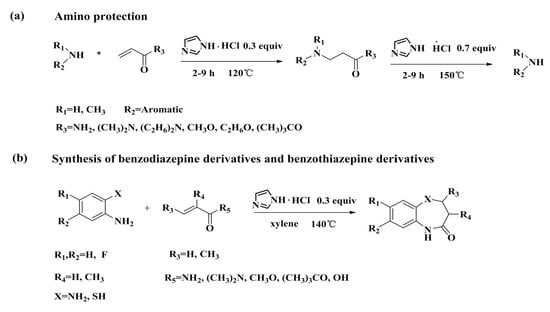
Synthesis of 1,3,4,5-Tetrahydro-2H-benzo[b][1,4]diazepin-2-one (6c)
To 20 mL of schlenk tube with a magnetic stir bar, o-phenylenediamine (4.6 mmol, 0.5 g, 1 eq), methyl acrylate (7.9 mmol, 0.68 g, 1.7 eq) and imidazolium hydrochloride (1.4 mmol, 0.15 g, 0.3 eq) were successively added. Then xylene (1 mL) was added to the reaction mixture. The mixture was stirred at 90 °C for 5 h and then at 140 °C for 8 h. The progress of the reaction was monitored by TLC. After the reaction completed, water (15 mL) was added to the reaction mixture. The solution was extracted with ethyl acetate (3 × 15 mL) and dried using anhydrous Na
2SO
4. The crude product was purified by flash chromatography using silica gel to obtain the corresponding product (
6c) (
Figures S1–S74).
N,N-Dimethyl-3-phenylamino-propionamide (
3a): The product was obtained as a white solid in a yield of 74% (0.34 g), MP: 103–106 °C [
30].
1HNMR (600 MHz, CDCl
3) δ 7.17 (dd,
J = 8.4, 7.4 Hz, 2H), 6.69 (t,
J = 7.3 Hz, 1H), 6.64 (d,
J = 7.7 Hz, 2H), 4.33 (s, 1H), 3.48 (t,
J = 6.0 Hz, 2H), 2.95 (d,
J = 3.5 Hz, 6H), 2.59 (t,
J = 6.0 Hz, 2H).
13C-NMR (151 MHz, CDCl
3) δ 171.62, 147.72, 129.29, 117.57, 113.26, 39.58, 37.09, 35.28, 32.20.
3-(2-Amino-phenylamino)-N,N-dimethyl-propionamide (3b): The product was obtained as a black liquid in a yield of 72% (0.39 g). 1H NMR (600 MHz, CDCl3) δ 6.84 – 6.75 (m, 1H), 6.75 – 6.64 (m, 3H), 3.59 (s, 2H), 3.46 (t, J = 6.2 Hz, 2H), 2.96 (d, J = 9.6 Hz, 6H), 2.63 (t, J = 6.1 Hz, 2H). 13C-NMR (151 MHz, CDCl3) δ171.73, 136.94, 135.20, 120.21, 119.03, 116.33, 112.65, 40.06, 37.11, 35.31, 32.44.
2-Amino-N-(2-dimethylcarbamoyl-ethyl)-benzamide (3c): The product was obtained as a white solid in a yield of 43% (0.23 g). MP: 134–136 °C. 1H NMR (600 MHz, CDCl3) δ 8.04 (s, 1H), 7.41 (d, J = 7.9 Hz, 1H), 7.34 (t, J = 7.8 Hz, 1H), 6.80 (d, J = 8.4 Hz, 1H), 6.61 (t, J = 7.5 Hz, 1H), 5.84 (s, 2H), 3.55 (t, J = 7.3 Hz, 2H), 3.00 (s, 3H), 2.96 (s, 3H), 2.68 (t, J = 7.3 Hz, 2H). 13C NMR (151 MHz, CDCl3) δ 172.03, 171.13, 149.76, 133.62, 128.45, 114.89, 113.43, 112.02, 39.16, 37.25, 35.37, 32.86.
