The Role of Growth Hormone and Insulin-Like Growth Factor-I in the Liver
Abstract
:1. Non-Alcoholic Fatty Liver Disease (NAFLD)
2. Endocrine Abnormalities and NAFLD
3. The Metabolic Action of Growth Hormone (GH) and Pathophysiology in Adult Growth Hormone Deficiency (GHD)
4. NAFLD Is an Important Complication in Adult GHD
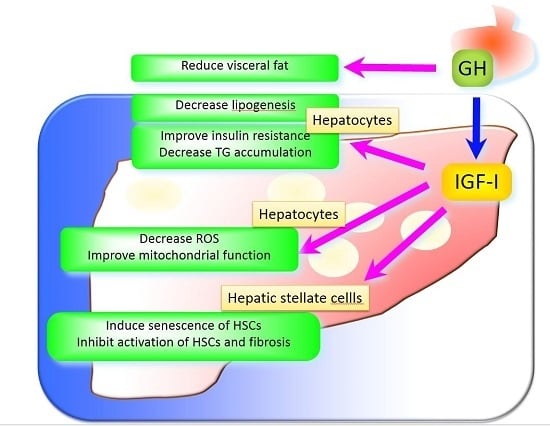
5. The Underlying Mechanisms
6. The Clinical Applications of GH and IGF-I in NASH and Cirrhosis
7. Conclusions
Acknowledgments
Conflicts of Interest
References
- Angulo, P. Nonalcoholic fatty liver disease. N. Engl. J. Med. 2002, 346, 1221–1231. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, E.; Tokushige, K. Prevalence, gender, ethnic variations, and prognosis of NASH. J. Gastroenterol. 2011, 46, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Italian Association for the Study of the Liver (AISF). Aisf position paper on nonalcoholic fatty liver disease (nafld): Updates and future directions. Dig. Liver Dis. 2017, 49, 471–483. [Google Scholar]
- Cordoba-Chacon, J.; Majumdar, N.; List, E.O.; Diaz-Ruiz, A.; Frank, S.J.; Manzano, A.; Bartrons, R.; Puchowicz, M.; Kopchick, J.J.; Kineman, R.D. Growth hormone inhibits hepatic de novo lipogenesis in adult mice. Diabetes 2015, 64, 3093–3103. [Google Scholar] [CrossRef] [PubMed]
- Adams, L.A.; Talwalkar, J.A. Diagnostic evaluation of nonalcoholic fatty liver disease. J. Clin. Gastroenterol. 2006, 40, S34–S38. [Google Scholar] [PubMed]
- Ballestri, S.; Nascimbeni, F.; Romagnoli, D.; Baldelli, E.; Lonardo, A. The role of nuclear receptors in the pathophysiology, natural course, and drug treatment of nafld in humans. Adv. Ther. 2016, 33, 291–319. [Google Scholar] [CrossRef] [PubMed]
- Lonardo, A.; Bellentani, S.; Argo, C.K.; Ballestri, S.; Byrne, C.D.; Caldwell, S.H.; Cortez-Pinto, H.; Grieco, A.; Machado, M.V.; et al. Epidemiological modifiers of non-alcoholic fatty liver disease: Focus on high-risk groups. Dig. Liver Dis. 2015, 47, 997–1006. [Google Scholar] [CrossRef] [PubMed]
- Adams, L.A.; Angulo, P.; Lindor, K.D. Nonalcoholic fatty liver disease. CMAJ 2005, 172, 899–905. [Google Scholar] [CrossRef] [PubMed]
- Wree, A.; Broderick, L.; Canbay, A.; Hoffman, H.M.; Feldstein, A.E. From nafld to NASH to cirrhosis-new insights into disease mechanisms. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 627–636. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Gea, V.; Friedman, S.L. Pathogenesis of liver fibrosis. Annu. Rev. Pathol. 2011, 6, 425–456. [Google Scholar] [CrossRef] [PubMed]
- Angulo, P.; Kleiner, D.E.; Dam-Larsen, S.; Adams, L.A.; Bjornsson, E.S.; Charatcharoenwitthaya, P.; Mills, P.R.; Keach, J.C.; Lafferty, H.D.; Stahler, A.; et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology 2015, 149, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.; Muller, H.L. Novel perspectives on hypothalamic-pituitary dysfunction as a risk factor for non-alcoholic fatty liver disease. Minerva Endocrinol. 2017, 42, 132–144. [Google Scholar] [PubMed]
- Lonardo, A.; Carani, C.; Carulli, N.; Loria, P. ‘Endocrine nafld’ a hormonocentric perspective of nonalcoholic fatty liver disease pathogenesis. J. Hepatol. 2006, 44, 1196–1207. [Google Scholar] [CrossRef] [PubMed]
