Investigating the Effect of Ni Loading on the Performance of Yttria-Stabilised Zirconia Supported Ni Catalyst during CO2 Methanation
Abstract
:1. Introduction
2. Results and Discussion
2.1. Catalyst Characterisation
2.1.1. Textural Properties
2.1.2. XRD Analysis
2.1.3. SEM Measurement
2.1.4. Catalyst Reducibility
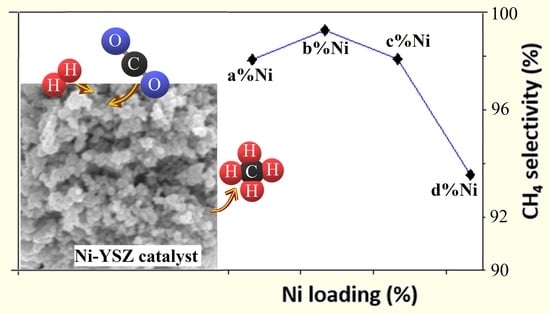
2.2. Catalyst Activity Test
3. Materials and Methods
3.1. Catalyst Synthesis
3.2. Catalyst Characterisation
3.3. Catalyst Testing
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- U.S. Energy Information Administration. International Energy Outlook 2021. Analysis & Projections. 2021. Available online: https://www.eia.gov/outlooks/ieo/consumption/sub-topic-01.php (accessed on 3 November 2021).
- NBC. News Greenhouse Gas Levels Hit a New Record, Cuts Fall Short, U.N. Finds. Climate in Crisis. 2021. Available online: https://www.nbcnews.com/science/environment/greenhouse-gas-levels-hit-new-record-cuts-fall-short-un-finds-rcna3728 (accessed on 3 November 2021).
- Barbera, E.; Mantoan, F.; Bertucco, A.; Bezzo, F. Hydrogenation to convert CO2 to C1 chemicals: Technical comparison of different alternatives by process simulation. Can. J. Chem. Eng. 2020, 98, 1893–1906. [Google Scholar] [CrossRef]
- Wang, S.; Tarroja, B.; Schell, L.S.; Shaffer, B.; Samuelsen, S. Prioritizing among the end uses of excess renewable energy for cost-effective greenhouse gas emission reductions. Appl. Energy 2019, 235, 284–298. [Google Scholar] [CrossRef]
- Chauvy, R.; Verdonck, D.; Dubois, L.; Thomas, D.; De Weireld, G. Techno-economic feasibility and sustainability of an integrated carbon capture and conversion process to synthetic natural gas. J. CO2 Util. 2021, 47, 101488. [Google Scholar] [CrossRef]
- Salomone, F.; Giglio, E.; Ferrero, D.; Santarelli, M.; Pirone, R.; Bensaid, S. Techno-economic modelling of a Power-to-Gas system based on SOEC electrolysis and CO2 methanation in a RES-based electric grid. Chem. Eng. J. 2019, 377, 120233. [Google Scholar] [CrossRef]
- Hussain, I.; Jalil, A.A.; Izan, S.M.; Azami, M.S.; Kidam, K.; Ainirazali, N.; Ripin, A. Thermodynamic and experimental explorations of CO2 methanation over highly active metal-free fibrous silica-beta zeolite (FS@SiO2-BEA) of innovative morphology. Chem. Eng. Sci. 2021, 229, 116015. [Google Scholar] [CrossRef]
- Beuls, A.; Swalus, C.; Jacquemin, M.; Heyen, G.; Karelovic, A.; Ruiz, P. Methanation of CO2: Further insight into the mechanism over Rh/γ-Al2O3 catalyst. Appl. Catal. B Environ. 2012, 113–114, 2–10. [Google Scholar] [CrossRef]
- Gao, J.; Liu, Q.; Gu, F.; Liu, B.; Zhong, Z.; Su, F. Recent advances in methanation catalysts for the production of synthetic natural gas. RSC Adv. 2015, 5, 22759–22776. [Google Scholar] [CrossRef]
- González-Castaño, M.; de Miguel, J.C.N.; Penkova, A.; Centeno, M.A.; Odriozola, J.A.; Arellano-Garcia, H. Ni/YMnO3 perovskite catalyst for CO2 methanation. Appl. Mater. Today 2021, 23, 101055. [Google Scholar] [CrossRef]
- Duyar, M.S.; Treviño, M.A.A.; Farrauto, R.J. Dual function materials for CO2 capture and conversion using renewable H2. Appl. Catal. B Environ. 2015, 168–169, 370–376. [Google Scholar] [CrossRef]