N,N-Dimethyl-3-(naphthalen-1-ylamino)-propionamide (3d): The product was obtained as a colorless liquid in a yield of 73% (0.52 g). 1H-NMR (600 MHz, CDCl3) δ 7.77 (d, J = 7.9 Hz, 1H), 7.69 (d, J = 7.8 Hz, 1H), 7.39 – 7.28 (m, 2H), 7.26 (t, J = 7.8 Hz, 1H), 7.18 – 7.10 (m, 1H), 6.57 (d, J = 7.5 Hz, 1H), 5.64 – 4.87 (m, 1H), 3.56 (t, J = 5.8 Hz, 2H), 2.86 (d, J = 6.3 Hz, 6H), 2.62 (t, J = 5.8 Hz, 2H).13C-NMR (151 MHz, CDCl3) δ 171.86, 143.27, 134.42, 128.48, 126.53, 125.77, 124.76, 123.87, 120.40, 117.37, 104.31, 39.73, 37.15, 35.36, 31.92.
3-(4-Methoxy-phenylamino)-N,N-dimethyl-propionamide (3e): The product was obtained as a white solid in a yield of 81% (0.5 g). MP: 85-87 °C. 1H-NMR (600 MHz, CDCl3) δ6.78 (d, J = 8.6 Hz, 2H), 6.64 (d, J = 8.6 Hz, 2H), 3.75 (s, 3H), 3.43 (t, J = 5.9 Hz, 2H), 2.96 (d, J = 6.5 Hz, 6H), 2.59 (t, J = 5.9 Hz, 2H). 13C NMR (151 MHz, CDCl3) δ 171.72, 152.41, 141.82, 114.92, 114.91, 55.80, 40.83, 37.12, 35.30, 32.18.
3-(2-Chloro-phenylamino)-N,N-dimethyl-propionamide (3f): The product was obtained as a yellow liquid in a yield of 82% (0.52 g). 1H NMR (600 MHz, CDCl3) δ 7.24 (dd, J = 7.9, 1.5 Hz, 1H), 7.15 – 7.11 (m, 1H), 6.71 (dd, J = 8.2, 1.1 Hz, 1H), 6.61 (tt, J = 14.6, 7.3 Hz, 1H), 4.92 (s, 1H), 3.54 (t, J = 6.4 Hz, 2H), 2.96 (d, J = 2.3 Hz, 6H), 2.62 (t, J = 6.4 Hz, 2H). 13C NMR (151 MHz, CDCl3) δ 170.16, 142.73, 128.25, 126.74, 118.56, 116.21, 110.12, 38.30, 36.08, 34.30, 31.33.
3-(4-Bromo-phenylamino)-N,N-dimethyl-propionamide (3g): The product was obtained as a white solid in a yield of 55% (0.47 g). MP: 110–113 °C. 1H NMR (600 MHz, CDCl3) δ7.24 (d, J = 8.8 Hz, 2H), 6.52 (d, J = 8.8 Hz, 2H), 4.65 (s, 1H), 3.44 (t, J = 5.9 Hz, 2H), 2.96 (d, J = 8.4 Hz, 6H), 2.58 (t, J = 5.9 Hz, 2H).13C NMR (151 MHz, CDCl3) δ 171.46, 146.80, 131.97, 114.83, 109.08, 39.58, 37.11, 35.32, 31.95.
3-Phenylamino-propionamide (
3i): The product was obtained as a white solid in a yield of 84% (0.39 g). MP: 53–56 °C [
31].
1H NMR (600 MHz, CDCl
3) δ7.18 (t,
J = 7.9 Hz, 2H), 6.73 (t,
J = 7.3 Hz, 1H), 6.63 (d,
J = 7.9 Hz, 2H), 5.91 (s, 2H), 3.44 (t,
J = 6.1 Hz, 2H), 2.49 (t,
J = 6.1 Hz, 2H).
