interactions of Ionic Liquids; Zhang, S., Wang, J., Lu, X., Zhou, Q., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 107–139. [Google Scholar]
- Bernardes, C.E.S.; Shimizu, K.; Lopes, J.N.C.; Marquetand, P.; Heid, E.; Steinhauser, O.; Schroder, C. Additive polarizabilities in ionic liquids. Phys. Chem. Chem. Phys. 2016, 18, 1665–1670. [Google Scholar] [CrossRef] [PubMed]
- Armand, M.; Endres, F.; R MacFarlane, D.; Ohno, H.; Scrosati, B. Ionic-liquid materials for the electrochemical challenges of the future. Mater. Sustain. Energy 2009, 8, 621–629. [Google Scholar]
- Hunt, P.A.; Ashworth, C.R.; Matthews, R.P. Hydrogen bonding in ionic liquids. Chem. Soc. Rev. 2015, 44, 1257–1288. [Google Scholar] [CrossRef] [PubMed]
- Ren, H.; Du, Y.; Su, Y.; Guo, Y.; Zhu, Z.; Dong, A. A review on recent achievements and current challenges in antibacterial electrospun n-halamines. Colloid Interface Sc. Commun. 2018, 24, 24–34. [Google Scholar] [CrossRef]
- Hou, J.; Zhang, Z.; Madsen, L.A. Cation/anion associations in ionic liquids modulated by hydration and ionic medium. J. Phys. Chem. B 2011, 115, 4576–4582. [Google Scholar] [CrossRef] [PubMed]
- Kowalczyk, P.; Borkowski, A.; Czerwonka, G.; Cłapa, T.; Cieśla, J.; Misiewicz, A.; Borowiec, M.; Szala, M. The microbial toxicity of quaternary ammonium ionic liquids is dependent on the type of lipopolysaccharide. J. Mol. Liquids 2018, 266, 540–547. [Google Scholar] [CrossRef]
- Hough, W.L.; Smiglak, M.; Rodriguez, H.; Swatloski, R.P.; Spear, S.K.; Daly, D.T.; Pernak, J.; Grisel, J.E.; Carliss, R.D.; Soutullo, M.D.; et al. The third evolution of ionic liquids: Active pharmaceutical ingredients. New J. Chem. 2007, 31, 1429–1436. [Google Scholar] [CrossRef]
- Zakrewsky, M.; Lovejoy, K.S.; Kern, T.L.; Miller, T.E.; Le, V.; Nagy, A.; Goumas, A.M.; Iyer, R.S.; Del Sesto, R.E.; Koppisch, A.T.; et al. Ionic liquids as a class of materials for transdermal delivery and pathogen neutralization. Proc. Natl. Acad. Sci. USA 2014, 111, 13313–13318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cole, M.R.; Li, M.; El-Zahab, B.; Janes, M.E.; Hayes, D.; Warner, I.M. Design, synthesis, and biological evaluation of β-lactam antibiotic-based imidazolium- and pyridinium-type ionic liquids. Chem. Biol. Drug Des. 2011, 78, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Sommer, J.; Fister, S.; Gundolf, T.; Bromberger, B.; Mester, P.J.; Witte, A.K.; Kalb, R.; Rossmanith, P. Virucidal or not virucidal? That is the question-predictability of ionic liquid's virucidal potential in biological test systems. Int. J. Mol. Sci. 2018, 19, 790. [Google Scholar] [CrossRef] [PubMed]
- Fister, S.; Mester, P.; Sommer, J.; Witte, A.K.; Kalb, R.; Wagner, M.; Rossmanith, P. Virucidal influence of ionic liquids on phages p100 and ms2. Front. Microbiol. 2017, 8, 1608. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.X.; Li, J.S.; Xu, R.; Yang, S.Y. An environmental friendly approach for the synthesis of the ionic liquid 1-ethyl-3-methylimidazolium acetate and its dissolubility to 1, 3, 5-triamino-2, 4, 6-trinitrobenzene. J. Mol. Liquids 2012, 165, 173–176. [Google Scholar] [CrossRef]
- Srour, H.; Rouault, H.; Santini, C.C.; Chauvin, Y. A silver and water free metathesis reaction: A route to ionic liquids. Green Chem. 2013, 15, 1341–1347. [Google Scholar] [CrossRef]
