1. Introduction
Cerebral stroke, commonly known as “stroke”, is an acute cerebrovascular disease caused by sudden rupture or blockage of blood vessels in the brain, resulting in normal blood flow into the brain and brain tissue damage. It has the characteristics of high rates of disability, fatality, and recurrence, and is one of the most lethal diseases in the world [
1]. Stroke includes ischemic and hemorrhagic stroke. About 80% of these strokes are ischemic in nature, also known as ischemic stroke or cerebral infarction, and are caused by a transient or permanent reduction in cerebral blood flow (CBF), which is limited to the main areas of cerebral arteries, such as middle cerebral artery occlusion (MCAO). Blockage of blood vessels results in a lack of oxygen and energy in brain tissue, leading to inflammation that can lead to irreversible tissue damage [
2]. The clinical manifestations are mainly characterized by sudden dizziness, unconsciousness, or the sudden appearance of slanted eyes, hemiplegia, and intellectual disability. The study reported that it may be salvageable if appropriate treatment is administered within a specified golden therapeutic window. At present, reperfusion and neuroprotection were mainly developed to treat ischemic stroke. The latter treatment strategy was to re-drain blood vessels with thrombolytic drugs or mechanical devices to restore perfusion as quickly as possible. However, when cerebral ischemia persists for a long time, treatment to restore blood flow and reperfusion can lead to brain damage, cerebral edema, cerebral hemorrhage, and neuronal death, etc., known as cerebral ischemia-reperfusion injury (CIRI) [
3]. At present, the drugs used in the treatment of ischemic stroke include thrombolytic drugs (t-PA, urokinase, etc.), antithrombotic drugs, antiplatelet drugs, fibrinolytic drugs, microcirculation-improving drugs, etc. t-PA (tissue plasminogen activator) was the first drug approved by FDA for the treatment of stroke in 1996, but due to the strict treatment window of ischemic stroke (within 3 h), many patients could not be treated, and there was a high risk of intracranial hemorrhage rate. There are many drugs in the domestic market, such as nimodipine, flunarizine, yatan, karan and ginkgo biloba extract, which are used to improve cerebral circulation, brain metabolism and brain function. Although they all have certain therapeutic effects, their efficacy in the treatment of stroke cannot be fully affirmed.
At present, the representative clinical drug is butylphthalide (NBP) for the treatment of ischemic stroke, a class 1 new drug developed in China. Butylphthalide is widely found in Umbelliferae, such as Chuanxiong, Chaxiong, and so on. Therefore, other more widely available treatment options are urgently needed. Chaxiong, also known as Fuxiong, is the dried rhizome of
Ligusticum sinense Oliv.cv. Chaxiong [
4]. Chaxiong is mainly produced in the Jiujiang area of Jiangxi Province, where it has a long history of cultivation and medicinal use. Chaxiong is warm in nature and acrid in taste and has the effects of expelling wind and relieving pain, promoting qi and promoting blood circulation. Chaxiong volatile oil (volatile oil from
Ligusticum sinense Oliv.cv. Chaxiong, VOC) is the volatile oil obtained from the rhizome of Chaxiong by steam POPE molecular distillation. Luo Yongming et al. [
5] used the GC-MS technique to separate and identify the volatile oil of Chaxiong and identified 21 chemical components, including five phthalide components, Yangchuanxiong lactone A, n-butylphthalide, new Cnidolide, Z-ligustilide and butenylphthalide, which accounted for about 90% of the total volatile oil. The study showed that the volatile oil of Chaxiong has the effects of antagonizing the neurotoxicity of glutamic acid, inhibiting the heartbeat, enhancing coronary blood flow and dilating blood vessels, etc. It can be used for cerebral thrombosis and stroke and cerebral edema caused by focal cerebral ischemia [
6].
The research group conducted preliminary pharmacodynamic research on the volatile oil of Chaxiong in the early stage, and it has been preliminarily confirmed that the volatile oil of Chaxiong is the main pharmacodynamic part of the anti-ischemic stroke of Chaxiong. The study found that the volatile oil of Chaxiong has a good protective effect on the primary cultured rat neurons mediated by H2O2 and glutamate and also has a certain protective effect on the rat MCAO reperfusion model. However, the volatile oil of Chaxiong is highly volatile, insoluble in water and has low oral bioavailability, which limits its clinical application in ischemic stroke.