13C NMR (151 MHz, CDCl
3) δ 174.26, 147.54, 129.38, 118.09, 113.40, 39.99, 34.91.
3-(4-Bromo-phenylamino)-propionamide (3j): The product was obtained as a white solid in a yield of 79% (0.68 g). MP: 122–125 °C. 1H NMR (600 MHz, CDCl3) δ7.29 – 7.28 (m, 1H), 7.27 (d, J = 2.1 Hz, 1H), 6.64 – 6.47 (m, 2H), 5.57 (d, 2H), 3.48 – 3.41 (m, 2H), 2.59 – 2.50 (m, 2H). 13C NMR (151 MHz, CDCl3) δ 173.48, 146.05, 132.10, 115.26, 110.16, 40.19, 34.28.
N,N-Diethyl-3-phenylamino-propionamide (
3k)[
32]: The product was obtained as a yellow liquid in a yield of 83% (0.39 g).
1H NMR (600 MHz, CDCl
3) δ 7.21 – 7.11 (m, 2H), 6.68 (t,
J = 7.3 Hz, 1H), 6.64 – 6.61 (m, 2H), 4.10 (m, 1H), 3.46 (t,
J = 6.2 Hz, 2H), 3.36 (q,
J = 7.1 Hz, 2H), 3.23 (q,
J = 7.2 Hz, 2H), 2.57 (t,
J = 6.2 Hz, 2H), 1.10 (td,
J = 7.1, 5.6 Hz, 6H).
13C NMR (151 MHz, CDCl
3) δ170.75, 148.04, 129.25, 117.35, 113.18, 41.90, 40.13, 39.75, 32.00, 14.16, 13.11.
N,N-Diethyl-3-p-tolylamino-propionamide (3l): The product was obtained as a white solid in a yield of 72% (0.39 g). MP: 52–55 °C. 1H NMR (600 MHz, CDCl3) δ6.98 (d, J = 8.3 Hz, 2H), 6.57 (d, J = 8.3 Hz, 2H), 3.45 (t, J = 6.1 Hz, 2H), 3.37 (q, J = 7.1 Hz, 2H), 3.25 (q, J = 7.2 Hz, 2H), 2.58 (t, J = 6.1 Hz, 2H), 2.23 (s, 3H), 1.11 (td, J = 7.1, 3.1 Hz, 6H). 13C NMR (151 MHz, CDCl3) δ170.77, 145.45, 129.78, 126.96, 113.66, 41.87, 40.41, 40.09, 31.96, 20.41, 14.17, 13.11.
3-(2-Amino-phenylamino)-N,N-diethyl-propionamide (3m): The product was obtained as a black liquid in a yield of 63% (0.34 g). 1H NMR (600 MHz, CDCl3) δ 6.82 – 6.77 (m, 1H), 6.75 – 6.69 (m, 3H), 3.85 (s, 2H), 3.47 (t, J = 6.2 Hz, 2H), 3.39 (q, J = 7.1 Hz, 2H), 3.29 (q, J = 7.2 Hz, 2H), 2.65 (t, J = 6.2 Hz, 2H), 1.14 (dt, J = 13.9, 7.1 Hz, 6H). 13C NMR (151 MHz, CDCl3) δ170.87, 136.52, 135.44, 120.22, 119.48, 116.43, 113.28, 41.94, 40.62, 40.17, 32.06, 14.21, 13.12.
3-(3-Chloro-phenylamino)-N,N-diethyl-propionamide (3o): The product was obtained as a white solid in a yield of 76% (0.48 g). MP: 58–61 °C. 1H NMR (600 MHz, CDCl3) δ 7.04 (t, J = 8.0 Hz, 1H), 6.65 – 6.61 (m, 1H), 6.59 (t, J = 2.1 Hz, 1H), 6.49 (dd, J = 8.2, 2.2 Hz, 1H), 4.58 (m, 1H), 3.49 – 3.41 (m, 2H), 3.37 (q, J = 7.1 Hz, 2H), 3.25 (q, J = 7.2 Hz, 2H), 2.57 (t, J = 6.0 Hz, 2H), 1.18 – 0.99 (m, 6H). 13C NMR (151 MHz, CDCl3) δ 171.73, 136.94, 135.20, 120.21, 119.03, 116.33, 112.65, 40.06, 37.11, 35.31, 32.44.
