Effect of the Drying Process on the Intensification of Phenolic Compounds Recovery from Grape Pomace Using Accelerated Solvent Extraction
Abstract
:1. Introduction
2. Results and Discussion
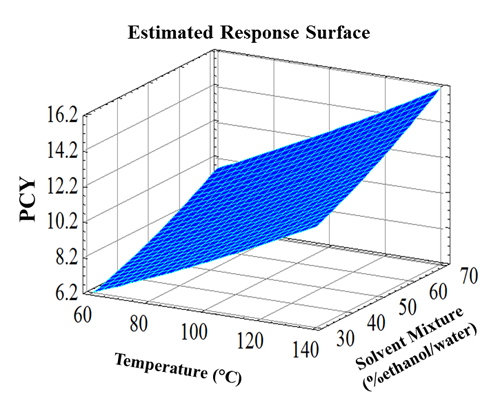
2.1. Total Phenolic Content Optimization by Response Surface Methodology
| Run | Blocks | Variables Levels Uncoded (Coded) | |||
|---|---|---|---|---|---|
| Temperature (°C) | Solvent Mixture (%etdanol/water) | Phenolic Compounds Yield (g GAE/100 g DM) | |||
| Experimental | Predicted | ||||
| 1 | 1 | 60 (−1) | 30 (−1) | 6.02 | 6.23 |
| 2 | 1 | 60 (−1) | 70 (+1) | 9.94 | 9.94 |
| 3 | 1 | 140 (+1) | 30 (−1) | 11.62 | 11.62 |
| 4 | 1 | 140 (+1) | 70 (+1) | 16.31 | 16.1 |
| 5 | 2 | 60 (−1) | 50 (0) | 7.56 | 7.35 |
| 6 | 2 | 140 (+1) | 50 (0) | 13.72 | 13.93 |
| 7 | 2 | 100 (0) | 30 (−1) | 9.31 | 9.1 |
| 8 | 2 | 100 (0) | 70 (+1) | 12.18 | 12.39 |
| 9 | 3 | 100 (0) | 50 (0) | 10.50 | 10.49 |
| 10 | 3 | 100 (0) | 50 (0) | 10.68 | 10.49 |
| 11 | 3 | 100 (0) | 50 (0) | 10.64 | 10.49 |
| 12 | 3 | 100 (0) | 50 (0) | 10.18 | 10.469 |



2.2. Effect of the Raw Material Treatment on the Total Phenolic Content

2.3. Free Radical Scavenging Properties of the Extracted Phenolic Compounds
| Wet Grape Pomace | Dry Grape Pomace | |||
|---|---|---|---|---|
| Extraction Temperatures | Inhibition Percentage Compared to BHT 50 µg/mL | Inhibition Percentage Compared to Resveratrol 50 µg/mL | Inhibition Percentage Compared to BHT 50 µg/mL | Inhibition Percentage Compared to Resveratrol 50 µg/mL |
| 40 °C | 23 ± 1.01 | 14 ± 0.4 | 11.4 ± 2.1 | 11.2 ± 0.2 |
| 60 °C | 25.9 ± 0.59 | 16.8 ± 0.8 | 23.5 ± 0.6 | 14 ± 0.1 |
| 80 °C | 30 ± 0.56 | 21.3 ± 1.4 | 29.7 ± 0.41 | 21 ± 0.2 |
| 100 °C | 35.7 ± 0.78 | 27.7 ± 0.9 | 30 ± 0.35 | 21.8 ± 0.1 |
| 120 °C | 42 ± 0.89 | 34.8 ± 1.01 | 48 ± 2.2 | 41.5 ± 0.9 |
| 140 °C | 46.4 ± 1.2 | 39.8 ± 2.1 | 54.73 ± 0.44 | 49.12 ± 1 |
2.4. Characterization, Identification and Quantification of Phenolic Compounds by HPLC-DAD


3. Experimental Section
3.1. Reagents
3.2. Sample Preparation
3.3. Dry Matter Content
3.4. ASE Extraction
3.5. Total Phenolic Compounds Determination
3.6. Free Radical Scavenging Activity
3.7. HPLC Analysis:
3.8. Experimental Design
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Galanakis, C.M. Recovery of high added-value components from food wastes: Conventional, emerging technologies and commercialized applications. Trends Food Sci. Technol. 2012, 26, 68–87. [Google Scholar] [CrossRef]
- Xia, E.-Q.; Deng, G.-F.; Guo, Y.-J.; Li, H.-B. Biological activities of polyphenols from grapes. Int. J. Mol. Sci. 2010, 11, 622–646. [Google Scholar] [CrossRef]
- Femenia, A.; Waldron, K. High-value co-products from plant foods: Cosmetics and pharmaceuticals. In Handbook of Waste Management and Co-Product Recovery in Food Processing; Waldron, K., Ed.; Woodhead Publishing Limited: Cambridge, UK, 2007; Volume 1, pp. 470–501. [Google Scholar]
- Yilmaz, Y.; Toledo, R.T. Oxygen radical absorbance capacities of grape/wine industry byproducts and effect of solvent type on extraction of grape seed polyphenols. J. Food Compos. Anal. 2006, 19, 41–48. [Google Scholar] [CrossRef]
- Laufenberg, G.; Kunz, B.; Nystroem, M. Transformation of vegetable waste into value added products: (A) the upgrading concept; (B) practical implementations. Bioresour. Technol. 2003, 87, 167–198. [Google Scholar] [CrossRef]