- Loria, P.; Carulli, L.; Bertolotti, M.; Lonardo, A. Endocrine and liver interaction: The role of endocrine pathways in NASH. Nat. Rev. Gastroenterol. Hepatol. 2009, 6, 236–247. [Google Scholar] [CrossRef] [PubMed]
- Rockall, A.G.; Sohaib, S.A.; Evans, D.; Kaltsas, G.; Isidori, A.M.; Monson, J.P.; Besser, G.M.; Grossman, A.B.; Reznek, R.H. Hepatic steatosis in cushing’s syndrome: A radiological assessment using computed tomography. Eur. J. Endocrinol. 2003, 149, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Sumida, Y.; Yonei, Y.; Kanemasa, K.; Hara, T.; Inada, Y.; Sakai, K.; Imai, S.; Hibino, S.; Yamaguchi, K.; Mitsuyoshi, H.; et al. Lower circulating levels of dehydroepiandrosterone, independent of insulin resistance, is an important determinant of severity of non-alcoholic steatohepatitis in japanese patients. Hepatol. Res. 2010, 40, 901–910. [Google Scholar] [CrossRef] [PubMed]
- Volzke, H.; Aumann, N.; Krebs, A.; Nauck, M.; Steveling, A.; Lerch, M.M.; Rosskopf, D.; Wallaschofski, H. Hepatic steatosis is associated with low serum testosterone and high serum dheas levels in men. Int. J. Androl. 2010, 33, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Kalinchenko, S.Y.; Tishova, Y.A.; Mskhalaya, G.J.; Gooren, L.J.; Giltay, E.J.; Saad, F. Effects of testosterone supplementation on markers of the metabolic syndrome and inflammation in hypogonadal men with the metabolic syndrome: The double-blinded placebo-controlled moscow study. Clin. Endocrinol. 2010, 73, 602–612. [Google Scholar] [CrossRef] [PubMed]
- Mu, Y.; She, R.; Zhang, H.; Dong, B.; Huang, C.; Lin, W.; Li, D.; Li, X. Effects of estrogen and androgen deprivation on the progression of non-alcoholic steatohepatitis (NASH) in male sprague-dawley rats. Hepatol. Res. 2009, 39, 910–920. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Wu, Z.; Zhao, Y.; Meng, Y.; Si, Y.; Fu, X.; Mu, Y.; Han, W. Differential induction of lrp16 by liganded and unliganded estrogen receptor α in SKOV3 ovarian carcinoma cells. J. Endocrinol. 2009, 202, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Targher, G.; Rossini, M.; Lonardo, A. Evidence that non-alcoholic fatty liver disease and polycystic ovary syndrome are associated by necessity rather than chance: A novel hepato-ovarian axis? Endocrine 2016, 51, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Kargi, A.Y.; Merriam, G.R. Diagnosis and treatment of growth hormone deficiency in adults. Nat. Rev. Endocrinol. 2013, 9, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Gazzaruso, C.; Gola, M.; Karamouzis, I.; Giubbini, R.; Giustina, A. Cardiovascular risk in adult patients with growth hormone (GH) deficiency and following substitution with GH—An update. J. Clin. Endocrinol. Metab. 2014, 99, 18–29. [Google Scholar] [CrossRef] [PubMed]
- Lonardo, A.; Ballestri, S.; Guaraldi, G.; Nascimbeni, F.; Romagnoli, D.; Zona, S.; Targher, G. Fatty liver is associated with an increased risk of diabetes and cardiovascular disease—Evidence from three different disease models: Nafld, HCV and HIV. World J. Gastroenterol. 2016, 22, 9674–9693. [Google Scholar] [CrossRef] [PubMed]