3-Benzylamino-N,N-diethyl-propionamide (3p): The product was obtained as a white solid in a yield of 84% (0.45 g). MP: 119–121 °C. 1H NMR (600 MHz, DMSO-d6) δ 9.44 (s, 1H), 7.68 – 7.51 (m, 2H), 7.51 – 7.37 (m, 3H), 4.26 – 4.04 (m, 2H), 3.27 (q, J = 7.1 Hz, 4H), 3.05 (t, J = 7.1 Hz, 2H), 2.83 (t, J = 7.2 Hz, 2H), 1.11 (t, J = 7.1 Hz, 3H), 1.02 (t, J = 7.1 Hz, 3H). 13C NMR (151 MHz, DMSO-d6) δ 168.78, 132.51, 130.46, 129.32, 129.12, 50.27, 43.13, 41.74, 40.38, 40.24, 40.11, 29.02, 14.40, 13.47.
3-Phenylamino-propionic acid methyl ester (
3q): The product was obtained as a white solid in a yield of 88% (0.41 g). MP: 32–35 °C [
33].
1H NMR (600 MHz, CDCl
3) δ 7.17 (t,
J = 7.6 Hz, 2H), 6.71 (t,
J = 7.3 Hz, 1H), 6.61 (d,
J = 8.1 Hz, 2H), 4.02 (s, 1H), 3.68 (s, 3H), 3.43 (t,
J = 6.4 Hz, 2H), 2.60 (t,
J = 6.4 Hz, 2H).
13C NMR (151 MHz, CDCl
3) δ 172.85, 147.60, 129.52, 129.35, 129.15, 117.91, 117.75, 117.54, 113.24, 113.06, 112.83, 99.99, 51.78, 39.43, 33.73.
3-p-Tolylamino-propionic acid ethyl ester (
3r) [
34]: The product was obtained as a yellow liquid in a yield of 82% (0.44 g).
1H NMR (600 MHz, CDCl
3) δ 7.02 – 6.91 (m, 2H), 6.58 – 6.51 (m, 2H), 4.14 (qd,
J = 7.5, 2.3 Hz, 2H), 3.85 (s, 1H), 3.41 (td,
J = 6.4, 1.0 Hz, 2H), 2.58 (td,
J = 6.4, 0.8 Hz, 2H), 2.23 (d,
J = 2.3 Hz, 3H), 1.25 (ddd,
J = 7.1, 4.5, 1.8 Hz, 3H).
13C NMR (151 MHz, CDCl
3) δ 172.44, 145.32, 129.96, 129.78, 126.92, 113.30, 60.56, 60.52, 39.83, 33.92, 20.37, 20.19, 14.19.
3-(Pyridin-2-ylamino)-propionic acid ethyl ester (
3s): The product was obtained as a grey solid in a yield of 33% (0.15 g). MP: 46–48 °C [
35].
1H NMR (600 MHz, CDCl
3) δ 8.08 (dd,
J = 4.8, 0.9 Hz, 1H), 7.45 – 7.31 (m, 1H), 6.56 (dd,
J = 6.7, 5.4 Hz, 1H), 6.40 (d,
J = 8.4 Hz, 1H), 4.96 (s, 1H), 4.15 (q,
J = 7.1 Hz, 2H), 3.64 (d,
J = 5.1 Hz, 2H), 2.63 (t,
J = 6.2 Hz, 2H), 1.26 (t,
J = 7.1 Hz, 3H).
13C NMR (151 MHz, CDCl
3) δ 172.61, 158.22, 147.93, 137.37, 112.94, 107.64, 60.60, 37.36, 34.23, 14.20.