The inability of therapeutic agents to cross the blood–brain barrier (BBB) and blood–cerebrospinal fluid barrier (CSF) represents a major obstacle in the treatment of ischemic stroke. One possibility to overcome this hurdle is to use novel drug-delivery systems to deliver drugs to the brain. Intranasal delivery routes can enhance drug delivery to the brain through the use of appropriate vehicle systems (e.g., liposomes, nanoparticles, nanoemulsions, etc.), thereby enhancing CNS permeability to the drug and allowing sustained drug delivery. Mukesh Kumar et al. prepared risperidone-loaded nanoemulsions (RNE) and muco-adherent nanoemulsions (RMNE) for the treatment of schizophrenia, and their radiolabel-labeled and gamma scintigraphy imaging studies demonstrated that muco-adherent risperidone nanoemulsions provided rapid and efficient intranasal drug delivery to the brain. As a non-invasive topical-drug-delivery method, the nasal–brain pathway not only bypasses the blood–brain barrier but also avoids the first-pass effect of the liver. Nasal administration has gradually become one of the research hotspots of drug administration routes due to its advantages of no hepatic first-pass metabolism, fast drug absorption, and high bioavailability. As a local or systemic absorption drug, the nasal mucosa has the advantages of relatively large drug absorption surface area, abundant capillaries and lymphatic capillaries, and abundant total blood flow. These advantages enable the nasal route to significantly increase drug absorption, significantly increase the amount of drug delivered to the brain, and improve the therapeutic effect of brain diseases. Additionally, many studies have shown that drugs can be effectively transferred directly from the nasal cavity to the brain via the nasal route. Therefore, it is considered to develop the volatile oil of Chaxiong into a preparation for intranasal administration, which is used for intranasal administration to transport the drug to the brain through the nose.
In recent years, drug delivery by nanoemulsion has proven to be a promising strategy for improving the bioavailability of lipophilic drugs and has received increasing attention from researchers [
7,
8]. Nanoemulsion is a thermodynamically, kinetically stable, isotropic transparent dispersion system composed of water phase, oil phase, emulsifier and co-emulsifier [
9]. Nanoemulsion has the advantages of small particle size, high solvent capacity and excellent stability. Nanoemulsions can not only increase the solubility of poorly soluble drugs but also carry drugs with different solubilities, achieving synergistic effects between drugs and prolonging the effect. They improve the bioavailability of the drug and play a sustained-release and targeted role [
10]. The emulsifier and co-emulsifier in the nanoemulsion can reduce the interfacial tension between the oil and water of the emulsion, and at the same time, reducing the particle size to the nanometer level can reduce the aggregation and flocculation of droplets, thereby significantly improving the stability of the emulsion. However, the rapid clearance of numerous cilia on the nasal mucosa limits intranasal drug delivery. The direct nasal administration of nanoemulsion may cause leakage or irritation to the nasal mucosa. Therefore, the use of nasal nanoemulsion in situ gel in this paper can effectively avoid or reduce the clearance of nasal cilia. The volatile oil of Chaxiong is embedded in the nanoemulsion, which can increase the solubility of the drug, improve the stability of the volatile oil, and prevent the drug from being degraded before reaching the action site. The in situ gel can prolong the retention time of the drug in the nasal mucosa, promote the dissolution and penetration of drugs in the nasal mucosa, avoid drug leakage, reduce drug drips into the back of the throat, and improve bioavailability [
11].
Studies have shown that Chaxiong volatile oil was a potential anti-ischemic stroke drug, and its further application is limited due to its instability and low water solubility, which seriously affect its accumulation in the brain. The aim of this study is to develop a brain drug-delivery system loaded with Chaxiong volatile oil nanoemulsion in situ gel for the effective treatment of ischemic stroke by direct nasal–brain administration. In this study, a temperature-sensitive nanoemulsion in situ gel formulation (VOC-NE-ISG) loaded with volatile oil of Chaxiong was developed by phase transformation and “cold” methods. In addition, this study also aimed to evaluate VOC-NE-ISG physicochemical properties, pharmacokinetic studies, and in vivo anti-ischemic stroke effects, as well as preliminary brain targets for ischemic stroke therapy by nasal-to-brain administration towards ability.
2. Results and Discussion
2.1. Screening of Nanoemulsion Formulations
2.1.1. Screening of Cosurfactant
Because cosurfactants are amphiphilic, they can accumulate in the interface layer and reduce the interfacial tension, which enables them to increase the fluidity of the interface film and penetrate the single layer of the surfactant [
12]. It can be seen from
Figure S3 (Supplementary Materials) that the solubility of the five components of the volatile oil of chaxiong in ethanol absolute was the highest, and the solubility in PEG400, propylene glycol, and n-butanol was relatively high, while it was almost immiscible in glycerol. Therefore, in this experiment, anhydrous ethanol was selected as the co-surfactant to further prepare the volatile oil of Chaxiong nanoemulsion.