- Puértolas, E.; López, N.; Condón, S.; Álvarez, I.; Raso, J. Potential applications of PEF to improve red wine quality. Trends Food Sci. Technol. 2010, 21, 247–255. [Google Scholar]
- Tiwari, B.K.; O’Donnell, C.P.; Cullen, P.J. New challenges in food science and technology: An industrial perspective. Trends Food Sci. Technol. 2009, 20, 180–181. [Google Scholar] [CrossRef]
- Richter, B.E.; Jones, B.A.; Ezzell, J.L.; Porter, N.L.; Avdalovic, N.; Pohl, C. Accelerated solvent extraction: A technique for sample preparation. Anal. Chem. 1996, 68, 1033–1039. [Google Scholar] [CrossRef]
- Lorenzo, R.A.; Carro, A.M.; Alvarez-Lorenzo, C.; Concheiro, A. To remove or not to remove? The challenge of extracting the template to make the cavities available in molecularly imprinted polymers (MIPs). Int. J. Mol. Sci. 2011, 12, 4327–4347. [Google Scholar] [CrossRef]
- Yrjönen, T. Extraction and Planar Chromatographic Separation Techniques in the Analysis of Natural Products; University of Helsinki: Helsinki, Finland, 2004. [Google Scholar]
- Delgado-Torre, M.P.; Ferreiro-Vera, C.; Priego-Capote, F.; Pérez-Juan, P.M.; Luque de Castro, M.D. Comparison of accelerated methods for the extraction of phenolic compounds from different vine-shoot cultivars. J. Agric. Food Chem. 2012, 60, 3051–3060. [Google Scholar] [CrossRef]
- Plaza, M.; Amigo-Benavent, M.; del Castillo, M.D.; Ibáñez, E.; Herrero, M. Facts about the formation of new antioxidants in natural samples after subcritical water extraction. Food Res. Int. 2010, 43, 2341–2348. [Google Scholar] [CrossRef] [Green Version]
- Myers, R.; Montgomery, D.C. Response Surface Methods; Allyn-Bacon: Boston, MA, USA, 1971. [Google Scholar]
- Ratti, C. Hot air and freeze-drying of high-value foods: A review. J. Food Eng. 2001, 49, 311–319. [Google Scholar] [CrossRef]
- Lin, T.M.; Durance, T.D.; Scaman, C.H. Characterization of vacuum microwave, air and freeze dried carrot slices. Food Res. Int. 1998, 31, 111–117. [Google Scholar] [CrossRef]
- Kwok, B.H.L.; Hu, C.; Durance, T.; Kitts, D.D. Dehydration techniques affect phytochemical contents and free radical scavenging activities of Saskatoon berries (Amelanchier alnifolia Nutt.). J. Food Sci. 2004, 69, SNQ122–SNQ126. [Google Scholar]
- Larrauri, J.A.; Rupérez, P.; Saura-Calixto, F. Effect of drying temperature on the stability of polyphenols and antioxidant activity of red grape pomace peels. J. Agric. Food Chem. 1997, 45, 1390–1393. [Google Scholar] [CrossRef]
- Katsube, T.; Tsurunaga, Y.; Sugiyama, M.; Furuno, T.; Yamasaki, Y. Effect of air-drying temperature on antioxidant capacity and stability of polyphenolic compounds in mulberry (Morus alba L.) leaves. Food Chem. 2009, 113, 964–969. [Google Scholar] [CrossRef]
- Jeong, S.-M.; Kim, S.-Y.; Kim, D.-R.; Jo, S.-C.; Nam, K.C.; Ahn, D.U.; Lee, S.-C. Effect of heat treatment on the antioxidant activity of extracts from citrus peels. J. Agric. Food Chem. 2004, 52, 3389–3393. [Google Scholar] [CrossRef]
- Chemat, F.; Vian, M.A.; Cravotto, G. Green extraction of natural products: Concept and principles. Int. J. Mol. Sci. 2012, 13, 8615–8627. [Google Scholar] [CrossRef]
- El Hajj, Y.; Debs, E.; Nguyen, C.; Maroun, R.G.; Louka, N. Antioxidants from syrah grapes (Vitis vinifera L. cv. Syrah). Extraction process through optimization by response surface methodology. Food Nutr. Sci. 2013, 4, 1209–1220. [Google Scholar]