- Agatemor, C.; Ibsen, K.N.; Tanner, E.E.L.; Mitragotri, S. Ionic liquids for addressing unmet needs in healthcare. Bioeng. Transl. Med. 2018, 3, 7–25. [Google Scholar] [PubMed]
- Vander Hoogerstraete, T.; Jamar, S.; Wellens, S.; Binnemans, K. Determination of halide impurities in ionic liquids by total reflection x-ray fluorescence spectrometry. Anal. Chem. 2014, 86, 3931–3938. [Google Scholar] [CrossRef] [PubMed]
- Sainz-Diaz, C.I.; Francisco-Marquez, M.; Soriano-Correa, C. Polymorphism, intermolecular interactions, and spectroscopic properties in crystal structures of sulfonamides. J. Pharm. Sci. 2018, 107, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Docherty, K.M.; Joyce, M.V.; Kulacki, K.J.; Kulpa, C.F. Microbial biodegradation and metabolite toxicity of three pyridinium-based cation ionic liquids. Green Chem. 2010, 12, 701–712. [Google Scholar] [CrossRef]
- Wasserscheid, P.; Welton, T. (Eds.) Synthesis and purification of ionic liquids. In Ionic Liquids in Synthesis; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- Hough-Troutman, W.L.; Smiglak, M.; Griffin, S.; Matthew Reichert, W.; Mirska, I.; Jodynis-Liebert, J.; Adamska, T.; Nawrot, J.; Stasiewicz, M.; Rogers, R.D.; et al. Ionic liquids with dual biological function: Sweet and anti-microbial, hydrophobic quaternary ammonium-based salts. New J. Chem. 2009, 33, 26–33. [Google Scholar] [CrossRef]
- Stoimenovski, J.; MacFarlane, D.R.; Bica, K.; Rogers, R.D. Crystalline vs. Ionic liquid salt forms of active pharmaceutical ingredients: A position paper. Pharm. Res. 2010, 27, 521–526. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Xu, Q.; Guo, J.; Qin, J.; Mao, H.; Wang, B.; Yan, F. Structure-antibacterial activity relationships of imidazolium-type ionic liquid monomers, poly(ionic liquids) and poly(ionic liquid) membranes: Effect of alkyl chain length and cations. ACS Appl. Mater. Interfaces 2016, 8, 12684–12692. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Liu, D.; Dong, Y.; Chen, J.; Liu, H. Effect of ionic liquids with different cations and anions on photosystem and cell structure of scenedesmus obliquus. Chemosphere 2018, 195, 437–447. [Google Scholar] [CrossRef] [PubMed]
- Ibsen, K.N.; Ma, H.; Banerjee, A.; Tanner, E.E.L.; Nangia, S.; Mitragotri, S. Mechanism of antibacterial activity of choline-based ionic liquids (cage). ACS Biomater. Sci. Eng. 2018, 4, 2370–2379. [Google Scholar] [CrossRef]
- Abouserie, A.; Zehbe, K.; Metzner, P.; Kelling, A.; Günter, C.; Schilde, U.; Strauch, P.; Körzdörfer, T.; Taubert, A. Alkylpyridinium tetrahalidometallate ionic liquids and ionic liquid crystals: Insights into the origin of their phase behavior. Eur. J. Inorg. Chem. 2017, 2017, 5640–5649. [Google Scholar] [CrossRef]
- Jing, B.; Lan, N.; Qiu, J.; Zhu, Y. Interaction of ionic liquids with a lipid bilayer: A biophysical study of ionic liquid cytotoxicity. J. Phys. Chem. B 2016, 120, 2781–2789. [Google Scholar] [CrossRef] [PubMed]
- Witos, J.; Russo, G.; Ruokonen, S.-K.; Wiedmer, S.K. Unraveling interactions between ionic liquids and phospholipid vesicles using nanoplasmonic sensing. Langmuir 2017, 33, 1066–1076. [Google Scholar] [CrossRef] [PubMed]