2.1.2. Screening of Surfactant
In general, non-ionic surfactants have high surface activity, good solubility, and are less toxic and irritating than ionic surfactants, so all surfactants and cosurfactants used in this study were selected as non-ionic type. Emulsifiers promote the formation of nanoemulsions by reducing interfacial tension, thereby promoting the free energy loss associated with nanodroplet formation [
13]. An emulsifier is a kind of surface-active agent, which can improve the surface tension between the various constituent phases in the emulsion body and form a uniform emulsion body. Each non-aqueous component has a desired HLB value; the emulsification characteristics of emulsifier depend on its hydrophilic and oleophilic equilibrium value (HLB value). The higher the HLB value, the stronger the hydrophilicity, and vice versa. The optimum HLB value for the emulsifier is close to the HLB value required for the oil. When several emulsifiers are used in combination, the HLB value of the composite emulsifier has the additivity according to the mass fraction.
According to the results in
Table S4 (Supplementary Materials), it can be concluded that when RH40 is used as an emulsifier, a nanoemulsion can be formed, and the appearance is clear and transparent, the particle size is 23.11 ± 0.12 nm, and the PDI is 0.474 ± 0.23. The HLB value of O/W type nanoemulsion is generally 8~18, while the HLB value of Span 80 is 4.3, which is not suitable for forming O/W type nanoemulsion. VOC has high solubility in surfactant cremoophor RH 40, and its HLB value is between 14 and 16. Therefore, RH40 was selected as an emulsifier for the study of nanoemulsions.
2.1.3. Pseudo-Ternary Phase Diagram
The pseudo ternary phase diagram was developed to determine the concentration range of components forming nano emulsion. It can be seen from
Figure S4 (Supplementary Materials) that when the ratio of oil to Smix is 9:1, 8:2, and 7:3, 6:4, 5:5, 4:6, the prepared emulsion system is a turbid yellow-white emulsion. The test results show that the maximum area of O/W nanoemulsion region was observed when Km was 2:1.
2.1.4. Optimization of VOC-Nanoemulsion
According to the experimental results, Design Expert 8.0.6 software (Stat-Ease Inc., Minneapolis, MN, USA) as used to perform multiple linear regression fitting on the data to analyze the main effect, interaction effect and quadratic effect of three independent variables on three responses. Visual analysis and ANOVA were performed on the test results to screen out the optimal nanoemulsion prescription. The results are shown in
Table 1. The ANOVA results of the three response values obtained by regression analysis are shown in
Table S5 (Supplementary Materials). The
p value of lack of fit of the three response values was >0.05, indicating that the lack of fit test was insignificant, implying that there was a significant model correlation between the independent variables and the response values. The polynomial equations for size, PDI and ZP were as follows:
The analysis of variance (ANOVA) was performed on the above particle size model. The analysis shows that the F value of the model is 8, the
p value is 0.006, reaching a very significant level (
p < 0.01), and the lack of fit (Lack of Fit)
p value is 0.0539 > 0.05, indicating that the model is less interfered by other factors, and the model fits well. The results of the analysis of variance of the potential model are shown in
Table S5 (Supplementary Materials). The
p value of the model is 0.0129, indicating that the model has reached a significant level (
p < 0.05), and the
p value of the lack-of-fit term is >0.05, indicating that the model fits well.
According to the above data and the model fitting results, using Design-Expert 8.0.6 software (Stat-Ease Inc., Minneapolis, MN, USA) for mapping, we can obtain the 3D ratio of oil ratio (A), Smix ratio (B), stirring time (C) to response value, particle size and potential. The effect surface map and contour map are shown in
Figure S5 (Supplementary Materials) and
Figure S6 (Supplementary Materials). It can be seen from the results that A (oil phase ratio) and C (stirring time) have a very significant effect on particle size Y1, while B (Smix ratio) has a small effect on particle size Y1, and A and B have a significant interaction effect on the particle size; B (Smix ratio) has a more significant effect on the potential Y2, and A, B, and C all have significant interaction effects.
RH40 has a good hydrophile lipophilic balance (14~16), which is conducive to the formation of O/W nanoemulsion. In addition, RH40 is generally recognized as safe (GRAS) and can be used as pharmaceutical and food surfactants. After the preparation of nano emulsion, the growth of droplet may occur due to Ostwald maturation or coalescence process, depending on the composition of surfactant and oil. The 3D surface results showed that the increase in the concentration of Smix used in the preparation of NE reduced the average particle size. With the moderate increase in surfactant concentration, the particle size will also decrease significantly, because additional surfactant can stabilize more of the interface area. Because the newly formed interface cannot be completely stabilized under the condition of insufficient surfactant, the coalescence rate of liquid drops is very significant.