- Zhao, K.; Li, Z.; Bian, L. CO2 methanation and co-methanation of CO and CO2 over Mn-promoted Ni/Al2O3 catalysts. Front. Chem. Sci. Eng. 2016, 10, 273–280. [Google Scholar] [CrossRef]
- Ahn, J.Y.; Chang, S.W.; Lee, S.M.; Kim, S.S.; Chung, W.J.; Lee, J.C.; Cho, Y.J.; Shin, K.S.; Moon, D.H.; Nguyen, D.D. Developing Ni-based honeycomb-type catalysts using different binary oxide-supported species for synergistically enhanced CO2 methanation activity. Fuel 2019, 250, 277–284. [Google Scholar] [CrossRef]
- Lin, J.; Ma, C.; Wang, Q.; Xu, Y.; Ma, G.; Wang, J.; Wang, H.; Dong, C.; Zhang, C.; Ding, M. Enhanced low-temperature performance of CO2 methanation over mesoporous Ni/Al2O3-ZrO2 catalysts. Appl. Catal. B Environ. 2019, 243, 262–272. [Google Scholar] [CrossRef]
- Karelovic, A.; Ruiz, P. Improving the hydrogenation function of Pd/γ-Al2O3 catalyst by Rh/γ-Al2O3 Addition in CO2 methanation at low temperature. ACS Catal. 2013, 3, 2799–2812. [Google Scholar] [CrossRef]
- Younas, M.; Loong Kong, L.; Bashir, M.J.K.; Nadeem, H.; Shehzad, A.; Sethupathi, S. Recent Advancements, Fundamental Challenges, and Opportunities in Catalytic Methanation of CO2. Energy Fuels 2016, 30, 8815–8831. [Google Scholar] [CrossRef]
- Nizio, M.; Albarazi, A.; Cavadias, S.; Amouroux, J.; Galvez, M.E.; Da Costa, P. Hybrid plasma-catalytic methanation of CO2 at low temperature over ceria zirconia supported Ni catalysts. Int. J. Hydrogen Energy 2016, 41, 11584–11592. [Google Scholar] [CrossRef]
- Lee, C.J.; Lee, D.H.; Kim, T. Enhancement of methanation of carbon dioxide using dielectric barrier discharge on a ruthenium catalyst at atmospheric conditions. Catal. Today 2017, 293–294, 97–104. [Google Scholar] [CrossRef]
- Ab Halim, A.Z.; Ali, R.; Bakar, W.A.W.A. CO2/H2 methanation over M*/Mn/Fe-Al2O3 (M*: Pd, Rh, and Ru) catalysts in natural gas; optimization by response surface methodology-central composite design. Clean Technol. Environ. Policy 2015, 17, 627–636. [Google Scholar] [CrossRef]
- Kustov, A.L.; Frey, A.M.; Larsen, K.E.; Johannessen, T.; Nørskov, J.K.; Christensen, C.H. CO methanation over supported bimetallic Ni-Fe catalysts: From computational studies towards catalyst optimization. Appl. Catal. A Gen. 2007, 320, 98–104. [Google Scholar] [CrossRef]
- Liu, Q.; Yang, H.; Dong, H.; Zhang, W.; Bian, B.; He, Q.; Yang, J.; Meng, X.; Tian, Z.; Zhao, G. Effects of preparation method and Sm2O3 promoter on CO methanation by a mesoporous NiO-Sm2O3/Al2O3 catalyst. New J. Chem. 2018, 42, 13096–13106. [Google Scholar] [CrossRef]
- Yan, X.; Liu, Y.; Zhao, B.; Wang, Z.; Wang, Y.; Liu, C. Methanation over Ni/SiO2: Effect of the catalyst preparation methodologies. Int. J. Hydrogen Energy 2013, 38, 2283–2291. [Google Scholar] [CrossRef]
- Liu, Q.; Tian, Y. One-pot synthesis of NiO/SBA-15 monolith catalyst with a three-dimensional framework for CO2 methanation. Int. J. Hydrogen Energy 2017, 42, 12295–12300. [Google Scholar] [CrossRef]