3-p-Tolylamino-propionic acid tert-butyl ester (3t): The product was obtained as a yellow liquid in a yield of 82% (0.44 g). 1H NMR (600 MHz, CDCl3) δ6.91 (d, J = 7.9 Hz, 2H), 6.48 (d, J = 8.2 Hz, 2H), 3.83 (s, 1H), 3.29 (t, J = 6.3 Hz, 2H), 2.43 (t, J = 6.3 Hz, 2H), 2.16 (s, 3H), 1.37 (s, 9H). 13C NMR (151 MHz, CDCl3) δ 170.79, 144.49, 128.74, 125.82, 112.29, 79.75, 39.02, 34.08, 28.67, 27.09, 26.95, 19.35.
3-Phenylamino-propionic acid tert-butyl ester (
3u) [
36]: The product was obtained as a yellow liquid in a yield of 80% (0.37 g).
1H NMR (600 MHz, CDCl
3) δ 7.16 (dd,
J = 11.1, 4.2 Hz, 2H), 6.69 (t,
J = 7.3 Hz, 1H), 6.60 (d,
J = 7.9 Hz, 2H), 4.03 (s, 1H), 3.37 (t,
J = 6.3 Hz, 2H), 2.50 (t,
J = 6.3 Hz, 2H), 1.44 (s, 9H).
13C NMR (151 MHz, CDCl
3) δ 171.79, 147.88, 129.33, 129.30, 117.62, 117.59, 113.10, 113.07, 80.86, 39.69, 35.17, 28.17, 28.14.
3-(4-Chloro-phenylamino)-propionic acid tert-butyl ester (3v): The product was obtained as a white solid in a yield of 83% (0.53 g). MP: 33–36 °C. 1H NMR (600 MHz, CDCl3) δ 7.13 (d, J = 8.7 Hz, 2H), 6.57 (d, J = 8.7 Hz, 2H), 4.41 (s, 1H), 3.37 (t, J = 6.3 Hz, 2H), 2.52 (t, J = 6.3 Hz, 2H), 1.45 (s, 9H). 13C NMR (151 MHz, CDCl3) δ 171.60, 146.03, 129.13, 122.51, 114.40, 81.07, 39.98, 34.76, 28.11.
3-(Methyl-phenyl-amino)-propionamide (3w): The product was obtained as a white solid in a yield of 80% (0.43 g). MP: 88–90 °C. 1H NMR (600 MHz, DMSO-d6) δ 7.38 (s, 1H), 7.21 – 7.10 (m, 2H), 6.85 (s, 1H), 6.70 (d, J = 8.0 Hz, 2H), 6.65 – 6.54 (m, 1H), 3.56 – 3.49 (m, 2H), 2.84 (s, 3H), 2.29 – 2.09 (m, 2H). 13C NMR (151 MHz, DMSO-d6) δ 173.82, 149.12, 129.54, 116.60, 116.13, 112.78, 112.43, 48.84, 38.13, 32.50.
3-(pyrrolidin-1-yl)propanamide (
3x) [
37]: The product was obtained as a yellow liquid in a yield of 96% (0.48 g).
1H NMR (600 MHz, CDCl
3) δ 8.21 (s, 1H), 5.88 (s, 1H), 2.79 (s, 2H), 2.62 (s, 4H), 2.45 (s, 2H), 1.82 (t,
J = 3.5 Hz, 4H).
13C NMR (151 MHz, CDCl
3) δ 175.51, 53.84, 53.43, 51.62, 34.13, 23.50, 22.75.
3-(4-methylpiperidin-1-yl)propanamide (
3y): The product was obtained as a white solid in a yield of 95% (0.47 g). MP: 116–117 °C [
38].