- Tran, C.D.; Prosenc, F.; Franko, M. Facile synthesis, structure, biocompatibility and antimicrobial property of gold nanoparticle composites from cellulose and keratin. J. Colloid Interface Sci. 2018, 510, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Ubeda-Torres, M.T.; Ortiz-Bolsico, C.; Garcia-Alvarez-Coque, M.C.; Ruiz-Angel, M.J. Gaining insight in the behaviour of imidazolium-based ionic liquids as additives in reversed-phase liquid chromatography for the analysis of basic compounds. J. Chromatogr. A 2015, 1380, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Payagala, T.; Armstrong, D.W. Chiral ionic liquids: A compendium of syntheses and applications (2005–2012). Chirality 2012, 24, 17–53. [Google Scholar] [CrossRef] [PubMed]
- Baraniak, A.; Izdebski, R.; Herda, M.; Fiett, J.; Hryniewicz, W.; Gniadkowski, M.; Kern-Zdanowicz, I.; Filczak, K.; Lopaciuk, U. Emergence of klebsiella pneumoniae st258 with kpc-2 in poland. Antimicrob. Agents Chemother. 2009, 53, 4565–4567. [Google Scholar] [CrossRef] [PubMed]
- Monaco, M.; Giani, T.; Raffone, M.; Arena, F.; Garcia-Fernandez, A.; Pollini, S.; Network Eu, S.-I.; Grundmann, H.; Pantosti, A.; Rossolini, G.M. Colistin resistance superimposed to endemic carbapenem-resistant klebsiella pneumoniae: A rapidly evolving problem in italy, november 2013 to april 2014. Eurosurveillance 2014, 19, 20939. [Google Scholar] [CrossRef] [PubMed]
- Beyrouthy, R.; Robin, F.; Dabboussi, F.; Mallat, H.; Hamze, M.; Bonnet, R. Carbapenemase and virulence factors of enterobacteriaceae in north lebanon between 2008 and 2012: Evolution via endemic spread of oxa-48. J. Antimicrob. Chemother. 2014, 69, 2699–2705. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, K.L.; Paterson, D.L. Long-term mortality following pseudomonas aeruginosa bloodstream infection. J. Hosp. Infect. 2017, 95, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Król, J.E.; Wojtowicz, A.J.; Rogers, L.M.; Heuer, H.; Smalla, K.; Krone, S.M.; Top, E.M. Invasion of E. coli biofilms by antibiotic resistance plasmids. Plasmid 2013, 70, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Yong, D.; Toleman, M.A.; Giske, C.G.; Cho, H.S.; Sundman, K.; Lee, K.; Walsh, T.R. Characterization of a new metallo-beta-lactamase gene, bla(ndm-1), and a novel erythromycin esterase gene carried on a unique genetic structure in klebsiella pneumoniae sequence type 14 from india. Antimicrob. Agents Chemother. 2009, 53, 5046–5054. [Google Scholar] [CrossRef] [PubMed]
- Nordmann, P. Carbapenemase-producing enterobacteriaceae: Overview of a major public health challenge. Med. Mal. Infect. 2014, 44, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Rolain, J.M.; Parola, P.; Cornaglia, G. New delhi metallo-beta-lactamase (ndm-1): Towards a new pandemia? Clin. Microbiol. Infect. 2010, 16, 1699–1701. [Google Scholar] [CrossRef] [PubMed]
- Abbaszadegan, A.; Gholami, A.; Abbaszadegan, S.; Aleyasin, Z.S.; Ghahramani, Y.; Dorostkar, S.; Hemmateenejad, B.; Ghasemi, Y.; Sharghi, H. The effects of different ionic liquid coatings and the length of alkyl chain on antimicrobial and cytotoxic properties of silver nanoparticles. Iran. Endod. J. 2017, 12, 481–487. [Google Scholar] [PubMed]
- Pendleton, J.N.; Gilmore, B.F. The antimicrobial potential of ionic liquids: A source of chemical diversity for infection and biofilm control. Int. J. Antimicrob. Agents 2015, 46, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Busetti, A.; Crawford, D.E.; Earle, M.J.; Gilea, M.A.; Gilmore, B.F.; Gorman, S.P.; Laverty, G.; Lowry, A.F.; McLaughlin, M.; Seddon, K.R. Antimicrobial and antibiofilm activities of 1-alkylquinolinium bromide ionic liquids. Green Chem. 2010, 12, 420–425. [Google Scholar] [CrossRef]