- Mota, F.M.; Kim, D.H. From CO2 methanation to ambitious long-chain hydrocarbons: Alternative fuels paving the path to sustainability. Chem. Soc. Rev. 2019, 48, 205–259. [Google Scholar] [CrossRef]
- Abate, S.; Mebrahtu, C.; Giglio, E.; Deorsola, F.; Bensaid, S.; Perathoner, S.; Pirone, R.; Centi, G. Catalytic Performance of γ-Al2O3-ZrO2-TiO2-CeO2 Composite Oxide Supported Ni-Based Catalysts for CO2 Methanation. Ind. Eng. Chem. Res. 2016, 55, 4451–4460. [Google Scholar] [CrossRef]
- Cheng, C.; Shen, D.; Xiao, R.; Wu, C. Methanation of syngas (H2/CO) over the different Ni-based catalysts. Fuel 2017, 189, 419–427. [Google Scholar]
- Moghaddam, S.V.; Rezaei, M.; Meshkani, F.; Daroughegi, R. Synthesis of nanocrystalline mesoporous Ni/Al2O3–SiO2 catalysts for CO2 methanation reaction. Int. J. Hydrogen Energy 2018, 43, 19038–19046. [Google Scholar] [CrossRef]
- Jia, X.; Zhang, X.; Rui, N.; Hu, X.; Liu, C. jun Structural effect of Ni/ZrO2 catalyst on CO2 methanation with enhanced activity. Appl. Catal. B Environ. 2019, 244, 159–169. [Google Scholar] [CrossRef]
- Alves, L.M.N.C.; Almeida, M.P.; Ayala, M.; Watson, C.D.; Jacobs, G.; Rabelo-Neto, R.C.; Noronha, F.B.; Mattos, L.V. CO2 methanation over metal catalysts supported on ZrO2: Effect of the nature of the metallic phase on catalytic performance. Chem. Eng. Sci. 2021, 239, 116604. [Google Scholar] [CrossRef]
- Ilsemann, J.; Murshed, M.M.; Gesing, T.M.; Kopyscinski, J.; Bäumer, M. On the support dependency of the CO2 methanation—Decoupling size and support effects. Catal. Sci. Technol. 2021, 11, 4098–4114. [Google Scholar] [CrossRef]
- Kouva, S.; Honkala, K.; Lefferts, L.; Kanervo, J. Review: Monoclinic zirconia, its surface sites and their interaction with carbon monoxide. Catal. Sci. Technol. 2015, 5, 3473–3490. [Google Scholar] [CrossRef]
- Fan, Z.; Wang, Y.; Zhang, Y.; Liu, J. Influence of oxygen vacancy compensation on the structure, electronic and mechanical properties of yttrium stabilized tetragonal zirconia. Mater. Sci. Semicond. Process. 2021, 135, 106082. [Google Scholar] [CrossRef]
- Najafi, S.; Soltanali, S.; Nazemi, A.H. Effect of Synthesis Parameters on Zirconia Phases: Tetragonal or Monoclinic? ECS J. Solid State Sci. Technol. 2021, 10, 043003. [Google Scholar] [CrossRef]
- Kalita, P.; Ghosh, S.; Gutierrez, G.; Rajput, P.; Grover, V.; Sattonnay, G.; Avasthi, D.K. Grain size effect on the radiation damage tolerance of cubic zirconia against simultaneous low and high energy heavy ions: Nano triumphs bulk. Sci. Rep. 2021, 11, 10886. [Google Scholar] [CrossRef] [PubMed]
- Comba, A.; Baldi, A.; Tempesta, R.M.; Carossa, M.; Perrone, L.; Saratti, C.M.; Rocca, G.T.; Femiano, R.; Femiano, F.; Scotti, N. Do chemical-based bonding techniques affect the bond strength stability to cubic zirconia? Materials 2021, 14, 3920. [Google Scholar] [CrossRef] [PubMed]
- de Souza, E.F.; Appel, L.G. Oxygen vacancy formation and their role in the CO2 activation on Ca doped ZrO2 surface: An ab-initio DFT study. Appl. Surf. Sci. 2021, 553, 149589. [Google Scholar] [CrossRef]
- Ricca, C.; Ringuedé, A.; Cassir, M.; Adamo, C.; Labat, F. A comprehensive DFT investigation of bulk and low-index surfaces of ZrO2 polymorphs. J. Comput. Chem. 2015, 36, 9–21. [Google Scholar] [CrossRef]