- Carroll, P.V.; Christ, E.R.; Bengtsson, B.A.; Carlsson, L.; Christiansen, J.S.; Clemmons, D.; Hintz, R.; Ho, K.; Laron, Z.; Sizonenko, P.; et al. Growth hormone deficiency in adulthood and the effects of growth hormone replacement: A review. Growth hormone research society scientific committee. J. Clin. Endocrinol. Metab. 1998, 83, 382–395. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, Y. Essential roles of growth hormone (GH) and insulin-like growth factor-I (IGF-I) in the liver. Endocr. J. 2012, 59, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Takano, S.; Kanzaki, S.; Sato, M.; Kubo, T.; Seino, Y. Effect of growth hormone on fatty liver in panhypopituitarism. Arch. Dis. Child. 1997, 76, 537–538. [Google Scholar] [CrossRef] [PubMed]
- Kaji, H.; Sakurai, T.; Iguchi, G.; Murata, M.; Kishimoto, M.; Yoshioka, S.; Iida, K.; Okimura, Y.; Chihara, K. Adult growth hormone deficiency in Japan: Results of investigation by questionnaire. Endocr. J. 2002, 49, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, T.; Hamasaki, K.; Ishikawa, H.; Ejima, E.; Eguchi, K.; Nakao, K. Non-alcoholic steatohepatitis and hepatic steatosis in patients with adult onset growth hormone deficiency. Gut 2003, 52, 914. [Google Scholar] [CrossRef] [PubMed]
- Adams, L.A.; Feldstein, A.; Lindor, K.D.; Angulo, P. Nonalcoholic fatty liver disease among patients with hypothalamic and pituitary dysfunction. Hepatology 2004, 39, 909–914. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, I.; Hizuka, N.; Yasumoto, K.; Morita, J.; Kurimoto, M.; Takano, K. Metabolic co-morbidities revealed in patients with childhood-onset adult gh deficiency after cessation of GH replacement therapy for short stature. Endocr. J. 2008, 55, 977–984. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, Y.; Iida, K.; Takahashi, K.; Yoshioka, S.; Fukuoka, H.; Takeno, R.; Imanaka, M.; Nishizawa, H.; Takahashi, M.; Seo, Y.; et al. Growth hormone reverses nonalcoholic steatohepatitis in a patient with adult growth hormone deficiency. Gastroenterology 2007, 132, 938–943. [Google Scholar] [CrossRef] [PubMed]
- Nishizawa, H.; Iguchi, G.; Murawaki, A.; Fukuoka, H.; Hayashi, Y.; Kaji, H.; Yamamoto, M.; Suda, K.; Takahashi, M.; Seo, Y.; et al. Nonalcoholic fatty liver disease in adult hypopituitary patients with gh deficiency and the impact of GH replacement therapy. Eur. J. Endocrinol. 2012, 167, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, R.; Fukuoka, H.; Iguchi, G.; Nishizawa, H.; Bando, H.; Suda, K.; Takahashi, M.; Takahashi, Y. Long-term effects of growth hormone replacement therapy on liver function in adult patients with growth hormone deficiency. Growth Horm. IGF Res. 2014, 24, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Gardner, C.J.; Irwin, A.J.; Daousi, C.; McFarlane, I.A.; Joseph, F.; Bell, J.D.; Thomas, E.L.; Adams, V.L.; Kemp, G.J.; Cuthbertson, D.J. Hepatic steatosis, GH deficiency and the effects of GH replacement: A liverpool magnetic resonance spectroscopy study. Eur. J. Endocrinol. 2012, 166, 993–1002. [Google Scholar] [CrossRef] [PubMed]