Using the process optimization function of Design-Expert 8.0.6 software, the smallest particle size and the lowest potential are substituted into the analog linear equation so that the particle size and potential are as small as possible and the best prescription process can be predicted: the ratio of oil (A) was 6.67%, the ratio of Smix (B) was 26.67%, and stirring time (C) was 1.27 h. The predicted particle size was 21.38 nm and the zeta potential was −21.33 mV. The test results are shown in
Table S6 (Supplementary Materials). It was verified that the particle size of three batches of VOC-NE was 21.02 ± 0.17 nm, PDI was 0.15 ± 0.02, and zeta potential was −22.87 ± 1.02 mV. The deviation rate of the particle size and zeta potential was 1.71% and 1.72%, respectively. This indicates that the model has a high degree of fit.
2.2. Characterization of VOC-NE
2.2.1. Traits and Types
Through observation, it was found that the volatile oil of Chaxiong nanoemulsion was a light yellow, clear liquid with Tyndall phenomenon, with no turbidity, flocculation, stratification, or other phenomena (
Figure S7, Supplementary Materials).
According to the diffusion speed of water-soluble dye methylene blue and oil-soluble dye Sudan red III in nanoemulsion, the type of Chaxiong volatile oil nanoemulsion was determined. The identification results of the staining method are shown in
Figure S8 (Supplementary Materials). The water-soluble dye methylene blue diffuses very quickly in the Chaxiong volatile oil nanoemulsion. After a period of time, the nanoemulsion is dyed blue by methylene blue, while Sudan red III cannot diffuse in the nanoemulsion, which settles directly at the bottom. This indicates that the VOC-NE formulation in this experiment was an oil-in-water nanoemulsion.
The conductivity measurement depends on the poor conductivity of oil compared to water. In the water-in-oil nanoemulsion with oil as the continuous phase, the conductivity is very low, close to the conductivity of oil. On the contrary, oil-in-water nanoemulsion was close to the conductivity of water with a higher conductivity [
14]. Therefore, the conductivity value of the nanoemulsion can help to judge the type of the prepared nanoemulsion. The conductivity of Chaxiong volatile oil nanoemulsion was measured to be 31.62 ± 0.17 µS/cm, the conductivity of water was 10.8 µS/cm, and the conductivity of oil was 0.65 µS/cm. The experimental results show that the VOC-NE was an oil-in-water nanoemulsion. This is consistent with the results of the dye identification test.
2.2.2. Particle Size, PDI and Zeta Potential
For nanoparticle drug-delivery systems, the particle size has a significant effect on the absorption and penetration of the drug or particle. The particle size is a key value for evaluating nanoemulsions. The smaller particle size provides a larger interfacial surface area for drug absorption. PDI reflects the uniformity of the nanoemulsion particle size and can also reflect the stability of nanoemulsion. The smaller the polydispersity coefficient, the more uniform the size of the prepared nanoemulsion droplets and the more stable the prepared nanoemulsion [
15].The low surface charge may be related to the intrinsic properties of nonionic surfactants. The higher the absolute value of the potential, the higher the surface charge of the droplets, the greater the repulsive force between the droplets, and the more stable the prepared nanoemulsion [
11]. The optimized VOC-NE has a particle size of 21.02 ± 0.25 nm, a PDI of 0.150 ± 0.02, and a potential of −22.87 ± 1.02 mV.
Figure S9 (Supplementary Materials) shows the particle size distribution and potential distribution of the VOC-NE.
2.2.3. pH, Viscosity, Refractive Index and Transmittance
The pH value of VOC-NE was 4.52 ± 0.02, which is well tolerated in the range of the human nasal pH range (4.5–6.5), so it will not cause nasal irritation when administered intranasally.
The viscosity of the formulation may affect the absorption of the entire nasal mucosa. The formulation with high viscosity tends to stay longer on the mucosa and increase the stay time in the nasal cavity. However, higher viscosity may also lead to delayed drug release from the carrier to the mucosal surface. These formulations have Newtonian rheological properties because they have a constant viscosity. The viscosity of the volatile oil of Chaxiong nanoemulsion was 32 ± 0.5 mPa·s, which is low and very close to the viscosity of water. The low viscosity value ensures easy handling, packaging, and trouble-free administration of the formulation.