- Chevalier, J.; Gremillard, L.; Virkar, A.V.; Clarke, D.R. The tetragonal-monoclinic transformation in zirconia: Lessons learned and future trends. J. Am. Ceram. Soc. 2009, 92, 1901–1920. [Google Scholar] [CrossRef]
- Hofer, T.S.; Kilchert, F.M.; Tanjung, B.A. An effective partial charge model for bulk and surface properties of cubic ZrO2, Y2O3 and yttrium-stabilised zirconia. Phys. Chem. Chem. Phys 2019, 21, 25635. [Google Scholar] [CrossRef]
- Cousland, G.P.; Cui, X.Y.; Ringer, S.; Smith, A.E.; Stampfl, A.P.J.; Stampfl, C.M. Electronic and vibrational properties of yttria-stabilised zirconia from first-principles for 10–40 mol% Y2O3. J. Phys. Chem. Solids 2014, 75, 1252–1264. [Google Scholar] [CrossRef]
- Gac, W.; Zawadzki, W.; Rotko, M.; Greluk, M.; Słowik, G.; Kolb, G. Effects of support composition on the performance of nickel catalysts in CO2 methanation reaction. Catal. Today 2020, 357, 468–482. [Google Scholar] [CrossRef]
- Traitangwong, A.; Guo, X.; Meeyoo, V.; Li, C. XNi/Ni0.05Ce0.20Zr0.75O2 Solid Solution over a CO2 Methanation Reaction. Ind. Eng. Chem. Res. 2020, 59, 13440–13449. [Google Scholar] [CrossRef]
- Kosaka, F.; Yamaguchi, T.; Ando, Y.; Mochizuki, T.; Takagi, H.; Matsuoka, K.; Fujishiro, Y.; Kuramoto, K. Effect of Ni content on CO2 methanation performance with tubular-structured Ni-YSZ catalysts and optimization of catalytic activity for temperature management in the reactor. Int. J. Hydrogen Energy 2020, 45, 12911–12920. [Google Scholar] [CrossRef]
- Kesavan, J.K.; Luisetto, I.; Tuti, S.; Meneghini, C.; Iucci, G.; Battocchio, C.; Mobilio, S.; Casciardi, S.; Sisto, R. Nickel supported on YSZ: The effect of Ni particle size on the catalytic activity for CO2 methanation. J. CO2 Util. 2018, 23, 200–211. [Google Scholar] [CrossRef]
- Al-Fatesh, A.S.; Ibrahim, A.A.; Osman, A.I.; Albaqi, F.; Arasheed, R.; Francesco, F.; Serena, T.; Anojaid, K.; Lanre, M.S.; Abasaeed, A.E.; et al. Effect of Holmium Oxide Loading on Nickel Catalyst Supported on Yttria-Stabilized Zirconia in Methane Dry Reforming. ACS Omega 2022, 7, 43700–43709. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.; Zhang, X.; Luo, D. Evolution and effect on electrolysis performance of pores in YSZ electrolyte films prepared by screen-printing. Ceram. Int. 2022. [Google Scholar] [CrossRef]
- Chang, H.; Chen, H.; Shao, Z.; Shi, J.; Bai, J.; Li, S.D. In situ fabrication of (Sr,La)FeO4 with CoFe alloy nanoparticles as an independent catalyst layer for direct methane-based solid oxide fuel cells with a nickel cermet anode. J. Mater. Chem. A 2016, 4, 13997–14007. [Google Scholar] [CrossRef]
- Wong-Ng, W.; Holomany, M.; McClune, W.F. The JCPDS Data Base-Present and Future. In Advances in X-ray Analysis; Cambridge University Press: Cambridge, UK, 1982; Volume 26, pp. 87–88. Available online: https://www.cambridge.org/core/product/identifier/S0376030800012313/type/journal_article (accessed on 28 April 2022).
- JCPDS Powder Diffraction File; International Centre for Diffraction Data: Swarthmore, PA, USA, 2000.