- Meienberg, F.; Yee, M.; Johnston, D.; Cox, J.; Robinson, S.; Bell, J.D.; Thomas, E.L.; Taylor-Robinson, S.D.; Godsland, I. Liver fat in adults with gh deficiency: Comparison to matched controls and the effect of GH replacement. Clin. Endocrinol. 2016, 85, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Vernon, G.; Baranova, A.; Younossi, Z.M. Systematic review: The epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment. Pharmacol. Ther. 2011, 34, 274–285. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.W.; Kim, J.Y.; Kim, Y.E.; Lee, E.J. Metabolic parameters and nonalcoholic fatty liver disease in hypopituitary men. Horm. Metab. Res. 2011, 43, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, T.; Nakao, K.; Hamasaki, K.; Furukawa, R.; Tsuruta, S.; Ueda, Y.; Taura, N.; Shibata, H.; Fujimoto, M.; Toriyama, K.; et al. Role of growth hormone, insulin-like growth factor 1 and insulin-like growth factor-binding protein 3 in development of non-alcoholic fatty liver disease. Hepatol. Int. 2007, 1, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Chishima, S.; Kogiso, T.; Matsushita, N.; Hashimoto, E.; Tokushige, K. The relationship between the growth hormone/insulin-like growth factor system and the histological features of nonalcoholic fatty liver disease. Intern. Med. 2017, 56, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Dichtel, L.E.; Corey, K.E.; Misdraji, J.; Bredella, M.A.; Schorr, M.; Osganian, S.A.; Young, B.J.; Sung, J.C.; Miller, K.K. The association between IGF-I levels and the histologic severity of nonalcoholic fatty liver disease. Clin. Transl. Gastroenterol. 2017, 8, e217. [Google Scholar] [CrossRef] [PubMed]
- Sumida, Y.; Yonei, Y.; Tanaka, S.; Mori, K.; Kanemasa, K.; Imai, S.; Taketani, H.; Hara, T.; Seko, Y.; Ishiba, H.; et al. Lower levels of insulin-like growth factor-I standard deviation score are associated with histological severity of non-alcoholic fatty liver disease. Hepatol. Res. 2015, 45, 771–781. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.L.; Yakar, S.; LeRoith, D. Conditional knockout of mouse insulin-like growth factor-I gene using the cre/loxp system. Proc. Soc. Exp. Biol. Med. 2000, 223, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Yakar, S.; Rosen, C.J.; Beamer, W.G.; Ackert-Bicknell, C.L.; Wu, Y.; Liu, J.L.; Ooi, G.T.; Setser, J.; Frystyk, J.; Boisclair, Y.R.; et al. Circulating levels of IGF-I directly regulate bone growth and density. J. Clin. Investig. 2002, 110, 771–781. [Google Scholar] [CrossRef] [PubMed]
- Yakar, S.; Liu, J.L.; Stannard, B.; Butler, A.; Accili, D.; Sauer, B.; LeRoith, D. Normal growth and development in the absence of hepatic insulin-like growth factor I. Proc. Natl. Acad. Sci. USA 1999, 96, 7324–7329. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Menon, R.K.; Cohen, P.; Hwang, D.; Clemens, T.; DiGirolamo, D.J.; Kopchick, J.J.; Le Roith, D.; Trucco, M.; Sperling, M.A. Liver-specific deletion of the growth hormone receptor reveals essential role of growth hormone signaling in hepatic lipid metabolism. J. Biol. Chem. 2009, 284, 19937–19944. [Google Scholar] [CrossRef] [PubMed]
- Ekberg, S.; Luther, M.; Nakamura, T.; Jansson, J.O. Growth hormone promotes early initiation of hepatocyte growth factor gene expression in the liver of hypophysectomized rats after partial hepatectomy. J. Endocrinol. 1992, 135, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Krupczak-Hollis, K.; Wang, X.; Dennewitz, M.B.; Costa, R.H. Growth hormone stimulates proliferation of old-aged regenerating liver through forkhead box m1b. Hepatology 2003, 38, 1552–1562. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, P.A.; Kopchick, J.J.; Thorgeirsson, S.; LeRoith, D.; Yakar, S. Role of growth hormone (GH) in liver regeneration. Endocrinology 2004, 145, 4748–4755. [Google Scholar] [CrossRef] [PubMed]