- Zeeb, M.; R Ganjali, M.; Norouzi, P. Modified ionic liquid cold-induced aggregation dispersive liquid-liquid microextraction combined with spectrofluorimetry for trace determination of ofloxacin in pharmaceutical and biological samples. DARU J. Pharm. Sci. 2011, 19, 446–454. [Google Scholar]
- Thuy Pham, T.P.; Cho, C.-W.; Yun, Y.-S. Environmental fate and toxicity of ionic liquids: A review. Water Res. 2010, 44, 352–372. [Google Scholar] [CrossRef] [PubMed]
- Gulen, T.A.; Guner, R.; Celikbilek, N.; Keske, S.; Tasyaran, M. Clinical importance and cost of bacteremia caused by nosocomial multi drug resistant acinetobacter baumannii. Int. J. Infect. Dis. 2015, 38, 32–35. [Google Scholar] [CrossRef] [PubMed]
- Phoon, H.Y.P.; Hussin, H.; Hussain, B.M.; Lim, S.Y.; Woon, J.J.; Er, Y.X.; Thong, K.L. Distribution, genetic diversity and antibiotic resistance of clinically important bacteria from the environment of a tertiary hospital. J. Glob. Antimicrob. Resist. 2018, 14, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Ellis, D.; Cohen, B.; Liu, J.; Larson, E. Risk factors for hospital-acquired antimicrobial-resistant infection caused by acinetobacter baumannii. Antimicrob. Resist. Infect. Control 2015, 4, 40. [Google Scholar] [CrossRef] [PubMed]
- Sunenshine, R.H.; Wright, M.O.; Maragakis, L.L.; Harris, A.D.; Song, X.; Hebden, J.; Cosgrove, S.E.; Anderson, A.; Carnell, J.; Jernigan, D.B.; et al. Multidrug-resistant acinetobacter infection mortality rate and length of hospitalization. Emerg. Infect. Dis. 2007, 13, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Dai, X.; Ni, L.; Chen, B.; Luo, Z.; Yao, Y.; Wu, X.; Li, H.; Huang, S. Molecular epidemiology and virulence characteristics of staphylococcus aureus nasal colonization in medical laboratory staff: Comparison between microbiological and non-microbiological laboratories. BMC Infect. Dis. 2018, 18, 122. [Google Scholar] [CrossRef] [PubMed]
- van Hal, S.J.; Jensen, S.O.; Vaska, V.L.; Espedido, B.A.; Paterson, D.L.; Gosbell, I.B. Predictors of mortality in staphylococcus aureus bacteremia. Clin. Microbiol. Rev. 2012, 25, 362–386. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.T.; Liao, C.H.; Chen, S.Y.; Hsu, H.S.; Teng, L.J.; Hsueh, P.R. Emergence of multidrug-resistant sequence type 45 strains among borderline oxacillin-resistant meca-positive staphylococcus aureus causing bacteraemia in a medical centre in taiwan. Int. J. Antimicrob. Agents 2018, 52, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Ma, X.X.; Takeuchi, F.; Okuma, K.; Yuzawa, H.; Hiramatsu, K. Novel type v staphylococcal cassette chromosome mec driven by a novel cassette chromosome recombinase, ccrc. Antimicrob. Agents Chemother. 2004, 48, 2637–2651. [Google Scholar] [CrossRef] [PubMed]
- Osaka, S.; Okuzumi, K.; Koide, S.; Tamai, K.; Sato, T.; Tanimoto, K.; Tomita, H.; Suzuki, M.; Nagano, Y.; Shibayama, K.; et al. Genetic shifts in methicillin-resistant staphylococcus aureus epidemic clones and toxin gene profiles in japan: Comparative analysis among pre-epidemic, epidemic and post-epidemic phases. J. Med M.icrobiol. 2018, 67, 392–399. [Google Scholar] [CrossRef] [PubMed]