1H NMR (600 MHz, CDCl
3) δ 8.24 (s, 1H), 5.43 (s, 1H), 2.93 (d,
J=11.7, 2H), 2.61 (td,
J = 6.4, 3.8, 2H), 2.40 (t,
J = 6.2, 2H), 2.00 (t,
J = 11.0, 2H), 1.68–1.56 (m, 2H), 1.48–1.30 (m, 1H), 1.21 (ddd,
J = 15.6, 12.5, 3.7, 2H), 0.88 (d,
J = 6.5, 3H).
13C NMR (151 MHz, CDCl
3) δ 174.94, 53.92, 53.27, 42.62, 37.18, 33.88, 31.92, 30.49, 21.63.
Phenylamine (
1a) [
39]: The product was obtained as a colorless liquid in a yield of 63% (0.12 g).
1H NMR (600 MHz, CDCl
3) δ 7.11 (dd,
J = 8.4, 7.4 Hz, 2H), 6.72 (t,
J = 7.4 Hz, 1H), 6.58 (dd,
J = 8.5, 1.0 Hz, 2H), 3.49 (s, 2H).
13C NMR (151 MHz, CDCl
3) δ 146.69, 129.47, 118.61, 115.29.
o-phenylenediamine (
1b): The product was obtained as a colorless solid in a yield of 82% (0.17 g). MP: 100–102 °C [
40].
1H NMR (600 MHz, CDCl
3) δ 6.87 – 6.50 (m, 4H), 3.35 (s, 4H).
13C NMR (151 MHz, CDCl
3) δ 134.71, 120.22, 116.70.
1-Aminonaphthalene (
1c): The product was obtained as a white solid in a yield of 85% (0.2 g). MP: 45–48 °C [
41].
1H NMR (600 MHz, CDCl
3) δ 7.79 (tt,
J = 7.0, 4.2 Hz, 2H), 7.51 – 7.37 (m, 2H), 7.37 – 7.24 (m, 2H), 6.76 (dd,
J = 7.1, 1.2 Hz, 1H), 4.12 (s, 2H).
13C NMR (151 MHz, CDCl
3) δ 142.10, 134.43, 128.59, 126.38, 125.89, 124.90, 123.69, 120.83, 119.02, 109.72.
4-Methoxy-phenylamine (
1d): The product was obtained as a brown solid in a yield of 72% (0.16g). MP: 56–59 °C [
42].
1H NMR (600 MHz, CDCl
3) δ 6.86 – 6.68 (m, 2H), 6.68 – 6.57 (m, 2H), 3.85 – 3.61 (m, 3H), 3.39 (s, 2H).
13C NMR (151 MHz, CDCl
3) δ 152.78, 139.92, 116.41, 114.79, 55.72.
2-Chloro-phenylamine (
1e) [
43]: The product was obtained as a colorless liquid in a yield of 40% (0.09 g).
1H NMR (600 MHz, CDCl
3) δ 7.31 – 7.19 (m, 1H), 7.04 (dd,
J = 10.2, 4.2 Hz, 1H), 6.68 (dd,
J = 16.0, 6.6 Hz, 2H), 4.00 (s, 2H).
13C NMR (151 MHz, CDCl
3) δ 143.12, 129.53, 127.82, 119.32, 119.12, 116.06.
4-Bromo-phenylamine (
1f): The product was obtained as a grey solid in a yield of 70% (0.19 g). MP: 59–62 °C [
44].
1H NMR (600 MHz, CDCl
3) δ 7.29 – 7.09 (m, 2H), 6.61 – 6.42 (m, 2H), 3.66 (d,
J = 36.7 Hz, 2H).
13C NMR (151 MHz, CDCl
3) δ 145.41, 131.95, 116.68, 110.09.
Phenylamine (1g): The product was obtained as a colorless liquid in a yield of 73% (0.12 g). 1H NMR (600 MHz, CDCl3) δ7.11 (dd, J = 8.4, 7.4 Hz, 2H), 6.72 (t, J = 7.4 Hz, 1H), 6.58 (dd, J = 8.5, 1.0 Hz, 2H), 3.49 (s, 2H). 13C NMR (151 MHz, CDCl3) δ 146.69, 129.47, 118.61, 115.29.