- Sos, B.C.; Harris, C.; Nordstrom, S.M.; Tran, J.L.; Balazs, M.; Caplazi, P.; Febbraio, M.; Applegate, M.A.; Wagner, K.U.; Weiss, E.J. Abrogation of growth hormone secretion rescues fatty liver in mice with hepatocyte-specific deletion of jak2. J. Clin. Investig. 2011, 121, 1412–1423. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Hosui, A.; Sun, R.; Shen, K.; Gavrilova, O.; Chen, W.; Cam, M.C.; Gao, B.; Robinson, G.W.; Hennighausen, L. Loss of signal transducer and activator of transcription 5 leads to hepatosteatosis and impaired liver regeneration. Hepatology 2007, 46, 504–513. [Google Scholar] [CrossRef] [PubMed]
- Barclay, J.L.; Nelson, C.N.; Ishikawa, M.; Murray, L.A.; Kerr, L.M.; McPhee, T.R.; Powell, E.E.; Waters, M.J. GH-dependent STAT5 signaling plays an important role in hepatic lipid metabolism. Endocrinology 2011, 152, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Cordoba-Chacon, J.; Kineman, R.D.; Cronstein, B.N.; Muzumdar, R.; Gong, Z.; Werner, H.; Yakar, S. Growth hormone control of hepatic lipid metabolism. Diabetes 2016, 65, 3598–3609. [Google Scholar] [CrossRef] [PubMed]
- Laron, Z.; Ginsberg, S.; Webb, M. Nonalcoholic fatty liver in patients with laron syndrome and gh gene deletion - preliminary report. Growth Horm. IGF Res. 2008, 18, 434–438. [Google Scholar] [CrossRef] [PubMed]
- Donaghy, A.; Ross, R.; Gimson, A.; Hughes, S.C.; Holly, J.; Williams, R. Growth hormone, insulinlike growth factor-I, and insulinlike growth factor binding proteins 1 and 3 in chronic liver disease. Hepatology 1995, 21, 680–688. [Google Scholar] [PubMed]
- Cuneo, R.C.; Hickman, P.E.; Wallace, J.D.; Teh, B.T.; Ward, G.; Veldhuis, J.D.; Waters, M.J. Altered endogenous growth hormone secretory kinetics and diurnal GH-binding protein profiles in adults with chronic liver disease. Clin. Endocrinol. 1995, 43, 265–275. [Google Scholar] [CrossRef]
- Clemmons, D.R.; Klibanski, A.; Underwood, L.E.; McArthur, J.W.; Ridgway, E.C.; Beitins, I.Z.; Van Wyk, J.J. Reduction of plasma immunoreactive somatomedin c during fasting in humans. J. Clin. Endocrinol. Metab. 1981, 53, 1247–1250. [Google Scholar] [CrossRef] [PubMed]
- Caro, J.F.; Poulos, J.; Ittoop, O.; Pories, W.J.; Flickinger, E.G.; Sinha, M.K. Insulin-like growth factor i binding in hepatocytes from human liver, human hepatoma, and normal, regenerating, and fetal rat liver. J. Clin. Investig. 1988, 81, 976–981. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.O.; Park, J.G.; Lee, Y.I. Increased expression of the insulin-like growth factor i (IGF-I) receptor gene in hepatocellular carcinoma cell lines: Implications of IGF-I receptor gene activation by hepatitis b virus X gene product. Cancer Res. 1996, 56, 3831–3836. [Google Scholar] [PubMed]
- Tao, X.; Shen, D.; Ren, H.; Zhang, X.; Zhang, D.; Ye, J.; Gu, B. Hepatitis B virus X protein activates expression of IGF-IR and VEGF in hepatocellular carcinoma cells. Zhonghua Gan Zang Bing Za Zhi 2000, 8, 161–163. [Google Scholar] [PubMed]