- Rajha, H.N.; el Darra, N.; Vorobiev, E.; Louka, N.; Maroun, R.G. An environment friendly, low-cost extraction process of phenolic compounds from grape byproducts. Optimization by multi-response surface methodology. Food Nutr. Sci. 2013, 4, 650–659. [Google Scholar]
- Rajha, H.N.; el Darra, N.; Hobaika, Z.; Boussetta, N.; Vorobiev, E.; Maroun, R.G.; Louka, N. Extraction of total phenolic compounds, flavonoids, anthocyanins and tannins from grape byproducts by response surface methodology. Influence of solid-liquid ratio, particle size, time, temperature and solvent mixtures on the optimization process. Food Nutr. Sci. 2014, 5, 397–409. [Google Scholar]
- Rajha, H.N.; Louka, N.; el Darra, N.; Hobaika, Z.; Boussetta, N.; Vorobiev, E.; Maroun, R.G. Multiple response optimization of high temperature, low time aqueous extraction process of phenolic compounds from grape byproducts. Food Nutr. Sci. 2014, 5, 351–360. [Google Scholar] [CrossRef]
- Wijngaard, H.; Hossain, M.B.; Rai, D.K.; Brunton, N. Techniques to extract bioactive compounds from food by-products of plant origin. Food Res. Int. 2012, 46, 505–513. [Google Scholar] [CrossRef]
- Ibañez, E.; Herrero, M.; Mendiola, J.A.; Castro-Puyana, M. Extraction and characterization of bioactive compounds with health benefits from marine resources: macro and micro algae, cyanobacteria, and invertebrates. In Marine Bioactive Compounds; Hayes M., Springer: New York, NY, USA, 2012; pp. 55–98. [Google Scholar]
- Monrad, J.K.; Howard, L.R.; King, J.W.; Srinivas, K.; Mauromoustakos, A. Subcritical solvent extraction of anthocyanins from dried red grape pomace. J. Agric. Food Chem. 2010, 58, 2862–2868. [Google Scholar] [CrossRef]
- Monrad, J.K.; Howard, L.R.; King, J.W.; Srinivas, K.; Mauromoustakos, A. Subcritical solvent extraction of procyanidins from dried red grape pomace. J. Agric. Food Chem. 2009, 58, 4014–4021. [Google Scholar] [CrossRef]
- Piñeiro, Z.; Palma, M.; Barroso, C.G. Determination of catechins by means of extraction with pressurized liquids. J. Chromatogr. A 2004, 1026, 19–23. [Google Scholar] [CrossRef]
- Singh, P.P.; Saldaña, M.D.A. Subcritical water extraction of phenolic compounds from potato peel. Food Res. Int. 2011, 44, 2452–2458. [Google Scholar] [CrossRef]
- Ju, Z.; Howard, L.R. Subcritical water and sulfured water extraction of anthocyanins and other phenolics from dried red grape skin. J. Food Sci. 2005, 70, S270–S276. [Google Scholar]
- Hartonen, K.; Parshintsev, J.; Sandberg, K.; Bergelin, E.; Nisula, L.; Riekkola, M.-L. Isolation of flavonoids from aspen knotwood by pressurized hot water extraction and comparison with other extraction techniques. Talanta 2007, 74, 32–38. [Google Scholar] [CrossRef]
- Achanta, S.; Okos, M.R. Impact of drying on biological product quality. In Food Preservation and Moisture Control. Fundamentals and Applications; Barbosa-Cánovas, G.V., Welti-Chanes, J., Eds.; Technomic Pub. Co.: Lancaster, Basel, Switzerland, 1995; pp. 637–657. [Google Scholar]
- Cha, K.H.; Kang, S.W.; Kim, C.Y.; Um, B.H.; Na, Y.R.; Pan, C.-H. Effect of pressurized liquids on extraction of antioxidants from Chlorella vulgaris. J. Agric. Food Chem. 2010, 58, 4756–4761. [Google Scholar] [CrossRef]
- Vashisth, T. Evaluation of Drying Technologies for Muscadine Pomace to Produce an Antioxidant-Rich Functional Food Ingredient; University of Georgia: Athens, GA, USA, 2009. [Google Scholar]