4-Bromo-phenylamine (1h): The product was obtained as a grey solid in a yield of 70% (0.17 g). MP: 59––62 °C. 1H NMR (600 MHz, CDCl3) δ 7.29 – 7.09 (m, 2H), 6.61 – 6.42 (m, 2H), 3.66 (d, J = 36.7 Hz, 2H). 13C NMR (151 MHz, CDCl3) δ 145.41, 131.95, 116.68, 110.09.
o-phenylenediamine (1i): The product was obtained as a colorless solid in a yield of 81% (0.19 g). MP: 100–102 °C. 1H NMR (600 MHz, CDCl3) δ 6.87 – 6.50 (m, 4H), 3.35 (s, 4H). 13C NMR (151 MHz, CDCl3) δ 134.71, 120.22, 116.70.
3-Chloro-phenylamine (
1j) [
45]: The product was obtained as a colorless liquid in a yield of 35% (0.09 g).
1H NMR (600 MHz, CDCl
3) δ 7.01 (t,
J = 8.0 Hz, 1H), 6.69 (dd,
J = 7.9, 1.1 Hz, 1H), 6.59 (t,
J = 2.1 Hz, 1H), 6.47 (dd,
J = 8.1, 2.2 Hz, 1H), 3.65 (s, 2H).
13C NMR (151 MHz, CDCl
3) δ 147.85, 134.82, 130.47, 118.42, 114.97, 113.36.
p-Tolylamine (
1k): The product was obtained as a colorless solid in a yield of 72% (0.17 g). MP: 43–45 °C [
46].
1H NMR (600 MHz, CDCl
3) δ 7.07 – 6.83 (m, 2H), 6.58 (d,
J = 8.0 Hz, 2H), 3.46 (s, 2H), 2.23 (s, 3H).
13C NMR (151 MHz, CDCl
3) δ 143.91, 129.82, 127.80, 115.32, 20.52.
p-Tolylamine (1l): The product was obtained as a colorless solid in a yield of 76% (0.18 g). MP: 43–45 °C. 1H NMR (600 MHz, CDCl3) δ 7.07 – 6.83 (m, 2H), 6.58 (d, J = 8.0 Hz, 2H), 3.46 (s, 2H), 2.23 (s, 3H). 13C NMR (151 MHz, CDCl3) δ 143.91, 129.82, 127.80, 115.32, 20.52.
Phenylamine (1m): The product was obtained as a colorless liquid in a yield of 72% (0.16 g). 1H NMR (600 MHz, CDCl3) δ7.11 (dd, J = 8.4, 7.4 Hz, 2H), 6.72 (t, J = 7.4 Hz, 1H), 6.58 (dd, J = 8.5, 1.0 Hz, 2H), 3.49 (s, 2H). 13C NMR (151 MHz, CDCl3) δ 146.69, 129.47, 118.61, 115.29.
Chloro-phenylamine (
1o): The product was obtained as a white solid in a yield of 82% (0.21 g). MP: 65–67 °C [
47].
1H NMR (600 MHz, CDCl
3) δ 7.09 (d,
J = 8.6 Hz, 2H), 6.60 (d,
J = 8.6 Hz, 2H), 3.64 (s, 2H).
13C NMR (151 MHz, CDCl
3) δ 144.97, 129.14, 123.16, 116.25.
2,3,4,5-tetrahydro-1H-1,5-benzodiazepin-2-one (
6a, 6b and
6c): The products were obtained as a tan solid in yields of 74% (0.24 g), 68% (0.22 g) and 34% (0.17 g), respectively. MP: 138–140 °C [
48,
49].
1H NMR (600 MHz, CDCl
3) δ 8.36 (s, 1H), 6.96 (t,
J = 7.5 Hz, 1H), 6.90 (d,
J = 7.8 Hz, 1H), 6.82 (t,
J = 7.5 Hz, 1H), 6.74 (d,
J = 7.9 Hz, 1H), 3.70 – 3.63 (m, 2H), 2.80 – 2.69 (m, 2H).