- O'Toole, G.A.; Wathier, M.; Zegans, M.E.; Shanks, R.M.; Kowalski, R.; Grinstaff, M.W. Diphosphonium ionic liquids as broad-spectrum antimicrobial agents. Cornea 2012, 31, 810–816. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.D.; Liu, Q.P.; Smith, T.J.; Li, N.; Zong, M.H. Evaluation of toxicity and biodegradability of cholinium amino acids ionic liquids. PLoS ONE 2013, 8, e59145. [Google Scholar] [CrossRef] [PubMed]
- Torrecilla, J.S.; Garcia, J.; Rojo, E.; Rodriguez, F. Estimation of toxicity of ionic liquids in leukemia rat cell line and acetylcholinesterase enzyme by principal component analysis, neural networks and multiple lineal regressions. J. Hazard. Mater. 2009, 164, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Hodyna, D.; Kovalishyn, V.; Semenyuta, I.; Blagodatnyi, V.; Rogalsky, S.; Metelytsia, L. Imidazolium ionic liquids as effective antiseptics and disinfectants against drug resistant s. Aureus: In silico and in vitro studies. Comput. Biol. Chem. 2018, 73, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Zhang, S.; Dai, Y.; Lu, X.; Lei, Q.; Fang, W. Antimicrobial activity and cytotoxicity of piperazinium- and guanidinium-based ionic liquids. J. Hazard. Mater. 2016, 307, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Abbaszadegan, A.; Nabavizadeh, M.; Gholami, A.; Aleyasin, Z.S.; Dorostkar, S.; Saliminasab, M.; Ghasemi, Y.; Hemmateenejad, B.; Sharghi, H. Positively charged imidazolium-based ionic liquid-protected silver nanoparticles: A promising disinfectant in root canal treatment. Int. Endod. J. 2015, 48, 790–800. [Google Scholar] [CrossRef] [PubMed]
- Holland, T.L.; Arnold, C.; Fowler, V.G., Jr. Clinical management of staphylococcus aureus bacteremia: A review. JAMA 2014, 312, 1330–1341. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.W.; Park, J.S.; Stolte, S.; Yun, Y.S. Modelling for antimicrobial activities of ionic liquids towards escherichia coli, staphylococcus aureus and candida albicans using linear free energy relationship descriptors. J. Hazard. Mater. 2016, 311, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Elyasi, S.; Khalili, H.; Dashti-Khavidaki, S.; Emadi-Koochak, H.; Mohammadpour, A.; Abdollahi, A. Elevated vancomycin trough concentration: Increased efficacy and/or toxicity? Iran. J. Pharm. Res. 2014, 13, 1241–1247. [Google Scholar] [PubMed]
- Arning, J.; Stolte, S.; Boschen, A.; Stock, F.; Pitner, W.-R.; Welz-Biermann, U.; Jastorff, B.; Ranke, J. Qualitative and quantitative structure activity relationships for the inhibitory effects of cationic head groups, functionalised side chains and anions of ionic liquids on acetylcholinesterase. Green Chem. 2008, 10, 47–58. [Google Scholar] [CrossRef]
- Ling, L.L.; Schneider, T.; Peoples, A.J.; Spoering, A.L.; Engels, I.; Conlon, B.P.; Mueller, A.; Schaberle, T.F.; Hughes, D.E.; Epstein, S.; et al. A new antibiotic kills pathogens without detectable resistance. Nature 2015, 517, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Moellering, R.C., Jr. Ndm-1—A cause for worldwide concern. N. Engl. J. Med. 2010, 363, 2377–2379. [Google Scholar] [CrossRef] [PubMed]
- Firoozeh, F.; Mahluji, Z.; Shams, E.; Khorshidi, A.; Zibaei, M. New delhi metallo-beta-lactamase-1-producing klebsiella pneumoniae isolates in hospitalized patients in kashan, iran. Iran. J. Microbiol. 2017, 9, 283–287. [Google Scholar] [PubMed]
- Hussein, N.H. Emergence of ndm-1 among carbapenem-resistant klebsiella pneumoniae in iraqi hospitals. Acta Microbiol. Immunol. Hung. 2018, 65, 211–227. [Google Scholar] [CrossRef] [PubMed]