- Stefano, J.T.; Correa-Giannella, M.L.; Ribeiro, C.M.; Alves, V.A.; Massarollo, P.C.; Machado, M.C.; Giannella-Neto, D. Increased hepatic expression of insulin-like growth factor-I receptor in chronic hepatitis c. World J. Gastroenterol. 2006, 12, 3821–3828. [Google Scholar] [CrossRef] [PubMed]
- Aleem, E.; Nehrbass, D.; Klimek, F.; Mayer, D.; Bannasch, P. Upregulation of the insulin receptor and type I insulin-like growth factor receptor are early events in hepatocarcinogenesis. Toxicol. Pathol. 2011, 39, 524–543. [Google Scholar] [CrossRef] [PubMed]
- Nishizawa, H.; Takahashi, M.; Fukuoka, H.; Iguchi, G.; Kitazawa, R.; Takahashi, Y. Gh-independent IGF-I action is essential to prevent the development of nonalcoholic steatohepatitis in a GH-deficient rat model. Biochem. Biophys. Res. Commun. 2012, 423, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Bailyes, E.M.; Nave, B.T.; Soos, M.A.; Orr, S.R.; Hayward, A.C.; Siddle, K. Insulin receptor/IGF-I receptor hybrids are widely distributed in mammalian tissues: Quantification of individual receptor species by selective immunoprecipitation and immunoblotting. Biochem. J. 1997, 327, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.W.; Adams, L.A. Non-alcoholic fatty liver disease. Crit. Rev. Clin. Lab. Sci. 2011, 48, 97–113. [Google Scholar] [CrossRef] [PubMed]
- Yakar, S.; Liu, J.L.; Fernandez, A.M.; Wu, Y.; Schally, A.V.; Frystyk, J.; Chernausek, S.D.; Mejia, W.; Le Roith, D. Liver-specific IGF-I gene deletion leads to muscle insulin insensitivity. Diabetes 2001, 50, 1110–1118. [Google Scholar] [CrossRef] [PubMed]
- Hao, C.N.; Geng, Y.J.; Li, F.; Yang, T.; Su, D.F.; Duan, J.L.; Li, Y. Insulin-like growth factor-I receptor activation prevents hydrogen peroxide-induced oxidative stress, mitochondrial dysfunction and apoptosis. Apoptosis 2011, 16, 1118–1127. [Google Scholar] [CrossRef] [PubMed]
- Puche, J.E.; Garcia-Fernandez, M.; Muntane, J.; Rioja, J.; Gonzalez-Baron, S.; Castilla Cortazar, I. Low doses of insulin-like growth factor-I induce mitochondrial protection in aging rats. Endocrinology 2008, 149, 2620–2627. [Google Scholar] [CrossRef] [PubMed]
- Perez, R.; Garcia-Fernandez, M.; Diaz-Sanchez, M.; Puche, J.E.; Delgado, G.; Conchillo, M.; Muntane, J.; Castilla-Cortazar, I. Mitochondrial protection by low doses of insulin-like growth factor-I in experimental cirrhosis. World J. Gastroenterol. 2008, 14, 2731–2739. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Guo, S.; Copps, K.; Dong, X.; Kollipara, R.; Rodgers, J.T.; Depinho, R.A.; Puigserver, P.; White, M.F. Foxo1 integrates insulin signaling with mitochondrial function in the liver. Nat. Med. 2009, 15, 1307–1311. [Google Scholar] [CrossRef] [PubMed]
- Lee, U.E.; Friedman, S.L. Mechanisms of hepatic fibrogenesis. Best Pract. Res. Clin. Gastroenterol. 2011, 25, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Fujii, H.; Kawada, N. Inflammation and fibrogenesis in steatohepatitis. J. Gastroenterol. 2012, 47, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Scharf, J.G.; Knittel, T.; Dombrowski, F.; Muller, L.; Saile, B.; Braulke, T.; Hartmann, H.; Ramadori, G. Characterization of the IGF axis components in isolated rat hepatic stellate cells. Hepatology 1998, 27, 1275–1284. [Google Scholar] [CrossRef] [PubMed]