- Patterson, A.L. The scherrer formula for X-ray particle size determination. Phys. Rev. 1939, 56, 978–982. [Google Scholar] [CrossRef]
- Zhou, G.; Jin, P.; Wang, Y.; Pei, G.; Wu, J.; Wang, Z. X-ray diffraction analysis of the yttria stabilized zirconia powder by mechanical alloying and sintering. Ceram. Int. 2020, 46, 9691–9697. [Google Scholar] [CrossRef]
- Villarba, M.; Jónsson, H. Diffusion mechanisms relevant to metal crystal growth: Pt/Pt(111). Surf. Sci. 1994, 317, 15–36. [Google Scholar] [CrossRef]
- Ruckenstein, E.; Pulvermacher, B. Growth kinetics and the size distributions of supported metal crystallites. J. Catal. 1973, 29, 224–245. [Google Scholar] [CrossRef]
- Ghaani, M.R.; Catti, M. Investigation on the kinetic mechanism of the reduction of Fe2O3/CoO-decorated carbon xerogels: A non-isothermal study. J. Solid State Chem. 2019, 277, 368–375. [Google Scholar] [CrossRef]
- Lee, H.H. Kinetics of sintering of supported metal catalysts: The mechanism of atom diffusion. J. Catal. 1980, 63, 129–137. [Google Scholar] [CrossRef]
- Yi, H.; Xue, Q.; Lu, S.; Wu, J.; Wang, Y.; Luo, G. Effect of pore structure on Ni/Al2O3 microsphere catalysts for enhanced CO2 methanation. Fuel 2022, 315, 123262. [Google Scholar] [CrossRef]
- Omoregbe, O.; Danh, H.T.; Nguyen-Huy, C.; Setiabudi, H.D.; Abidin, S.Z.; Truong, Q.D.; Vo, D.-V.N. Syngas production from methane dry reforming over Ni/SBA-15 catalyst: Effect of operating parameters. Int. J. Hydrogen Energy 2017, 42, 11283–11294. [Google Scholar] [CrossRef]
- Shahid, M.; He, C.; Sankarasubramanian, S.; Ramani, V.K.; Basu, S. Co3O4-Impregnated NiO-YSZ: An Efficient Catalyst for Direct Methane Electrooxidation. ACS Appl. Mater. Interfaces 2020, 12, 32578–32590. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Liu, J.; Liu, F.; Wu, D. Reaction mechanism of CO2 methanation over Rh/TiO2 catalyst. Fuel 2020, 276, 118093. [Google Scholar] [CrossRef]
- Ashok, J.; Pati, S.; Hongmanorom, P.; Tianxi, Z.; Junmei, C.; Kawi, S. A review of recent catalyst advances in CO2 methanation processes. Catal. Today 2020, 356, 471–489. [Google Scholar] [CrossRef]
- Mebrahtu, C.; Krebs, F.; Abate, S.; Perathoner, S.; Centi, G.; Palkovits, R. Chapter 5—CO2 Methanation: Principles and Challenges. In Horizons in Sustainable Industrial Chemistry and Catalysis; Albonetti, S., Perathoner, S., Quadrelli, E.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 178, pp. 85–103. ISBN 0167-2991. [Google Scholar]
- El-Salamony, R.A.; El-Sharaky, S.A.; Al-Temtamy, S.A.; Al-Sabagh, A.M.; Killa, H.M. CO2 valorization into synthetic natural gas (SNG) using a Co–Ni bimetallic Y2O3 based catalysts. Int. J. Chem. React. Eng. 2021, 19, 571–583. [Google Scholar] [CrossRef]
- Razzaq, R.; Zhu, H.; Jiang, L.; Muhammad, U.; Li, C.; Zhang, S. Catalytic methanation of CO and CO2 in coke oven gas over Ni-Co/ZrO2-CeO2. Ind. Eng. Chem. Res. 2013, 52, 2247–2256. [Google Scholar] [CrossRef]