- Cornaglia, G.; Giamarellou, H.; Rossolini, G.M. Metallo-beta-lactamases: A last frontier for beta-lactams? Lancet Infect. Dis. 2011, 11, 381–393. [Google Scholar] [CrossRef]
- Jamal, W.Y.; Albert, M.J.; Rotimi, V.O. High prevalence of new delhi metallo-beta-lactamase-1 (ndm-1) producers among carbapenem-resistant enterobacteriaceae in kuwait. PLoS ONE 2016, 11, e0152638. [Google Scholar] [CrossRef] [PubMed]
- Pendleton, J.N.; Gorman, S.P.; Gilmore, B.F. Clinical relevance of the ESKAPE pathogens. Anti. Infect. Ther. 2013, 11, 297–308. [Google Scholar] [CrossRef] [PubMed]
- Henke, E.; Schuster, S.; Yang, H.; Bornscheuer, U.T. Lipase-catalyzed resolution of ibuprofen. Monatsh. Chem./Chem. Mon. 2000, 131, 633–638. [Google Scholar] [CrossRef]
- Zhao, D.; Wu, M.; Kou, Y.; Min, E. Ionic liquids: Applications in catalysis. Catal. Today 2002, 74, 157–189. [Google Scholar] [CrossRef]
- Gomes de Azevedo, R.; Esperança, J.M.S.S.; Szydlowski, J.; Visak, Z.P.; Pires, P.F.; Guedes, H.J.R.; Rebelo, L.P.N. Thermophysical and thermodynamic properties of ionic liquids over an extended pressure range: [bmim][ntf2] and [hmim][ntf2]. J. Chem. Thermodyn. 2005, 37, 888–899. [Google Scholar] [CrossRef]



| Pathogen | Strain Characteristics | Number Of Detected Cases | Confirmed Cases Of ICU* Acquired Infections | Percentage Of Selected Strains Detected Is Surgical Site Infections | Drug Resistance | Comorbidity Index | Mortality | Reference |
|---|---|---|---|---|---|---|---|---|
| Acinetobacter baumanii | Nosocomial | 712 | 541 | 4.1% | 3rd eneration cephalosporins | 2.8 | 18% | [85,87] |
| MDR+ | 3.9 | 26% | [88] | |||||
| Klebsiella pneumoaniae | New Delhi blaNDM-1 | 1367 | 561 | 4.7% | 3rd generation Cephalosporins, Meropenem, Vancomycin | 6.4 | 72% | BIOCONTAM Unit |
| Pseudomonas aeruginosa | PAPI+ | 2 269 | 516 | 7.1% | Aminoglycosides, Gyrase inhibitors, Penicillin with β-lactamase inhibitor | 4.8 | 38% | [75,80,89] EARS-Net |
| Staphylococcus aureus | MRSA | 1996 | 631 | 38.1% | β-lactams, Lincosamides, Fluoroquinolones | 4.5 | 30% | [90] |
| Enterococcus species | VRE | 492 | 340 | 20.4% | Vancomycin, Teicoplanin | 2.7 | 43.1% | [85] ECDC |
| other Enterobacteriaceae | ESBL+ | 1367 | 479 | 3.3% | β-lactams, 3rd generation Cephalosporins, Trimethoprim/Sulfomethaxazole | 1.8 | 18.2% | BIOHAZ team, EARS-Net |
| Bacterial Strain | Strain Specification | Antibiotic Resistance Chracteristics | Ionic Liquid | Principal Mechanism of Action | MIC [Mmol L−1] | MBC [Mmol L−1] | References |
|---|---|---|---|---|---|---|---|
| Staphylococcus aureus | HA-MRSA | MGEs+ | [C8mim][Cl] | Collapse of liposomes, localized perforations in dioleoylphosphatidylcholine bilayers | 116 | 170 | [51,86,89,91,92,93,94,95,97,98,101] |
| MZ100 | SCCmec+IV-V | [C4C1Im] | Perforation of dipalmitoylophosphatidylcholine with phospholipid bilayers [Tf2N], isotopic substitution of hydrogen | 188 | 250 | ||
| tsst-1 | |||||||
| BORSA | pvl+ | [Chol][Cl] | 125 | 188 | |||
| USA300 | icaD+ | Di-But C6 | Leakage of cellular liposomes, alternation of apolar regions by protic ionic liquids | 1000 | |||
| CA-MRSA | hlα-γ+ | Di-Hex C6 | 8 | 4 | |||
| ATCC25923 | NorA efflux pump | BTFLA | 23 | 43 | |||
| MDRSA | |||||||
| 1,3-dialkiloimidazolinum | Docking complex formation with tubulin FtsZ PC190723, Increased affinity to the membrane of cancerous cells—interaction with phosphatydilserine | 2.12 | 6.14 | ||||