The refractive index (RI) is a characteristic of a medium to light volatile oil, which is used to characterize the isotropy of nanoemulsions. The refractive index of the volatile oil of Chaxiong nanoemulsion was measured by Abbe refractometer to evaluate the isotropy of the nanoemulsion. The results show that no significant difference was observed in the RI values between the various VOC-NE formulations, which indicated that the nanoemulsion formulations were chemically stable and remained isotropic in nature.
The percent transmittance is the net value of each component of the nanoemulsion, indicating the isotropic nature of the formulation. The experimental results showed that the %T values of all the VOC-NE were 99.63% ± 0.40, between 97% and 100%, indicating that the volatile oil nanoemulsions of Chaxiong were optically transparent.
2.2.4. Result of TEM
The morphology of VOC-NE formulation was characterized by TEM.
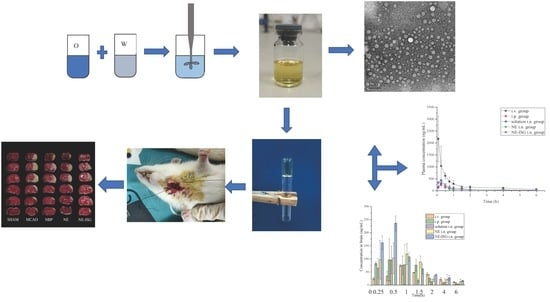
Figure 1 shows TEM images of the optimized VOC-NE formulation. The nanoemulsion droplets were spherical and the particle size ranged from 20 nm to 50 nm. The TEM results of particle size analysis are similar to the average particle size obtained by particle size analyzer.
2.2.5. Result of Stability Studies
The results of the centrifugation stability test showed that the appearance of the VOC-NE did not change after centrifugation, and no stratification, demulsification, or other phenomena occurred, and it was still a light-yellow, clear liquid. The results of freeze–thaw stability showed that after four freeze–thaw cycles, the VOC-NE did not appear display delamination or demulsification, the particle size ranged from 18 to 31 nm, and the PDI was 0.14 to 0.20, indicating that the VOC-NE formulation has a good freeze–thaw stability. The heating–cooling stability results showed that the VOC-NE did not display delamination or demulsification after four heating and cooling cycles, the particle size ranged from 18 to 35 nm, and the PDI was 0.15 to 0.20, which indicated that the VOC-NE has good freeze–thaw stability. In summary, the volatile oil of Chaxiong nanoemulsion has a good centrifugation, freeze–thaw and heating–cooling stability.
2.3. Screening of VOC-NE-ISG Gel Framework Materials
2.3.1. The Effect of P407 on the Gelling Temperature of In Situ Gels
The results of the gelation temperature are shown in
Figure S10 (Supplementary Materials). It can be seen from the figure that the poloxamer 407 solution can undergo phase transformation to form a gel after being heated. Additionally, in the range of P407 concentration from 16% to 24%, the gelation temperature of in situ gels decreased gradually with the increase in P407 concentration.
2.3.2. The Effect of P188 on the Gelling Temperature of In Situ Gels
It can be seen from
Figures S10 and S11 (Supplementary Materials) that the concentrations of P407 and P188 have a greater influence on the gelation temperature of the in situ gel. The results showed that the gelation temperature of the in situ gel decreased with the increase in the concentration of P407, and increased with the increase in the concentration of P188. This is consistent with literature reports.
2.3.3. Screening Prescription Ratio of VOC-NE-ISG
The previous preliminary experiments found that the gel matrix could not be fully swelled by directly adding VOC-NE. Therefore, the total volume of the solvent was fixed, and the ratio of fixed water and Chaxiong volatile oil nanoemulsion was 1:1. The experimental results show that when the ratio of water and VOC-NE was 1:1, the ratio of P407 was 12%, and the ratio of P188 was 10%, the gelation temperature at this time was 33 °C (
Figure S12, Supplementary Materials).
2.4. In Vitro Evaluation of VOC-NE-ISG
2.4.1. Measurement of pH and Viscosity of the Gel
The experimental results showed that the three batches of VOC-NE-ISG samples were all pale-yellow, transparent liquids, the pH value was around 5.85, and the pH of the nasal cavity was 4.5–6.5. The viscosity of VOC-NE-ISG at room temperature is about 1799 m·Pa s, and the gel viscosity at physiological temperature is about 4500 m·Pa s. The viscosity of the formulation can ensure prolonged contact and effectively improve the nasal absorption level of drugs and nanoparticles. However, higher viscosity may also result in delayed drug release from the carrier to the mucosal surface.
2.4.2. Measurement of Gelation Temperature and Gelation Time
The temperature of the nasal cavity is generally 29–34 °C. If the gelling temperature of the in situ gel is too high, it may not be converted into a gel after administration. If the gelling temperature is too low, it may not be possible to administer in liquid form. The experimental results show that the gelation temperatures of the three batches of VOC-NE-ISG samples prepared are basically the same, about 33 °C.