- Panagiotopoulou, P.; Kondarides, D.I.; Verykios, X.E. Selective methanation of CO over supported Ru catalysts. Appl. Catal. B Environ. 2009, 88, 470–478. [Google Scholar] [CrossRef]
- Choi, C.; Khuenpetch, A.; Zhang, W.; Yasuda, S.; Lin, Y.; Machida, H.; Takano, H.; Izumiya, K.; Kawajiri, Y.; Norinaga, K. Determination of Kinetic Parameters for CO2 Methanation (Sabatier Reaction) over Ni/ZrO2 at a Stoichiometric Feed-Gas Composition under Elevated Pressure. Energy Fuels 2021, 35, 20216–20223. [Google Scholar] [CrossRef]
- Wu, H.C.; Chang, Y.C.; Wu, J.H.; Lin, J.H.; Lin, I.K.; Chen, C.S. Methanation of CO2 and reverse water gas shift reactions on Ni/SiO2 catalysts: The influence of particle size on selectivity and reaction pathway. Catal. Sci. Technol. 2015, 5, 4154–4163. [Google Scholar] [CrossRef]
- Li, B.; Watanabe, R.; Maruyama, K.; Kunimori, K.; Tomishige, K. Effect of Ni Loading on Catalyst Bed Temperature in Oxidative Steam Reforming of Methane over r-Al2O3-Supported Ni Catalysts. Ind. Eng. Chem. Res. 2005, 44, 485–494. [Google Scholar] [CrossRef]
- Li, Y.; Men, Y.; Liu, S.; Wang, J.; Wang, K.; Tang, Y.; An, W.; Pan, X.; Li, L. Remarkably efficient and stable Ni/Y2O3 catalysts for CO2 methanation: Effect of citric acid addition. Appl. Catal. B Environ. 2021, 293, 120206. [Google Scholar] [CrossRef]










| Samples | BET Surface Area (m2 g−1) | Pore Size (nm) a | Pore Volume (cm3 g−1) b | Crystallite Size (nm) c | |
|---|---|---|---|---|---|
| dNiO | dNi | ||||
| YSZ | 5.825 | 5.639 | 0.0082 | - | - |
| 5% Ni/YSZ | 5.246 | 6.029 | 0.0079 | 21.73 | 25.74 |
| 10% Ni/YSZ | 5.335 | 6.356 | 0.0085 | 31.36 | 34.39 |
| 20% N/YSZ | 6.221 | 5.922 | 0.0092 | 31.24 | 36.52 |
| 30% N/YSZ | 6.081 | 5.846 | 0.0089 | 33.73 | 36.22 |
| 40% N/YSZ | 4.237 | 5.675 | 0.0060 | 35.75 | 39.11 |
| 50% Ni/YSZ | 4.842 | 6.064 | 0.0073 | 35.83 | 40.30 |
| 75% Ni/YSZ | 4.731 | 5.929 | 0.0070 | 35.92 | 41.08 |
| Samples | Reduction Peak Temperatures (K) * | |
|---|---|---|
| Peak 1 | Peak 2 | |
| 5% Ni/YSZ | 629 | - |
| 10% Ni/YSZ | 641 | 734 |
| 20% N/YSZ | 645 | 724 |
| 30% N/YSZ | 654 | 748 |
| 40% N/YSZ | 601 | 706 |
| 50% Ni/YSZ | 654 | 778 |
| 75% Ni/YSZ | 659 | 764 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Omoregbe, O.; Majewski, A.J.; Steinberger-Wilckens, R.; El-kharouf, A. Investigating the Effect of Ni Loading on the Performance of Yttria-Stabilised Zirconia Supported Ni Catalyst during CO2 Methanation. Methane 2023, 2, 86-102. https://doi.org/10.3390/methane2010007
Omoregbe O, Majewski AJ, Steinberger-Wilckens R, El-kharouf A. Investigating the Effect of Ni Loading on the Performance of Yttria-Stabilised Zirconia Supported Ni Catalyst during CO2 Methanation. Methane. 2023; 2(1):86-102. https://doi.org/10.3390/methane2010007
Chicago/Turabian StyleOmoregbe, Osaze, Artur J. Majewski, Robert Steinberger-Wilckens, and Ahmad El-kharouf. 2023. "Investigating the Effect of Ni Loading on the Performance of Yttria-Stabilised Zirconia Supported Ni Catalyst during CO2 Methanation" Methane 2, no. 1: 86-102. https://doi.org/10.3390/methane2010007