- Svegliati-Baroni, G.; Ridolfi, F.; Di Sario, A.; Casini, A.; Marucci, L.; Gaggiotti, G.; Orlandoni, P.; Macarri, G.; Perego, L.; Benedetti, A.; et al. Insulin and insulin-like growth factor-I stimulate proliferation and type I collagen accumulation by human hepatic stellate cells: Differential effects on signal transduction pathways. Hepatology 1999, 29, 1743–1751. [Google Scholar] [CrossRef] [PubMed]
- Brenzel, A.; Gressner, A.M. Characterization of insulin-like growth factor (IGF)-I-receptor binding sites during in vitro transformation of rat hepatic stellate cells to myofibroblasts. Eur. J. Clin. Chem. Clin. Biochem. 1996, 34, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Sanz, S.; Pucilowska, J.B.; Liu, S.; Rodriguez-Ortigosa, C.M.; Lund, P.K.; Brenner, D.A.; Fuller, C.R.; Simmons, J.G.; Pardo, A.; Martinez-Chantar, M.L.; et al. Expression of insulin-like growth factor I by activated hepatic stellate cells reduces fibrogenesis and enhances regeneration after liver injury. Gut 2005, 54, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Skrtic, S.; Wallenius, V.; Ekberg, S.; Brenzel, A.; Gressner, A.M.; Jansson, J.O. Insulin-like growth factors stimulate expression of hepatocyte growth factor but not transforming growth factor β1 in cultured hepatic stellate cells. Endocrinology 1997, 138, 4683–4689. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, Y.; Matsumoto, K.; Yamada, A.; Ichida, T.; Asakura, H.; Komoriya, Y.; Nishiyama, E.; Nakamura, T. Preventive and therapeutic effects in rats of hepatocyte growth factor infusion on liver fibrosis/cirrhosis. Hepatology 1997, 26, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Nishizawa, H.; Iguchi, G.; Fukuoka, H.; Takahashi, M.; Suda, K.; Bando, H.; Matsumoto, R.; Yoshida, K.; Odake, Y.; Ogawa, W.; et al. IGF-I induces senescence of hepatic stellate cells and limits fibrosis in a p53-dependent manner. Sci. Rep. 2016, 6, 34605. [Google Scholar] [CrossRef] [PubMed]
- Krizhanovsky, V.; Yon, M.; Dickins, R.A.; Hearn, S.; Simon, J.; Miething, C.; Yee, H.; Zender, L.; Lowe, S.W. Senescence of activated stellate cells limits liver fibrosis. Cell 2008, 134, 657–667. [Google Scholar] [CrossRef] [PubMed]
- Handayaningsih, A.E.; Takahashi, M.; Fukuoka, H.; Iguchi, G.; Nishizawa, H.; Yamamoto, M.; Suda, K.; Takahashi, Y. IGF-I enhances cellular senescence via the reactive oxygen species-p53 pathway. Biochem. Biophys. Res. Commun. 2012, 425, 478–484. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, R.; Fukuoka, H.; Iguchi, G.; Odake, Y.; Yoshida, K.; Bando, H.; Suda, K.; Nishizawa, H.; Takahashi, M.; Yamada, S.; et al. Accelerated telomere shortening in acromegaly; IGF-I induces telomere shortening and cellular senescence. PLoS ONE 2015, 10, e0140189. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Chen, C.C.; Monzon, R.I.; Lau, L.F. Matricellular protein CCN1 promotes regression of liver fibrosis through induction of cellular senescence in hepatic myofibroblasts. Mol. Cell. Biol. 2013, 33, 2078–2090. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.; Feng, D.; Wang, H.; Hong, F.; Bertola, A.; Wang, F.S.; Gao, B. Interleukin-22 induces hepatic stellate cell senescence and restricts liver fibrosis in mice. Hepatology 2012, 56, 1150–1159. