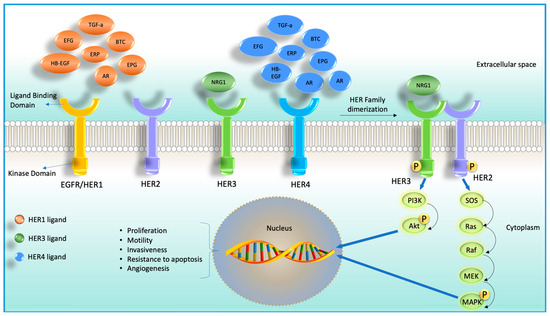
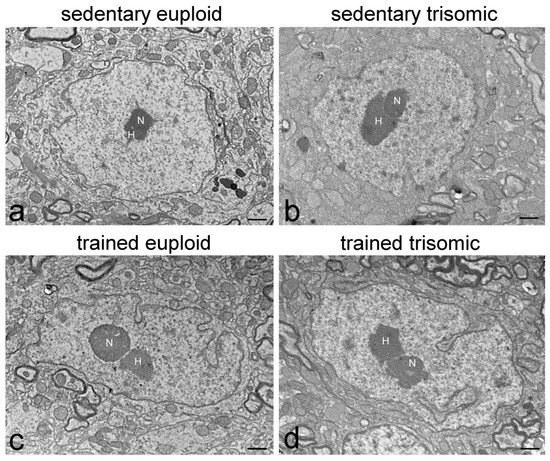
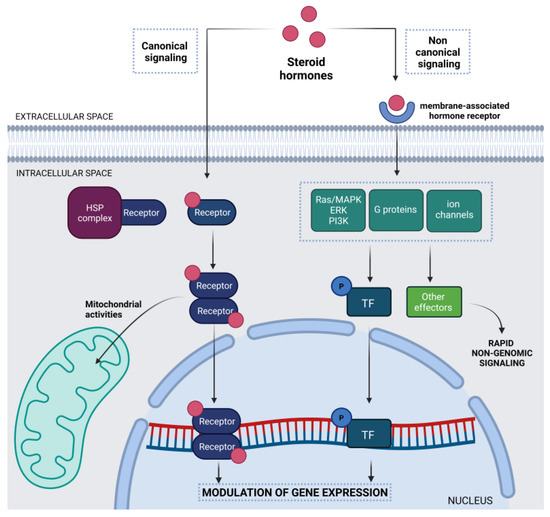
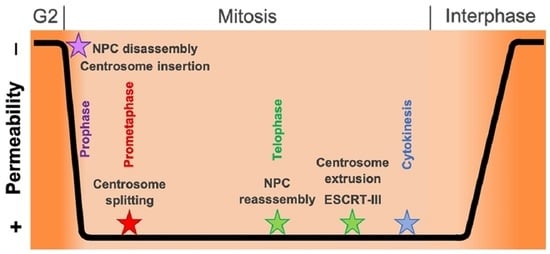
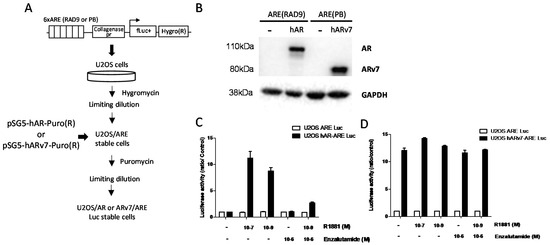
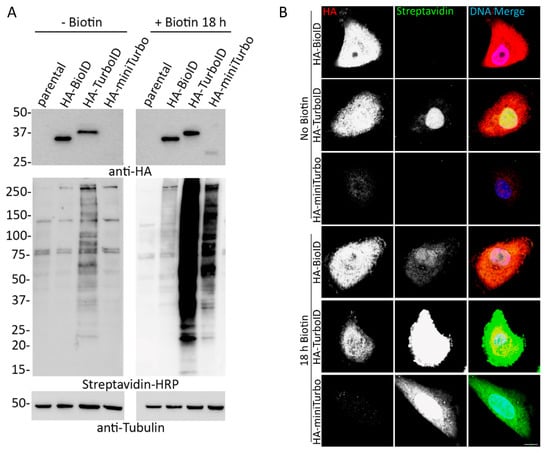
Feature Papers in Cell Nuclei: Function, Transport and Receptors
A topical collection in Cells (ISSN 2073-4409). This collection belongs to the section "Cell Nuclei: Function, Transport and Receptors".
Viewed by 39864Editor
Interests: nuclear hormone receptors; androgen receptor; glucocorticoid receptor; antiandrogens; glucocorticoids; urothelial cancer; prostate cancer; genitourinary pathology
Special Issues, Collections and Topics in MDPI journals
Topical Collection Information
Dear Colleagues,
This Topical Collection “Feature Papers in Cell Nuclei: Function, Transport and Receptors” aims to collect high-quality research articles, review articles, and communications on all aspects of cell nuclei, with a focus on cell biology research. Since the goal of this Topical Collection is to illustrate, through selected works, frontier research in cell nuclei, I encourage Editorial Board Members of Cells to contribute papers reflecting the latest progress in their research field or to invite relevant experts and colleagues to do so. We also welcome senior experts in this field to make contributions to this Topical Collection.
Topics include, without being limited to:
- Gene regulation
- Transcription
- Translation
- Cell growth
- Cell reproduction
- Nuclear structures
- Nuclear transport and assembly
- Nuclear receptors
Dr. Hiroshi Miyamoto
Collection Editor
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 100 words) can be sent to the Editorial Office for announcement on this website.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Cells is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2700 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.