13C NMR (151 MHz, CDCl
3) δ 174.06, 138.75, 126.36, 125.36, 122.19, 120.57, 119.89, 45.55, 36.19.
4-Methyl-4,5-dihydro-1H-benzo[b][1,4]diazepin-2(3H)-one (
6d): The product was obtained as a white solid in a yield of 93% (0.30 g). MP: 182–184 °C [
50,
51].
1H NMR (600 MHz, CDCl
3) δ 8.60 (s, 1H), 7.01 – 6.97 (m, 1H), 6.95 (dd,
J = 7.7, 1.0, 1H), 6.88 (t,
J = 7.5, 1H), 6.78 (d,
J = 7.8, 1H), 4.03 (dt, 1H), 3.55 (s, 1H), 2.64 (dd,
J = 13.6, 4.0, 1H), 2.45 (dd,
J = 13.7, 7.9, 1H), 1.33 (d,
J = 6.3, 3H).
13C NMR (151 MHz, CDCl
3) δ 173.07, 138.25, 128.00, 125.64, 122.19, 121.41, 121.00, 54.14, 41.44, 23.62.
7,8-difluoro-2-methyl-1,2,3,5-tetrahydro-1,5-benzodiazepin-4-one (6e): The product was obtained as a white solid in a yield of 90% (0.29 g). MP: 193.5–195 °C. 1H NMR (600 MHz, CDCl3) δ 8.46 (s, 1H), 6.82 (dd, J = 10.6, 7.9, 1H), 6.64 (dd, J = 11.0, 7.5, 1H), 4.05 (dd, J = 12.1, 6.3, 1H), 3.38 (s, 1H), 2.63 (dd, J=13.5, 4.5, 1H), 2.40 (dd, J = 13.5, 7.6, 1H), 1.72 (s, 1H), 1.33 (d, J = 6.3, 3H).13C NMR (151 MHz, CDCl3) δ = 172.99, 148.33, 148.24, 146.69, 146.60, 145.90, 145.84, 144.30, 144.21, 134.80, 124.80, 111.04, 110.91, 109.69, 109.56, 55.07, 40.76, 23.43.
2,3-dihydro-1,5-benzothiazepine-4(5H)-one (
6f): The product was obtained as a brown solid in a yield of 34% (0.11 g). Mp: 215–216°C[
52].
1H NMR (600 MHz, CDCl
3) δ 8.55 (s, 1H), 7.61 (d,
J = 7.7, 1H), 7.37 (td,
J = 7.7, 1.2, 1H), 7.17 (t,
J = 7.6, 1H), 7.14 (d,
J = 7.8, 1H), 3.46 (t,
J = 6.9, 2H), 2.65 (t,
J = 6.9, 2H).
13C NMR (151 MHz, CDCl
3) δ 173.71, 141.41, 135.51, 129.83, 126.97, 126.49, 123.30, 34.29, 33.54.
3-Methyl-2,3-dihydrobenzo[b][1,4]thiazepin-4(5H)-one (
6g): The product was obtained as a white solid in a yield of 31% (0.10 g). Mp: 176–178 °C [
53].
1H NMR (600 MHz, CDCl
3) δ 8.32 (s, 1H), 7.60 (d,
J = 7.6, 1H), 7.36 (t,
J = 6.9, 1H), 7.17 (t,
J = 8.2, 1H), 7.13 (d,
J = 7.8, 1H), 3.49 (dd,
J = 11.3, 6.0, 1H), 3.05 – 2.98 (m, 1H), 2.79 (dt,
J = 12.6, 6.3, 1H), 1.19 (d,
J = 6.6, 3H).
13C NMR (151 MHz, CDCl
3) δ 175.74, 141.12, 135.15, 129.72, 127.72, 126.46, 123.47, 41.51, 36.33, 15.47.