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Meng, F.; Wu, N.; Zhou, T.; Venter, J.; Francis, H.; Kennedy, L.; Glaser, T.; Bernuzzi, F.; Invernizzi, P.; et al. Substance P increases liver fibrosis by differential changes in senescence of cholangiocytes and hepatic stellate cells. Hepatology 2017. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Bai, J.; Dong, F.; Fang, H.; Zhang, Y.; Meng, W.; Liu, B.; Luo, Y.; Liu, M.; Bai, Y.; et al. Hepatic Dsba-L protects mice from diet-induced hepatosteatosis and insulin resistance. FASEB J. 2017, 31, 2314–2326. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, M.; Zhang, Z.; Luo, Z.; Liu, F.; Liu, J. Celecoxib derivative OSU-03012 inhibits the proliferation and activation of hepatic stellate cells by inducing cell senescence. Mol. Med. Rep. 2015, 11, 3021–3026. [Google Scholar] [CrossRef] [PubMed]
- Franco, C.; Brandberg, J.; Lonn, L.; Andersson, B.; Bengtsson, B.A.; Johannsson, G. Growth hormone treatment reduces abdominal visceral fat in postmenopausal women with abdominal obesity: A 12-month placebo-controlled trial. J. Clin. Endocrinol. Metab. 2005, 90, 1466–1474. [Google Scholar] [CrossRef] [PubMed]
- Donaghy, A.; Ross, R.; Wicks, C.; Hughes, S.C.; Holly, J.; Gimson, A.; Williams, R. Growth hormone therapy in patients with cirrhosis: A pilot study of efficacy and safety. Gastroenterology 1997, 113, 1617–1622. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Zhou, L.; Zhang, B.; Dong, P.; Lin, W.; Wang, H.; Xu, R.; Ding, H. Recombinant human growth hormone increases albumin and prolongs survival in patients with chronic liver failure: A pilot open, randomized, and controlled clinical trial. Dig. Liver Dis. 2008, 40, 554–559. [Google Scholar] [CrossRef] [PubMed]
- Conchillo, M.; de Knegt, R.J.; Payeras, M.; Quiroga, J.; Sangro, B.; Herrero, J.I.; Castilla-Cortazar, I.; Frystyk, J.; Flyvbjerg, A.; Yoshizawa, C.; et al. Insulin-like growth factor I (IGF-I) replacement therapy increases albumin concentration in liver cirrhosis: Results of a pilot randomized controlled clinical trial. J. Hepatol. 2005, 43, 630–636. [Google Scholar] [CrossRef] [PubMed]


| Receptor/Signaling | Function | References |
|---|---|---|
| GH receptor/JAK2/STAT5 | ||
| Metabolism | Lipogenesis↓, Steatosis↓ | [3,43,46,47,48,49,59] |
| Fibrosis | Fibrosis↓ | [59] |
| Regeneration | Regeneration↑ | [43,44,45] |
| NAFLD/NASH (human) | improves NAFLD/NASH | [23,25,28,29] |
| IGF-I receptor | ||
| Metabolism | Steatosis↓ | [59,62,75] |
| Fibrosis | Fibrosis↓↓ | [59,65,72,75] |
| Regeneration | Regeneration↑ | [72,73,75] |
| NAFLD/NASH (human) | improves NASH/cirrhosis | [85,86,87] |
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takahashi, Y. The Role of Growth Hormone and Insulin-Like Growth Factor-I in the Liver. Int. J. Mol. Sci. 2017, 18, 1447. https://doi.org/10.3390/ijms18071447
Takahashi Y. The Role of Growth Hormone and Insulin-Like Growth Factor-I in the Liver. International Journal of Molecular Sciences. 2017; 18(7):1447. https://doi.org/10.3390/ijms18071447
Chicago/Turabian StyleTakahashi, Yutaka. 2017. "The Role of Growth Hormone and Insulin-Like Growth Factor-I in the Liver" International Journal of Molecular Sciences 18, no. 7: 1447. https://doi.org/10.3390/ijms18071447