Figure S13 (
Supplementary Materials) shows the liquid–solid transition state of VOC-NE-ISG. As shown in the figure, when the ambient temperature is lower than the gelation temperature, the VOC-NE-ISG has good fluidity; when the ambient temperature is equal to the gelation temperature, the VOC-NE-ISG transforms into an immobile gel state. The gelation time of three batches of VOC-NE-ISG samples was about 52 s.
2.4.3. Determination of Gel Strength
The gel strength measurement results showed that the gel strength of the VOC-NE-ISG was 47~49 s. The two most important metrics for in situ gels are viscosity and strength [
16]. Studies have shown that the appropriate viscosity and strength of nasal gel can ensure the long-term retention of the gel at the administration site, increase the drug retention time, improve the absorption efficiency of the nasal cavity, and then improve the transport efficiency of the drug from the nose to the brain.
2.4.4. Determination of Bioadhesion
The mucoadhesion strength of VOC-NE-ISG was (5.69 ± 0.51) × 102 dynes/cm2. The stronger the mucoadhesion, the better the ability to prevent the gel solution from flowing out of the nasal cavity and into the nasopharynx.
2.4.5. Determination of Expansion Coefficient and Gel Water Retention Capacity
The volume of the human nasal cavity was limited, so the volume expansion coefficient of the gel after gelation was required to be less than 5% [
17]. The experimental results show that the expansion coefficient of VOC-NE-ISG was 1.96%, less than 5%, and there is no obvious volume expansion. Therefore, when the Chaxiong volatile oil nanoemulsion in situ gel is instilled into the nasal cavity, it will not affect the patient’s compliance. The water retention capacity of in situ gels can be used to evaluate the storage and transport stability of gels [
18]. The water retention capacity of VOC-NE-ISG was 97.82 ± 0.36%, indicating its good storage and transportation stability.
2.4.6. Rheological Studies
It can be seen from
Figure S14A (Supplementary Materials) that the strain (γ) < 1%, G′ > G″, and elasticity dominates, that is, VOC-NE-ISG exhibits a gel structure, when γ > 1%, G′ gradually decreases, and when G′ = G″, the gel structure of VOC-NE-ISG is completely destroyed [
19], and then with the gradual increase in γ, G′ gradually decreases, and G″ gradually increases. At this time, G′ < G″, viscosity predominates, showing fluid properties. Therefore, all dynamic oscillation experiments need to control the strain γ within 1%.
Frequency sweeps were used to characterize the time-dependent properties of the samples in the non-destructive deformation range and to characterize the stability of the internal structure of the gel. The frequency scanning of VOC-NE-ISG was performed in the oscillation mode of the rheometer to investigate the stability of the internal structure of the in situ gel. In this experiment, the strain is 1% and the frequency sweep range is 0.1 to 100 Hz for the frequency sweep. The test results are shown in
Figure S14B (Supplementary Materials). The test results show that in the scanning range of 0.1 to 100 Hz, G′ is always greater than G″, showing obvious elastic-dominated characteristics, and the variation of G′ is small and stable, and there is no obvious frequency dependence, which indicates that VOC-NE-ISG always has a stable 3D network structure.
Using the rotational mode of the rheometer, the VOC-NE-ISG was scanned at a shear rate (0.1–100 s
−1) at 25 and 33 °C, respectively, to preliminarily determine the fluid type of the sample.
Figure S14C,D (Supplementary Materials) were the change curves of the shear rate (γ′), shear stress (τ) and viscosity (η) of VOC-NE-ISG at 25 °C and 33 °C. It can be seen from
Figure S14C,D (Supplementary Materials) that in the continuous shear rheological measurement, the shear stress and shear rate of the in situ gel of Chaxiong volatile oil nanoemulsion showed a nonlinear relationship, and τ gradually increased with the overall change in γ′, which is large, and η has the property of decreasing and leveling off with the increase in γ′, which belongs to pseudoplastic fluid and is a typical characteristic of a hydrophilic polymer system. A higher degree of pseudoplasticity facilitates their smearing on biological surfaces [
20]. The test results show that VOC-NE-ISG exhibits stable pseudoplastic fluid characteristics at 25 and 33 °C.
2.4.7. In Vitro Release Studies
The in vitro release model fitting results of each component of VOC-NE-ISG are shown in
Table S7 (Supplementary Materials), and the in vitro release curves are shown in
Figure S15 (Supplementary Materials). According to the fitting results of the release model, the release mechanism of the five components in VOC-NE-ISG is closer to the Ritger–Peppas release, 0.45 <
n < 0.89, and the release mechanism is the combined effect of diffusion and matrix erosion.