| [C1C14 pi][BF4 ]2 | 5.6 | 19.3 | |||||
| [Phpi][BF4] | 1.2 | 4.8 | |||||
| [(C2)2(C1)2(C1)2 3gu][C2OSO3] | >100 | 342 | |||||
| ATCC6538 | C16M1Im][Br] | 23 | NA | ||||
| [C16M1Im][Amp]* | 7.9 | 7.9 ** | |||||
| Enterococcus species | COM12-15 E1071 E4452-E4453 AUS0004 | VRE | [C12Py] | De-stabilising effect on lipid structure | 8.1 | 8.1 ** | [38,66,69,80,99] |
| [C18Py] | 8.5 | 8.5 ** | |||||
| [EMIm+Tf2N-] | Mismatch between ionic liquid cations and lipids in the layer | 16.8 | 34.6 | ||||
| [C12Im] | Long-tail cation mediated cytotoxicity, electrostatic signature interacting with peptidoglycan | 7.1 | 7.1 ** | ||||
| [C18Im] | 8.1 | 8.1 ** | |||||
| [BMIm+Cl-] | 7.5 | 7.5 ** | |||||
| Ag+C3H5N2-p | 5.7 × 10−10 | 5.7 × 10−10 ** | |||||
| Klebsiella pneumoniae | ATCC4352 | Metallo-β-lactamse-1 | [C2mim][Cl] | Inhibition of acetylcholineesetrase | 178 | 263 | [51,96] |
| [CBP] | Interaction with phosphatidic acid, apoptosis triggering | 13 | NA | ||||
| [CPB][AMP] * | Antibiotic donor | 4.7 | 9 ** | ||||
| [C16M2Im][Br] | Interaction with cytoskeleton protein subunits | 15 | NA | ||||
| [C16M2Im][AMP] * | Antibiotic donor | 7.8 | 7.8 ** | ||||
| Enterobacteriaceae genus | W3110 | OXA-48 carbapenemase | [Chol]+ Thre | Interaction with biomembranes surrounding cellular organelles | 31.3 | 62.5 | [94,95,102] |
| [Chol]+ Pro | 46.9 | 62 | |||||
| β-lactamase encoding genes K1-2 capsular serotypes | Di-But C10 | Decreased flexural rigidity and reduced interfacial tension between the bilayer and ionic liquid, lysis of bacterial outer membrane | 40 | 40 ** | |||
| Di-Hex C10 | 8 | 8 ** | |||||
| PTLFS | 91 | 470 | |||||
| [(C2)2 2(C1)2(C1)2 3gu][C2OSO3] | 12.5 | 39.5 | |||||
| [C2pi][BF4] | 2.38 | 11.8 | |||||
| [C2C1C14pi][I] | >50 | 185 | |||||
| Pseudomonas aeruginosa | PA14 | OM-proteins | Di-Hex C10 | Coagulation of cytoplasm | 9 | 20 | [50,103] |
| ATCC 27853 | MDEP | 1,3 dialkiloimidazolinum | Asymetric absorbtion of ionic liquid cation by leaflets of phospholipid bilayer | 18.4 | 45.8 | ||
| BMP-NTf2 | 24 | 65.6 | |||||
| HMIM-Cl | 12.4 | 61.5 | |||||
| [P(C14H29)(C6H13)3]+ | 8.4 | 20 | |||||
| (ZnCl2)2 - BZBN | 12.3 | 31 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miskiewicz, A.; Ceranowicz, P.; Szymczak, M.; Bartuś, K.; Kowalczyk, P. The Use of Liquids Ionic Fluids as Pharmaceutically Active Substances Helpful in Combating Nosocomial Infections Induced by Klebsiella Pneumoniae New Delhi Strain, Acinetobacter Baumannii and Enterococcus Species. Int. J. Mol. Sci. 2018, 19, 2779. https://doi.org/10.3390/ijms19092779
Miskiewicz A, Ceranowicz P, Szymczak M, Bartuś K, Kowalczyk P. The Use of Liquids Ionic Fluids as Pharmaceutically Active Substances Helpful in Combating Nosocomial Infections Induced by Klebsiella Pneumoniae New Delhi Strain, Acinetobacter Baumannii and Enterococcus Species. International Journal of Molecular Sciences. 2018; 19(9):2779. https://doi.org/10.3390/ijms19092779
Chicago/Turabian StyleMiskiewicz, Andrzej, Piotr Ceranowicz, Mateusz Szymczak, Krzysztof Bartuś, and Paweł Kowalczyk. 2018. "The Use of Liquids Ionic Fluids as Pharmaceutically Active Substances Helpful in Combating Nosocomial Infections Induced by Klebsiella Pneumoniae New Delhi Strain, Acinetobacter Baumannii and Enterococcus Species" International Journal of Molecular Sciences 19, no. 9: 2779. https://doi.org/10.3390/ijms19092779