- kerget, M.; Kotnik, P.; Hadolin, M.; Hraš, A.R.; Simonič, M.; Knez, Ž. Phenols, proanthocyanidins, flavones and flavonols in some plant materials and their antioxidant activities. Food Chem. 2005, 89, 191–198. [Google Scholar]
- Maier, T.; Schieber, A.; Kammerer, D.R.; Carle, R. Residues of grape (Vitis vinifera L.) seed oil production as a valuable source of phenolic antioxidants. Food Chem. 2009, 112, 551–559. [Google Scholar]
- Yilmaz, Y.; Toledo, R.T. Major flavonoids in grape seeds and skins: Antioxidant capacity of catechin, epicatechin, and gallic acid. J. Agric. Food Chem. 2004, 52, 255–260. [Google Scholar] [CrossRef]
- Yoshimura, Y.; Nakazawa, H.; Yamaguchi, F. Evaluation of the NO scavenging activity of procyanidin in grape seed by use of the TMA-PTIO/NOC 7 ESR system. J. Agric. Food Chem. 2003, 51, 6409–6412. [Google Scholar] [CrossRef]
- Hagerman, A.E.; Riedl, K.M.; Jones, G.A.; Sovik, K.N.; Ritchard, N.T.; Hartzfeld, P.W.; Riechel, T.L. High molecular weight plant polyphenolics (tannins) as biological antioxidants. J. Agric. Food Chem. 1998, 46, 1887–1892. [Google Scholar]
- Ghiselli, A.; Nardini, M.; Baldi, A.; Scaccini, C. Antioxidant activity of different phenolic fractions separated from an Italian red wine. J. Agric. Food Chem. 1998, 46, 361–367. [Google Scholar] [CrossRef]
- El Darra, N.; Tannous, J.; Mouncef, P.B.; Palge, J.; Yaghi, J.; Vorobiev, E.; Louka, N.; Maroun, R.G. A comparative study on antiradical and antimicrobial properties of red grapes extracts obtained from different Vitis vinifera varieties. Food Nutr. Sci. 2012, 3, 1420–1432. [Google Scholar]
- Slinkard, K.; Singleton, V.L. Total phenol analysis: Automation and comparison with manual methods. Am. J. Enol. Vitic. 1977, 28, 49–55. [Google Scholar]
- Gyamfi, M.A.; Yonamine, M.; Aniya, Y. Free-radical scavenging action of medicinal herbs from Ghana: Thonningia sanguinea on experimentally-induced liver injuries. Gen. Pharmacol.: Vasc. Syst. 1999, 32, 661–667. [Google Scholar] [CrossRef]
- Ho, P.; Hogg, T.A.; Silva, M.C.M. Application of a liquid chromatographic method for the determination of phenolic compounds and furans in fortified wines. Food Chem. 1999, 64, 115–122. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rajha, H.N.; Ziegler, W.; Louka, N.; Hobaika, Z.; Vorobiev, E.; Boechzelt, H.G.; Maroun, R.G. Effect of the Drying Process on the Intensification of Phenolic Compounds Recovery from Grape Pomace Using Accelerated Solvent Extraction. Int. J. Mol. Sci. 2014, 15, 18640-18658. https://doi.org/10.3390/ijms151018640
Rajha HN, Ziegler W, Louka N, Hobaika Z, Vorobiev E, Boechzelt HG, Maroun RG. Effect of the Drying Process on the Intensification of Phenolic Compounds Recovery from Grape Pomace Using Accelerated Solvent Extraction. International Journal of Molecular Sciences. 2014; 15(10):18640-18658. https://doi.org/10.3390/ijms151018640
Chicago/Turabian StyleRajha, Hiba N., Walter Ziegler, Nicolas Louka, Zeina Hobaika, Eugene Vorobiev, Herbert G. Boechzelt, and Richard G. Maroun. 2014. "Effect of the Drying Process on the Intensification of Phenolic Compounds Recovery from Grape Pomace Using Accelerated Solvent Extraction" International Journal of Molecular Sciences 15, no. 10: 18640-18658. https://doi.org/10.3390/ijms151018640