2.4.8. Histological Evaluation of Nasal Mucosa
The test results are shown in
Figure 2. A large amount of damage and inflammatory cell infiltration appeared in the nasal mucosa of the rats in the isopropanol nasal administration group. The nasal mucosa of the rats in the VOC-NE-ISG group and the normal saline group showed similar microstructures, no cell necrosis was observed, and no significant damage and inflammatory cell infiltration occurred, indicating that the prepared VOC-NE -ISG is safe and has no obvious irritation to the nasal mucosa.
2.5. Results of Pharmacokinetic Study
The plasma concentration–time curve was shown in
Figure 3, and the main pharmacokinetic parameters were shown in
Table 2.
It can be obtained from the experimental results that after oral administration of the volatile oil of Chaxiong to rats, the peak time Tmax of its five components is between 0.21 and 0.50 h, indicating that the oral absorption of the five components is faster. Compared with the intravenous injection group, its peak concentration Cmax is lower, and the five components of Chaxiong volatile oil are Senkyunolide A, neocnidolide, n-butylphthalide, Z-ligustilide and butenylbenzene. The absolute bioavailability of phthalein was 17.19%, 19.19%, 18.32%, 9.54% and 10.91%, respectively. Compared with the intravenous injection group, the Tmax and t1/2 of each component of the volatile oil of Chaxiong were prolonged in the nasal solution group. The absolute bioavailability of ligustilide and butenylphthalide was 26.91%, 25.53%, 26.13%, 21.28% and 23.10%, respectively. Compared with intragastric administration of Chaxiong volatile oil, after preparing Chaxiong volatile oil into a nanoemulsion in situ gel preparation, its peak time Tmax and t1/2 were prolonged, and the degrees of absolute bioavailability of the five components of Chaxiong volatile oil were 42.16%, 40.69%, 34.74%, 29.93% and 31.63%, which were significantly greater than 25%, indicating that the absolute bioavailability of the five components in the VOC-NE-ISG was improved to varying degrees. Compared with the nasal solution group, the Tmax and t1/2 of the VOC-NE group were significantly increased, indicating that the modification of the volatile oil of Chaxiong with nanoemulsion could increase the amount of drug absorption. Compared with the VOC-NE group, the Tmax of the VOC-NE-ISG group was decreased, and the t1/2 was significantly increased (p < 0.05), indicating that the preparation of nanoemulsion into an in situ gel nasal preparation can significantly increase the retention time of the drug in the nasal cavity, thereby increasing drug absorption and improving bioavailability. Compared with the nasal solution group, the absolute bioavailability of the five components of Chaxiong volatile oil in the VOC-NE-ISG group increased by 1.57, 1.59, 1.33, 1.41 and 1.36 times, respectively, indicating that the volatile oil of Chaxiong was prepared into nanoemulsion in situ coagulation. The glue can significantly improve the bioavailability of each component and increase the transport from the nose to the brain.
2.6. Results of Brain Tissue Distribution Studies
The measured drug concentrations in the rat brain tissue were averaged, and the brain tissue drug concentration–time curves of each group were drawn. The plasma pharmacokinetic parameters and brain tissue pharmacokinetic parameters are shown in
Table 3. The brain tissue drug time curve is shown in
Figure 4 and the blood drug time curve is shown in
Figure 5.
It can be seen from the experimental results that compared with the gavage group and the intravenous injection group, after intranasal administration of VOC-NE-ISG, the Tmax of the drug in the rat brain tissue was significantly reduced, while the t1/2, Cmax and AUC0–t were significantly increased, indicating that intranasal administration of VOC-NE-ISG can facilitate drug delivery into the brain. Compared with the nasal solution group, the AUC0–t of the VOC-NE group was significantly increased, indicating that the modification of VOC-NE could increase the transport amount of the drug absorbed through the nose into the brain. The AUC0–t value of the VOC-NE-ISG group was significantly different from that of the nasal solution group (p < 0.05), indicating that the in situ gel can prolong the retention time of the drug in the nasal cavity and increase the absorption of the drug.
According to the experimental results, the order of the brain targeting index in each group was: NE-ISG group > NE group > nasal solution group > intravenous injection group. The BTIs of the three components Senkyunolide A, neocnidolide and n-butylphthalide in the intranasal administration of VOC-NE-ISG were 1.73, 1.56, and 2.01, respectively, all greater than 1.0, indicating that VOC-NE -ISG formulations have obvious brain-targeting properties. Additionally, the larger the BTI, the more obvious its brain targeting.
2.7. Results of Pharmacodynamic Study
2.7.1. Results of Neurological Deficit Score
In this experiment, VOC-NE and VOC-NE-ISG were administered intranasally for evaluating the therapeutic efficacy in MCAO rats. The cerebral infarct size and neurological score were used to assess the therapeutic efficacy of the prepared formulations. In this experiment, the rat middle artery occlusion model was established by the suture method. This model is consistent with human cerebral ischemic infarction and has good repeatability. The rat MCAO model established by the suture method does not require craniotomy and is less traumatic, thus avoiding damage to the skull structure of the brain (
Figure S16, Supplementary Materials). In addition, the MCAO model has the advantages of large infarct volume, high reproducibility, and precise and controllable duration of reperfusion and ischemia. Therefore, the MCAO model is considered to be the most suitable for reproducing the brain injury after ischemic stroke and reperfusion [
21]. Additionally, in the test, it was found that the model control group rats had a mortality rate of 25%.
Twenty-four hours after MCAO modeling, the rats were scored for neurological deficits; dead rats were excluded from the experiment, and the neurological function score of 1 to 3 points was selected for the experiment. The rats were tested: the higher the neurological score, the more serious the behavioral disorder of the rats. The neurological deficit scores after administration of each group in the experiment are shown in
Table S8 (Supplementary Materials) below. The neurological function scores of each group are as follows: the score of the sham operation group was 0 ± 0.00, the score of the model group was 2.50 ± 0.50, the score of the NBP group was 1.20 ± 0.60, the score of NE group was 1.60 ± 0.92, and the score of NE-ISG group was 1.30 ± 0.78. The results of the study showed that the rats in the sham-operated group did not have any symptoms of neurological loss. Rats in the model group, NE group, NE-ISG group and NBP group were successfully modeled, and they all developed symptoms of neurological deficits, which were manifested as paralysis of the left forelimb, turning to the left or tilting to the left when walking, turning to the left when lifting, etc. After continuous administration for 7 days in each group, the results of neurological deficit score showed that both the Chaxiong volatile oil nanoemulsion group and the Chaxiong volatile oil nanoemulsion in situ gel group had a certain improvement effect on behavioral disorders caused by cerebral ischemia in rats, and the improvement effect after NBP administration is equal. Chaxiong volatile oil nanoemulsion also has a certain improvement effect on neurobehavioral disorders caused by cerebral ischemia in rats.
2.7.2. TTC Staining
After the rat brain tissue was stained with TTC, the normal brain tissue was red, and the cerebral infarction was white. The percentage of cerebral infarction area in each experimental group was measured by Image J software (Wayne Rasband and contributors National Institutes of Health, Bethesda, MD, USA): 0 ± 0.00 in the sham operation group, 41.36 ± 4.83% in the model group, 23.02 ± 2.30% in the NBP group, 31.65 ± 2.60% in the NE group, and 25.14 ± 4.26% in the NE-ISG group.
The measurement results of the cerebral infarction area are shown in
Figure 6A and
Table S8 (Supplementary Materials). The brain tissue of the rats in the sham operation group was red, and no infarction was found; the brain tissue of the rats in the model group and each drug group appeared as white infarct. From the staining results of brain tissue in
Figure 6A, it can be seen that compared with the model group, the infarct area of the rats in the NE-ISG group was significantly reduced (
p < 0.01), indicating that the volatile oil nanoemulsion in situ gel preparation of Chaxiong obviously improved the effect of cerebral ischemia. Additionally, there was a significant difference between the NE group and the model group (
p < 0.05), indicating that after intranasal administration of Chaxiong volatile oil nanoemulsion, the area of cerebral infarction in MCAO rats was reduced, and it had an improvement effect on cerebral infarction. After intranasal administration of NBP, the area of cerebral infarction in rats was reduced, and there was a significant difference compared with the model group (
p < 0.01). The results of cerebral infarction size determination are as follows: NBP group > NE-ISG group > NE group. NBP is a clinical drug for the treatment of ischemic stroke in China, and the effect is significant. Nasal administration of VOC-NE-ISG can achieve drug delivery from nose to brain, increase the transport of drugs to brain tissue, and improve the efficiency of ischemic stroke treatment. After statistical analysis, the NE-ISG group was compared with the model group,
p < 0.01; compared with the model group, the NE group was
p < 0.05, indicating a significant difference, indicating that the cerebral infarction area of the rats was improved, and the efficacy of the VOC-NE-ISG group was better than that of the NE group.